Abstract
Varicella zoster virus (VZV) is the only human virus known to replicate in arteries. After the acute infection, the virus persists in a noninfectious latent form in ganglia along the neuraxis, with intermittent periods of reactivation. Both primary and secondary reactivation are associated with stroke in children. These patients, regardless of the chosen treatment, have a high risk of recurrence, particularly those with worsening arterial stenosis. There are no specific therapy protocols for varicella-associated stroke in children, and the use of steroids or antiviral drugs is still controversial. We present a series of 4 children with stroke following varicella infection, with no recurrence and stable vascular stenosis at a mean follow-up of 18 months without steroid treatment. We also analyze possible correlations between anti-protein C, protein S and protein Z autoantibodies, and post-varicella arteriopathy.
Introduction
There is growing evidence in literature about the association between a history of varicella and arterial ischemic stroke during the following year in otherwise healthy children. 1 Varicella zoster virus (VZV) is the only human virus known to replicate in arteries, spreading both through the sensory nerves and blood vessels. Vasculopathy seems to result from infection and inflammation of cerebral arteries. After the acute infection, which is usually recognized by the appearance of the typical rash, the virus persists in a noninfectious latent form in ganglia along the neuraxis, with intermittent periods of reactivation. Varicella zoster virus infection may result in a wide spectrum of neurologic complications. Both primary and secondary reactivation are associated with stroke in children and adults. 2
The diagnostic criteria proposed by Lanthier and colleagues 3 for post-varicella arteriopathy are (a) first arterial ischemic stroke following varicella infection within 12 months; (b) unilateral vascular disease affecting the supraclinoid internal carotid artery, A1 or A2 segments of the anterior cerebral artery, or M1 or M2 segments of the middle cerebral artery; (c) no other possible etiology.
