Abstract
Previously, our group showed a prothrombotic state in asymptomatic patients with chronic Chagas disease. The current paper studies the inflammatory status and endothelial function in these patients. Methods: In 40 patients and 40 healthy volunteers, we evaluated prothrombotic state, blood parasitemia (molecular biology: polymerized chain reaction [PCR]-amplification), tissue factor pathway inhibitor antibodies (aTFPI), interleukin 6 (IL-6), and vascular cell adhesion molecule-1 (VCAM-1). Endothelial function was determined by reactive hyperemia (pulse plethysmography). Results: In patients, prothrombin fragment 1 + 2,
Introduction
Chagas disease, caused by Trypanosoma cruzi (T cruzi), is transmitted in Argentina by Triatoma infestans and is currently a major public health problem in South America. An estimated 18 million individuals are infected, 1 with an incidence of 200,000 new cases each year. Chagasic cardiomyopathy is observed in 30% of the patients. The epidemiology of Chagas disease has changed in the recent decades due to migratory flux and has become a potential public health threat in countries where the disease is not endemic.
The natural progression of the disease presents an acute oligosymptomatic state, an intermediate asymptomatic state and a symptomatic state. 2 The pathological changes consistent with myocardial damage are observed early in the progression of Chagas disease 3 and frequently include pulmonary and systemic thromboembolic complications. 4
The etiopathogenesis of the chronic damage in Chagas disease is multifactorial. Several factors have been implicated in the beginning and the progression of injury in the organs. 4,5 In this sense, the cardiac damage has been postulated to be depending not only on the direct effect of the parasite but also on the effect of an autoimmune damage, the presence of a dysautonomia, and the dysfunction of the vascular endothelium. Previous to organ damage could exist metabolic alterations. Our group showed that a prothrombotic state exists in the intermediate state of chronic Chagas disease 6 and that this state is associated with thrombotic risk factors and with the significant presence of antiphospholipid antibodies (aFL)
Increased plasma levels of endothelial markers of inflammation have been seen in asymptomatic patients, 8 suggesting that prolonged exposure to these markers could affect the progression of Chagas cardiomyopathy. Although the role of endothelial dysfunction in Chagas disease has not been well established, a correlation between endothelial dysfunction and the plasma levels of different cytokines and adhesion molecules has been reported. 9
The aim of this study was to evaluate the inflammatory state and endothelial function in chronic asymptomatic patients with Chagas disease.
Materials and Methods
From March 2004 to December 2006, 40 asymptomatic chronic patients with Chagas disease, including 14 men and 26 women, were evaluated by outpatient consultation at the “Zenón J. Santillán” Hospital Health Center in Tucumán, Argentina. All patients included in this study were in functional class I of chagasic cardiomyopathy. This clinical classification of functional class was taken from Storino et al, which is specific to patients with chronic Chagas. 10 These cases were compared to a randomized selection of 40 healthy volunteers, comprising 19 men and 21 women.
All individuals underwent a physical examination, teleradiography of the chest, electrocardiogram, routine laboratory measurements, lipid profiling, and coagulogram. The study was approved by the Research and Teaching Committee of the “Zenón J Santillán” Hospital Health Center. Prior to the start of the study, informed consent was obtained from all patients and controls. In patients, the inclusion criteria were as follows: an immunoglobulin G (IgG) >1:32 (the assay used for this determination was hemaglutination method modified by Cerisola) 11 in 2 separate serological tests and a functional class I of chagasic cardiomyopathy. 10 The exclusion criteria were the presence of risk factors for atherosclerosis disease, risk factors for venous thromboembolism, abnormal routine laboratory values, an altered coagulogram (prothrombin time; activated partial thromboplastin time and thrombin time), and/or the presence of other concomitant diseases with or without medication.
In blood samples, the prothrombotic state was evaluated by
The presence of parasites in the blood was determined by polymerized chain reaction (PCR) amplification, using a thermostable DNA polymerase. The sensitivity of the method is 90%, and the specificity of the probe depends upon the primers used. In the current study, infection was detected using the primers 121/122, which are specific for T cruzi. This primer set amplified a 330-base pair (bp) region of the kinetoplast. 12 To test potential reaction inhibitors in the negative samples for T cruzi, a fragment of the human β-actin gene was coamplified.
