Abstract
Background: Preclinical and clinical studies indicated that endothelial progenitor cells (EPCs) enhanced blood vessel formation in many clinical situations. However, whether transplantation of EPCs would enhance chronic venous thrombus recanalization and resolution is unknown. Methods: Mononuclear cells were isolated from bone marrow of immature rats by density gradient centrifugation, cultured, and then transplanted into inferior vena cava of rats with experimentally induced thrombi. Vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), messenger RNA (mRNA), and protein expression levels were measured through real-time quantitative polymerase chain reaction and Western blotting of thrombi and adjacent caval walls 14 days following transplantation. Results: Transplantation of bone marrow-derived EPCs led to an increase in VEGF, bFGF, mRNA, and protein expression. In addition, transplantation of bone marrow-derived EPCs also resulted in reduced thrombus size and increased neovascularization in the specimen. Conclusions: Transplanted bone marrow-derived EPCs may be a therapeutic option for treating deep venous thrombosis.
Keywords
Introduction
Deep vein thrombosis (DVT) is a significant health care problem associated with serious short-term and long-term physical, social, and economic sequelae for patients. Apart from the immediate adverse consequences such as pulmonary embolism and mortality, long-term sequalae include venous outflow restriction, valvular damage and reflux, and ambulatory venous hypertension. These can result in postthrombotic syndrome (PTS), which is characterized clinically by leg pain, swelling, chronic ulceration, and lipoderamatosclerosis. 1 Approximately 1 in 3 patients would develop PTS within 5 years of developing DVT. However, standard treatment of DVT with anticoagulants has not shown any effect on thrombus resolution or on the development of PTS. 2
The resolution of venous thrombi after development of DVT is a complex and spontaneous process that requires orchestrated action of different cell types. 3 Studies have shown that in a process similar to wound healing, monocytic cells and myofibroblasts penetrate into thrombi and new capillaries form during the resolution phase. 4 It has also been shown that endothelial progenitor cells (EPCs) play a key role in neovascularization in the context of peripheral arterial disease and coronary heart disease.5,6 and help reestablish myocardial tissue after myocardial infarction. 7 Increasing evidence suggests that EPCs are recruited into the thrombus and contribute to venous thrombi organization and resolution.8,9
In the present experimental study, our goal was to determine whether the activity of EPCs can promote the formation of new blood vessels (neovascularization) in the thrombus, thereby enhancing the recanalization and resolution of thrombi, improving clinical outcome, and building a platform for a novel interventional therapy for the treatment for subacute and chronic DVT.
Materials and Methods
Endothelial Progenitor Cell Isolation, Culture, Identification, and Expansion
Ex vivo expansion of EPCs was performed using the method described in detail in Ref 10. Bone marrow was harvested from both femurs and tibias of 80, 3-week-old (about 50-100 g) male Wistar rats. Density gradient centrifugation was used to fractionate the mononuclear cells (density, 1.083). The cells were plated on human-fibronectin-coated (Sigma-Aldrich China Inc, Shanghai, China) culture dishes and maintained in an extracellular (EC) basal medium 2 ([EBM-2] Clonetics, San Diego, California) supplemented with 5% fetal bovine serum, human vascular endothelial growth factor A (VEGF-A), human fibroblast growth factor 2, human epidermal growth factor, insulin-like growth factor 1, and ascorbic acid. On day 4, the adherent cells were extensively washed to remove the unattached cells, and fresh culture medium was added. The culture medium was changed every 3 days and then cultured for a total of 14 days. The adherent cells were collected for immunohistochemical and immunofluorescent analyses, which were performed by staining with fluorescein-isothiocyanate-labeled Ulex europaeus agglutinin 1 (Sigma-Aldrich China Inc) and 1,1′-dioctadecyl-3,3,3′,3′- tetramethylindo carbocyanine (Dil)-labeled acetylated low-density lipoprotein (Biomedical Technologies, Stoughton, Massachusetts).
Animal and Thrombosis Model and Cell Transplantation
All procedures were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication no. 85-23, revised 1996). Laminated thrombi were produced in the inferior vena cava of rats (280-320 g) using models of venous thrombosis previously described in Ref 10. Endothelial progenitor cells were transplanted through the femoral vein 10 days after venous thrombosis was induced. The experimental rats were divided into 3 groups: A (n = 15), sham-operated group; B (n = 15), medium-injected group, injected with 1 mL EBM 2; and C (n = 15), EPC-transplantation group, received 1 mL (106) of EPCs. The animals were sacrificed on the 14th day following transplantation, and the thrombi and adjacent caval walls were harvested.
