Abstract
Background:
Intestinal barrier dysfunction would lead to a rigorous inflammatory reaction due to the translocation of intestinal lumen-derived bacteria and endotoxins. The aim of the present study was to investigate whether intestinal barrier dysfunction occurs in patients with acute Stanford type A aortic dissection (ATAAD) and to determine its potential relationship with the plasma levels of several inflammatory biomarkers in the progression of ATAAD.
Design and Methods:
Serum samples from a total of 46 patients with ATAAD and 36 healthy volunteers were prospectively collected and analyzed. The serum levels of diamine oxidase (DAO), lactate dehydrogenase (LDH), interleukin 6 (IL-6), tumor necrosis factor α (TNF-α), and C-reactive protein (CRP) were measured using colorimetric assay, enzyme-linked immunosorbent assay, and immunoturbidimetric assay.
Results:
Serum levels of DAO, LDH, IL-6, TNF-α, and CRP in patients with ATAAD were significantly higher than those in healthy participants. A significantly positive correlation between DAO activity and IL-6 (r = .56, P < .001), TNF-α (r = .63, P < .001), and CRP (r = .53, P < .001) was observed. Moreover, the activity of DAO correlated negatively with the Pa
Conclusions:
Intestinal barrier dysfunction, reflected by an increased level of serum DAO, may play an important role in the development of systemic inflammatory responses in patients with ATAAD. Therefore, strategies of preserving a normal intestinal barrier function may open new horizons in the treatment of inflammation-related adverse events in the setting of ATAAD.
Keywords
Background
Acute aortic dissection is a life-threatening emergency associated with severe mortality and morbidity. 1 –3 Untreated mortality in patients with acute Stanford type A aortic dissection (ATAAD) has been reported to be up to 1% to 2% per hour after an acute event, and the death rate reaches as high as 35% to 68% during the first 24 to 48 hours. 4 Recent clinical and basic research have revealed a significant involvement of inflammatory mechanism in the pathogenesis of aortic dissection and have also demonstrated a close relationship between the increased plasma level of inflammatory markers and the poor prognosis of such patients. 5 –8 Although there is plenty of evidence that inflammation actively participates in the formation of this acute aortic syndrome, 7,9,10 gaps remain in our knowledge of the potential mechanisms of systemic inflammatory responses (SIRS) since the onset of dissection.
Intestinal epithelial barrier functions allow the passage of nutrients/electrolytes and, concurrently, control the passage of luminal bacterium and its pathogenic components such as endotoxin. 11 Dysfunction of the intestinal barrier is believed to result in an excessive translocation of the gram-negative bacteria into the bowel wall and microcirculation, and subsequent development of SIRS, leading to adult respiratory distress syndrome, multiple organ failure, and death. 12 Diamine oxidase (DAO) exists mostly in intestinal mucosa, an elevated blood DAO level reflects gut mucosal damage at an early stage and has been proven to be an excellent marker in the disruption of intestinal defense function. 13 –15 Our previous study has demonstrated increased gut permeability and endotoxemia in a canine model of acute aortic dissection 8 ; however, it remains unknown whether intestinal barrier dysfunction plays a role in the development of inflammatory responses in patients with ATAAD. The present study was designed to detect the changes in serum DAO levels in patients with ATAAD and to explore its potential association with SIRS in the setting of ATAAD.
Methods
Study Population
The present study was a prospective, single-center, observational study. From August 2013 to January 2014, 128 patients with ATAAD were admitted to our institution. Patients with renal dysfunction and history of autoimmune, systemic inflammatory diseases, gastrointestinal diseases, hepatic tumor, lung, or renal disease were excluded. Of 128 patients screened for participation, 23 patients refused participation in the study and 59 patients met at least 1 exclusion criteria, and the remaining 46 patients enrolled in this study (ATAAD group). Meanwhile, 36 healthy volunteers selected among those attending our outpatient department were enrolled as controls.
Stanford type A aortic dissection was diagnosed in all patients on the basis of typical clinical symptoms, echocardiography, and computed tomography. The acute phase of dissection is defined as the time from symptom onset to admission within 14 days. 1 The study was approved by the institutional review board of West China Hospital (reference number: 2012150), and all patients provided written informed consent.
