Abstract
Presence of high fever may cause confusion in differential diagnosis of pulmonary embolism (PE) versus pneumonia. The aim of this study is to investigate the diagnostic value of serum procalcitonin (PCT) in differential diagnosis of PE and community-acquired pneumonia (CAP). A total of 24 patients with proven PE and 22 patients with CAP were included in the study. The study population was subdivided as PE patients with fever (group 1, n = 8) and without fever (group 2, n = 16); and CAP (group 3, n = 22). Serum PCT and systemic inflammatory markers were measured at the initial diagnosis and the third day of the treatment. The relation of PCT level with the other systemic inflammatory markers was investigated in each measurement point. The initial mean serum PCT level in group 3 (2.24 ± 0.99 ng/mL) was statistically higher than group 1 (0.48 ± 0.77 ng/mL) and group 2 (0.14 ± 0.17 ng/mL; P = .000, .000, respectively). Procalcitonin level at the initial (2.24 ± 0.99 ng/mL) and the third day of treatment (0.92 ± 0.62 ng/mL) in group 3 showed a statistically significant reduction (P = .000). There were no statistically significant reduction in PCT levels by anticoagulation in groups 1 and 2 (P = .262, .119, respectively). Other systemic inflammatory markers including interleukin 6 (IL-6), C-reactive protein (CRP), and tumor necrosis factor α (TNF-α) levels statistically significantly decreased with anticoagulant and antimicrobial therapy. This study suggested that serum PCT level may be valuable for differentiating PE patients with or without fever from patients with CAP.
Keywords
Introduction
Pulmonary embolism (PE) and community-acquired pneumonia (CAP) are common diseases with high annual incidences. 1 In many cases, the differential diagnosis is still difficult because of the overlap signs and symptoms such as dyspnea, fever, cough, hemoptysis, and pleuritic chest pain. The classical laboratory markers (eg, blood leukocyte count, C-reactive protein [CRP], erythrocyte sedimentation rate [ESR]) have low specificity for the differential diagnosis of the PE and CAP.
Procalcitonin (PCT) is a prohormone of the calcitonin that is produced by the C cells of the thyroid gland. Procalcitonin is secreted as a part of the systemic inflammatory response to the infection. Serum values of PCT changes mostly based on the severity and type of infection. 2 Its level increases rapidly in bacterial infection but remains low in viral diseases. 3
Currently, the role of PCT in PE is not well studied. Therefore, it is not proven that PCT is not elevated in PE. In a previous study, Delèvaux et al suggested that patients with PE did not show elevated level of PCT even in febrile ones. 4 This study is designed to investigate the diagnostic value of PCT in differential diagnosis of PE particularly for PE patients with fever from patients with CAP. The second goal of the study is to demonstrate possible relation of PCT with other systemic markers in appropriately treated patients with PE and CAP.
Materials and Methods
Study Population
In all, 24 patients (11 male and 13 female, mean age = 54.95 ± 14.95 years) with PE and 22 patients (15 male and 7 female, mean age = 55.09 ± 17.92 years) with CAP were included in the study. Patients with PE were divided into 2 groups with 8 patients (2 male and 6 female, mean age: 54 ± 22.27 years) with fever (group 1) and 16 patients (9 male and 7 female, mean age = 55.43 ± 10.50 years) without fever (group 2). Patients with CAP were served as group 3.
Definition of PE and CAP
The diagnosis of PE was performed according to high or moderate probability ventilation-perfusion scan obtained according to previously published protocol by Prospective Investigation of Pulmonary Embolism Diagnosis (PIOPED) investigators 5 or diagnostic computerized tomography (CT; single- or multislice) findings with appropriate clinical probability. 6,7
In case of proven PE, patients received a regular care of standardized unfractionated heparin and coumarin combination or low-molecular-weight heparin.
The diagnosis of CAP was established according to the Infectious Diseases Society of America (IDSA) guideline. 8 An acute illness with respiratory symptoms and infiltrate on chest X-ray or auscultatory findings consistent with CAP was defined as CAP. 8 Patients with CAP who were included in the study were treated with appropriate antibiotic therapy according to the guidelines.
