Abstract
Objectives:
A high loading dose of atorvastatin has been confirmed to reduce postprocedural events in patients undergoing percutaneous coronary intervention (PCI). In this study, we sought to investigate the protective effects of rosuvastatin in patients with acute coronary syndromes (ACS) undergoing PCI and to determine the effect of rosuvastatin pretreatment on the postprocedural levels of high-sensitivity C-reactive protein (hs-CRP), interleukin 6 (IL-6), and monocyte chemotactic protein 1 (MCP-1).
Methods:
A total of 125 patients with non-ST-segment elevation ACS were randomized to pretreatment with rosuvastatin (20 mg 2-4 hours before PCI [n = 62]) or placebo (n = 63). All the patients received subsequent long-term rosuvastatin treatment (10 mg/d). The main end point of the trial was the 30-day incidence of major adverse cardiac events (death, myocardial infarction, or unplanned revascularization). Plasma levels of hs-CRP, IL-6, and MCP-1 were detected before PCI and 6 hours, 24 hours, and 3 days after PCI.
Results:
The primary end point occurred in 8.1% of the patients in the rosuvastatin arm and 22.2% in the placebo arm (P < .01); this difference was entirely attributed to a reduced incidence of myocardial infarction (8.1% vs 22.2%; P < .01). The postprocedural elevation in creatine kinase-MB and troponin I was also significantly lower in the rosuvastatin group at 6 hours, 24 hours, and 3 days. Plasma levels of hs-CRP, IL-6, and MCP-1 increased significantly after PCI in both the rosuvastatin and control groups; however, the postprocedural elevations in hs-CRP and IL-6 levels were significantly lower in the rosuvastatin group than the control group.
Conclusions:
A single, high dose (20 mg) of rosuvastatin prior to PCI reduces postprocedural myocardial injury in patients with ACS, with a concomitant attenuation of the postprocedural increase in hs-CRP and IL-6 levels.
Keywords
Introduction
Statins reduce low-density lipoprotein cholesterol (LDL-C) by blocking 3-hydroxy-3-methylglutaryl coenzyme A reductase in the liver, which suppresses cholesterol biosynthesis, resulting in a dramatic reduction in circulating LDL-C. Multiple landmark clinical trials have demonstrated the beneficial effects of statin therapy for the primary and secondary prevention of cardiovascular disease.1,2 These benefits can be explained not only by the lipid-lowering potential of this therapy but also by nonlipid-related mechanisms, called pleiotropic effects, including inhibition of inflammation, modulation of endothelial function, stabilization of atherosclerotic plaques, and attenuation of thrombosis. 3 Observational studies have suggested that pretreatment with statins may reduce the incidence of myocardial infarction after coronary intervention and prevent myocardial injury.4,5 Even more recently, additional randomized controlled trials have confirmed that a high loading dose of a statin before elective percutaneous coronary intervention (PCI) reduces postprocedural myocardial injury,6–8 but most of these studies were conducted using atorvastatin.
To date, only 2 studies have used rosuvastatin. The researchers found that a single high dose of rosuvastatin prior to PCI reduced postprocedural myocardial injury and improved 12-month clinical outcomes in patients with acute coronary syndrome (ACS)9,10 or stable angina. 11 However, the potential mechanisms of action of rosuvastatin are still not clear. Being a more effective statin when compared to atorvastatin,12,13 rosuvastatin has become the subject of increasing interest in recent years. Here, we designed a prospective, randomized, double-blind, placebo-controlled study to investigate whether pretreatment with rosuvastatin can reduce procedural myocardial damage in patients with ACS undergoing elective PCI and to determine whether variations in the postprocedural levels of high-sensitivity C-reactive protein (hs-CRP), interleukin 6 (IL-6), and monocyte chemotactic protein 1 (MCP-1) are influenced by pretreatment with rosuvastatin.