In order to monitor the transplanted EPCs, the EPCs were stained with fluorescent carbocyanine Dil (Biomedical Technologies, Madrid, Spain) before transplantation. The EPCs in suspension were washed with PBS and incubated with PBS containing Dil (2.5 µg/mL) for 5 minutes at 37°C and 15 minutes at 4°C. Then the cells were washed twice in PBS and resuspended in EBM 2 medium. On the 14th day after EPC transplantation, 4 rats from the EPC-transplantation group were sacrificed, and the thrombi and adjacent caval walls were harvested and snap frozen in liquid nitrogen.
Measurement of Thrombus Size
Tissue sections were fixed in 10% formalin saline and embedded in paraffin. Sections of 5 µm were taken at 0.5 mm intervals along the whole thrombus and stained with hematoxylin and eosin (H&E). Digital images of stained sections were taken at ×50 magnification (Nikon Imaging, shanghai, China). Acquisition, spatial calibration, and analysis of captured images were achieved in a blinded manner using Image-Pro Plus analysis software (Media Cybernetics UK, Berkshire, UK). Thrombus size was estimated by measuring the cross-sectional area of the thrombus in sections and adding the areas obtained along the entire length of the specimen.
Immunohistochemistry
The number of neovascular channels was evaluated at the microvascular level using light microscopy to analyze the sections harvested from the thrombi. The tissue sections were embedded in the optimal cutting temperature compound, snap-frozen in liquid nitrogen, and cut into 5-µm-thick sections. The total number of endothelial cells in the tissues was determined by immunohistochemical staining using the von Willebrand factor (vWF) antibody (1:200; Santa Cruz Biotechnology, Califronia), and the number of neovascular channels of vWF-positive cells was determined by counting the number of cells in 5 random high-power (×2 magnification) fields. The area of the fields was measured using an NIH Image analysis system.
Real-Time Reverse Transcriptase–Polymerase Chain Reaction
Total RNA was isolated and purified using the RNeasy Mini Kit (Roche, Reinach Switzerland), according to the manufacturer’s recommendations and stored at −80°C until use. A quantitative real-time reverse transcriptase–polymerase chain reaction (RT-PCR) assay was performed to measure the rat VEGF and bFGF messenger RNA (mRNA). Polymerase chain reaction primers for rat VEGF, bFGF, and β-actin for the control were designed by Shengong Biotechnology Company, Shanghai, China. The primers used were as follows: for VEGF, forward ATCTTCCAGGAGTACCCCGA and reverse TCACCGCCTTGGCTTGTCACA; for bFGF, forward CTTATTAAGCTTATGGCCGCCGGGAGC and reverse GGCCGAATTCGTCA GCTCTTAGCAGAC; and for β-actin, forward GAGCAAGCGTGGCATCCTGA and reverse CCAGAGGCGTAGAGGGACAG. All the real-time PCR data were measured and recorded with the ABI Prism 7700 sequence detector system (Applied Biosystems, Foster City, California). Data are presented as relative induction of VEGF or bFGF normalized to β-actin. The detailed information for isolation of RNA and protein is available in the article we previously published. 10
Western Blot
The protein expression of VEGF and bFGF were analyzed by Western blot. Two hundred micrograms of protein were separated on sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) under reducing and denaturing conditions and transferred to Trans-Blot nitrocellulose membranes. The membranes were incubated in a 1:500 dilution of polyclonal antirat VEGF (Abcam, 1 Kendall Square, Cambridge, Massachusetts) or a 1:500 dilution of polyclonal antirat bFGF (Abcam, 1 Kendall Square), followed by incubation with horseradish peroxidase-conjugated secondary antibody (1:2000; Abcam, 1 Kendall Square) or β-actin (1:10 000; Abcam, 1 Kendall Square) antibodies.
Statistical Analysis
All experiments were repeated twice to confirm the reproducibility of the results, and all the data were presented as means ± SE. The results for the 3 groups were compared using single-factor analysis of variance (1-way ANOVA), and the difference was considered significant when the P value was lower than .05. The statistical analysis was performed using the Statistical Package for Social Sciences (SPSS, Chicago, Illinois), version 13.
