Abstract
Keywords
Introduction
Patients with cardiovascular disease, particularly those who have undergone percutaneous coronary intervention (PCI), are often administered antiplatelet agents, such as clopidogrel. These patients also require additional medications to manage comorbidities, such as hypertension, diabetes, and hyperlipidemia. Clopidogrel can interact with other drugs, affecting its efficacy and safety. 1
Clopidogrel is a prodrug that is primarily converted by cytochrome P450 (CYP) 3A4 and 2C19 in the liver into its active metabolite. The active metabolite exerts its pharmacological effects by blocking the adenosine diphosphate P2Y12 receptor and inhibiting platelet activation and recruitment. 2 The potential interaction between clopidogrel and calcium channel blockers (CCBs) has been a topic of interest in clinical research.3–5 The findings of several studies that investigated the influence of CCBs on the effectiveness of clopidogrel were conflicting. Some have suggested a potential interaction between CCBs and clopidogrel, whereas others have reported the absence of any substantial association between the two.6–8
Previous research has shown that coadministration of CCBs is associated with decreased platelet inhibition by clopidogrel, indicating that CCBs may reduce the effectiveness of clopidogrel through CYP3A4-mediated interactions. 4 Furthermore, a study assessing ADP-inducible platelet reactivity by light transmission aggregometry and the VerifyNow P2Y12 assay, found that patients with concomitant CCB therapy showed significantly higher on-treatment platelet reactivity than those without CCB medication, confirming CCB therapy as an independent predictor of reduced clopidogrel-mediated platelet inhibition. 5 However, these studies were performed on a limited number of patients and large-scale clinical studies are lacking. Additionally, studies examining the impact on major adverse cardiac and cerebrovascular event (MACCE) with long-term follow-up are also lacking. Population-based cohort study in western Denmark involving 13 001 patients found that concomitant use of CCBs did not modify the protective effect of clopidogrel and was not associated with increased cardiovascular risk among patients using clopidogrel after coronary stent implantation. 6 Similarly, in a cohort study of 1608 consecutive patients, it was found that concomitant CCB therapy did not alter clopidogrel-mediated platelet aggregation and did not have a measurable impact on stent thrombosis and mortality after coronary stenting. 7 Therefore, we aimed to examine the potential interactions between CCBs and clopidogrel using a large-scale national registry of patients who underwent PCI.
Methods and Materials
Data Collection
The Platelet function and genoType-Related long-term proGnosis in Drug-Eluting Stent-treated patients with coronary artery disease (PTRG-DES) consortium conducted a prospective multicenter registry (ClinicalTrials.gov identifier: NCT04734028). This study included 13 160 patients who underwent successful PCI for significant coronary artery disease in nine registries in Korea between July 2003 and August 2018. 9
In the PTRG-DES cohort, 11 714 patients underwent the VerifyNow P2Y12 test during antiplatelet therapy. Patients at each center who had been successfully treated with one or more drug-eluting stents (DES), which were adequately loaded with aspirin and clopidogrel, regardless of the complexity of their medical condition or lesion were enrolled in the study. All PCI procedures were performed according to the standard technique.10,11 Following PCI, patients were administered 100 mg of aspirin and 75 mg of clopidogrel daily. Patients were recommended to continue taking aspirin indefinitely and clopidogrel for at least one year, and all other treatments were per standard of care. The clinical outcomes were evaluated at the last outpatient visit.
The patient population taking dihydropyridine (DHP) or non-DHP calcium channel blockers (CCB) at the time of hospitalization was defined as the CCB users. The institutional review board of each participating center approved the registry and waived the requirement for written informed consent for access to institutional registries. This study was performed in accordance with the Good Clinical Practice Guidelines and principles of the Declaration of Helsinki.
Platelet Function Test
Platelet reactivity was measured after an adequate period to ensure the full antiplatelet effect using the VerifyNow assay (Accriva, San Diego, CA, USA) during the peri-procedural period (from “just after the insertion of the arterial sheath” to “within 24 h after DES implantation”). 12 Aspirin and clopidogrel were administered according to standardized guidelines for loading and maintenance doses. 13 Aspirin was given as either 1) a coated oral dose of 300 mg at least 6 h; or 2) a dose of 100 mg at least 5 days before PCI. Clopidogrel was given as either 1) a dose of 600 mg at least 6 h; 2) a dose of 300 mg at least 12 h; or 3) a dose of 75 mg at least 5 days before PCI.