Fever was defined as an axillary body temperature measured within 24 hours of the admission, which exceeded 37.2°C. 9 Pulmonary embolism patients with fever did not receive antibiotics.
Inclusion and exclusion criteria
Pulmonary embolism patients without fever were consecutively included if they had remained afebrile in the whole course of disease during the hospitalization. Pulmonary embolism patients with fever were evaluated with repeated blood, sputum, throat, and urine cultures to detect the origin of fever. Patients with PE who had high fever originating from other identifiable reasons were excluded from the study. Patients with PE who developed symptoms consistent with CAP and/or who had received empiric antibiotics and/or underwent mechanical ventilation were excluded from the study.
Patients with CAP who had other site of infection, who had signs and symptoms of deep venous thrombosis (DVT), and a past history of venous thromboembolism (VTE) were excluded from the study.
Patients with systemic diseases that may influence the serum levels of PCT such as lung cancer, extensive surgery (eg, aortic, cardiac, and colonic), trauma, and extrapulmonary infectious diseases were also excluded from the study. 10
Study Design
Patients were evaluated at the initial diagnosis and the third day of the treatment of the hospitalization days. The highest body temperature was recorded daily. Blood leukocyte count, CRP, ESR, PCT, interleukin 6 (IL-6), and tumor necrosis factor α (TNF-α) levels were measured at defined measurement points. The difference between the groups at the initial level was compared. Hence, an ingroup analysis was performed to see how those parameters were influenced by appropriate treatment.
Laboratory workup
Standard hematological and biochemical laboratory tests were measured routinely. C-reactive protein was measured using a nephelometric assay (Specific Protein Analyzer, Beckman, Marburg, Germany), with the normal range defined as 0.0 to 6 mg/L.
Blood samples for PCT, IL-6, and TNF-α were immediately centrifuged at 3500 rpm for 10 minutes and subsequently stored at −70°C, according to the manufacturer’s recommendations.
Procalcitonin was measured using serum samples by the immunofluorescent assay (BRAHMS PCT-sensitive Kryptor; BRAHMS AG; Hennigsdorf, Germany). BRAHMS Kryptor uses TRACE (Time Resolved Amplified Cryptate Emission) technology, based on a nonradiative transfer of energy. The method based on a sheep polyclonal anti-calcitonin (CT) antibody and a monoclonal anti-katacalcin antibody, which bind to the CT and katacalcin sequence of CT precursor molecules. The assay has a functional assay sensitivity of 0.06 µg/L. 11
Serum IL-6 levels were measured with a solid phase enzyme linked-immuno-sorbent assay (ELISA) using human IL-6 (hIL-6) kit (Biosource International, Inc, Camarillo, CA-USA). The minimum detectable dose of hIL-6 is 2.0 pg/mL with this assay protocol. 12
Plasma TNF-α levels were measured with a solid phase sandwich ELISA using human TNF-α (hTNF-α) kit (Biosource International, Inc). The minimum detectable dose of hTNF-α is 1.7 pg/mL with this assay protocol. 13
Statistical Analysis
Statistical results are presented as means ± standard deviation. SPSS for Windows Release 11.5 (2002) statistical software was used for statistical analysis. P < .05 was considered statistically significant. Chi-square and Fisher’s exact chi-square tests were performed to compare selected subgroups. Kruskal-Wallis test were used for a comparison of 3 groups. When the difference was found statistically significant by the Kruskal-Wallis test, the Mann-Whitney U test was performed in coupled subgroups. Bonferroni correction was used for the calculation of P values for these selected groups.
Results
The flow diagram of study population was shown in Figure 1 . Of the 81 consecutive patients with PE, 57 patients were excluded from the study in 1 year period. Seven patients of group 1 were diagnosed with thorax CT scan, whereas 1 patient was diagnosed with high-probability lung perfusion scan. Nine patients from group 2 were diagnosed with high-probability lung perfusion scan, whereas 7 patients were diagnosed with thorax CT scan.