Methods
Study Population and Design
This study was a randomized, prospective, double-blind, placebo-controlled trial. From March 2011 to June 2012, a total of 215 consecutive patients who had non-ST-segment elevation ACS and received diagnostic coronary angiography were recruited. We excluded patients with a history of previous statin therapy, with current cardiogenic shock, in whom medical treatment was recommended, in whom coronary artery bypass grafting (CABG) was recommended, and with severe hepatic (history of liver cirrhosis or alanine aminotransferase levels >2.5 times the upper limit of normal) or renal (serum creatinine >177 µmol/L) disease.
A total of 48 patients were excluded, 34 because of previous or current treatment with statins, 11 because of emergency angiography due to cardiogenic shock or ongoing pain, and 3 because of renal or hepatic disease. Eligible patients were randomly assigned to receive placebo pretreatment (control group) or rosuvastatin (20 mg) as a loading treatment (rosuvastatin group) before PCI. Physicians performing the procedure and the follow-up assessments were blinded to the randomization assignment. Randomization was performed in a 1:1 ratio. After coronary angiography, 42 patients who did not receive PCI were excluded from the study; thus, 125 patients with significant coronary artery disease who underwent PCI were enrolled.
All of the patients gave informed consent according to a protocol approved by the ethical committee of Qingdao Municipal Hospital. A flowchart of this study is provided in Figure 1.

Flowchart of the study. NSTE-ACS indicates non-ST-segment elevation acute coronary syndrome; PCI, percutaneous coronary intervention; CABG, coronary artery bypass grafting; CAG, coronary angiography.
Percutaneous Coronary Angioplasty
All of the participants underwent standard PCI via radial or femoral artery puncture. All of the participants were given oral acetylsalicylic acid (100 mg/d) and clopidogrel (75 mg/d) daily, with a total dose of at least 300 mg before PCI. In some cases, glycoprotein IIb/IIIa receptor antagonists were administered during PCI as deemed necessary by the operators. The PCI operation was considered to be successful if the post-PCI residual stenosis was less than 30%.
Postprocedural Management
After PCI, the patients were treated with low-molecular-weight heparin for 3 to 5 days, clopidogrel (75 mg/d) for at least 12 months, rosuvastatin (10 mg every night) for at least 1 month, and acetylsalicylic acid (100 mg/d) as long as possible. In addition, patients without contraindications were given beta-receptor antagonists and angiotensin-converting enzyme inhibitors or receptor blockers.
Blood Sample Collection and Laboratory Examination
Venous blood samples were collected from each patient at 4 time points (before PCI and 6 hours, 24 hours, and 3 days after PCI). The creatine kinase MB (CK-MB) level was assayed using a fluorescence immunoassay, cardiac troponin I (cTnI) was tested using chemiluminescence, hs-CRP was evaluated using immunoturbidimetry, and IL-6 and MCP-1 levels were assessed using an enzyme-linked immunosorbent assay.
Primary and Secondary End Points
The primary end point events included major adverse cardiac events (MACEs) that occurred within 30 days after PCI, including cardiac death, myocardial infarction, and revascularization treatment. Myocardial infarction in patients with normal pre-PCI cardiac marker levels was defined by post-PCI levels of CK-MB or cTnI reaching 300% of the upper limit of normal (ULN); in patients whose pre-PCI levels were above the normal ranges, myocardial infarction was defined by a post-PCI level of CK-MB or cTnI reaching 300% of the baseline level before PCI. 14 Revascularization treatment included secondary PCI or CABG of the target vessel. Secondary end point events included an elevation in cardiac markers above the ULN within 30 days after PCI.
Statistical Analyses
Continuous data are expressed as the mean ± standard deviation. Intergroup analysis was conducted using an independent t test. To compare the change in cardiac enzymes and inflammatory factors (hs-CRP, IL-6, and MCP-1) before and after PCI, we used analysis of variance. The categorical data are expressed as n (%). Proportions were compared using Fisher exact test when the expected frequency was <5; otherwise, the Yates-corrected chi-square test was used. P < .05 was considered to indicate statistical significance. All of the analyses were performed using SPSS version 17.0 (SPSS Inc, Chicago, Illinois).