The VerifyNow assay is a whole-blood, point-of-care, turbidimetric optical detection assay designed to measure agonist-induced platelet aggregation. Blood samples were collected in 3.2% citrate Vacuette™ tubes (Greiner Bio-One Vacuette North America, Monroe, NC, USA). The measurement protocol followed the manufacturer's recommendations as previously described. 11 In this study, platelet function tests (PFTs) for clopidogrel were presented in terms of P2Y12 reaction units (PRU). Based on a previous study that established an optimal cutoff value for PRU related to ischemic events in Korea, 14 high platelet reactivity (HPR) on clopidogrel was defined as a PRU value of 252 or higher.
Clinical Outcomes and Definitions
An independent clinical events committee, blinded to the PFT and genotyping results, adjudicated all clinical events using the source documents. The primary ischemic endpoint encompassed significant adverse cardiac and cerebrovascular events (MACCEs), including all-cause mortality, myocardial infarction (MI), definite stent thrombosis (ST), or stroke up to 5 years. Major bleeding was defined as type 3-5 bleeding according to the Bleeding Academic Research Consortium. 15
All deaths were considered to be of cardiovascular (CV) cause unless a definite non-CV cause was established. MI was defined as increased cardiac troponin levels with ischemic symptoms or ischemic changes on electrocardiography, imaging evidence of recent loss of viable myocardium, or the appearance of a new regional wall motion abnormality unrelated to the procedure. ST (definite) was defined according to the Academic Research Consortium criteria. 12 Stroke was defined as the presence of neurological deficits necessitating hospitalization and clinically documented lesions observable on brain computed tomography or magnetic resonance imaging.
Statistical Analysis
Continuous variables are presented as mean ± standard deviation (SD), while categorical variables are presented as numbers and frequencies. Laboratory measurements and clinical parameters were analyzed using Student's t-test and Fisher's exact test. Survival analysis was conducted to evaluate time-to-event outcomes and estimate survival curves using the Kaplan–Meier method. The log-rank test was used to compare survival curves between groups and determine the statistical significance of the observed discrepancies. Additionally, Cox proportional hazards regression analysis was performed to investigate the independent impact of the variables on survival outcomes. Hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) were generated using Cox proportional hazards regression models. Multivariate analysis was conducted using variables from the univariate analysis, employing the stepwise forward selection method.
Given that this study was conducted as a registry analysis, propensity score matching (PSM) was employed to address potential associations between CCBs and clinical events. Control variables, including age, sex, body mass index (BMI), the clinical presentation of MI, smoking status, hypertension (HTN), diabetes mellitus (DM), dyslipidemia, chronic kidney disease (CKD), peripheral artery disease (PAD), congestive heart failure, prior history of MI or cerebrovascular events (CVAs), platelet reactivity unit, hemoglobin levels, creatinine levels, low-density lipoprotein (LDL) cholesterol levels, the complexity of coronary arterial disease (as categorized by ACC/AHA lesion type), risk of PCI assessed by the complex high-risk indicated percutaneous coronary intervention (CHIP-PCI) criteria, 16 and concomitant medications (aspirin, cilostazol, beta-blockers [BBs], renin-angiotensin system [RAS]-inhibitors, and statins), were considered.
The inverse probability of treatment weighting (IPTW) methodology was applied to account for potential confounding factors. Cox proportional hazards regression analysis stratified by propensity score was used to adjust for HRs associated with the events of interest. Statistical significance was set at
Results
Baseline Characteristics
Patients were divided into two subgroups based on CCB administration, with CCBs administered to 2817 patients (24.0%). Significant differences were observed among the baseline characteristics between the two groups, including age, sex, BMI, clinical presentation: predisposing factors such as HTN, DM, hyperlipidemia, CKD, and previous histories of myocardial infarction, PCI, CABG, and CVA. However, there were no significant differences in congestive heart failure and peripheral artery disease. Propensity score matching (PSM) significantly reduced the differences between the two groups (with a C-statistic of 0.675) regarding patient characteristics like age, sex, BMI, predisposing factors, laboratory tests, angiographic features, and medication use, as well as conditions, such as previous histories of MI, PCI, CABG, and CVA, but not PAD. This was further supported by IPTW analysis. For factors such as age, sex, congestive heart disease, PAD, and past medical history, the standardized mean differences (SMD) were below 0.1, indicating that the method effectively balanced these variables between the CCB and non-CCB groups (Table 1).