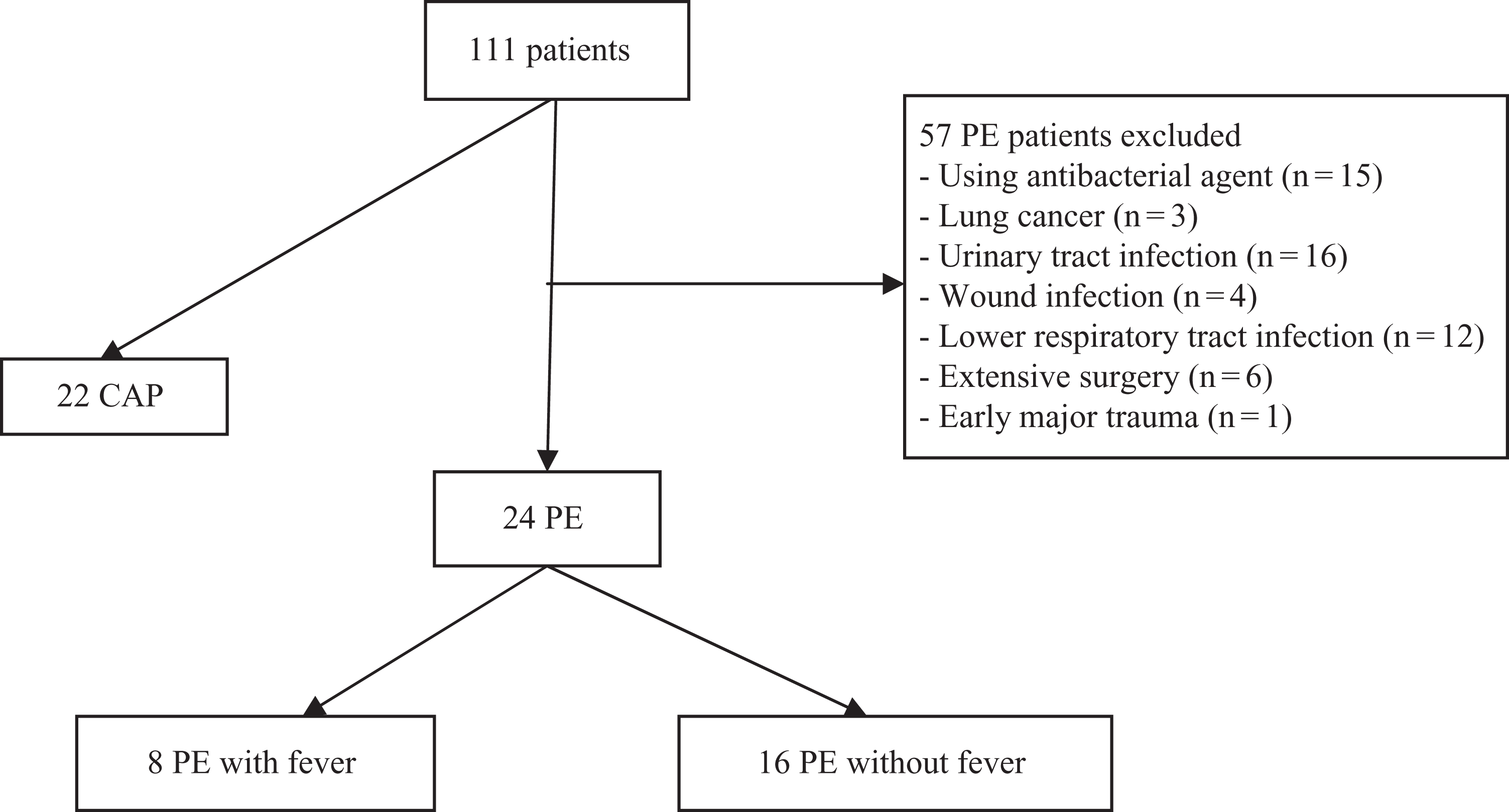
Flow diagram of study population. PE indicates pulmonary embolism; CAP, community-acquired pneumonia.