With the preanalytical conditions established, serum tissue factor pathway inhibitor antibodies (aTFPI) were determined using an enzyme-linked immunosorbent assay (ELISA) method optimized by Forastiero et al, 13 which uses a solution of 15 μg/mL of TFPI in a bicarbonate−carbonate buffer at pH 9.6. Data from bibliography described that levels of aTFPI-IgG >18 UmL−1 and aTFPI-IgM >15 UmL−1 were associated with the presence of prothrombotic state in antiphospholipid syndrome. 13 Blood was also obtained to test the serum levels of interleukin 6 (IL-6) 14 (reference range: 3.12-12.5 pg/mL) and vascular cell adhesion molecule 1 (VCAM-1) 15 (reference range: 34.9-99.1 pg/dL) by ELISA.
Endothelial function was evaluated by a noninvasive method using a digital pulse-wave plethysmography (FEAS Electrónica, Cordoba, Argentina). This method determines reactive hyperemia and discriminates between participants with and without endothelial dysfunction. 16 The plethysmograph system was connected to an electrocardiograph system to record data for subsequent analysis. A positive hyperemia response was correlated with a conserved endothelial function. 17 Briefly, systolic arterial pressure (SAP) was first determined with an aneroid sphygmomanometer, followed by the placement of a photoelectric transducer on the index finger of the left hand, being careful to place the cells of the light sensor parallel to the transmitter. The electrocardiograph was calibrated (1 mm = 0.1 mV) to obtain a standard chart recording at a velocity of 25 mm/sec. Prior to the start of the recording phase, the patient was asked to hold his/her breath for 10 seconds (preocclusion apnea), which is necessary to eliminate technical artifacts produced by the mechanics of respiration. The pressure cuff was then insufflated to 50 mm Hg above SAP, to achieve arterial occlusion (blockage phase), and the blockage was maintained for 5 minutes. Subsequently, the pressure cuff was deflated, and after 2 minutes, another 10-second apnea was required to obtain the postocclusion recording (postblockage apnea). To measure the height of the valley/peak, 10 waves from each phase (preblockage vs postblockage) were averaged.
Statistical Analysis
The data were expressed as average values ± the standard error. A statistical probability of less than 5% (P < .05) was considered significant.
The Arcus Quickstat Biomedical Research Solution (Addison Wesley Longman, Cambridge, UK) and the statistical software package Statistica 5.0 were used to calculate statistical parameters. The Student t test was used for paired and grouped data sets. Analysis of variance−multivariate analysis of variance (ANOVA-MANOVA) with or without repeated measures and the Newman-Keuls posttest, chi-square, Fisher exact, and/or Mann-Whitney U tests were used as needed.
Results
Clinical characteristics were the following: No differences were observed between patients (n = 40) and healthy volunteers (n = 40) in age (33.5 ± 5 vs 28.8 ± 6 years, respectively; P = NS), body mass index (23.2 ± 1 vs 23.7 ± 1; P = NS), cardiac rate (79 ± 2 vs 77 ± 3 lat/min; P = NS), SAP (111 ± 4 vs 112 ± 5 mm Hg; P = NS), and diastolic blood pressure (82 ± 3 vs. 72 ± 3 mm Hg; P = NS). In addition, healthy volunteers as well as patients showed normal values by biochemical analysis: cell blood count, erythrocyte sedimentation rate, blood glucose levels, and lipid profiles. Chest teleradiographies and electrocardiograms were normal in both groups.
In relation to coagulogram, no differences were found between patients and healthy volunteers in prothrombin time (14.3 ± 0.2 vs 14.3 ± 0.1 sec; P = NS), activated partial thromboplastin time (44.9 ± 0.7 vs 44.6 ± 0.5 seconds, respectively; P = NS), and thrombin time (19.2 ± 0.2 vs 19.3 ± 0.3 seconds, respectively; P = NS).