Baseline Characteristics.

Values are presented as the mean ± standard deviation or number with percentage (%).
Abbreviations: PSM, propensity score matching; IPTW, inverse probability of treatment weighting; CCB, calcium channel blocker; SMD, standard mean difference; BMI, body mass index; MI, myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; CVA, cardiovascular adverse event; LDL, low-density lipoprotein; ACC/AHA, the American College of Cardiology and American Heart Association; CHIP-PCI, Complex high-risk indicated percutaneous coronary intervention; RAS, renin-angiotensin system.
Platelet Function Test
PRU values were not significantly different between the CCB and non-CCB groups, with mean values of 215.8 ± 84.7 and 218.4 ± 76.7, respectively. Moreover, no substantial variations were observed within patients with HPR based on the utilization of CCB (Table 1 and Figure 1).

Clopidogrel responsiveness according to CCB administration.
Furthermore, after implementing the PSM and IPTW methods, no statistically significant disparities in patients with HPR were detected between the two groups. These findings indicate that the concurrent administration of CCB and clopidogrel effectively reduces the impact of other variables, facilitating a more precise evaluation of their influence on MACCE.
MACCEs According to Calcium Channel Blocker Administration
The occurrence of MACCEs was 6.84% (n = 801) in the total population and was not significantly different between the CCB-users and non-users (6.4% and 7.0%, respectively; HR: 0.988; 95% CI: 0.837-1.166;
Comparison of Major Adverse Cardiac and Cerebrovascular Events (MACCEs) Between Two Groups.

Abbreviations: CCB, calcium channel blocker; PSM, propensity score matching; IPTW, inverse probability of treatment weighting; MACCE, major adverse cardiovascular events; MI, myocardial infarction
