Abstract
Introduction
Atrial fibrillation is the most common arrhythmia in the elderly population. The prevalence of atrial fibrillation increases exponentially with age, with over 10% of individuals older than 80 years being affected.1,2 When rate control is unable to manage the functional symptoms associated with atrial fibrillation, rhythm control with antiarrhythmic agents is often considered in the elderly population.3,4 Dofetilide and sotalol are class III antiarrhythmic agents approved for converting to and maintaining normal sinus rhythm in persistent atrial fibrillation.5–8 Sotalol and dofetilide are both potassium channel antagonists which have the potential adverse effect of Torsades de pointes, a polymorphic ventricular tachycardia associated with prolongation of the corrected QT interval (QTc). Incidences of Torsades de pointes reported range from 0.6% to 2.4% for sotalol and 0.9% to 3.3% for dofetilide, with the majority of episodes presenting at the initiation of therapy.9,10 Due to the increased risk of fatal arrhythmias, FDA-approved manufacturing labeling recommends initiation of therapy in a facility that would allow for serial electrocardiographic monitoring for at least 3 days.11,12
To further minimize proarrhythmic risk, standard dosing strategies for dofetilide and sotalol involve dose adjustments based on the patient's baseline QTc and renal function.11,12 The physiologic cardiac changes with aging, alongside electrolyte abnormalities, polypharmacy, and concomitant comorbidities are all additional risk factors that may subject elderly patients to an increased risk for fatal arrhythmias with dofetilide and sotalol.13–15 The tolerability of dofetilide and sotalol in elderly patients has not been well established. Both the Symptomatic Atrial Fibrillation Investigative REsearch of Dofetilide Study and the Sotalol Amiodarone Atrial Fibrillation Efficacy Trial enrolled patients with a mean age of 67 years. As a result, there is limited published data on tolerability outcomes in elderly patients over 80 years of age.5,7 The objective of this study was to describe the tolerability of dofetilide and sotalol initiated in patients 80 years or older and to identify risk factors associated with safety-related events.
Materials and Methods
This was an IRB-exempt, multicenter, retrospective descriptive study conducted across the Mayo Clinic Health System Enterprise. All hospitalized patients 80 years or older who were administered at least one dose of dofetilide or sotalol between May 8, 2018 and July 31, 2021 were considered for inclusion. Patients were excluded if they received prior treatment with dofetilide or sotalol, had orders placed in the emergency department, had documented refusal of research authorization, or were incarcerated.
A list of patients with any documentation of the verbiage “dofetilide, sotalol, initiation, load, loading” in their electronic health record was generated using Mayo Data Explorer reporting. A manual chart review of the electronic health record was performed to confirm the new initiation of dofetilide and sotalol, collect clinical variables, administration of concomitant atrioventricular (AV) nodal blockers and QTc prolonging medications, and outcome-related data.16–18 Additional data parameters such as patient demographics and baseline laboratory results were obtained using electronic health record reporting.
The primary outcome was defined as the incidence of dofetilide or sotalol dose reduction or discontinuation due to safety-related events or clinical concerns during the initial loading hospitalization. Safety-related events or clinical concerns were characterized as episodes of QTc prolongation, Torsades de Pointes, bradycardia, PVC burden, or reduction in renal function. Secondary endpoints include a description of safety-related events leading to the primary tolerability outcome, percentage of patients with standard and nonstandard initial dosing strategy, risk factors for dose discontinuation or dose reduction, and median change in final QTc from baseline between patients who required discontinuation or dose reduction and those who did not. Standard initial dosing was defined by FDA-approved package insert labeling, considering the renal function and QTc for patient eligibility and dose selection (Figure 1).11,12

Standard package insert recommended dosing for treatment of atrial fibrillation/atrial flutter.