In relation to prothrombotic state, we found that chagasic patients presented increase of PAI-1 values (chagasic patients: 4.95 ± 0.45 vs healthy participants: 3.54 ± 0.24 nmol/L P < .01). Also, prothrombin fragment 1 + 2 and
All healthy volunteers were found negative for the presence of the parasite by PCR amplification primers 121/122 specific for T cruzi. However, in the chronic chagasic patients, we found that 18 (45%) were positive and 22 (55%) were negative for PCR amplification. In patients with negative PCR, reaction inhibitors were ruled out by the fact that in the absence of 330-bp PCR product produced by the primers 121/122, β-actin amplification product was present.
Figure 1 shows values of aTFPI-IgG and aTFPI-IgM in patients and healthy volunteers. Chagasic patients presented higher values of aTFPI-IgG (P < .05) and aTFPI-IgM (P < .001) compared to healthy volunteers (Figure 1A and B). No association was obtained in chagasic patients between presence of the parasite by PCR amplification and values of aTFPI-IgG (P = .135; NS) or aTFPI-IgM (P = .296; NS).
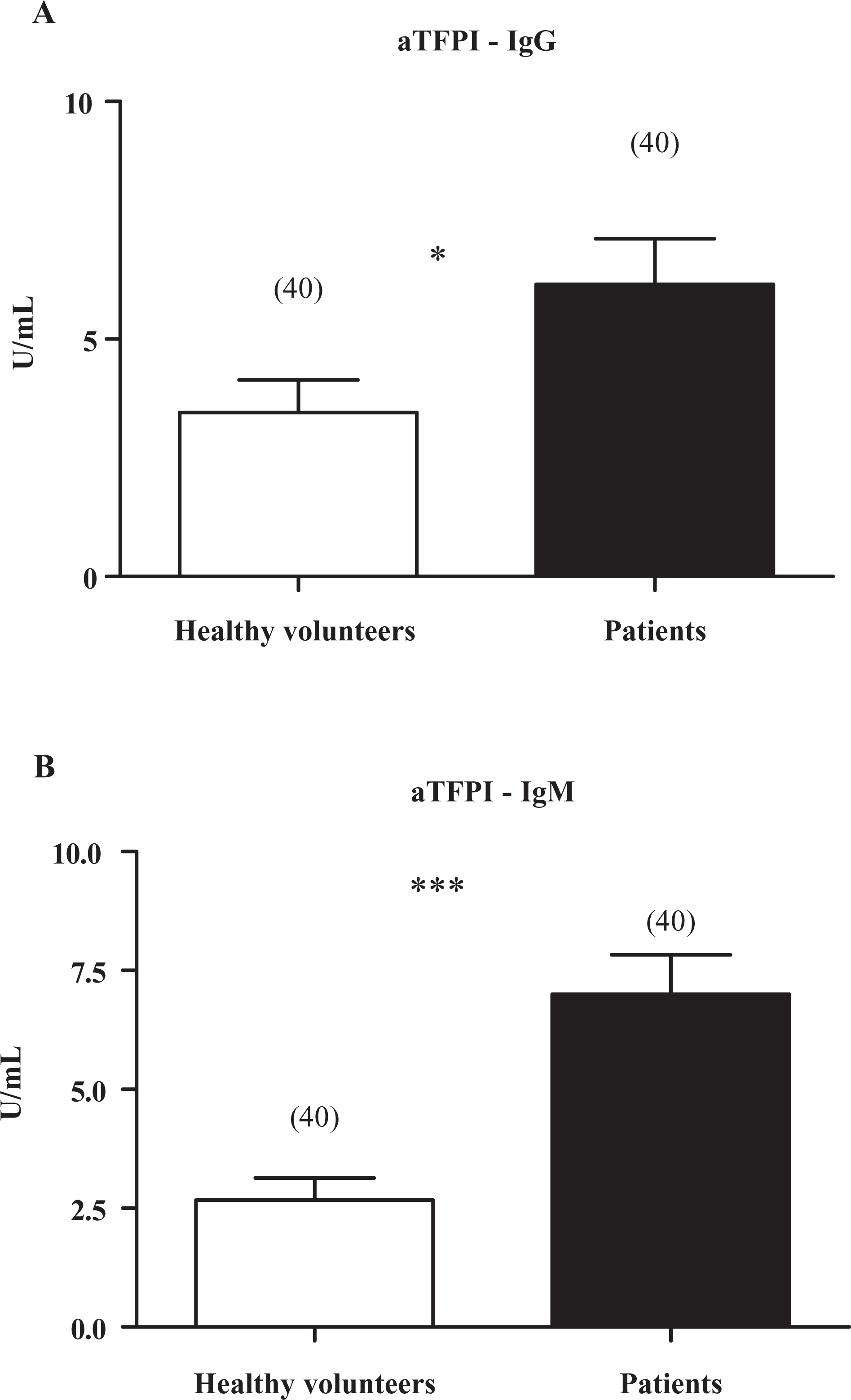