Assessment of Serum DAO and Inflammatory Biomarkers
Venous blood was drawn from all patients in the fasting state. Serum was obtained after centrifuging blood at 1000g for 15 minutes at 4°C and then stored at −80°C for further analysis. The DAO activity was determined by spectrophotography (Sigma-Aldrich, Louis, Missouri). Serum interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α) levels were measured and calculated using enzyme-linked immunosorbent assay technique (R&D Systems, Minneapolis, Minnesota) according to the manufacturer’s instructions. The level of C-reactive protein (CRP) was determined by immunoturbidimetric assay (Beckman Assay 360; Beckman, Brea, California).
Statistical Analysis
Data are presented as numbers, percentages, and means ± standard deviations. Continuous variables were compared using t test for normally distributed data. The chi-square test was used to evaluate differences between qualitative variables. Correlations between DAO activity, inflammatory mediators, and Pa
Results
Demographics of Study Population
A total of 82 individuals were included, and baseline characteristics of the study patients are summarized in Table 1. The patients’ distribution according to the time of admission since the symptoms onset was demonstrated in Figure 1. Apart from the Pa
Clinical Baseline of the ATAAD and Healthy Groups.

Abbreviations: TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; LDH, lactate dehydrogenase; ATAAD, acute Stanford type A aortic dissection; Fi
a P < .05.

The distribution patients admitted at different time courses of acute Stanford type A aortic dissection (ATAAD).
Serum Levels of DAO and Inflammatory Markers
The serum level of DAO was significantly elevated in patients with ATAAD compared to that in healthy controls (7.77 ± 4.49 vs 4.76 ± 2.20 U/L, P < .05; Figure 2A). The concentration of IL-6 was significantly higher in patients with ATAAD than those in the control group (69.26 ± 28.54 vs 7.05 ± 1.44 pg/mL, P < .05; Figure 2B). Similarly, a significantly increased plasma level of TNF-α (57.15 ± 24.31 vs 4.15 ± 2.21 pg/mL, P < .05) and CRP (65.31 ± 26.59 vs 4.07 ± 0.75 pg/mL, P < .05) was detected in patients with ATAAD (Figure 2C and D).
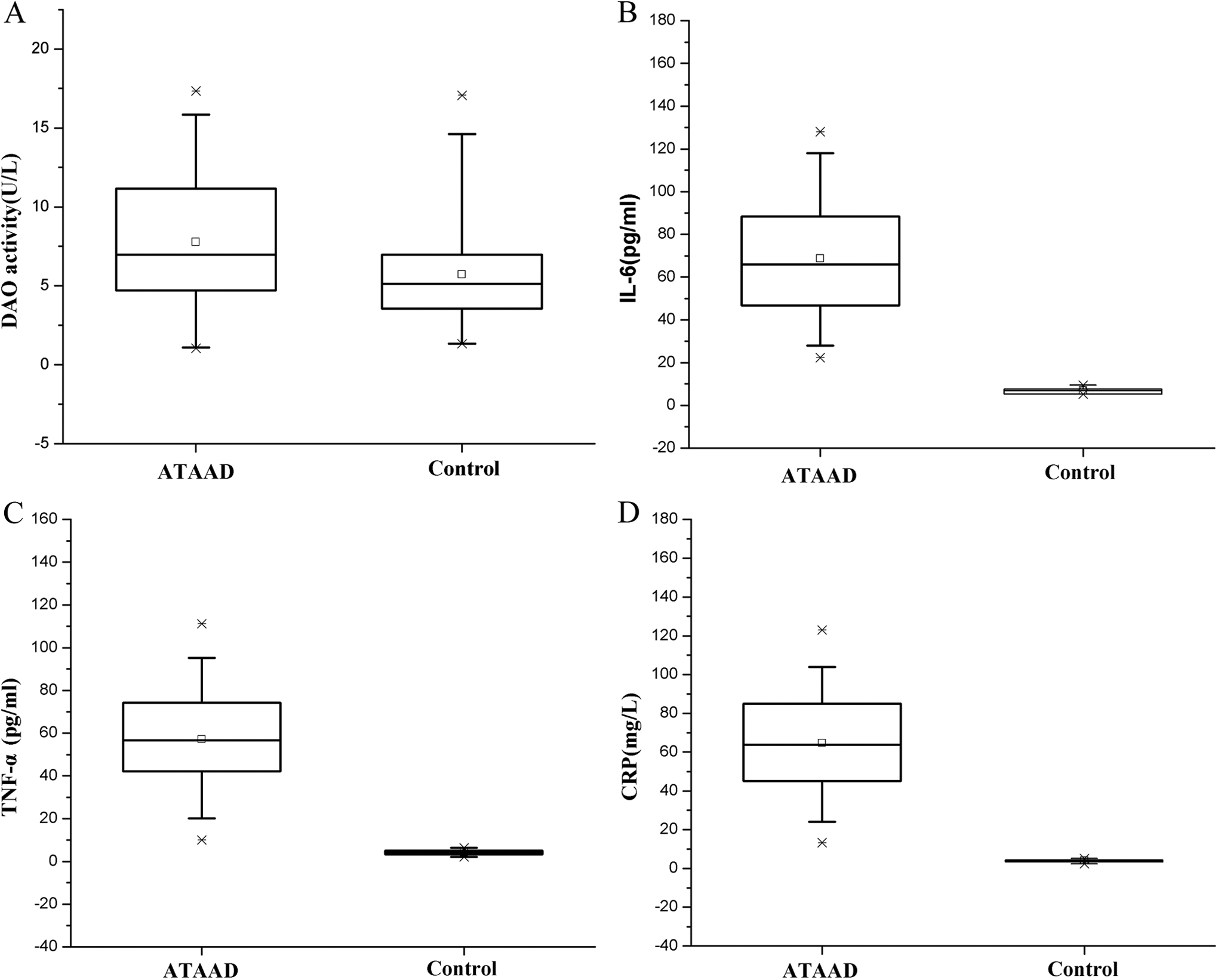