Results
Clinical Characteristics
The clinical features of the 2 groups are reported in Table 1. The 2 groups had similar baseline characteristics, including age, gender, body mass index, tobacco use, left ventricular ejection fraction, lipid levels, creatinine levels, clinical presentation, and in-hospital medication administration (all P > .05).
Baseline Clinical Characteristics and Laboratory Results.

Abbreviations: HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; NSTEMI, non-ST-segment elevation myocardial infarction; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker.
Angiographic and Procedural Characteristics
Coronary anatomy, procedure characteristics, the use of drug-eluting stents, implanted stent diameter and length, and periprocedural infusion of glycoprotein IIb/IIIa inhibitors were comparable between the 2 groups (Table 2). Angiographic complications during the procedure occurred in 8 (13.0%) patients in the rosuvastatin group and 9 (14.3%) patients in the control group (P = .82; Table 3).
Procedural Features in the Rosuvastatin and Control Groups.

Procedural Complications.
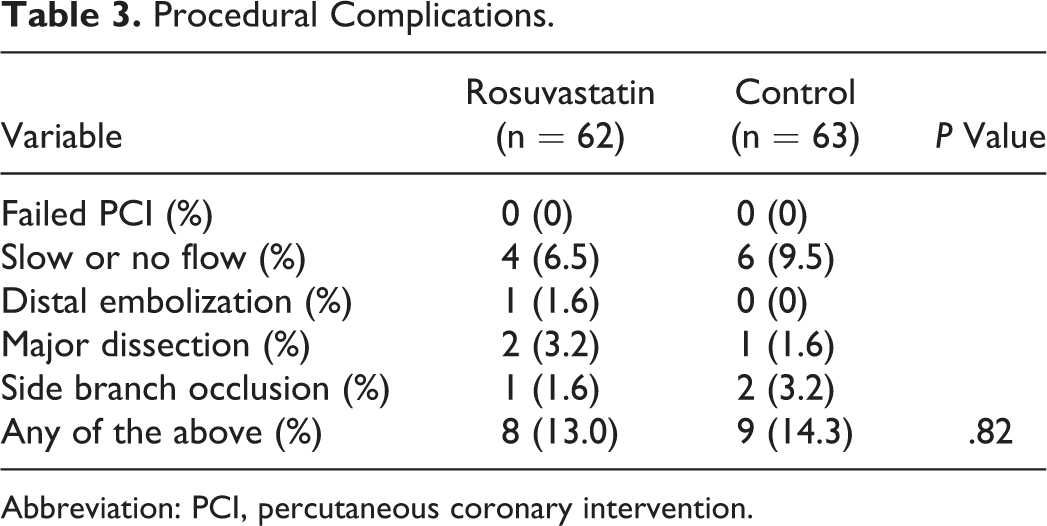
Abbreviation: PCI, percutaneous coronary intervention.
Primary End Point
The primary end point was evaluated at 30 days (Table 4). The composite primary end point of death, myocardial infarction, and target vessel revascularization occurred in 8.1% (5 of 62) of the patients in the rosuvastatin arm and 22.2% (14 of 63) in the placebo arm (P < .01). The incidence of MACE at 1 month was completely influenced by postprocedural myocardial infarction (8.1% vs 22.2%, P < .01).
The Incidence of Primary End Points at 1 Month in the 2 Groups.

Abbreviation: MACEs, major adverse cardiac events.
aThe rate compared with the rosuvastatin group was significantly different (P < .01).
Secondary End Points
The prevalence of patients with a preprocedural elevation in cardiac markers above the ULN was similar in the rosuvastatin and placebo groups. After PCI, the proportion of patients with an elevated CK-MB or cTnI level was significantly lower in the rosuvastatin arm (Table 5).