Values of tissue factor pathway inhibitor antibody (aTFPI) in plasma of healthy volunteers (□) and chagasic patients (▪). A, aTFPI- immunoglobulin G (IgG) determination. B, aTFPI-IgM determination. Data are expressed as mean ± standard error; n is given in parentheses; *P < .05; ***P < .0001; unpaired Student t test.
Figure 2 shows values of IL-6 in serum of patients and healthy volunteers. Interleukin 6 was able to be detected in serum of all individuals. However, significantly higher levels of IL-6 were observed in patients than in healthy volunteers (Figure 2).
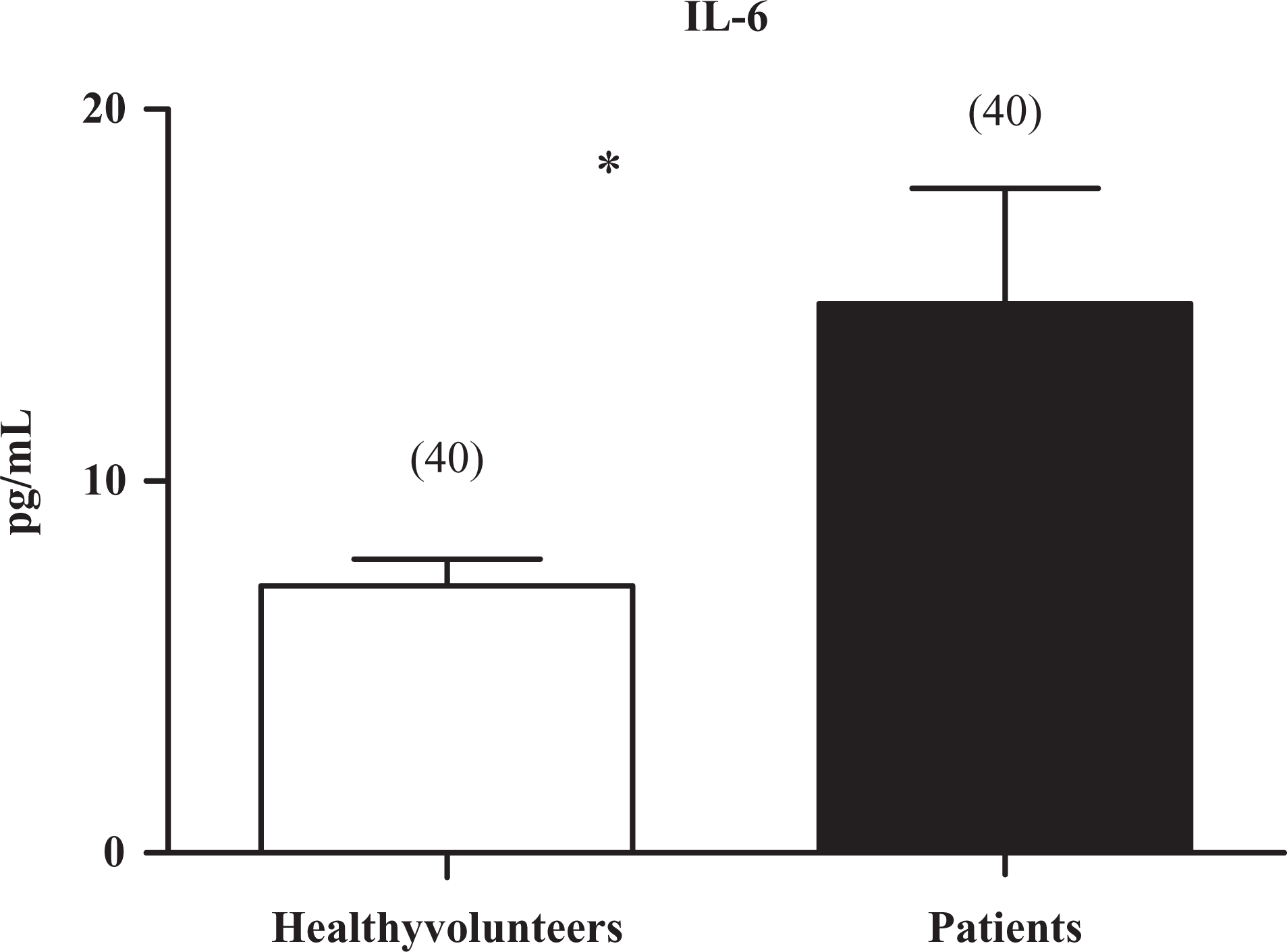
Values of Interleukin 6 (IL-6) in plasma of healthy volunteers (□) and chagasic patients (▪). Data are expressed as mean ± standard error; n is given in parentheses; *: P < .05; unpaired Student t test. NS indicates not significant.
Values of VCAM-1 in serum of patients and healthy volunteers are shown in Figure 3 . Similar to IL-6, the levels of VCAM-1 were elevated in the patients with respect to healthy volunteers.
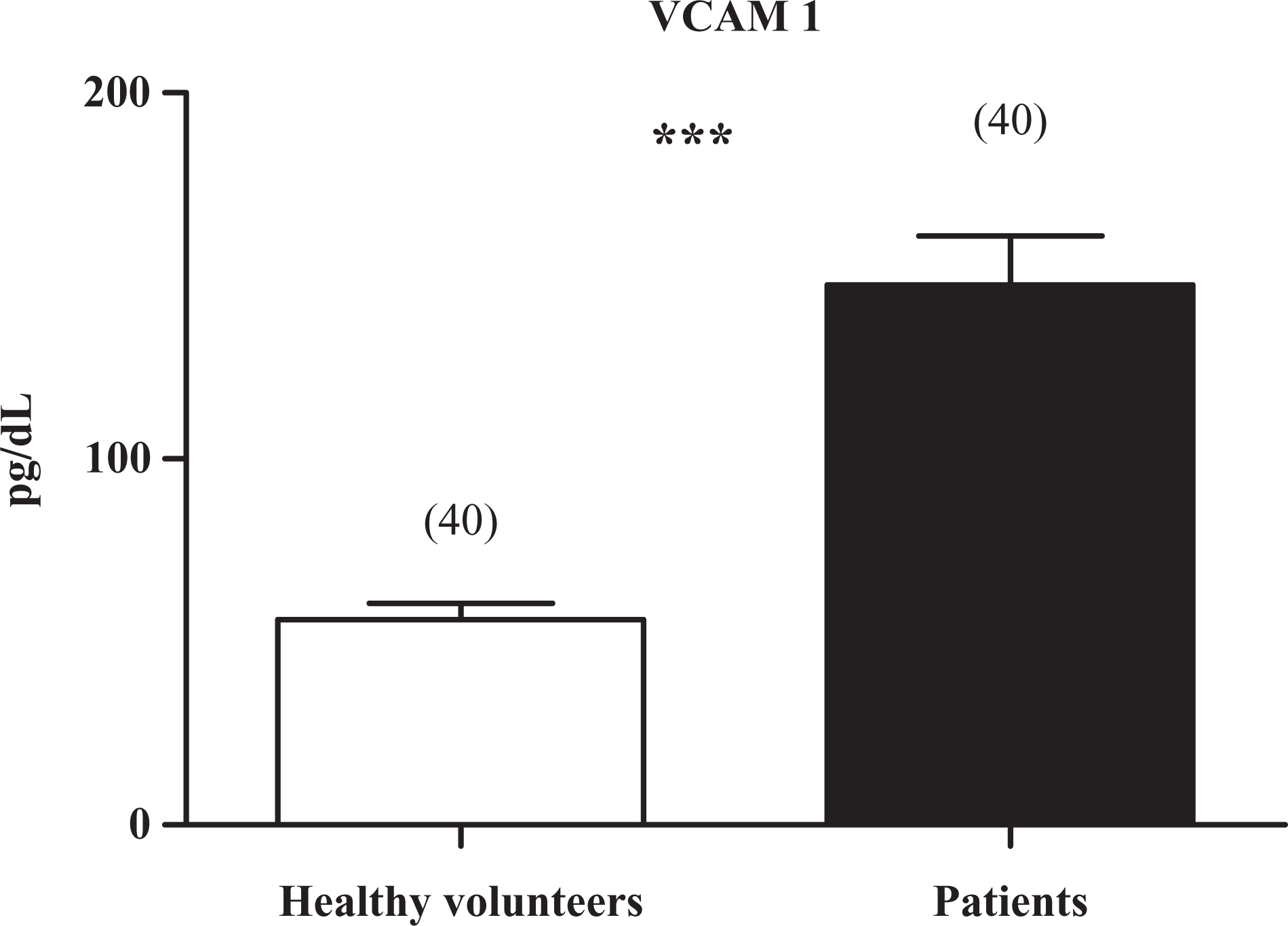
Values of vascular cell adhesion molecule-1 (VCAM-1) in plasma of healthy volunteers (□) and chagasic patients (▪). Data are expressed as mean ± standard error; n is given in parentheses; ***P < .001; unpaired Student t test.