Results
Effect of EPC Transplantation on Thrombus Resolution
Endothelial progenitor cell transplantation resulted in a significant reduction in thrombus size in group C compared with groups A and B (P < .05). No significant difference was observed between groups A and B (P > .05; Figure 1 ).
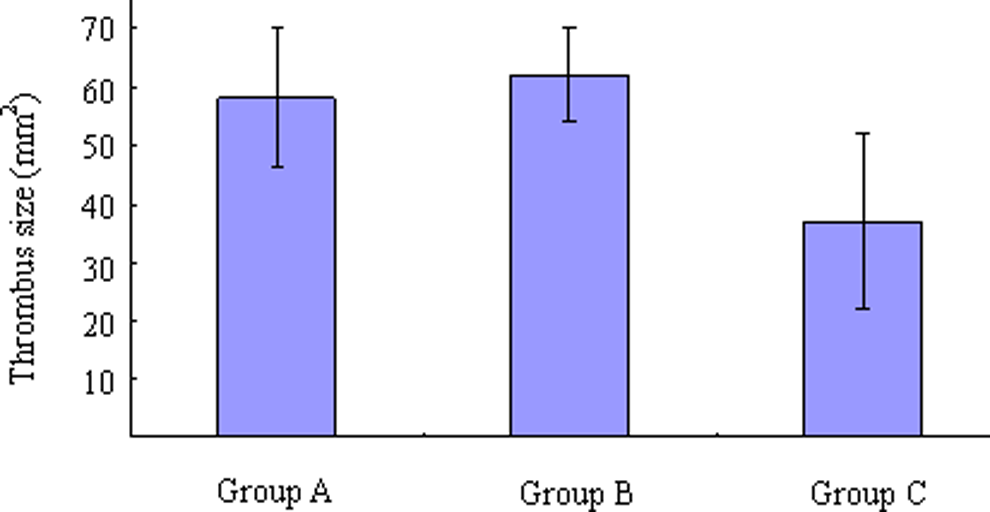
Quantitative assessment of thrombus size 14 days after transplantation in the 3 groups: A, sham-operated group (n = 15). B, medium-injected group (n = 15) injected with 1 mL EBM-2. C, EPC-transplantation group (n = 15), received 1 mL (106) of EPCs. EBM-2 indicates extracellular basal medium 2; EPCs, endothelial progenitor cells.
Histologic Findings
Neovascular channels developed in the periphery of the thrombus as part of the normal course of resolution in our induced model of DVT. The presence of capillary structures lined with vWF immunoreactive cells embedded in the thrombus illustrates the presence of true neovascularization in the organizing thrombi (Figure 2 ). The extent of thrombus neovascularization was estimated by counting vWF-positive channels under high-magnification (×400). Fourteen days after EPC transplantation, the number of channels increased significantly in group C compared to groups A and B (P < .05). No significant difference in the number of neovascular channels was found between groups A and B (P > .05; Figure 3 ).
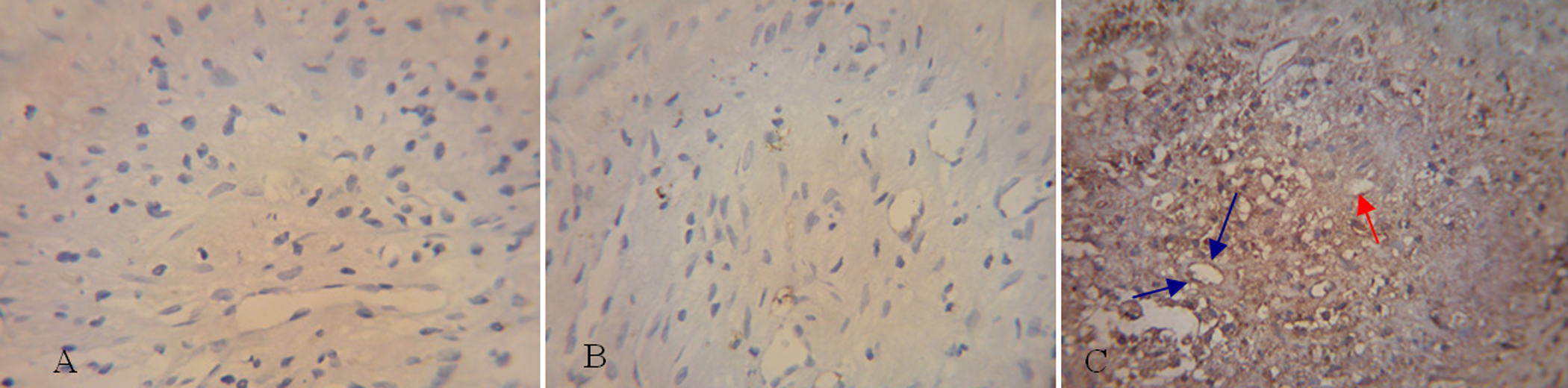
Transverse sections of experimental thrombi in the vena cava on the 14th day after transplantation in the 3 experimental groups show the presence of cell-lined recanalization channels (blue arrows). These channels stain immunohistochemically positive for the endothelial markers, vWF (100×) (red arrow). The channels are more abundant in group C. vWF indicates von Willebrand factor.