These patients, particularly those with evidence of worsening arterial stenosis, are reported to have a high risk of recurrence, irrespective of the chosen treatment. 3 It is still unclear whether corticosteroid or antiviral drugs in association with antithrombotic therapy are effective to improve the outcome.
Case Reports
Our series includes 4 children, aged between 26 months and 5 years, evaluated at the Department of Pediatrics, University Hospital of Padua, Italy, between August 2006 and November 2008, for arterial ischemic stroke. Table 1 summarizes the characteristics of our patients. All risk factors for stroke other than previous varicella infection have been ruled out with extensive tests, including complete screening for hereditary and acquired prothrombotic conditions, metabolic screening, serum iron and ferritin, and echocardiography. Three patients were also tested for specific anti-protein C, anti-protein S, and anti-protein Z autoantibodies using commercially available enzyme-linked immunosorbent assays (ELISAs; Zymutest anti-protein C immunoglobulin G [IgG] and IgM, Zymutest anti-protein S IgG and IgM, and Zymutest anti-protein Z IgG and IgM; Hyphen Biomed, Neuville sur Oise, France; Table 2 ). Tests for autoantibodies were performed according to the instructions of the manufacturer. Normal ranges were obtained in 60 healthy children of both sex aged 0 to 18 years. The value of 20 U (approximately, mean +2 standard deviation) was considered as a cutoff, and patients with a level >20 U were considered positive for the presence of autoantibodies.
Clinical, Radiological, and Therapeutic Data of Patients
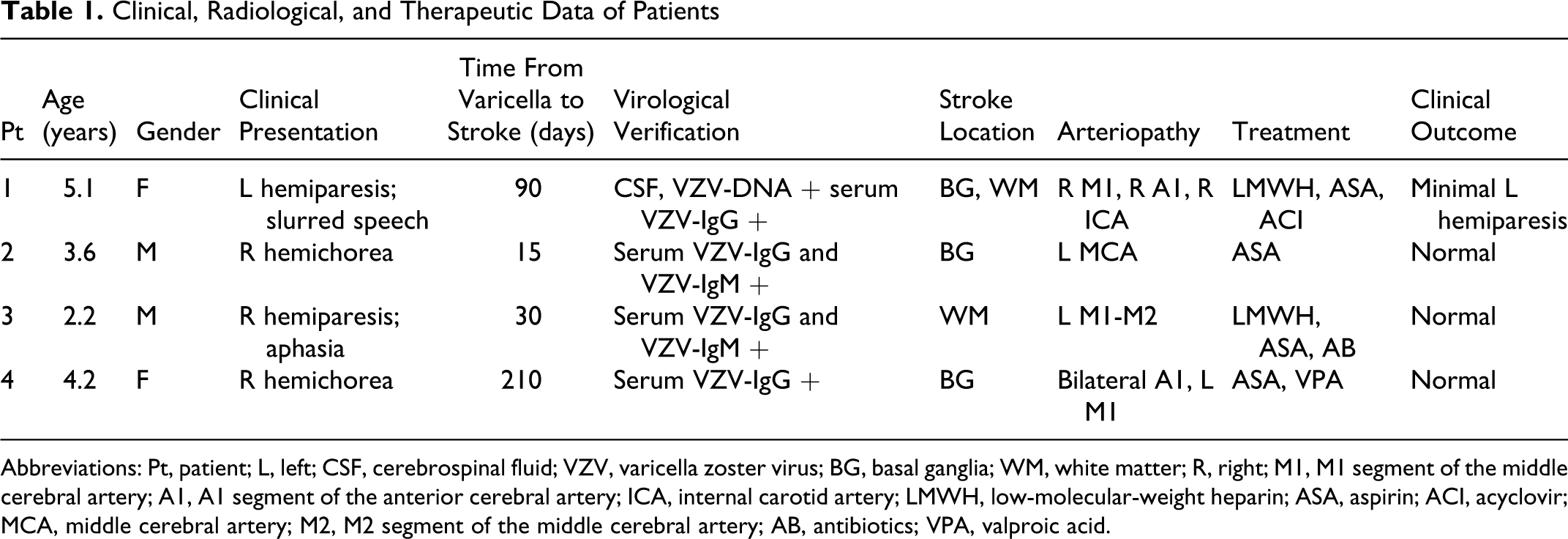
Abbreviations: Pt, patient; L, left; CSF, cerebrospinal fluid; VZV, varicella zoster virus; BG, basal ganglia; WM, white matter; R, right; M1, M1 segment of the middle cerebral artery; A1, A1 segment of the anterior cerebral artery; ICA, internal carotid artery; LMWH, low-molecular-weight heparin; ASA, aspirin; ACI, acyclovir; MCA, middle cerebral artery; M2, M2 segment of the middle cerebral artery; AB, antibiotics; VPA, valproic acid.
Anti-Protein C, S, and Z Levels in Our Patients a
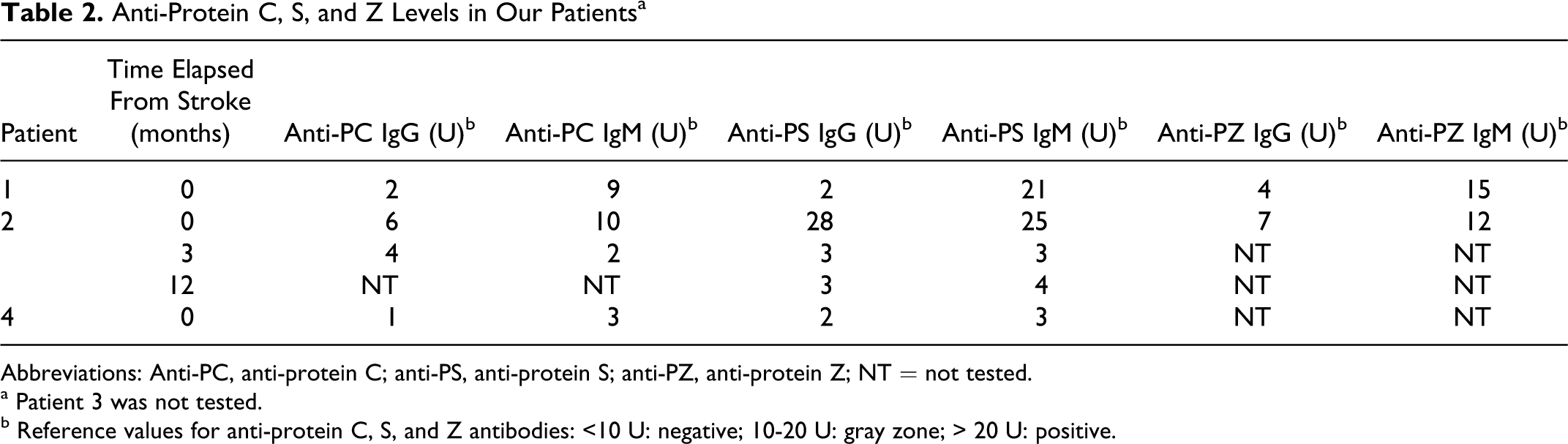
Abbreviations: Anti-PC, anti-protein C; anti-PS, anti-protein S; anti-PZ, anti-protein Z; NT = not tested.
a Patient 3 was not tested.
b Reference values for anti-protein C, S, and Z antibodies: <10 U: negative; 10-20 U: gray zone; > 20 U: positive.
