Abstract
Background
Clopidogrel is a pro-drug that requires metabolic activation by hepatic cytochrome P450 (CYP) enzymes in two steps. Previous studies have shown that the administration of continuous nitrate significantly affects the activity of hepatic CYP. In this pilot study, we assess the impact of intravenous nitrate treatment on the antiplatelet effects of clopidogrel in patients with non-ST-elevation myocardial infarction (NSTEMI).
Method
We included 20 NSTEMI patients: 15 in the nitrate group and five in the control group. All patients received a 300 mg acetylsalicylic acid and a 600 mg clopidogrel loading dose. The nitrate group was administered an intravenous glyceryl trinitrate infusion at 10 µg/min for 48 h. After the drugs were initiated, blood samples were collected from patients at intervals of 30th minutes, 60th minutes, 120th minutes, fourth hour, sixth hour, and 48th hour to assess platelet function. The antiplatelet effect of clopidogrel was evaluated using the platelet reactivity unit (PRU) from the Verify-Now P2Y12 assay.
Result
The PRU values at baseline and the 30th, 60th, 120th minutes, and 48th hour were similar between the two groups (P > .05). However, PRU values at the fourth and sixth hours were significantly higher in the nitrate group than in the control group (274.1 ± 53.9 vs 162.4 ± 39.9; P = .002 and 272.0 ± 67.0 vs 185.6 ± 18.1; P = .03, respectively).
Conclusion
Intravenous nitrate therapy in patients with NSTEMI delayed the onset of the antiplatelet effect of orally loaded clopidogrel by at least 4 h. This point should be kept in mind in patients using clopidogrel and nitrate therapy together. (NCT06878638)
Keywords
Introduction
Dual antiplatelet therapy that combines acetylsalicylic acid (ASA) with an oral P2Y12 inhibitor, such as ticagrelor, prasugrel, or clopidogrel, is the standard treatment for all patients with acute coronary syndrome (ACS), regardless of whether percutaneous coronary intervention (PCI) is performed. 1 Prasugrel and ticagrelor, both third-generation P2Y12 inhibitors, are superior to clopidogrel in preventing major adverse cardiovascular events after ACS due to their rapid onset of action, higher potency, and reduced interindividual variability.2,3 Therefore, guidelines recommend using prasugrel and ticagrelor instead of clopidogrel in patients undergoing PCI for ACS. 1 However, prasugrel and ticagrelor are associated with higher rates of major bleeding, increased costs, and greater discontinuation rates than clopidogrel, so clopidogrel remains the most commonly prescribed P2Y12 inhibitor for PCI and ACS.2-6 Clopidogrel is a prodrug that requires two sequential oxidative steps by several hepatic cytochrome P450 (CYP) enzymes to produce an active metabolite. CYP2C19 plays a crucial role in both oxidation steps and contributes the most among these enzymes.7,8 CYP2C19 activity is influenced by genetic polymorphisms, and about one in three patients have reduced metabolism due to the presence of CYP2C19 loss-of-function alleles.9,10 In addition to the CYP2C19 polymorphism, drug-drug interactions can also lead to reduced responsiveness to clopidogrel.11-15 Omeprazole and some drugs have been reported to reduce the antiplatelet effect of clopidogrel by inhibiting its CYP activation.11-13 Furthermore, using morphine in patients with ACS has been shown to reduce the absorption of oral P2Y12 receptor inhibitors, delay the onset of action, and decrease their antiplatelet efficacy.14,15 Clopidogrel hyporesponsiveness is associated with an increased risk of adverse cardiovascular events.16,17
The latest ESC guideline recommends ASA alone as antiplatelet therapy in patients with non-ST elevation ACS with unknown coronary anatomy and planned early invasive treatment (<24 h) and does not recommend routine pre-treatment with a P2Y12 receptor inhibitor. 1 This approach further enhances the importance of rapid-onset periprocedural P2Y12 therapy in preventing adverse cardiovascular events such as stent thrombosis. Additionally, ACS guidelines also recommend intravenous nitrate therapy for treating angina.1,18 Previous studies have shown that the administration of continuous nitrate significantly affects the activity of hepatic CYP. 19 However, the impact of intravenous nitrates on clopidogrel-induced platelet inhibition remains uncertain. This study aims to investigate whether intravenous nitrate given during non-ST-elevation myocardial infarction (NSTEMI) affects the platelet inhibition of clopidogrel.