Nonstandard dosing was defined as initial dosing that was higher or lower than the FDA-approved package insert dose recommendations based on renal function, QTc or both. Renal function was assessed by calculating the estimated CrCl (eCrCl) using the Cockroft-Gault equation and baseline serum creatinine. Baseline QTc measurements were obtained prior to sotalol or dofetilide initiation. Subsequent QTc measurements were collected from ECGs obtained after each dose. Final QTc measurements were collected either prior to discharge, among patients who were discharged on an antiarrhythmic, or the last QTc obtained prior to therapy discontinuation. QTc was calculated using the Bazett formula. In patients with a prolonged QRS, which was defined as > 120 msec, an adjustment to the QTc was made using the following equation: QTc (adjusted) = QTc – [QRS-120]. 19 Categorical data were reported using frequencies and percentages. Continuous data were reported using either means and standard deviations or medians and interquartile ranges. Patients with discontinuation of dofetilide or sotalol during the index hospitalization due to nontolerability reasons were excluded from the final regression analysis. Univariate logistic regression was utilized to assess risk factors for the primary outcome.
Results
From the initial screening process, 174 patients were evaluated for inclusion, and 104 patients were included in the final analysis. From this group, 47 received sotalol and 57 received dofetilide loading (Figure 2).

Patient inclusion.
Baseline characteristics are described in Table 1. The mean age of the cohort was 83.8 years. The majority of patients (73%) received antiarrhythmic therapy for atrial fibrillation, while fewer received therapy for atrial flutter (14%) or ventricular tachycardia (13%). Common comorbidities among patients undergoing drug loading included hypertension (78%), coronary artery disease (56%), congestive heart failure (49%), and chronic kidney disease (35%). The median [IRQ] baseline eCrCl among the cohort was 57 mL/min [47, 71] and median [IRQ] baseline QTc interval was 447 milliseconds [422, 470]. AV nodal blocking agents were coadministered in 59% of patients receiving dofetilide or sotalol while 36% of patients received concomitant QTc prolonging medications. There were 18 patients who received both concomitant QTc prolonging agents and AV nodal blockers during dofetilide or sotalol initiation. AV nodal blockers consisted of beta-blockers (eg, metoprolol, carvedilol) or nondihydropyridine calcium channel blockers (eg, diltiazem, verapamil). Antiemetics (eg, ondansetron) and antidepressants (eg, venlafaxine, sertraline) were the most common concomitant QTc prolonging medications coadministered. A full table of concomitant QTc prolonging drugs administered can be found in the supplement.