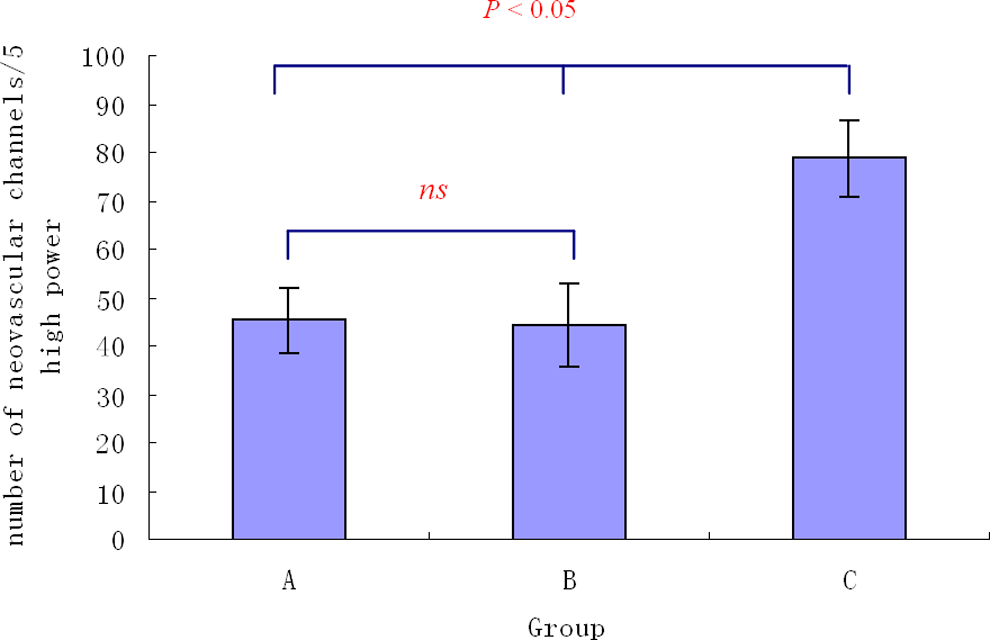
At 14 days after EPC transplantation, the number of channels increased significantly in group C compared to groups A (P < .05) and B (P < .05). There was no significance (ns) in the number of neovascular channels between groups A and B (P > .05). EPC, indicates endothelial progenitor cell.
Expression of VEGF and bFGF mRNA in the Thrombi and Adjacent Caval Walls After Transplantation
Samples of the thrombi and the adjacent caval walls were obtained 14 days after EPC transplantation. Figure 4 shows that VEGF and bFGF mRNA expression in group C were significantly higher than those in groups A and B (P < .05); groups A and B did not differ significantly from each other (P > .05; Figure 4).