Materials and Methods
Study Population
Twenty patients who were admitted to our hospital's emergency department with chest pain and diagnosed with NSTEMI were included in the study. Patients aged 18 to 75 years with positive troponin levels, blood pressure above 120/70 mm Hg, and who consented to participate were evaluated for inclusion. Patients who met any of the following criteria were excluded from the study: Hemodynamically unstable and requiring urgent intervention, ST elevation on electrocardiography, atrial fibrillation, recurrent angina, use of narcotic analgesics (eg, morphine), nitrates and glycoprotein IIb/IIIa inhibitors during hospitalization, history of coronary artery bypass graft (CABG) surgery, active infection, uncontrolled hypertension, and diabetes, cerebrovascular accident, use of nitrate, P2Y12 inhibitor, proton pump inhibitor, oral anticoagulant, statin, CYP enzyme-inducing or inhibiting drugs at the time of admission, active systemic disease (eg, malignancy, thyroid disorders chronic inflammatory diseases, and liver dysfunction), left ventricular systolic dysfunction (ejection fraction <50%), glomerular filtration rate <60 mL/min/1.73 m2, allergy to ASA or clopidogrel, coagulopathy, platelet counts below 100,000/μL and hematocrit levels below 29%, or exceeding 52%. The flowchart of the study is illustrated in Figure 1. The ethics committee of our hospital approved the study protocol, and informed consent was obtained from each patient.

Flow Diagram of the Study.
Study Protocol
All patients included in the study were admitted to the coronary intensive care unit, where vascular access was established in both arms. Routine blood tests were performed for each patient's baseline evaluation before drug administration, including a complete blood count, biochemistry, and troponin levels. All patients received a loading dose of 600 mg clopidogrel and 300 mg ASA. A maintenance treatment of ASA 100 mg/day and clopidogrel 75 mg/day was prescribed. Enoxaparin was started subcutaneously at 1 mg/kg twice daily, and during PCI, an additional 100 IU/kg intravenous bolus of unfractionated heparin was given. The nitrate group, consisting of 15 patients, received an intravenous glyceryl trinitrate infusion at 10 µg/min for 48 h. The solution obtained by adding 10 mg of glyceryl trinitrate to 500 mL of 0.9% sodium chloride was administered to the patients at a constant rate of 30 mL/h. In contrast, the control group (n = 5) received only 0.9% sodium chloride at a rate of 30 mL/h, with no nitrate administered. The study protocol is illustrated in Figure 2. After the drugs were initiated, in addition to routine enzyme monitoring, blood samples were collected from the patients at intervals of 30 min, 60 min, 120 min, 4 h, 6 h, and 48 h to assess platelet function. These blood samples were analyzed within 30 min to 4 h using the VerifyNow P2Y12 assay (Accumetrics, San Diego, CA, USA). All patients who had blood samples collected within the first 6 h underwent coronary angiography 24 h after the patients were included in the study, and PCI was performed in the same session if necessary. The radial approach was preferred for performing coronary procedures. Narcotic analgesics, nitrates, and glycoprotein IIb/IIIa inhibitors were administered at the operator's discretion during the procedures. However, patients who received these medications were excluded from the study.