Risk factors for PE were evaluated in each patient. In all, 6 (75%), 11 (68.8%), and 1 (4.5%) patients had a history of immobility in groups 1, 2, and 3, respectively. Two (25%), 4 (25%), and 1 (4.5%) patients had a history of past surgery in groups 1, 2, and 3, respectively. One (12.5%) patient had a history of trauma in group 1. Six (37.5%) patients of group 2 had a history of either current or past DVT. None of the patients with CAP had a sign of current or past DVT.
The mean levels of different parameters at initial level in 3 groups were summarized in Table 1 . Subgroup comparison analysis showed that only body temperature and ESR showed statistically significant difference between groups 1 (PE with fever) and 2 (PE without fever; P = .000 and P = .000, respectively). Procalcitonin level was the only statistically different parameter between groups 1 (PE with fever) and 3 (CAP; P = .001). When subgroup comparison was performed between groups 2 and 3, body temperature, CRP, ESR, PCT, and IL-6 levels were significantly different between the groups (0.000, 0.000, 0.005, 0.000, and 0.006, respectively).
Comparison of Valuables at Initial Evaluation in 3 Groups
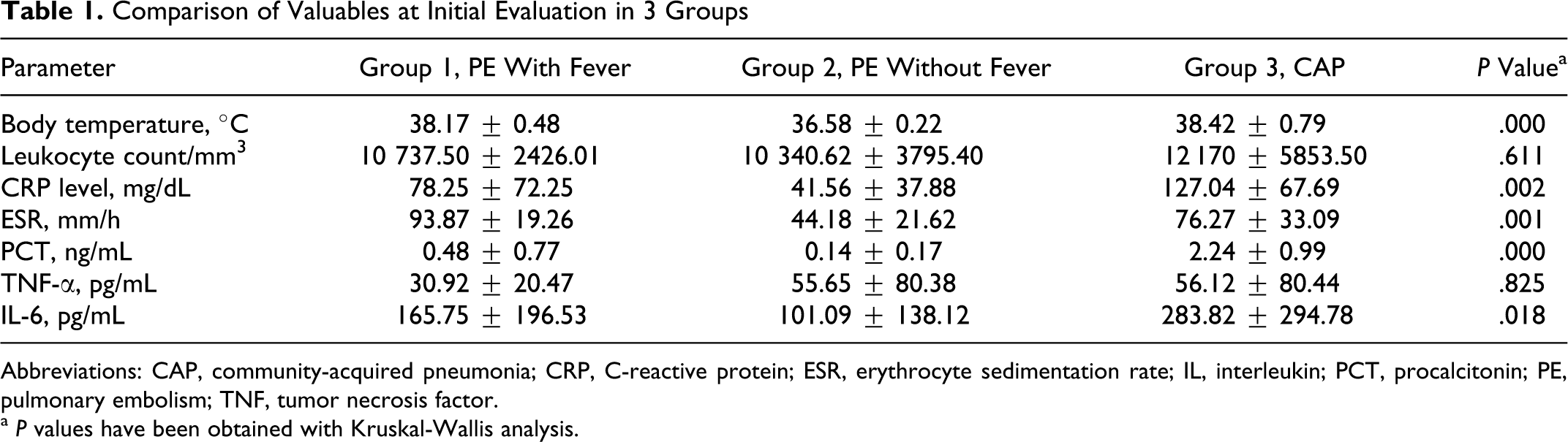
Abbreviations: CAP, community-acquired pneumonia; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IL, interleukin; PCT, procalcitonin; PE, pulmonary embolism; TNF, tumor necrosis factor.
a P values have been obtained with Kruskal-Wallis analysis.
The effect on treatment in different measurement points were analyzed in ingroup comparisons (Table 2 ). According to the statistical analysis, in group 1, except for the TNF-α and PCT level, other parameters were significantly decreased with anticoagulant treatment. In group 2, body temperature and PCT level were not statistically different between the initial and the third-day measurement, but other parameters were significantly decreased by anticoagulant therapy. In group 3, all parameters decreased significantly due to antimicrobial therapy. Comparative results were shown in Figures 2 and 3 .