This trend persisted even among patients with HPR, and CCB usage did not appear to affect MACCE, and major bleeding based on, as no significant differences were observed between the two groups, even after adjusting using the PSM and IPTW methods (Table 3).
Comparison of Adverse Clinical Events Based on the Use of CCBs in Patients with High Clopidogrel Responsiveness (≥252 PRU).
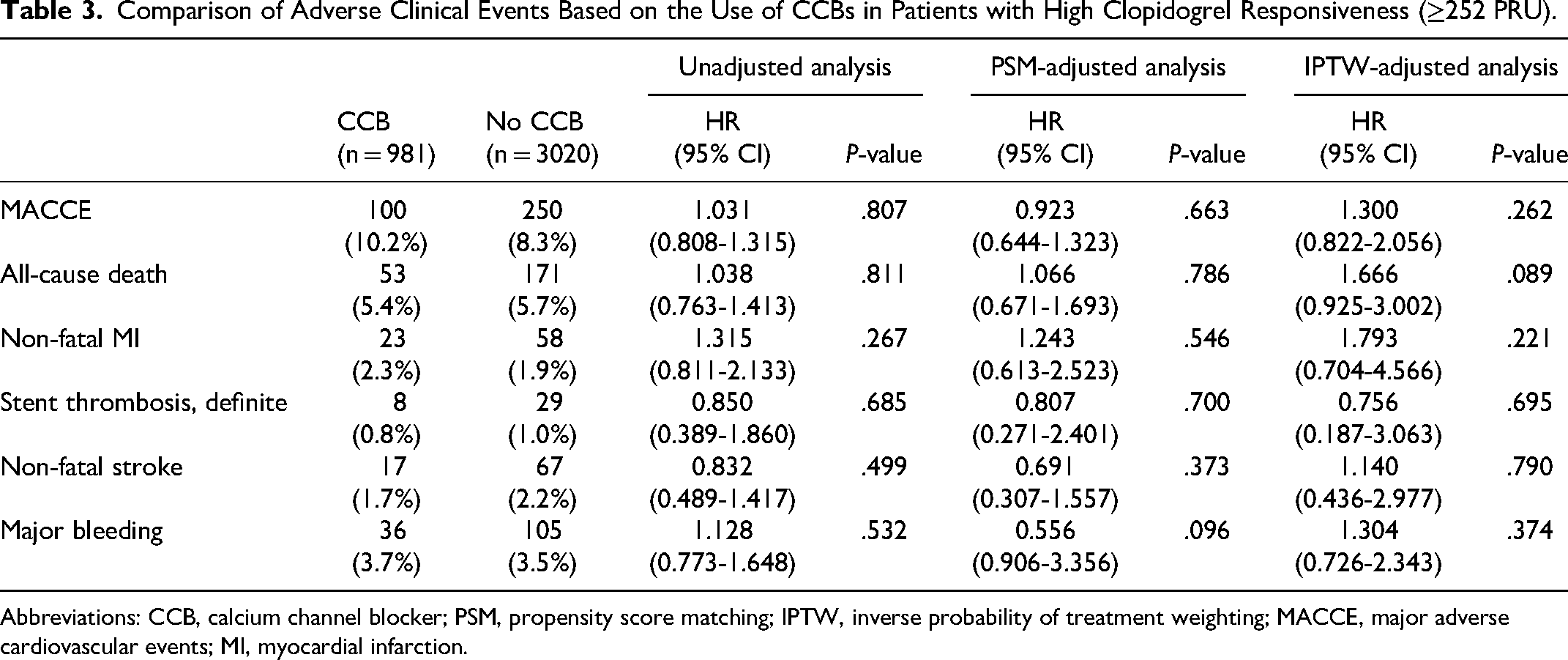
Abbreviations: CCB, calcium channel blocker; PSM, propensity score matching; IPTW, inverse probability of treatment weighting; MACCE, major adverse cardiovascular events; MI, myocardial infarction.
Table 4 presents the results of the univariate and multivariate Cox regression analysis for MACCEs. In the univariate analysis, several variables significantly impacted MACCEs, such as age, DM, HTN, CKD, and CVA. However, the multivariate analysis demonstrated that PAD, a history of CVA, and anemia, but not the use of CCB, significantly influenced MACCEs.
Univariable and Multivariable Cox Regression Analysis for MACCEs.

Abbreviations: BMI, body mass index; DM, diabetes mellitus; CKD, chronic kidney disease; PAD, peripheral artery disease; CHF, congestive heart failure; CVA, cardiovascular adverse event; MI, myocardial infarction; RAS, renin-angiotensin system.
As a subgroup analysis, we conducted further analysis to evaluate the impact of CCB use on platelet reactivity and the MACCE across various CYP2C19 genotype subgroups. A total of 6717 patients had both genotype data and PRU level. The CYP2C19 genotypes were classified into three genotypically predicted metabolizer status groups according to the number of CYP2C19 loss-of-function (LoF) alleles present : (1) extensive metabolizers (EMs) for individuals not carrying the LoF allele (*1/*1, *1/*17, or *17/*17); (2) intermediate metabolizers (IMs) for carriers of one LoF allele (*1/*2, *1/*3, *2/*17, or *3/*17); and (3) poor metabolizers (PMs) for carriers of two LoF alleles (*2/*2, *2/*3, or *3/*3).
Prior study has shown that the CYP2C19 genotype significantly influences 5-year MACCE outcomes in the PTRG registry.