Baseline characteristics.
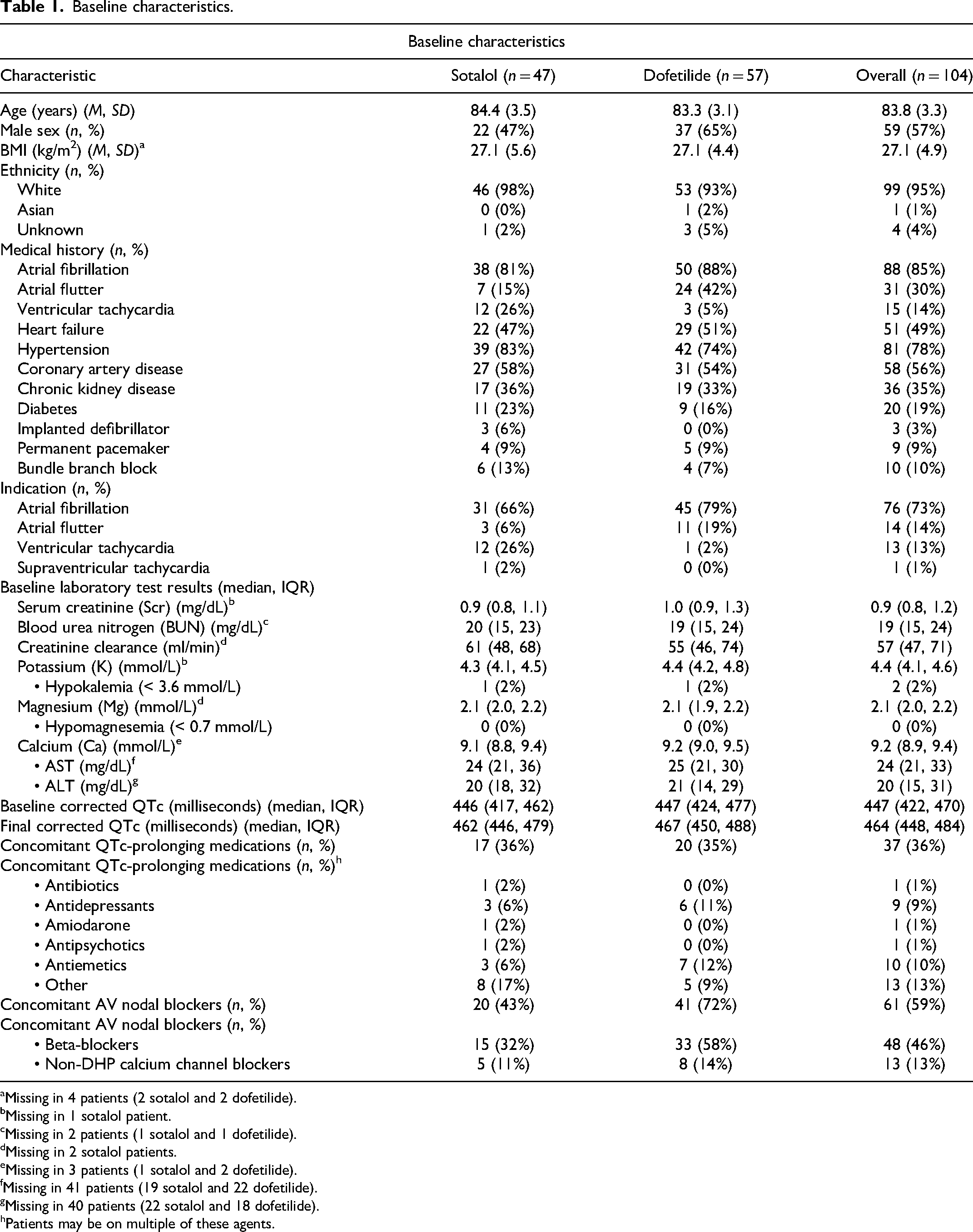
Missing in 4 patients (2 sotalol and 2 dofetilide).
Missing in 1 sotalol patient.
Missing in 2 patients (1 sotalol and 1 dofetilide).
Missing in 2 sotalol patients.
Missing in 3 patients (1 sotalol and 2 dofetilide).
Missing in 41 patients (19 sotalol and 22 dofetilide).
Missing in 40 patients (22 sotalol and 18 dofetilide).
Patients may be on multiple of these agents.
Medication dosing is described in Table 2. Patients received a median of 5 doses of dofetilide or sotalol during the loading period and remained hospitalized for a median of 3 days. A total of 30 (25%) patients received standard initial dosing based on baseline QTc or eCrCl, of which 12 were on sotalol and 18 were on dofetilide. The remaining 74 (75%) patients received nonstandard initial dosing that was higher than package insert recommendations. A total of 65 (63%) of patients received non-standard final dosing that was higher than package insert recommendations.
Medication dosing.

The primary outcome occurred in 41 (39%) patients—19 who received sotalol and 22 who received dofetilide (Table 3). A total of 17 (16%) patients discontinued therapy by discharge, of which 5 had received sotalol and 12 had received dofetilide. A total of 3 patients had therapy discontinued for nonsafety reasons—two in the sotalol group due to perceived inefficacy and one in the dofetilide group due to insurance coverage concerns. Figure 3 illustrates the sequential dose adjustments of patients who had therapy discontinued or dose reduced.

Primary composite tolerability outcome dose adjustments.
Tolerability outcomes.

Among patients who received dofetilide, 35% were started on the maximum dose of dofetilide 500 mcg BID, though only 19% were discharged on this dose. Similarly, among patients who received sotalol, 43% were started on the maximum dose of sotalol 120 mg BID, though only 21% were discharged on this dose. Of the 74 patients who received nonstandard initial dosing, 10 were reduced to a standard dose based on package insert recommendations by discharge. Among the other 64 patients who received nonstandard initial dosing, 11 required discontinuation of the agent, 3 required a dose increase, 17 required a dose reduction, and the remaining 33 patients required no dose adjustments by discharge.