The Proportion of Patients With Cardiac Markers Elevated Above the ULN.
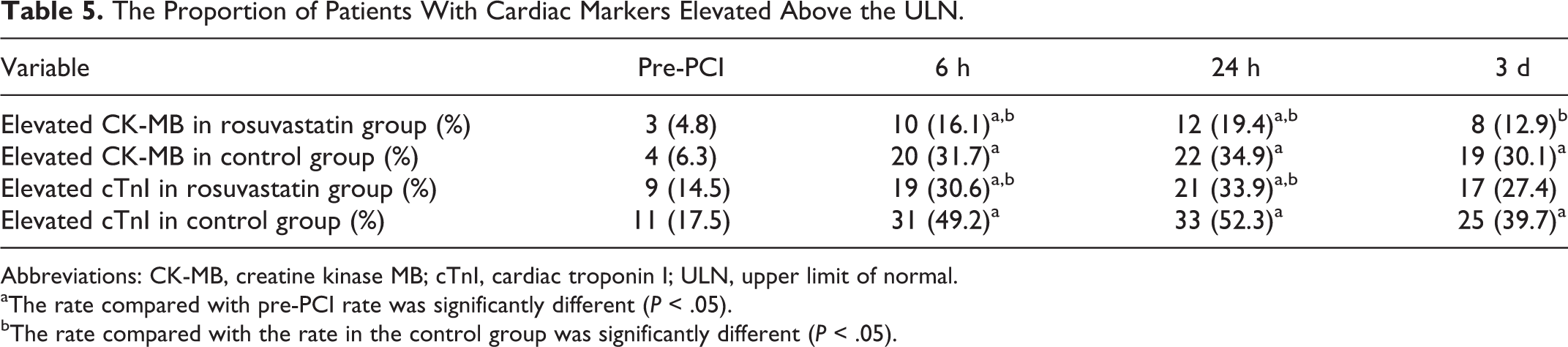
Abbreviations: CK-MB, creatine kinase MB; cTnI, cardiac troponin I; ULN, upper limit of normal.
aThe rate compared with pre-PCI rate was significantly different (P < .05).
bThe rate compared with the rate in the control group was significantly different (P < .05).
Changes in hs-CRP, IL-6, and MCP-I Levels in the 2 Groups
At baseline, there was no significant difference in hs-CRP, IL-6, and MCP-I levels between the rosuvastatin and the control groups. After PCI, hs-CRP markedly increased in both the groups at 6 hours, 24 hours, and 3 days, but less elevation was found in the rosuvastatin group compared to the control group (Figure 2). The same trend was also shown in IL-6 levels (Figure 3). Significant changes were also found in MCP-1 levels after PCI in the rosuvastatin and control groups, but there was no significant difference between the levels in these 2 groups after PCI (Figure 4).

Changes in hs-CRP levels in the 2 groups. *P < .01 versus pre-PCI; #P < .01 versus the rosuvastatin group. hs-CRP indicates high-sensitivity C-reactive protein.

Changes in IL-6 levels in the 2 groups. *P < .01 versus pre-PCI; #P < .01 versus the rosuvastatin group. IL-6 indicates interleukin 6.

Changes in MCP-1 levels in the 2 groups. *P < .01 versus pre-PCI. No significant difference was detected between the levels in the rosuvastatin and control groups at 6 hours, 24 hours, and 3 days after PCI. MCP-1, indicates monocyte chemotactic protein; PCI, percutaneous coronary intervention.
Discussion
Our study shows that a single, high dose of rosuvastatin prior to PCI reduces postprocedural myocardial injury in patients with ACS, with a concomitant attenuation of the postprocedural increase in hs-CRP and IL-6 levels, suggesting that the anti-inflammatory effect may contribute to the procedural myocardial protection provided by rosuvastatin.