In chagasic patients, no association between presence of the parasite by PCR amplification and values of IL-6 (P = .16; NS) or VCAM-1 (P = .36; NS) was obtained.
Figure 4 shows hyperemic response in patients and healthy volunteers. Hyperemic response was present in all studied individuals. In both cases, a statistically significant increase was observed in the relationship between valleys/peaks in postblockage vs preblockage (Figure 4). No differences were observed in the reactive hyperemia between healthy volunteers and chagasic patients (22.5% ± 3.5% vs 25.2% ± 6.9%, respectively; P = NS).
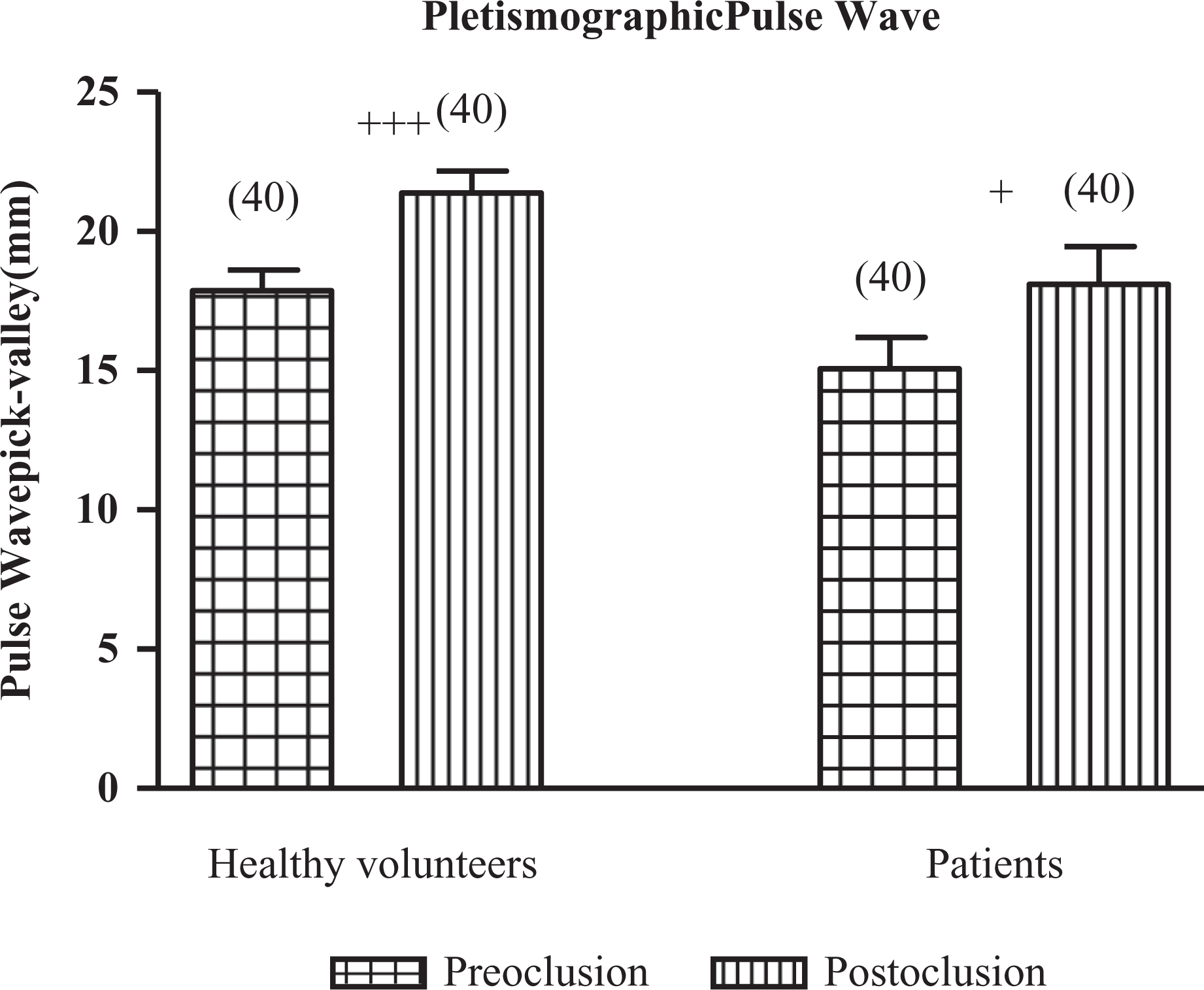
Pletismographic pulse-wave determination in healthy volunteers and chagasic patients. Data are expressed as mean ± standard error; n is given in parentheses; +++P < .001 preoclusion vs postoclusion in controls; +P < .02 preoclusion vs postoclusion in patients; ANOVA and Newman-Keuls post hoc comparision.
Discussion
In the current work, in asymptomatic patients with chronic Chagas disease, we found elevated levels of prothrombin fragment 1 + 2, fibrinogen,