Univariate regression analysis (Table 4) revealed that patients who were administered nonstandard initial dosing of dofetilide or sotalol were 4.7 times more likely to experience the primary outcome of dose reduction or discontinuation due to safety-related events or clinical concerns (OR 4.73, CI [1.64-13.71]; p = .004).
Univariate regression analysis for primary tolerability outcome.

The most frequently reported safety event or clinical concern resulting in discontinuation or a dose reduction was QTc prolongation (68%). Among patients who tolerated therapy and were discharged on their initial dose of dofetilide or sotalol, the median [IQR] QTc at discharge was 458 msec [439, 479], roughly 17 msec longer than baseline (5% increase from median [IQR] baseline of 437 msec [415, 459]). In contrast, the median [IQR] QTc at the time of dose reduction or discontinuation in patients who met the primary outcome was 512 msec [495, 536], approximately 51 msec longer than baseline (11% increase from the median [IQR] baseline of 461 msec [433-477]). Reduced renal function (11%), bradycardia (7%), and PVC burden (5%) were other frequently reported safety-related events requiring dose modification or discontinuation (Table 5). Torsades de Pointes occurred in one (2%) patient, who received a nonstandard initial dose of sotalol.
Reasons for dose reductions and/or discontinuation.

Discussion
In this retrospective study, we found that over one-third of patients required dose reduction or discontinuation of dofetilide or sotalol due to safety-related events or clinical concerns. To our knowledge, this is the first study that has described the tolerability of dofetilide and sotalol loading in elderly patients ≥ 80 years of age. It is well-established that elderly patients are at a higher arrhythmia risk compared to the general population. Age-related physiologic cardiac changes such as ventricular hypertrophy, natural pacemaker cell loss, and upregulation of L-type Ca2+ ion channels lengthen the duration of action potential in elderly patients at baseline. These changes are additionally theorized to alter the pharmacokinetic and pharmacodynamic properties of antiarrhythmic medications in the elderly, further increasing their proarrhythmic risk. 15 In our study, the most common safety event requiring dose modification was QTc prolongation, with one patient even experiencing an episode of Torsades de Pointes.
It has been well established that dofetilide and sotalol demonstrate a dose-response relationship with QTc prolongation, with an expected increase in QTc of 15 msec to 25 msec after the first dose of dofetilide and 25 msec to 40 msec after the first dose of sotalol.11,12 In our study, patients who tolerated their initial dosing seemed to demonstrate an expected and comparable degree of QTc prolongation; however, patients who required a dose reduction or discontinuation had a more pronounced QTc prolongation than adult patients in clinical trials, as we observed a prolongation of approximate 51 msec in this group.
Previously published studies investigating dofetilide and sotalol dose modifications due to QTc prolongation report rates of dose modifications between 10% and 31%.13,20–22 Patients included in these studies had a median age of 65 years, which is a significantly younger population than those included in our study, who averaged 83 years.20–22 Our study illustrated a higher dose reduction or discontinuation rate of 39%, which is higher than previously published real-world studies with younger patients, highlighting an enhanced degree of QTc prolongation in the elderly population.
It is important to recognize that approximately 75% of patients in our study were initiated on a dose that was higher than package insert recommendations based on baseline QTc and renal function. This is a significant finding, as the odds of patients experiencing a dose reduction or discontinuation due to safety-related event was 4.7 times higher in patients who had nonstandard initial dosing compared to standard dosing. In a prior study by Ting and colleagues that included younger adult patients with a median age of 65 years, about 21.7% of patients who received nonstandard dosing experienced a prolongation of QTc >15% of their baseline. 13 Of the elderly patients with a median age of 83 years who received nonstandard initial dosing in our study, only 14% experienced a prolongation of QTc >15% of their baseline. Despite only 14% experiencing a prolongation of QTc >15%, 49% of elderly patients in our study who received nonstandard dosing required a dose reduction or discontinuation; whereas only 37.6% of patients who received nonstandard dosing required a dose modification in the study by Ting et al. 13 While the rates of significant QTc prolongation among patients receiving nonstandard dosing were lower in our study compared to that by Ting and colleagues, we theorize that the elderly population included in our study are more intolerant to these electrophysio cardiac changes and subsequently have a lower threshold for dose reductions.