Percutaneous coronary intervention is the most prevalent revascularization strategy in patients with coronary artery disease. Although this procedure is safe, postprocedural myocardial infarction, as assessed by cardiac marker elevation, occurs in 5% to 40% of patients. 15 Large postprocedural ischemic events are associated with adverse outcomes; however, even small increases in cardiac enzymes have been associated with increased long-term mortality. 16 The reduction in ischemic events and enhancement of the safety of PCI are the primary goals of catheterization laboratories.
Statins inhibit hepatic 3-hydroxy-3-methylglutaryl coenzyme A reductase, with a subsequent suppression of cholesterol biosynthesis. The advent of these drugs has greatly impacted the treatment of cardiovascular disease, with well-documented benefits in primary and secondary disease prevention. 17 Their benefit has also been largely demonstrated in the setting of PCI by the prevention of postprocedural myocardial infarction. For example, in 2004, the Atorvastatin for Reduction of Myocardial Damage during Angioplasty (ARMYDA) study demonstrated a beneficial effect of statins in preventing myocardial damage after coronary angioplasty in patients undergoing PCI for stable angina. 6 Later, the ARMYDA-ACS trial confirmed the same benefit of short-term pretreatment with atorvastatin in patients with ACS undergoing an early invasive treatment. 7 Immediate high-dose statin loading before primary PCI in ST-segment elevation myocardial infarction (MI) also showed a benefit in terms of myocardial perfusion. 18 Considering the extensive use of statins in primary and secondary disease prevention, the ARMYDA RECAPTURE study was conducted and demonstrated that reloading with high-dose atorvastatin improves the clinical outcome in patients receiving chronic statin therapy and undergoing PCI. 8 In addition to the ARMYDA series of studies, numerous other trials have also confirmed the beneficial effects of statins in patients undergoing PCI, including the Naples IL 19 and PCI-PROVE IT 20 trials.
All of the above studies were conducted using atorvastatin and demonstrated the preventive benefits of atorvastatin on postprocedural myocardial infarction in the setting of PCI. But none of them has explored the plasma level of IL-6 and MCP-1 before and after statin loading. And to date, only 2 studies used rosuvastatin, which were performed by Korean and Italy researchers. They found that a single high dose usage of rosuvastatin prior to PCI could reduce postprocedural myocardial injury and improve 12-month clinical outcomes in patients with ACS9,10 or stable angina. 11 In these 2 trials, the loading doses of rosuvastatin were 40 mg and none of them has studied the changes in inflammatory factors before and after rosuvastatin loading. In our study, we found that only 20 mg loading dose of rosuvastatin prior to PCI could substantially decrease the incidence of MACE at 1 month and decrease the elevation of CK-MB and cTnI levels at 6 hours, 24 hours, and 3 days after PCI, suggesting that even a lower loading dosage (20 mg) of rosuvastatin before PCI could provide protective effect.