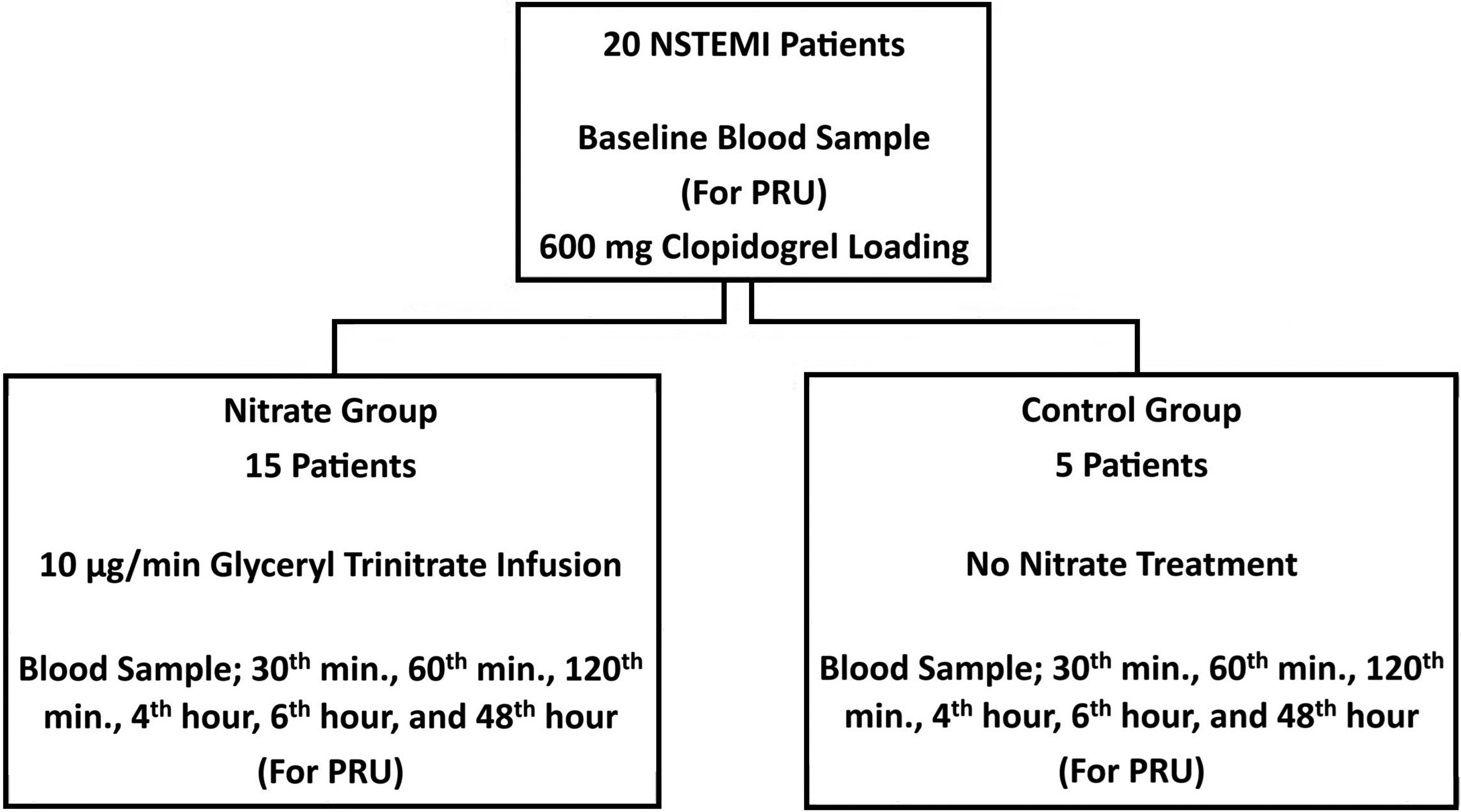
Study Design.