Clinical and Laboratory Parameters of All Groups on Admission and Third Day
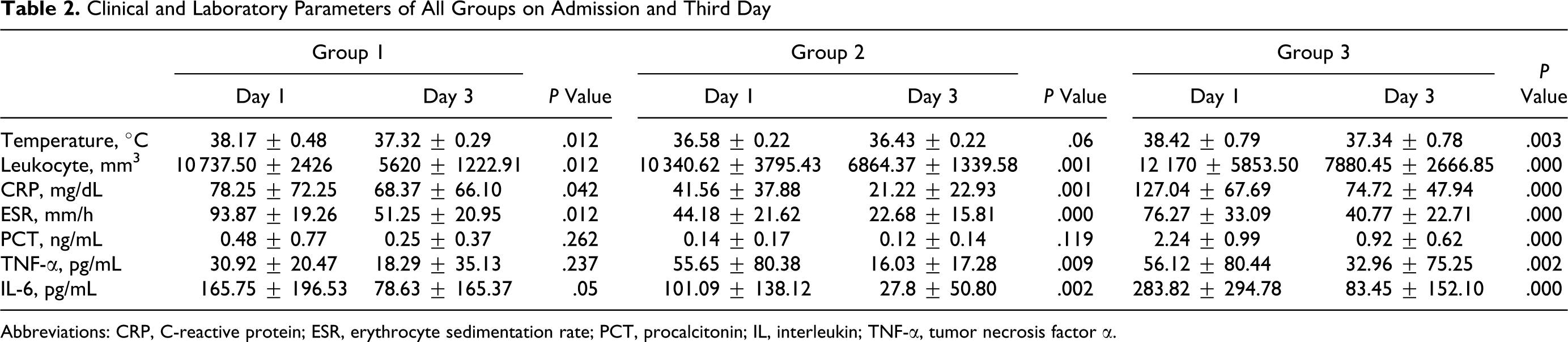
Abbreviations: CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; PCT, procalcitonin; IL, interleukin; TNF-α, tumor necrosis factor α.
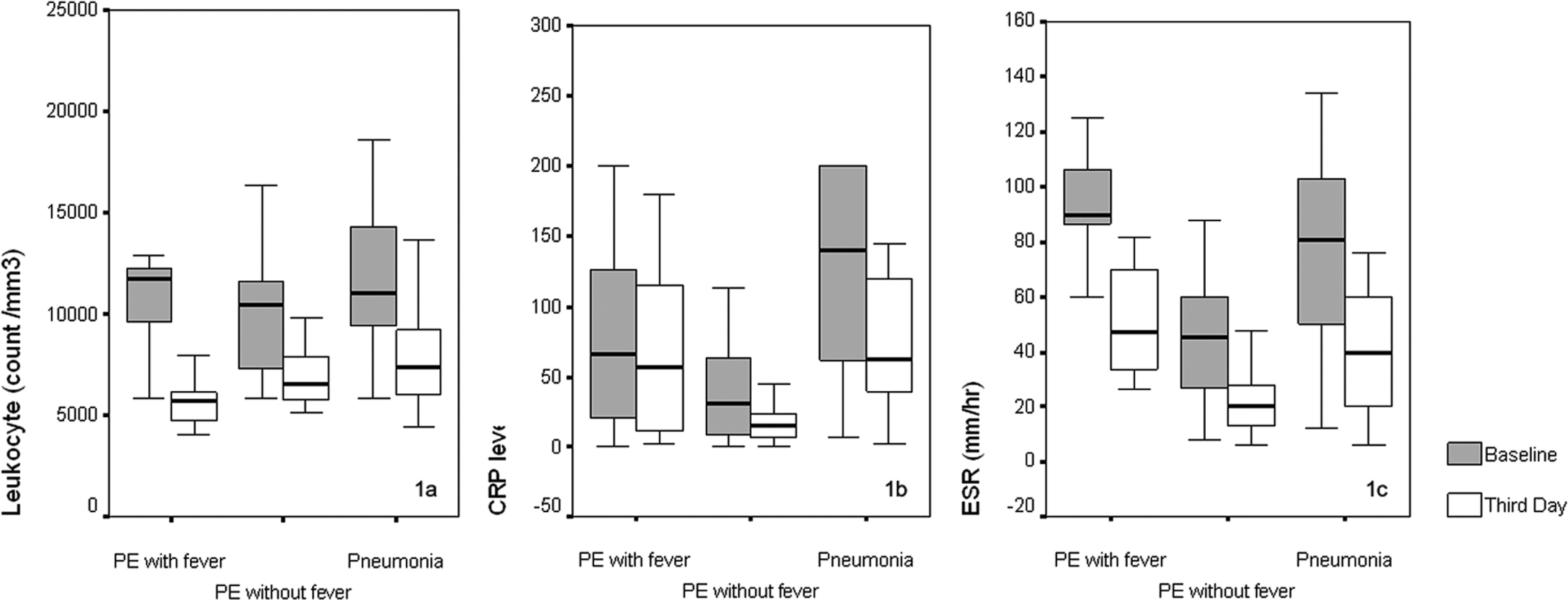