17
Therefore, we analyzed whether there were differences in platelet reactivity and MACCE based on CCB use within each CYP2C19 genotype subgroup. As shown in Supplementary Table 1, there were no significant differences in platelet reactivity between CCB users and non-users within each subgroup [197.0 ± 79.7 versus 193.6 ± 79.5 in the EM group (
Other Clinical Outcomes
Survival analysis was performed to evaluate additional clinical events, including all-cause death, MI, ST, stroke, and major bleeding events. Kaplan–Meier survival plots were generated based on the original patient information (Figure 2). As shown in Table 2, in the CCB and non-CCB groups, respectively, the incidence of all-cause death was 3.3% (n = 92) and 3.9% (n = 350), showing no significant difference (HR: 0.891; 95% CI: 0.708-1.121;
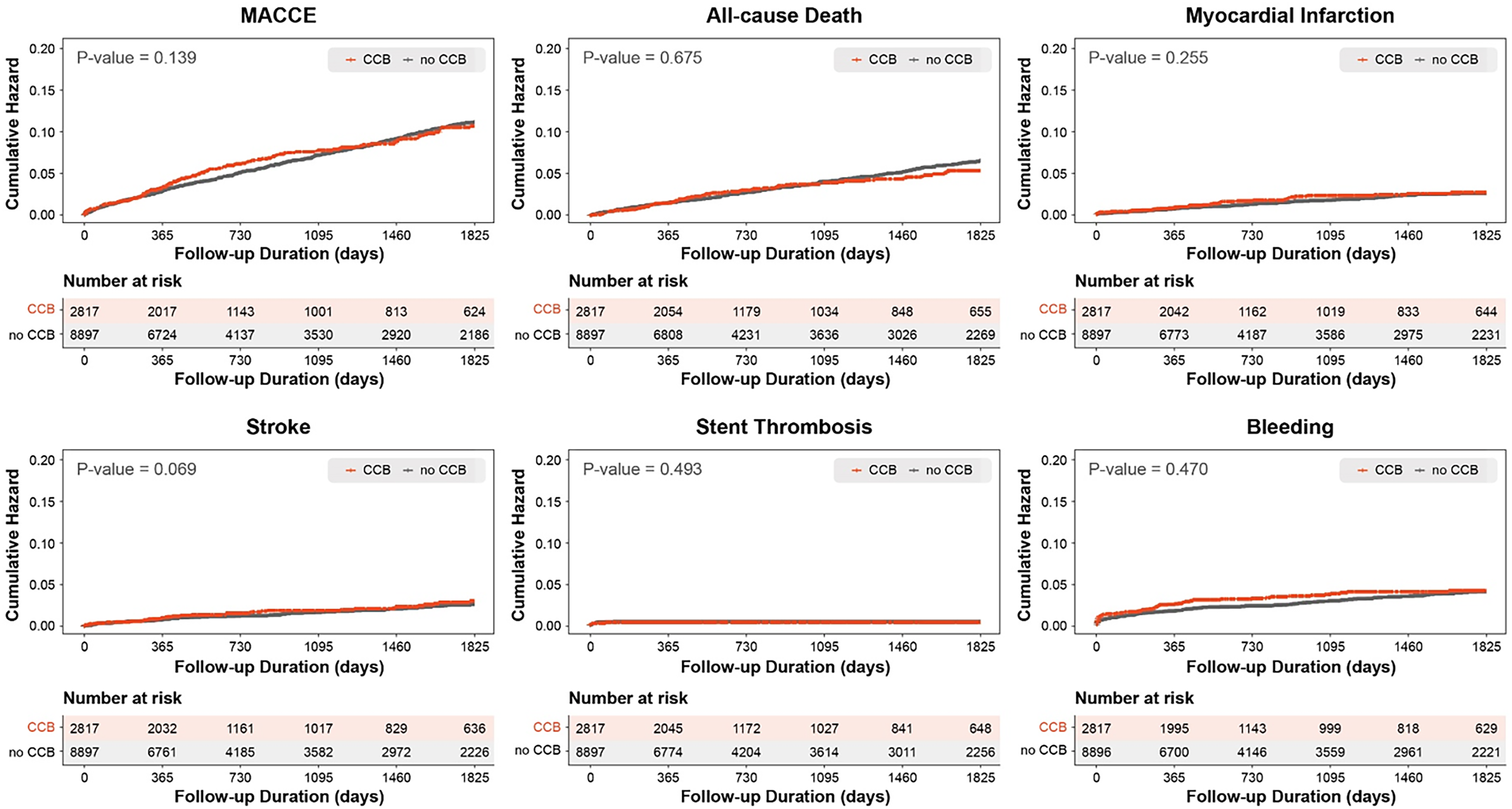
Kaplan–Meier analysis results of the cumulative incidence of events before PSM & IPTW. Abbreviations: PSM, propensity score matching; IPTW, inverse probability of treatment weighting; MACCE, major adverse cardiovascular events.