The serum levels of inflammatory cytokines and DAO at admission. The DAO activity was significantly higher in the ATAAD group than the control group (A), while the serum levels of interleukin 6 (IL-6), tumor necrosis factor α (TNF-α), and C-reactive protein (CRP) markedly elevated in patients with ATAAD compared to those in healthy controls (B-D). ATAAD indicates acute Stanford type A aortic dissection; DAO, diamine oxidase.
The Level of DAO in Relation to Inflammatory Markers and Pao
2–Fio
2 Ratio
In patients with ATAAD, a significant positive correlation was found between serum level of DAO and several inflammatory biomarkers, including IL-6 (r = .56, P < .001), TNF-α (r = .63, P < .001), and CRP (r = .53, P < .001; Figure 3A-C). However, the relationship between the serum level of DAO and the value of Pa

Correlations between the serum levels of DAO and inflammatory cytokines in patients with acute Stanford type A aortic dissection (ATAAD). Significant positive correlations were observed between the serum level of DAO and interleukin 6 (IL-6; r = .56, P < .001; A), tumor necrosis factor α (TNF-α; r = .63, P < .001; B), C-reactive protein (CRP; r = .53, P < .001; C), and the value of Pa
Discussion
The present study demonstrated a significant elevation in serum DAO level in patients with ATAAD. Such an increase in expression of DAO and its associated intestinal barrier dysfunction strongly correlated with the serum levels of several inflammatory biomarkers, including IL-6, TNF-α, and CRP in acute aortic dissection. We further revealed a close relationship between the DAO activity and the value of Pa
Interleukin 6, TNF-α, and CRP are common proinflammatory cytokines, and an overexpression of these inflammatory biomarkers in aortic dissection has been repeatedly described. 5 –8 In particular, CRP is an acute-phase protein that is nonspecifically elevated in several conditions (including aortic dissection) in response to inflammation. More importantly, the serum level of CRP may reflect the degree of inflammatory response in the dissected wall, and elevated CRP level is associated with poor prognosis in patients with ATAAD. 6,16 Our results showed that the serum levels of IL-6, TNF-α, and CRP markedly increased in patients with ATAAD, clearly indicating the involvement of inflammatory processes in the progression of ATAAD. However, gaps remain in our knowledge of the etiopathogenesis of inflammatory responses in aortic dissection.