In this study, the plasma levels of the cytokine IL-6 and the adhesion molecule VCAM-1 were used to investigate the existence of a subclinical vascular inflammatory state. Interleukin 6 is a potential marker of myocardial damage induced by T cruzi. Gao and Pereira showed that infection by T cruzi in animal models produces elevated serum and tissue levels of IL-6, 20 and the main inducer of IL-6 during T cruzi infections is the trans-sialidase enzyme produced by the parasite. 21 The relationship between IL-6 and the development of chronic Chagas damage, however, has not been completely defined. In this sense, few studies have shown a correlation between the serum levels of IL-6 and the progression of myocardial damage. 22,23 However, there are no studies that have addressed the role of VCAM-1 during asymptomatic chronic Chagas disease. The significantly higher levels of both IL-6 and VCAM-1 in the plasma of the patients suggest the presence of a subclinical inflammatory status during the asymptomatic state of this illness. A prolonged exposure to these inflammatory markers could influence the progression of Chagas cardiomyopathy. 24
The functional integrity of the endothelium is essential in the control of the vascular tone and the blood homeostasis. Endothelial cell cultures infected with T cruzi showed alterations in several mechanisms involved in the vascular dysfunction. 25,26 In humans, the analysis of endothelial function can be used as a clinical indicator of the presence of cardiovascular disease. There are several techniques that evaluate the behavior of the arterial and venous endothelium and determine the extent of dysfunction; digital pulse plethysmography is a noninvasive technique used to evaluate endothelial dysfunction. 27 At present, few studies evaluated the endothelial-dependent vasodilatation in Chagas disease, and they have shown different results, indicating that the degree of vascular dysfunction varies with the stage of the disease. In the coronary bed of patients with Chagas cardiomyopathy has been reported an altered endothelial-dependent function. 28 In agreement with our paper, Consolim-Colombo et al 29 studied peripheral endothelial function in patients with Chagas disease without cardiac insufficiency and they showed that it was preserved. In our study, reactive hyperemia, by digital pulse plethysmography, was present, indicating that, in this state of chronic Chagas disease, endothelial function was conserved.