The Kaplan–Meier survival curves for the PSM-matched groups are presented in Figure 3. No significant differences were found in other clinical outcomes, including all-cause death, MI, ST, stroke, and the risk of major bleeding. Furthermore, survival analysis after IPTW did not reveal any significant differences in the clinical outcomes (Figure 4). The adjusted HRs, 95% CIs, and P-values for the outcomes are provided in Table 2.

Kaplan–Meier analysis results of the cumulative incidence of events after PSM. Abbreviations: PSM, propensity score matching; MACCE, major adverse cardiovascular events.

Kaplan–Meier analysis results of the cumulative incidence of events after IPTW. Abbreviations: IPTW, inverse probability of treatment weighting; MACCE, major adverse cardiovascular events.
Discussion
In this study, we comprehensively analyzed the clinical data of patients treated with drug-eluting stents to evaluate the effects of CCBs on the efficacy of clopidogrel in terms of laboratory measurement and long-term clinical prognosis. To the best of our knowledge, this is the largest cohort study encompassing both PFTs and clinical outcome data that scrutinized the interplay between CCBs and clopidogrel. The key findings were as follows: (1) CCBs did not attenuate the ex vivo efficacy of clopidogrel; (2) the incidence of HPR did not differ between CCB users and non-users; and (3) the co-administration of CCBs with clopidogrel did not increase MACCEs and major bleeding episodes during long-term follow-up, regardless of HPR status.
Current guidelines for ST-segment elevation MI (STEMI) and non-ST-segment elevation acute coronary syndromes classify BBs as Class I indications. 18 CCBs are recommended for patients for whom BBs are contraindicated. Despite this recommendation, CCBs are frequently prescribed, particularly in regions such as Korea and Japan, where the incidence of variant angina is higher than that in Western countries, and the prevalence of acute coronary syndrome is increasing. 19
The latest recommendations for BB use in patients with chronic coronary disease (CCD) are as follows 20 : (1) Prolonged BB therapy is not recommended to enhance outcomes in patients with CCD who have not experienced an MI in the past year, have a left ventricular ejection fraction ≤50%, or lack another primary indication for BB therapy. (2) For initial antianginal therapy, either a CCB or BB is recommended. Given these recommendations, it is crucial to thoroughly investigate the potential interactions between CCBs and clopidogrel.
Recent review articles investigated the concurrent use of clopidogrel and CCB, demonstrating that although variability in clopidogrel-induced antiplatelet effects can impact clinical outcomes, especially in patients undergoing coronary stenting, most studies evaluating the clinical implications of clopidogrel-drug interactions have not shown significant effects. Guidelines have been established recommending the prescription of PPIs, which significantly interact with clopidogrel, to patients who are on dual antiplatelet therapy and have a history of gastrointestinal bleeding. However, clear guidelines on the concurrent use of CCB and clopidogrel have not yet been established. Based on these large-scale studies, the need for randomized trials to provide clarity in this area persists.21,22
CCBs are extensively metabolized in the liver, mainly by cytochrome P450 3A4, resulting in the production of inactive metabolites, thereby competing with clopidogrel for the enzyme. This reduces the antiplatelet effect of clopidogrel. Furthermore, drug transporter P-glycoprotein (P-gp) and the metabolizing enzyme CYP3A often share a wide range of substrates and modulators, and they are frequently co-located within cells. This dual action of transport and metabolism significantly influences the disposition of drugs.22,23
Siller-Matula et al showed that the platelet reactivity index and ADP-induced platelet aggregation were higher in patients receiving concomitant clopidogrel and CCB and that the intake of CCBs was associated with adverse clinical outcomes. 4 These findings are consistent with other studies, which showed that the concomitant use of P-gp-inhibiting and non-P-gp-inhibiting CCBs increases the antiplatelet effect of clopidogrel.5,24
Sarafoff et al investigated the relationship between platelet reactivity and outcomes after coronary stenting in 1608 patients. 7 They found that median platelet aggregation values, the clinical endpoint of death, and ST were not significantly different between patients receiving CCB and those not receiving them. Moreover, Olesen et al performed a study on 56 800 patients, of whom 24 923 were treated with clopidogrel and 13 380 were treated with CCBs. They found that the risk associated with the use of CCBs increased in both groups. 24 Additionally, a randomized, controlled, open-label trial showed that concomitant administration of amlodipine did not significantly alter PRU values or the degree of platelet inhibition. 25