Conventional wisdom that exposure of the media to blood elements initiates the inflammatory cascades and generates the SIRS in aortic dissection can partly fill the gap in our knowledge. However, the plasma levels of inflammatory marker were inconsistently well correlated with the duration of fever and the dynamic changes in size of the false lumen, 17 suggesting other mechanisms might contribute to the development of inflammatory responses after the onset of ATAAD. Our data showed a significant elevation in LDH and DAO serum levels in patients with ATAAD. Such an increase in LDH and DAO is generally considered to be associated with intestinal epithelial and mucosal injury. 13,14,18 However, the specificity of total LDH activity is low, and it has been demonstrated to be a marker of tissue damage in a wide spectrum of conditions. 18 Only the accuracy of isoenzyme LDH-2 at optimal cutoff value seems to be better than total LDH activity in detecting intestinal injuries. 18 Conversely, about 95% of DAO exists in the intestinal villi of human, 19 and only a small percentage of DAO originates from placenta, thymus, and kidneys. 20 Since no patient with pregnancy, thyroid disorders, and signs of renal dysfunction was recruited in the present study, we have reasons to believe that the serum level of DAO is a reliable parameter that reflects the degree of intestinal barrier impairment in this cohort. Therefore, in the present study, the close relationship between the levels of DAO and inflammatory biomarkers clearly indicates the involvement of intestinal barrier dysfunction in the pathogenesis of ATAAD-induced SIRS.
Previous study by our group had observed a significant intestinal mucosa epithelial damage and had also revealed its association with endotoxemia and subsequent inflammatory responses in a canine ATAAD model.
8
In this study, we further confirmed the relationship between the intestinal barrier dysfunction and the ATAAD-related inflammatory responses in human patients. More importantly, a significant correlation between the serum level of DAO and the value of Pa
Limitation
There are several limitations in the present study. First, because of the absence of serial blood samples from the same patient spanning the period of dissection progression, it is difficult for us to fully elucidate the dynamic changes in intestinal barrier function and its mechanistic role in the development of inflammatory responses in the setting of ATAAD. In addition, although elevated serum level of DAO was found to be an excellent biomarker of the increased gut epithelial permeability, the evidence of the disruption of intestinal mucosa integrity (histological examinations) and resultant toxic spread of luminal bacteria to systemic body sites was insufficient. However, to our best knowledge, this is the first study focusing on the intestinal barrier dysfunction in patients with ATAAD and may give new insight into future studies that aim to optimize therapeutic strategies for attenuation of ATAAD-related SIRS. Finally, this study is based on in vitro analysis of human blood samples with a relatively small patient population, and thus we cannot exclude the possibility that other confounders such as interactions between history of medications (eg, statins and β-blockers) and serum levels of biomarkers may have influenced our results.
Conclusion
In summary, our results highlight an important role for intestinal barrier dysfunction in the development of SIRS and associated end-organ injury in patients with ATAAD. Future therapeutic strategies for ATAAD-related adverse events might benefit from the protection of a normal intestinal barrier function.
Footnotes
Authors’ Contribution
G. Jun contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, and gave final approval; H. Jia contributed to design, contributed to interpretation, drafted manuscript, and critically revised manuscript; Q. Hong contributed to conception and design and critically revised manuscript; S. Yingkang contributed to design, contributed to analysis, and critically revised manuscript; Z. Eryong contributed to design and acquisition and analysis; G. Yingqiang contributed to design and acquisition; X. Zhenghua contributed to acquisition and analysis; F. Zhi contributed to acquisition and analysis; Z. Minghua contributed to acquisition and interpretation; Z. Hongwei contributed to acquisition, analysis, and interpretation; M. Wei contributed to design, contributed to analysis, drafted manuscript, critically revised manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Written informed consent was obtained from the patients for publication of this study. Copies of the written consent are available for review by the Editor-in-Chief of this journal. Jun Gu and Jia Hu equally contributed to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grant Nos. 81300155, 81370413, 81170288, and 81470481 from the National Research Foundation of Nature Science, China.