Comparative results of leukocyte, C-Reactive Protein (CRP), and erytrocyte sedimentation rate (ESR) levels between the groups.
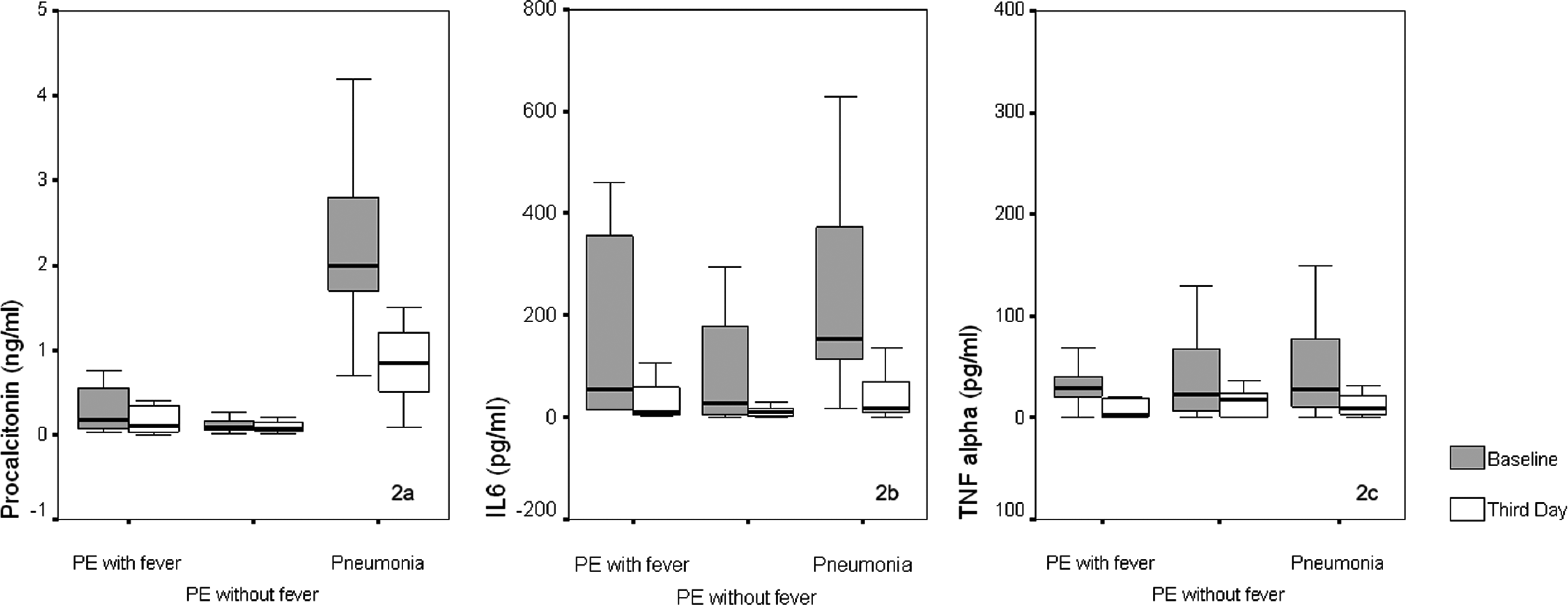
Comparative results of procalcitonin, interleukine -6 (IL-6) and tumor necrosis factor alpha (TNF-alpha) levels between the groups.