The development of diagnostic techniques that are more sensitive and specific, such as PCR, may detect the presence of the parasite in blood at any stage of the disease. Salomone et al 30 suggest that the periodic cycles of parasitemia could explain the evolution of endothelial dysfunction, the activation of immunity, and the eventual thrombosis. Despite these data, a direct relationship between the duration, frequency, and parasitic load of each cycle and the development of pathological lesions has not been elucidated. Much of the pathophysiological damage in chagasic patients is likely mediated by different acute-phase factors, adhesion molecules, metalloproteinases, cofactors, interleukins, and neural hormones. 31 –33 Because a significant presence of the parasite could not be shown by the PCR test, and no association between PCR and aTFPI levels was found, a defined role of periodic cycles of parasitemia in the prothrombotic state could not be implicated in our patients. This observation is in agreement with other studies that reported absence of the parasite in the blood during the chronic state 34 This issue could be addressed by future studies focused on determining the presence or absence of the parasite at different timepoints over the course of the disease. In recent decades, particular attention has been placed on cellular and humoral autoimmunity phenomena, resulting in the identification of a variety of antigens that can cross between myocardial fibers and the parasite. 35,36 Higuchi established a relationship between the immune response and the parasitic load. 37 The authors indicated that a good immune response would result in a decrease of parasitic load, which would be unable to stimulate the development of a late immune response, leaving the patient in the intermediate stage. In contrast, a poor initial immune response would lead to persistent parasitemia that would induce a late immune response, which, in turn, would result in the development of the lesions that are typical of the disease. 37 In our article, we did not find a significant presence of the parasite by PCR amplification in patients with chronic Chagas disease during the intermediate state of the disease, suggesting that a defined role of T cruzi in blood in this state cannot be involved.
In conclusion, we found that asymptomatic patients with chronic Chagas disease presented a prothrombotic status associated with the existence of a subclinical inflammatory state explained by elevated levels of the inflammatory markers IL-6 and VCAM-1. The fact that endothelial function is still preserved suggests that prothrombosis and inflammation are primarily implicated in the beginning of cardiovascular damage. Although more studies are needed to reveal the potential clinical relevance of our results, the findings of the current paper may suggest that in these patients, the thrombosis and the inflammation should be treated early to prevent the endothelial dysfunction and the organ damage.
Footnotes
Acknowledgments
We thank Drs Elsa B. Velázquez and Mario Fatala Chabén of the Department of Research of the Instituto Nacional de Parasitología Buenos Aires, Argentina, for conducting the PCR tests and Dr Ricardo R. Forastiero of the Hematology Services of the Fundación Favaloro Buenos Aires, Argentina, for providing the anti-TFPI antibodies.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
Research supported by grants from the Consejo de Investigaciones dela UNT, the Consejo de Investigaciones Científicas y Técnicas (CONICET) dela República Argentinaand the Fundación Universidad Carlos III (Madrid - España).