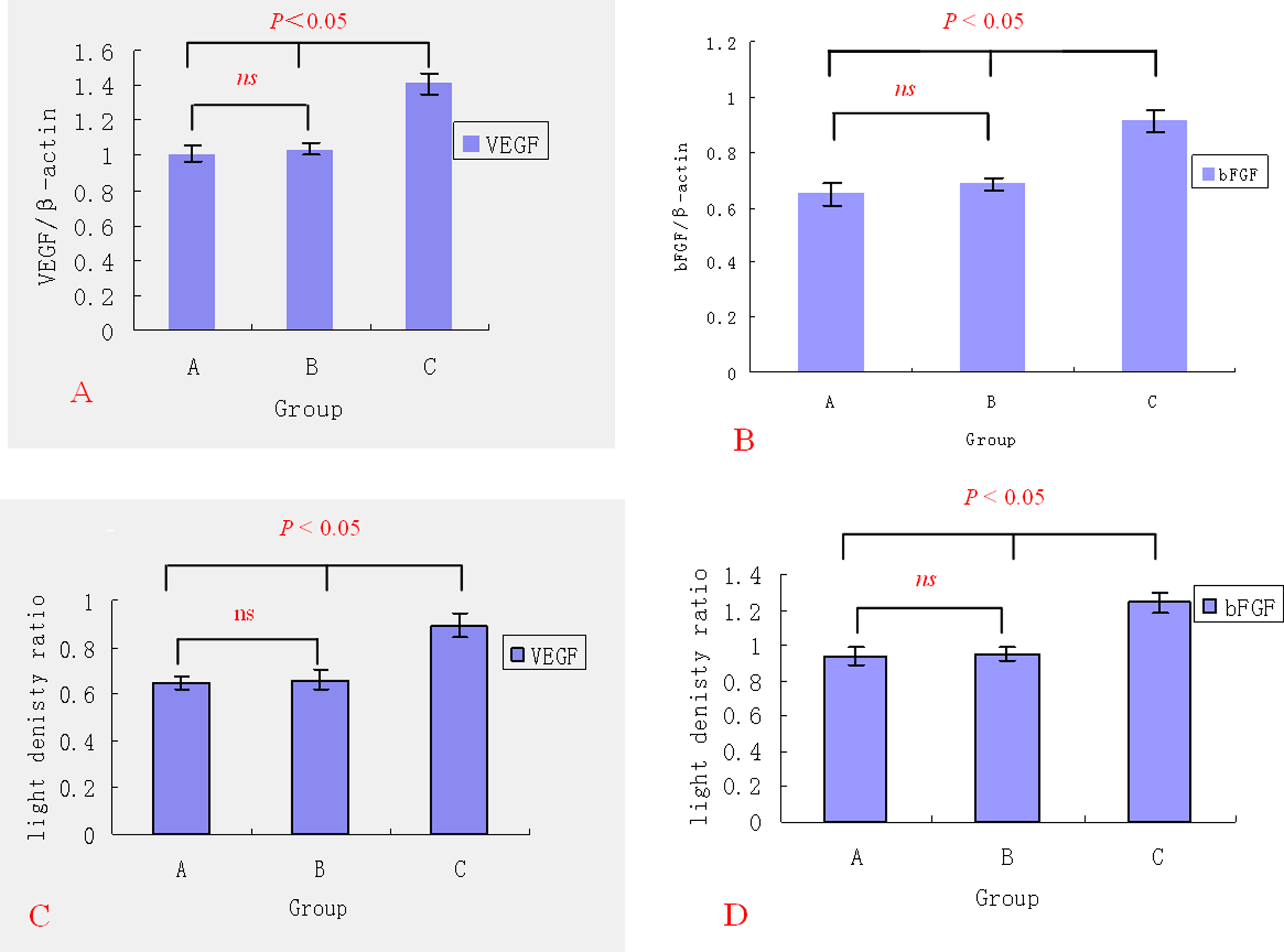
Expression of VEGF and bFGF mRNA in thrombi and adjacent caval walls. A, VEGF mRNA expression. B, bFGF mRNA expression. C, VEGF protein expression. D, bFGF protein expression. VEGF indicates vascular endothelial growth factor; bFGF, basic fibroblast growth factor; mRNA, messenger RNA.
Expression of VEGF and bFGF Protein in the Thrombi and Adjacent Caval Walls After Transplantation
As shown by the Western blot analysis, the expression of VEGF and bFGF protein significantly increased in group C compared to groups A and B 14 days after EPC transplantation (P < .05), and similar levels were found in groups A and B (P > .05; Figure 4).