Extensive thrombophilia screening including antithrombin, protein C and protein S antigen and activity, homocysteine levels, DNA analysis for factor V Leiden, methylenetetrahydrofolate reductase (MTHFR), and prothrombin G20210A mutation were also performed. Anticardiolipin and anti-β-2-glycoprotein I (GPI) antibodies and lupus anticoagulant were also tested.
Patient 1 was a 5-year-old girl with a history of transient left hemiparesis and subsequent brief episodes of dysesthesia in the following two days. After 3 days, the girl started to present slurred speech and left hemiparesis with VII cranial nerve palsy. She was then admitted to hospital. She did not have any previous medical condition, and she had varicella 3 months earlier. VZV-IgG titer was positive, with negative IgM. Magnetic resonance imaging (MRI) showed sub-acute ischemic lesions in the area of the right middle cerebral artery, and magnetic resonance angiography (MRA) revealed occlusion of the supraclinoid right internal carotid artery and severe stenosis of the M1 segment of the right middle cerebral artery and A1 segment of the right anterior cerebral artery. Cerebrospinal fluid was positive for VZV-DNA. Treatment with low-molecular-weight heparin was started, associated with antiviral treatment after 24 hours and followed by aspirin prophylaxis after 15 days. Blood tests revealed positive antinuclear antibodies ([ANA] 1:80) and anti-mycoplasma IgM (1:640) and negative antibodies to both extractable nuclear antigens (ENAs), antineutrophil cytoplasmic antibodies (ANCAs), myeloperoxidase antibodies (MPO), and anti-cardiolipin antibodies (ACAs). Serum neopterin level resulted 2.4 nmol/L (normal values 0.8-10) and a rheumatological evaluation excluded an autoimmune connective tissue disorder. The patient also had positive anti-protein S IgM antibodies (21 U). During the stay the child progressively improved and follow-up after 15 months showed minimal residual left hemiparesis, persistently positive ANA (1:320), and unchanged vascular stenosis.
Patient 2 was a 3-year-old boy who developed right hemichorea after 15 days from varicella infection. He had positive anti-VZV IgG- and IgM-specific antibodies. Cerebral imaging revealed ischemic lesions of the left caudate and lenticular nuclei and minimal left middle cerebral artery stenosis. He had positive anti-protein S IgG (28 U) and IgM antibodies (25 U). He received aspirin prophylaxis. After 2 years, he has a normal neurologic examination and MRA shows unchanged vascular stenosis. He also has normal anti-protein S IgG (3 U) and IgM (4 U) titers.
Patient 3 was a 2-year-old boy admitted because of right hemiparesis associated with aphasia during the course of a mild upper respiratory tract infection and after 1 month from varicella onset. Both IgG- and IgM-specific class antibodies were positive. Magnetic resonance imaging showed signal alteration at the left frontoparietal region and MRA revealed occlusion of M1 to M2 segments of the left middle cerebral artery. During the stay he received antibiotics and low-molecular-weight heparin, he showed progressive improvement, and he was discharged with aspirin. The last clinical and neuroradiological follow-up after 2 years and 8 months revealed complete neurologic recovery.
Patient 4 was a 4-year-old girl who developed right hemichorea approximately 7 months after varicella onset. Cerebral imaging showed ischemic lesions of left caudate and lenticular nuclei and internal capsule with bilateral A1 segment stenosis and left M1 segment stenosis. She had positive specific IgG and negative IgM. The child progressively improved with valproic acid and aspirin. One year after the stroke, she had a normal neurologic examination and unchanged vascular lesions.
In summary, all children had positive VZV-IgG and half of them had specific IgM class antibodies. Of the 4 patients, 2 presented with hemiparesis and 2 with hemichorea. Main vascular localization was the M1 segment of the middle cerebral artery (4 of 4) and ischemic lesions principally affected basal ganglia. Only 1 patient underwent lumbar puncture, and cerebrospinal fluid was positive for VZV-DNA. We tested 3 of the 4 patients for specific anti-protein S, protein C, and protein Z antibodies and 2 of the 3 patients had positive anti-protein S IgM and 1 of the 3 patients had also positive anti-protein S IgG. Neither inherited thrombophilia nor other antiphospholipid antibodies was detected in these patients. At the onset of the clinical manifestation, 2 children received low-molecular-weight heparin and 1 of them received also acyclovir. The other 2 patients received aspirin, at the dose of 2 mg/kg per d. All children continued aspirin until last follow-up examination. Valproic acid was administered to the child presenting with hemichorea. No children received steroids. In our case series, we observed no recurrence of stroke at a mean follow-up time of 18 months (range 7-32 months). Follow-up MRI was performed in all patients and 3 of the 4 patients also underwent MRA.