Our study had several limitations. First, it was based on an observational registry, which may have introduced biases and unmeasured factors influencing clinical outcomes. Efforts were made to overcome these limitations by using PSM or IPTW to adjust for potential confounders. Second, the registry only included PFT data at a single point using the VerifyNow method. However, previous studies have used PRU measured during the index hospitalization as a surrogate marker for predicting long-term prognosis after stenting.26,27 Third, medication information was obtained only from discharge records following the index admission, including the exact duration of CCB use, and prescription changes over time were not considered. Additionally, patient compliance and physician discretion might influence clinical outcomes, even though each patient adhered to standard therapy guidelines. This limitation is inherent in large-scale nationwide cohort studies. However, this limitation can be mitigated by the fact that most patients follow standard care regimens after PCI, maintaining consistent medication use without significant changes in the early post-PCI period. Fourth, this study lacked information regarding the specific types of CCBs used (DHP vs non-DHP). While some evidence suggests that CCB types may have different interactions with other medications, 28 previous studies have analyzed CCBs without distinguishing between these two types and there also have no significant interactions with clopidogrel.4,22 Our approach is consistent with existing studies, though further research may provide additional insights. Lastly, only East Asians were included in this cohort; therefore, our findings may not be generalizable to other ethnic groups.
Conclusion
In this large prospective multicenter cohort registry, we found no evidence of clinically significant drug-drug interactions between CCBs and clopidogrel. This result remained consistent even after minimizing confounding variables using PSM and IPTW. According to our study, the concomitant use of CCBs and clopidogrel does not seem to present safety concerns.
Supplemental Material
sj-docx-1-cpt-10.1177_10742484241298150 - Supplemental material for Effect of Calcium Channel Blockers on Antiplatelet Activity of Clopidogrel in Patients Undergoing Percutaneous Coronary Intervention: Insights from the PTRG-DES Consortium
Supplemental material, sj-docx-1-cpt-10.1177_10742484241298150 for Effect of Calcium Channel Blockers on Antiplatelet Activity of Clopidogrel in Patients Undergoing Percutaneous Coronary Intervention: Insights from the PTRG-DES Consortium by HoungBeom Ahn, MD, Hyun-Wook Chu, MD, Ae-Young Her, MD, PhD, Young-Hoon Jeong, MD, PhD, Byeong-Keuk Kim, MD, PhD, Hyung Joon Joo, MD, PhD, Kiyuk Chang, MD, PhD, Yongwhi Park, MD, PhD, Sung Gyun Ahn, MD, PhD, Sang Yeup Lee, MD, PhD, Jung Rae Cho, MD, PhD, Hyo-Soo Kim, MD, PhD, Moo Hyun Kim, MD, PhD, Do-Sun Lim, MD, PhD, Eun-Seok Shin, MD, PhD, Jung-Won Suh, MD, PhD, and On Behalf of the PTRG-DES Consortium Investigators in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
The study was designed by the principal investigator and executive committee and was sponsored by the Platelet-Thrombosis Research Group under the Korean Society of Intervention Cardiology.
Declaration of Conflicting Interests
Dr. Jeong has received honoraria for lectures from AstraZeneca, Daiichi Sankyo, Sanofi-Aventis, Han-mi Pharmaceuticals, and Yuhan Pharmaceuticals, as well as research grants or support from Yuhan Pharmaceuticals and U&I Corporation. Dr. Song has received honoraria for lectures from AstraZeneca, Daiichi Sankyo, Sanofi-Aventis, Bayer Korea, and Samjin Pharmaceutical. Dr. Joo has received honoraria for lectures from AstraZeneca, Hanmi, Samjin, Dong-A, HK Inno. N Pharmaceuticals, and DIO Medical Ltd. The other authors have no potential conflicts of interest to disclose.
Funding
This work was supported by the National Research Foundation of Korea (NRF), sponsored by the Ministry of Science, ICT and Future Planning [grant number NRF-2020R1F1A1075503], and the Platelet Thrombosis Research Group of the Korean Society of Interventional Cardiology.
Data Availability Statement
PTRG data underlying this article cannot be shared publicly because of data protection agreements but can be available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