Platelet Function
Blood samples for platelet function analysis were drawn from the antecubital vein using a 21-gauge needle into 2-mL vacutainer tubes containing 3.2% sodium citrate. Blood samples were assessed for clopidogrel antiplatelet activity using the VerifyNow P2Y12 assay within 30 min to 4 h. The VerifyNow test is a point-of-care assay that utilizes whole blood and features a single-use device containing the required biochemical reagents. The VerifyNow P2Y12 assay was developed to evaluate responsiveness to clopidogrel and other P2Y12 antagonists. The VerifyNow P2Y12 assay reports patient results in the platelet reactivity unit (PRU) within 5 min, utilizing a single blood sample. The impact of intravenous nitrate treatment on the antiplatelet effects of clopidogrel was examined by comparing the PRU values of the nitrate and control group patients at baseline and at the 30th minute, 60th minute, 120th minute, fourth hour, sixth hour, and 48th hour following clopidogrel treatment.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, Version 23 (IBM Corporation, Armonk, New York, USA). Continuous variables are presented as means ± standard deviations, whereas categorical variables are presented as percentages. Inter-group comparisons were made using the Mann–Whitney U-test. Categorical variables were analyzed with the chi-square test or Fisher's exact test. All statistical tests were two-sided, and P-values of < .05 were considered significant. According to a prior study, we calculated the number of patients and controls (3:1 ratio) necessary to detect a difference between the nitrate group and the control group for PRU value, with an α of 0.05, a β of 0.20, and a statistical power of 0.80. 20 The nitrate group required 14 patients, while the control group needed four. As a result, the nitrate group consisted of 15 patients, whereas the control group included five.
Results
A total of 33 patients with NSTEMI were screened. The study ruled out the following patients: two with glomerular filtration rates under 60 mL/min, three taking clopidogrel or nitrates, two with left ventricular systolic dysfunction, one on oral anticoagulants, two taking proton pump inhibitors, one receiving pethidine hydrochloride, one administered glycoprotein IIb/IIIa inhibitors during the procedure, and one with hematocrit levels below 29%. The remaining 20 patients were included in the nitrate (n = 15) and control (n = 5) groups.