Discussion
In the 1860s, Virchow showed that a thrombus resolves via organization and recanalization. The whole process was dynamic and influenced by many various microenvironmental factors including recruitment of inflammatory cells (initially neutrophils and subsequently monocytes), endothelial cells, and myofibroblasts.11,12 This process is very similar to the formation of granulation tissue that is essential for wound healing 13 and seems to depend on monocyte activity. Studies of venous thrombosis in animal models have shown that monocytes are recruited into maturing thrombi over time and that when their recruitment is reduced, thrombus resolution is severely restricted.14,15 Monocytes/macrophages are important mediators because they are capable of expressing a variety of proangiogenic factors such as VEGF, bFGF, and interleukin 8 (IL-8), which may generate an angiogenic drive within the thrombus.16,17 These proangiogenic growth factors can drive the development of new vascular channels in resolving thrombi. Neovascularization is the key process of venous thrombi resolution and recanalization. Restoration of blood flow through thrombi was identified in the early descriptions of venous thrombus resolution. 18 However, further research is still required to investigate whether new vascular channels can lead to blood flow restoration. Varma et al 19 thought thrombus vascularization alone was insufficient to induce thrombus resolution. However, new vascular channels within a thrombus are the critical element for the delivery of circulating chemokines and the recruitment of cells such as neutrophils and monocytes which are thought to contribute to the process of thrombus resolution.20,21 These new vascular channels are lined with endothelial-like cells which express many of the markers associated with endothelium, and the formation of these channels is analogous to angiogenesis. Furthermore, coverage with these endothelial-like cells prevents both activation of the clotting cascade and thrombus propagation by creating a barrier between blood and the thrombus surface. 22
Several studies were designed to evaluate neovascularization potential of EPCs in various animal models and in clinical trials.23,24 EPC-mediated therapeutic vasculogenesis has been well illustrated in cardiovascular disease models in which circulating EPCs preferentially home to ischemic tissue and are directly incorporated into vessel walls, thereby contributing to the natural mechanisms of blood vessel formation. Recognition of the therapeutic potential of these cells has already led to several pilot studies in humans.25,26 Clinically significant and consistent improvements were demonstrated with use of EPCs in these small nonrandomized trials.
Endothelial progenitor cells were also previously considered to play an important role in the process of resolution of venous thrombi. Normal tissues, including the vessel endothelium, have been shown to contain a certain set of resident progenitor cells that participate in the revascularization process when an appropriate stimulus and local conditions are present. Singh et al 15 reported that thrombus resolution is markedly delayed in urokinase gene-deleted animals but that they can be rescued by bone marrow transplantation. Modarai et al 8 showed that an increase in EPC number resulted in accelerated recanalization of thrombi. 27 Endothelial progenitor cell transplantation can change the microenvironment associated with venous thrombosis, as demonstrated in one of our previous studies. 10 Thus, it is likely that EPCs contribute to restore endothelial function by way of both direct incorporation and secretion of supportive factors that not only enhance their own survival but also help in recruiting other endogenous vascular progenitor tissues from remote tissues to the target site.
The results of our in vivo studies have demonstrated that EPC transplantation can decrease venous thrombus size and enhance venous thrombus resolution. The most interesting finding was that transplantation of EPCs significantly augmented the formation of vascular channels in the thrombi. Moreover, EPC transplantation led to changes of VEGF and bFGF in mRNA and protein expression within the thrombi. Vascular endothelial growth factor played a significant role in antithrombus agent by acting as a powerful endothelial cell mitogen and chemoattractant.28,29 Waltham et al 30 injected naked DNA gene of VEGF directly into a thrombus and found that it promoted thrombus recanalization and organization. In addition, animal studies have shown that administration of bFGF can substantially stimulate thrombus resolution. 19 Together, VEGF and bFGF could attract more EPCs into the thrombi to form new blood vessels by mobilizing the EPCs from the bone marrow and inducing the chemotactic movement of EPCs, thereby having a marked effect on thrombus resolution and recanalization. The decrease in thrombus size and increase in neovasularization induced by the use of transplanted EPCs in this study may offer a new therapeutic option for managing DVT.
Despite these encouraging findings, how these results will translate into actual clinical benefit will depend on how well the mechanisms of mobilization, integration, and functioning of these EPCS are elucidated in future. Because neovascularization is also a key component of tumor growth, malignant transformation with use of these EPCs is a major concern. Divergent opinions also occur about whether neovasuclarization actually accelerates thrombus resolution. 19 This study provides a starting point for future studies in this new therapeutic arena of DVT management.
Conclusion
It was indicated from the present study that transplanted bone marrow-derived EPCs may be a therapeutic option for treating DVT as transplantation of bone marrow-derived EPCs resulted in an increase in VEGF, bFGF, mRNA, protein expression, and neovascularization. Therefore, accelerating and promoting DVT resolution with the use of EPCs may be an attractive potential therapeutic option in the future to treat DVT-related symptoms.
Footnotes
Acknowledgment
This work was supported by Natural Science Foundation of Jiangsu Province (grant no. BK2007055).
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