While the purpose of our study was not to compare sotalol and dofetilide, it is worth mentioning that sotalol's package inserts dosing guidelines are less rigid in comparison to dofetilide, allowing for practitioners to have more latitude in sotalol dosing. Surprisingly in our study, we observed many elderly patients who received the highest recommended dose of dofetilide (500 mcg twice daily), but no elderly patients with preserved renal function received the highest studied dose of sotalol (160 mg twice daily).11,12,23 This observation raises particular interest, as it is rare for sotalol, also having beta-blocking properties, to exhibit its potassium-channel blocking effects before a dose of 120 mg twice daily in adult patients with normal renal function. 24 The dosing trends noticed in our study further support the question of whether the elderly population included in our study are more sensitive to the electrophysio cardiac changes seen with sotalol and warrants further investigation.
Although the prescribing patterns in our health system may not be reflective of practices at other institutions, the importance of not exceeding dofetilide and sotalol package insert recommendations in elderly patients can be extrapolated and endorsed at any practice site. Of the 38 patients who were both initiated on nonstandard dosing and required a dose reduction or discontinuation due to a safety-related event, 10 (28%) were reduced to a standard dose based on package insert recommendations on discharge, while 11 required discontinuation and 17 required dose reductions. This finding corroborates the significance of adhering to package insert dosing upon initiation of antiarrhythmic therapy to optimize patient tolerability at the time of initiation.
This is the first study to describe the tolerability of dofetilide and sotalol in elderly patients 80 years and older—a vulnerable population who is at a very high risk of proarrhythmias. We had a considerable sample size of 104 patients across various Mayo Clinic Health System sites, contributing to the study's external validity. However, some limitations should be noted to improve future studies in this area. First, atrial fibrillation/flutter dosing cutoffs were utilized in assessing nonstandard dosing in all our patients, despite a few patients having received sotalol for an indication of ventricular tachycardia. Because sotalol has different eCrCl cutoffs for dosing in ventricular tachycardia, there was a potential to generate some false “nonstandard dosing” results in the sotalol group. However, we believe that this was minimal in our study, as no patients in the sotalol group were initiated on extended dosing intervals (Q36-Q48 h), as recommended in the sotalol package insert for treatment of ventricular tachycardia when eCrCl <29 mL/min. Next, the computer ECG measurements were not confirmed by electrophysiology attending physician, and the Bazett formula used to correct QT inherently has some limitations in over- or undercorrecting QT when heart rate deviates significantly from average values. Third, given the retrospective nature of this study, we could not comprehensively assess the decision-making behind the selection of initial sotalol dosing, as this was left to provider preference. Additionally, laboratory tests were only obtained at baseline for study analysis and not on sequential days. Finally, there may have been patients who had dose modifications after discharge due to safety-related events, whom we did not follow. Because elderly patients are at a higher risk of arrhythmias at baseline, it is imperative to follow package insert-recommended initial dosing of sotalol and dofetilide to best optimize the tolerability of these agents in this vulnerable population.
Conclusion
In our descriptive study, 39% of elderly patients required a dose reduction or discontinuation of dofetilide or sotalol during their loading hospitalization due to safety-related events. About three-fourths of patients were initiated on higher-than-package insert recommended doses of dofetilide or sotalol based on baseline QTc and renal function, which was significantly associated with the need for dose modifications due to intolerability. Our study highlights the importance of adhering to package insert recommended dosing in elderly patients started on dofetilide and sotalol to optimize patient safety.
Supplemental Material
sj-docx-1-cpt-10.1177_10742484231224536 - Supplemental material for Assessment of Dofetilide or Sotalol Tolerability in the Elderly
Supplemental material, sj-docx-1-cpt-10.1177_10742484231224536 for Assessment of Dofetilide or Sotalol Tolerability in the Elderly by Nikitha Yagnala, Lindsay Moreland-Head, Joseph J Zieminski, Kristin Mara and Shea Macielak in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