All patients underwent coronary angiography. In the control group, one patient had a noncritical lesion, one had a critical lesion requiring medical follow-up, two had critical lesions treated with PCI, and one had critical lesions for which CABG was recommended. In the nitrate group, two patients had noncritical lesions, two had critical lesions requiring medical follow-up, seven had critical lesions treated with PCI, and four had critical lesions for which CABG was recommended (P = .94). No complications related to the procedures were observed in any of the patients.
The patients’ demographic characteristics and biochemical parameters were similar in both groups (Table 1). The groups had no differences in the initial blood pressure and heart rate values. Maximum troponin T values were similar in both groups (4.2 ± 5.1 vs 4.7 ± 5.6, P = .86). The PRU values at baseline and the 30th, 60th, and 120th minutes were similar between the two groups (P = .29, .63, .96, and .22, respectively) (Figure 3). However, PRU values at the fourth and sixth hours were significantly higher in the nitrate group than in the control group (274.1 ± 53.9 vs 162.4 ± 39.9; P = .002 and 272.0 ± 67.0 vs 185.6 ± 18.1; P = .03, respectively). At the 48th hour, the PRU values in both groups became similar again (P = .10) (Figure 3).
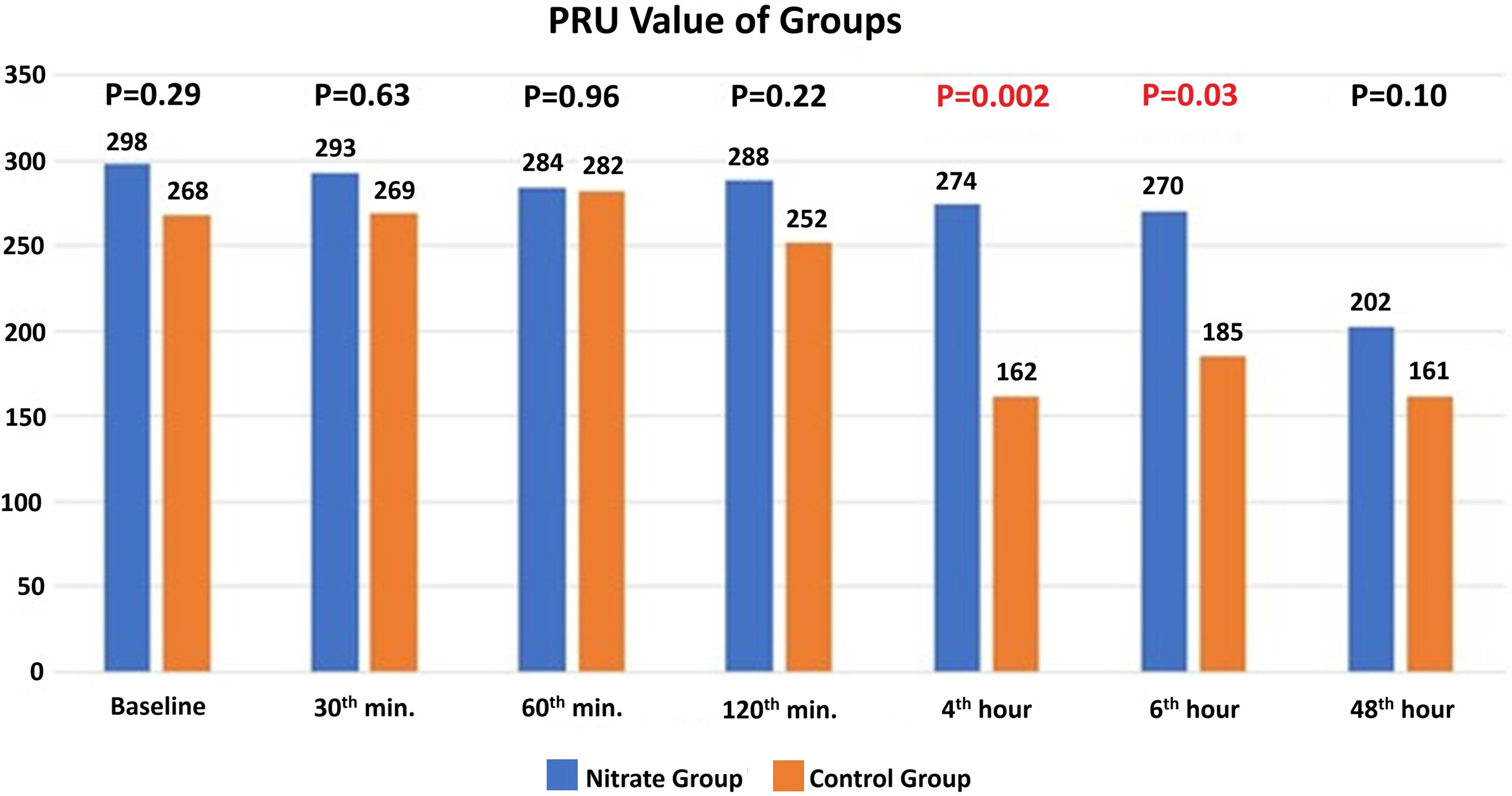
Platelet Reactivity Unit (PRU) Values of the Nitrate and Control Groups.
Baseline Characteristics of Nitrate and Control Groups.

Abbreviation: EF, ejection fraction.
The recently published International Consensus Statement on Platelet Function and Genetic Testing in PCI specified the high platelet reactivity (HPR) cut-off value as 208 PRU for the VerifyNow P2Y12 assay. 21 Four hours after clopidogrel loading, optimal platelet reactivity (PRU <208) was achieved in all patients in the control group, whereas it was attained in only three patients (20%) in the nitrate group. At the 48th hour, 80% of patients in the nitrate group and 100% of the patients in the control group had achieved the optimal platelet reactivity level (P = .53).
Discussion
In this pilot study, we examined the impact of intravenous nitrate on the platelet inhibition caused by clopidogrel. We showed that (i) intravenous nitrate therapy in patients with NSTEMI delayed the onset of the antiplatelet effect of orally loaded clopidogrel by at least 4 h; (ii) 4 h after clopidogrel loading, only 20% of patients achieved optimal platelet reactivity (PRU <208) in the intravenous nitrate treatment patients; and (iii) at the 48th hour, the PRU values in both groups became similar, and the achieved optimal platelet reactivity patient percent was similar.