The possible mechanisms underlying the early protective effect of statins in reducing the rate of postprocedural MI are unclear. Previous studies have suggested that the benefits may be mostly related to the nonlipid-related mechanisms of statins (so-called “pleiotropic effects”). These effects encompass several mechanisms that modify inflammatory responses, endothelial function, plaque stability, and thrombus formation.14,21 The anti-inflammatory effects of statins are independent of their lipid-lowering actions, but related to the reduced formation of isoprenoids (farnesil pyrophosphate and geranylgeranyl pyrophosphate), 22 which are responsible for the activation of nuclear transcription factors involved in proinflammatory mechanisms. In cultured vascular smooth muscle cells (VSMCs), atorvastatin inhibits nuclear factor κB activation by tumor necrosis factor alpha (TNF-α) or angiotensin II. 23 In rat aortic smooth muscle cells, atorvastatin negates thrombin-mediated increase in proinflammatory cytokine synthesis. 24 In an in vitro model of lipopolysaccharide-induced inflammation, statins significantly decreased VSMC–monocytes interaction and their synergistic production of proinflammatory cytokines such as IL-6. 25 In isolated T cells, lovastatin inhibited cytokine production of IL-2, IL-4, and interferon γ from activated cells. 26 In animal models of allograft atherosclerosis, statins reduce inflammatory cell infiltration in the arterial wall by reducing chemokine expression. 27 Simvastatin reduces the expression of proinflammatory cytokines such as IL-6, IL-8, and MCP-1 in peripheral blood mononuclear cells from patients with hypercholesterolemia, both in vitro and in vivo. 28
The benefits of lipid lowering with statins in primary and secondary prevention of CVD have already been established in large randomized clinical studies. However, some of these landmark clinical trials yielded the early clinical evidence for the beneficial effects of statins independent of their lipid-lowering properties. In the Long-Term Intervention with Pravastatin in Ischaemic Disease trial, cardiovascular risk reduction was significant throughout the continuum of LDL-C levels, even in patients with LDL-C <100 mg/dL. 29 In addition, the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS) trial demonstrated that lovastatin reduced cardiovascular events in patients with low LDL-C and high CRP levels, while it conferred no clinical benefit in patients with both low LDL-C and low CRP levels. 30 And the most convincing evidence for improvement of clinical outcome by statins due to their anti-inflammatory effect was provided by the JUPITER trial. 31 This study demonstrated that even in low- or intermediate-risk patients according to Framingham risk score, without hypercholesterolemia, suppression of background inflammation by rosuvastatin prevents CVD development and improves clinical outcome.
Vascular injury during PCI is associated with a systemically measurable inflammatory response, and the degree of inflammation has been shown to correlate with cardiovascular risk. Rebeiz et al found that systemic concentrations of CRP, IL-6, and MCP-1 increased substantially after PCI. 32 Sanchez-Margalet et al also found an increased serum CRP concentration following an increase in serum IL-6 levels by 12 to 36 hours, reaching a peak value by 24 hours after the procedure, 33 and the increase in serum IL-6 and CRP levels was more obvious in patients with a concomitant postprocedural troponin T elevation. 34 Although some other inflammatory factors, such as IL-1, TNF-α, 35 were also explored, CRP, IL-6, and MCP-1 were the most frequently studied inflammatory factors in the setting of PCI. Thus, we performed this study to evaluate whether a high loading dose of rosuvastatin could influence the serum levels of CRP, IL-6, and MCP-1 after PCI. The IL-6 is one of the most studied cytokines with both proinflammatory and proatherogenic activities. It is the main stimulant for the hepatic production of CRP and other reactant proteins, and it also has other important roles leading to increased endothelial cell adhesiveness by upregulating E-selectin, intercellular cell adhesion molecule 1, and vascular cell adhesion molecule 1 and releasing inflammatory mediators, including IL-6 itself. 36 In this study, we found that loading with rosuvastatin (20 mg) could substantially inhibit the elevation in hs-CRP and IL-6 in ACS after PCI. Anti-inflammatory effect of rosuvastatin has been confirmed to be the main mechanism that improved clinical outcome in JUPITER trial, so the attenuation of CRP and IL-6 in our study was thought to be involved in rosuvastatin-induced postprocedural cardioprotection. As the main stimulant for the hepatic production of CRP, decreased expression of IL-6 after rosuvastatin loading could further serve anti-inflammatory functions by inhibiting the expression and secretion of CRP and other inflammatory mediators.
Conclusion
In conclusion, our study confirms that high-dose rosuvastatin loading therapy before PCI may reduce postprocedural myocardial infarction in patients with ACS, possibly via the inhibition of both IL-6 and CRP. These results support the use of high-dose rosuvastatin loading therapy before PCI in patients with ACS and more efforts should be made to clarify the involved mechanisms.
Footnotes
Authors’ Note
The authors Zhengzhong Wang and Hongyan Dai contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Qingdao science and technology support program [2012-1-3-1-(2)-nsh].