Discussion
Varicella is an important risk factor for arterial ischemic stroke, even if the estimated absolute risk of varicella-associated stroke is only 1 in 15000 children. 1 A prospective cohort study including 70 children with arterial ischemic stroke between the ages of 6 months and 10 years showed an incidence of varicella infection in the preceding year of 31%, compared with 9% in the general population. 1 All our patients had a first arterial ischemic stroke within 12 months from varicella infection, with a mean time interval of approximately 80 days.
Parental reporting has proved to be an accurate method for identification of a recent varicella infection. 4 Other methods include measuring varicella titers, which cannot provide accurate information on the timing of the infection, 1 and examination of cerebrospinal fluid. However, cerebrospinal fluid findings are not always conclusive. In fact, pleocytosis may be absent in one third of cases, 5 the virus is almost never isolated by culture because it is highly cell-associated and grows to low titers, 6 and polymerase chain reaction for VZV-DNA has showed poor sensitivity, resulting negative in up to 70% of VZV vasculopathy cases. 5 Only the detection of anti-VZV IgG antibody in cerebrospinal fluid has proved to be a reliable method, with a sensitivity of 100% in adults, 7 while in the pediatric population data are still insufficient.
According to the diagnostic classification proposed by Lanthier and colleagues, 3 post-varicella arteriopathy is characterized by unilateral vascular stenosis; in our series, patient 4 had bilateral stenosis of the A1 tract and left M1 segment stenosis, nonetheless we ruled out every other possible etiology of stroke other than previous varicella infection. Moreover, this criterion is not reported by all authors.6,8
There is growing evidence in literature that VZV infection is correlated with a vigorous immune response resulting in the production of a wide spectrum of coagulation autoantibodies. 9 Particularly, it has been shown that serious complications of varicella, including purpura fulminans and thrombosis, are associated with the presence of anti-protein S antibodies, resulting in transient protein S deficiency. 10 In our series, patient 1 presented with positive anti-protein S IgM and patient 2 with a transient elevation of both anti-protein S IgG and IgM.
The association between anti-protein Z antibodies and thrombosis is still unclear and different studies have reported conflicting results.11,12 None of our patients had positive anti-protein Z antibodies.
Although the exact role of these autoantibodies is still to be determined, their importance in the pathogenesis of the coagulation abnormalities found in VZV vasculopathy could be prominent. 13 Interestingly enough, no other inherited or acquired thrombophilia sometimes associated with stroke in children could be identified in our patients.
There are no specific therapy protocols for varicella-associated stroke in children. Several treatment options in association with antithrombotic therapy have been proposed, including antiviral therapy and corticosteroids. However, it is still controversial whether their use actually improves the outcome. The rationale for using steroids would be to consider that vasculopathy is secondary to a certain degree of vascular inflammation following viral replication within large and small cerebral arteries. 2 In a recent article by Nagel et al, 5 30 patients with VZV vasculopathy were analyzed. Six Of the 30, 6 (20%) were children. Two of them were treated with intravenous acyclovir without steroids; 1 of them improved immediately and the other worsened until steroids were added, with subsequent improvement. Two children were treated with both acyclovir and steroids from the beginning, and both of them clinically improved. The last 2 children did not receive acyclovir or steroids; 1 stabilized and the other improved slowly. Unfortunately, no data concerning the neuroradiological follow-up are available and therefore the exact course of the vasculopathy in these patients is not known. In our series, only 1 patient received acyclovir and no one received steroids; nonetheless, all of them clinically improved and MRA showed stable vascular lesions at follow-up. Considering that the exact pathophysiology of VZV vasculopathy has not been completely defined yet, further prospective studies are needed to assess the efficacy of steroids in the treatment of this condition in children.
It has been suggested that in most children with typical post-varicella arteriopathy, vascular stenosis follows a monophasic course, with possible progression for up to 6 months, succeeded by spontaneous regression for as long as 48 months after presentation and no restenosis. 3 In our case series, we observed neurologic improvement in all patients, but no one presented a clear regression of vascular stenosis.
The same study reported that approximately one third of children with post-varicella arteriopathy experienced recurrent arterial ischemic stroke up to 33 weeks after presentation, despite antithrombotic prophylaxis. All our patients received antithrombotic prophylaxis and there were no relapses at a mean follow-up time of 18 months.
In conclusion, previous varicella infection should be ruled out in all children presenting with stroke, particularly in patients with stenosis of a large cerebral vessel in the proximal segment of the circle of Willis. Diagnostic workup should always include MRI, MRA, and cerebrospinal fluid examination for VZV IgG antibodies. Further prospective studies are needed to assess the most appropriate therapy for children with this condition.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