Correlation analysis showed that initial PCT level was correlated with body temperature, CRP, and IL-6 level (r = .653, P = .000; r = .455, P = .001; and r = .453, P = .002, respectively) in all study population.
Discussion
In the current study, we found that patients with CAP had significantly higher PCT levels than PE patients with or without fever on admission. Although, PCT levels did not change with anticoagulant treatment in patients with PE, antimicrobial therapy caused the decreasing level of PCT in CAP group. These findings suggested that serum PCT levels might be a useful marker to differentiate patients with CAP from patients with PE particularly the ones with fever.
Despite current extensive guidelines, the differential diagnosis of PE and CAP are still a dilemma in the clinical practice due to the same clinical presentations. Stein et al reported that fever occurs in 26% of patients with PE diagnosed by pulmonary angiography 14 ; hence, the presence of fever has been considered an argument against PE in favor of other diagnosis such as pneumonia. Blood leukocyte count, CRP, and ESR levels significantly increased in all febrile pulmonary conditions. Söderberg et al reported that CRP levels could help to differentiate CAP from PE. However, in certain patients with PE, very high values could be found without pneumonia. 1
As mentioned previously, febrile PE is particularly a profound clinical problem. In our previous study that was investigated, systemic inflammatory parameters in PE and CAP showed that in febrile PE patients there was a slight increase in ESR and blood leukocyte count. In that study, we concluded that the presence of slight leukocytosis and increased ESR may not securely differentiate PE patients with fever from patients with CAP. 9 In accordance with this, in this study the initial levels of blood leukocyte count, CRP, and ESR were not significantly different in PE patients with fever than patients with CAP. In addition, CRP, ESR, and blood leukocyte counts are significantly reduced by anticoagulant and antimicrobial therapy in PE patients with or without fever and in CAP patients. Consequently, CRP, ESR, and blood leukocyte count may not be a good marker for differentiating PE patients with fever from CAP patients.
Thrombosis has been shown to cause an inflammatory response in the vein wall, which involves inflammatory cytokines and neutrophil activations. 15 –17 As a result, multiple inflammatory cytokines such as IL-6, IL-8, and TNF-α are activated by procoagulant thrombin. 15,18 Taheri et al have found significantly increased concentration of plasma-soluble TNF receptor in patients with DVT as opposed to normal individuals. 19 IL-6 was known as a principal mediator of the acute-phase response and initiates expression in inflammation. 20 Roumen-Klappe et al have demonstrated an increased level of IL-6, CRP, and IL-8 at the initial diagnoses and a decreasing level in IL-6 and CRP by anticoagulant therapy in patients with DVT. 21 Despite these observations, none of the markers have been shown as strongly related to VTE. 22,23
Cytokine release such as TNF-α, IL-8, IL-12, and IL-6 are triggered by bacterial products. 24 There has been evidence that treatment failure and mortality due to pneumonia are related to the initial higher amount of cytokine levels. 25
To the best of our knowledge, there has been no comparative data on cytokine profile between PE and pneumonia. Hence, there is no data focus on the PE patients with fever. In the current study, despite there is no difference in the baseline serum TNF-α levels in any PE and the pneumonia groups, serum IL-6 levels were significantly higher in pneumonia group when compared with the group of PE without fever. Interestingly, IL-6 level was comparable between the group of pneumonia and the group of PE with fever. As a result, neither IL-6 nor TNF-α could be a differential marker for VTE and pneumonia. However, both TNF-α and IL-6 levels significantly decreased with anticoagulant and antimicrobial therapy probably due to reducing inflammation triggered by appropriate treatment.