Nitrates, such as glyceryl trinitrate (also known as nitroglycerin), have been used for many years to treat ACS, acute angina attacks, hypertensive crises, and acute heart failure with normal or elevated blood pressure. 22 They exert various hemodynamic effects, mainly on vasodilating capacitance veins and conductance arteries. Vasodilation occurs when smooth muscle cells relax in response to nitric oxide (NO) and cyclic guanosine monophosphate, which are produced through the bioactivation of nitrates.23,24 Nitrates are prodrugs that require bioactivation through complex mechanisms and are transformed into vasoactive molecules like NO through two distinct pathways: a high-potency pathway mediated by ALDH-2 and a low-potency pathway mediated by other enzymes (eg, CYP, deoxyhemoglobin, and xanthine oxidase) or low-molecular-weight reductants (eg, N-acetyl-cysteine and thiosalicylic acid). The ALDH-2 pathway is thought to play a more significant role in the biotransformation of nitrates at therapeutic doses.24-27
Our study found that simultaneous administration of intravenous nitrate and clopidogrel caused an interaction, and nitrate delayed the antiplatelet effect of clopidogrel. The primary mechanism that could explain this result may be that nitrate reduces the oxidation of clopidogrel to its active metabolites by influencing hepatic CYP enzymes. Minamiyama et al 19 investigated whether nitrates administered through continuous intravenous infusion for 24 to 96 h affected hepatic CYP levels in male Wistar rats. They found that hepatic CYP activity drastically decreased after 48 or 72 h of continuous nitrate infusion, but it recovered 48 h after cessation of nitrate administration. The authors point out that continuous nitrate administration may significantly impact hepatic CYP-dependent drug metabolism. Another study examined the effect of sustained-release nitrates in outpatients receiving ASA plus clopidogrel undergoing PCI. 20 Platelet reactivity was evaluated using the VerifyNow P2Y12 assay as in our study. They found that the PRU value was 251.6 ± 80.9 in the nitrate-using group, and the PRU value was 232.1 ± 73.5 in the nitrate-free group (P = .008). These results indicate that the PRU value was higher in the nitrate-using group. There are notable differences between the design of this study and ours. This study included stable outpatients who had been using oral clopidogrel, ASA, and sustained-release nitrates for a long time (mean observation period 17.7 ± 8.1 months). The study used seven different brands of sustained-release nitrates. Blood samples for the platelet function test were collected between 4 and 12 h after the last administration of routine medications, including clopidogrel, ASA, and sustained-release nitrates; however, the average blood draw time was not mentioned. 20 In our literature search, we could not find a study similar to ours that studied the effect of intravenous nitrate and clopidogrel loading on platelet functions during the ACS period, so we were unable to compare our results directly.
Morphine has been shown to reduce the absorption of oral P2Y12 receptor inhibitors, delay their onset of action, and diminish their antiplatelet effects in patients with ACS.14,15 This situation is explained by the increased transit time of the drug and the decreased absorption of antiplatelet agents, resulting from morphine's effects on intestinal motility and enzyme secretion through the activation of opioid receptors. Additionally, the observed emesis effect of morphine may also influence the absorption of platelet inhibitors. Similar mechanisms might also apply to nitrates. The primary effect of nitrates is vasodilation, resulting from the relaxation of smooth muscle in arteries and veins. Nonetheless, this effect is not restricted to the smooth muscle in blood vessels; it also induces relaxation of the bronchioles, gastrointestinal tract, ureters, and uterine smooth muscle to a lesser extent. 28 Therefore, some studies have examined the potential of nitrates to alleviate renal colic pain. 29 Nitrates can affect motility in the gastrointestinal tract, possibly decreasing clopidogrel absorption and delaying its antiplatelet effect. However, it should be noted that this effect may be dose-dependent.