Procalcitonin is a prohormone of the calcitonin that is produced by the C cells of the thyroid gland. It is secreted as a part of systemic inflammatory response to infection. 2 For the diagnosis of infections, the diagnostic accuracy of PCT and its optimum cutoffs are completely dependent on the use of a sensitive assay in a predefined clinical setting. In our study, a more sensitive, second-generation assay for PCT, time-resolved-amplified cryptate emission (Kryptor assay, BRAHMS) was used, which can detect mild elevations. Serum PCT level does not exceed 0.5 ng/mL in healthy participants and >0.5 ng/mL is a suggestive level to show the presence of bacterial infection. 11
In ProCAP study, it is shown that PCT is a good prognostic marker in patients with CAP. 26 They enrolled 300 patients to the study. Primary goal of their study was not to reduce antibiotic use on admission but to aid in the differential diagnosis of new or progressing infiltrates (eg, PE, malignancy, cryptogenic organizing pneumonia, viral pneumonia, and congestive heart failure) by measuring serum PCT levels. In this study, the authors concluded that in patients with diagnostic ambiguities, if the PCT level is between 0.25 and 0.5 ng/mL, physicians should keep in mind CAP in the differential diagnosis.
Another important randomized intervention, namely ProRESP study, showed that antibiotic therapy with PCT guidance reduced antimicrobial therapy almost 50% compared to antibiotic therapy with standard method guidance in patients with acute bronchitis and acute exacerbations of chronic obstructive pulmonary disease (COPD). 27
To the best of our knowledge, there is only 1 study that has evaluated the diagnostic value of PCT in febrile PE. In that study, Delevaux et al found that patients with PE did not show high level of PCT in PE group even in febrile patients. 4 In the current study, using the cutoff value of 0.5 ng/mL, none of the afebrile patients with PE showed positive results for PCT. In the febrile patients with PE, the mean level of PCT was 0.48 ng/mL. In this group, only 2 patients had higher levels of PCT (2.30 and 0.76 ng/mL). Both of those patients had pulmonary infarction demonstrated with computed tomography. We thought that, these elevations might be secondary to inflammation on infarction side due to inflammatory cell infiltration. The other hypothesis is that there might be a subsided secondary bacterial infection on infarction side. None of the patients, who had the criteria of infarction, received antibiotic therapy.
Our study also has a superiority to include a follow-up period to investigate the treatment response. Accordingly, PE patients did not show any significant changes in PCT level by anticoagulant therapy. However, the mean PCT value decreased from 2.24 to 0.92 ng/mL in the third day of antibiotic therapy in CAP group (P = .000).
In the previous studies, the diagnostic value of PCT has been compared with other inflammatory markers in CAP. In many studies, PCT was identified as a better discriminating marker than CRP to characterize the level of inflammation caused by infection. 11 Moreover, serum CRP but not PCT is affected by immunosuppressive treatment (ie, steroids). 28 Another superiority of PCT to CRP is that it rapidly increases in infectious conditions (within 6 hours) and has a better negative predictive value. 29,30 In accordance with this, in our study, CRP did not statistically differ in admission in febrile patients with PE and CAP. Hence, CRP has decreased in all groups with the treatment. Therefore, PCT but not CRP might have a value to differentiate febrile PE from CAP.
Small sample size is the main limitation of the study. This limitation might be the consequence of a careful selection of study population. Many patients were excluded from the study due to comorbidites that may affect the level of PCT such as lung cancer, acute trauma, and extensive surgery. Moreover, a considerable number of febrile patients with PE received empiric antibiotics before recruitment for the study.
In conclusion, measurement of serum PCT might be a reliable early marker for the differential diagnosis of PE and CAP. This finding is of particular importance to distinguish febrile PE from CAP, which is a current dilemma in the clinical practice. These findings need to be established in a larger number of febrile and afebrile patients with PE.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