Intravenous nitrate preparations generally contain 30% propylene glycol, with some variations. Propylene glycol is widely used as a drug solubilizer in topical, oral, and intravenous pharmaceutical formulations, and it is generally considered safe.30,31 Propylene glycol is primarily excreted through the kidneys, while the remainder is metabolized in the liver into lactate, acetate, and pyruvate. It has been reported that administering these drugs at high doses and/or for extended periods may lead to propylene glycol toxicity, which can manifest as serum hyperosmolality, lactic acidosis, hemolysis, central nervous system symptoms, and renal failure.30,31 However, we have not identified any substantial evidence in the literature indicating that propylene glycol interacts with any drugs or modifies their pharmacokinetic or pharmacodynamic effects. This indicates that the results of our study are solely due to the influence of nitrate.
In earlier studies assessing clopidogrel's inhibition of platelet aggregation, the cut-off value for HPR in the VerifyNow P2Y12 assay was > 240 PRU or < 40% platelet inhibition.32-34 The recently published International Consensus Statement on Platelet Function and Genetic Testing in PCI defines a therapeutic window between 85 and 208 PRU for the VerifyNow P2Y12 assay. 20 In our study, PRU values at the fourth hour were statistically significantly higher in the nitrate group than in the control group (274.1 ± 53.9 vs 162.4 ± 39.9; P = .002). The PRU values of all patients in the control group achieved the targeted therapeutic range by the fourth hour, while only three patients (20%) in the nitrate group reached this range. A similar situation occurred in the sixth hour. At 48 h, PRU values in both groups were statistically similar; however, three patients (20%) in the nitrate group had a PRU value exceeding 208. The inhibition of platelet aggregation by clopidogrel after loading begins within 2 to 8 h and reaches its peak effect between 6 to 8 h.35-36 Our control group's PRU values and course are consistent with established data on clopidogrel. There is a delay in the antiplatelet inhibition of clopidogrel in the nitrate group.
Clopidogrel is a prodrug that requires metabolic activation by hepatic CYP enzymes to form its active metabolite.7,8 In contrast, ticagrelor is a direct-acting P2Y12 inhibitor that does not require metabolic activation. Due to its pharmacokinetic and pharmacodynamic properties, ticagrelor provides faster, stronger, and more consistent inhibition of adenosine diphosphate receptors than clopidogrel.37,38 Based on our study's results, direct-acting P2Y12 inhibitors might be safer for ACS patients receiving nitrate therapy to reduce thrombotic complications.
Our study has three limitations that should be acknowledged. First, the study groups had few patients, and the allocation between the nitrate and control groups was imbalanced. While it may not be perfect, it is adequate for a pilot study. Our research could help guide more extensive clinical studies in the future. Secondly, the PRU values of the two groups measured at the fourth and sixth hours were statistically significant. However, this difference vanished by the 48th hour. It is unclear exactly when this difference between the two groups disappeared; we could have provided this information if we had planned additional measurements at intervals such as 12, 18, and 24 h between the sixth and 48th hours. Third, we did not assess plasma levels of clopidogrel's active metabolite, which would allow us to understand whether there are genetic differences in clopidogrel metabolism. However, guidelines do not recommend this assessment in routine practice.
Conclusions
Intravenous nitrate therapy in patients with NSTEMI delayed the onset of the antiplatelet effect of orally loaded clopidogrel by at least four hours. Direct-acting P2Y12 inhibitors may be a more reliable option for patients receiving nitrate treatment to prevent thrombotic complications. This pilot study will pioneer on major clinical studies to be performed about this subject.
Footnotes
Acknowledgements
This study was presented as an oral presentation at the fifth Uludag Cardiology Summit (6-9 March 2025, Uludag, Bursa), and it was selected as the third-best oral presentation.
Ethical Considerations
The Bursa Postgraduate Hospital Clinical Research Ethics Committee approved this study, which was conducted according to the principles of the Declaration of Helsinki.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
The authors affirm that participants in the human research provided informed consent for the publication of this article. All authors consent to the article's publication.
Authors’ Contributions
All authors contributed to the study's conception and design. HA, MM, AT, AK, and SA prepared materials and collected data. HA and MM analyzed and interpreted the patient data. HA, MM, AT, AK, SA, and TA wrote the first draft of the manuscript, and all authors provided feedback on this version. All authors read and approved the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data from this study are available from the corresponding author upon reasonable request.
