Abstract
Background:
Patients initiated on sotalol and dofetilide require inpatient monitoring and dose adjustments due to risks of corrected QT (QTc) prolongation and Torsades de pointes (TdP). Patients may receive higher initial doses than recommended due to close monitoring by specialized practitioners. The objective of this study was to describe prescribing practices of sotalol and dofetilide and to compare safety outcomes between standard and nonstandard dosing strategies.
Methods:
This was a single-center retrospective analysis of adult inpatients who underwent sotalol or dofetilide initiation between June 1, 2015, and August 1, 2018. The end points of this study included the percentage of patients who received standard and nonstandard dosing, incidence of QTc prolongation (≥500 milliseconds or ≥15% from baseline), incidence of TdP, and dose reduction or medication discontinuation.
Results:
A total of 379 patients (195 sotalol and 184 dofetilide) were included in this analysis. There were 110 (56.4%) patients in the sotalol group and 111 (58.4%) patients in the dofetilide group that received nonstandard initial dosing. Nonstandard dosing was associated with a greater incidence of QTc prolongation compared to standard dosing (57.5% vs 43.0%, P = .005). Only one patient in the nonstandard dosing group experienced TdP. Patients initiated on nonstandard dosing required dose reduction or therapy discontinuation (37.6% vs 23.4%, P = .003) more frequently.
Conclusion:
Higher than recommended initial doses of sotalol or dofetilide were associated with higher incidence of QTc prolongation and more frequent therapy modification.
Introduction
Atrial fibrillation is predicted to affect up to 12 million people in the United States by 2050 and potentially over 17 million in Europe by 2060. 1 Per the ACC/AHA guidelines for atrial fibrillation, in addition to rate control, patients with persistent symptomatic atrial fibrillation should be considered for a rhythm control strategy. 2 Sotalol is an antiarrhythmic medication approved for maintaining normal sinus rhythm in persistent atrial fibrillation or for life-threatening ventricular arrhythmias. 3,4 Dofetilide is effective for achieving or maintaining normal sinus rhythm in persistent atrial fibrillation. 5,6 The use of sotalol and dofetilide is associated with increased proarrhythmic risk due to their potassium channel-blocking properties. 7 Until 2016, dofetilide prescribing had extensive Risk Evaluation and Mitigation Strategies (REMS) requirements, including enrollment of health-care professionals, pharmacies, and health-care facilities into the Tikosyn and Authorized Generic REMS Program. The primary arrhythmia of concern is Torsades de pointes (TdP), a polymorphic ventricular tachycardia, which is associated with prolongation of the corrected QT (QTc) interval. Most episodes of TdP occur within the first 3 days of treatment and incidences reported 0.6% to 4% for sotalol and 0.9% to 3.3% for dofetilide. 8 -12 Due to these increased risks at initiation, both national guidelines and US Food and Drug Administration (FDA)–approved manufacturer labeling for sotalol and dofetilide recommend at least 3 days of monitoring in a facility capable of continuous cardiac monitoring. 11 -13 Metabolic factors that affect drug clearance such as renal dysfunction in addition to electrolyte abnormalities that may destabilize myocardial tissues may also increase the risk of TdP. 14 Sotalol and dofetilide should not be initiated if the baseline QTc interval is prolonged, and patients with reduced creatinine clearance should receive lower doses. 11,12 Additionally, cardiac-associated electrolytes should be within normal range prior to initiation.
Specialized electrophysiologists may prescribe more aggressive dosing strategies than recommended in the package insert in admitted patients as they will be closely monitored in the hospital setting. The objective of this study was to describe the prescribing practices of sotalol and dofetilide and to compare safety outcomes between standard and nonstandard dosing strategies at our institution.
Methods
This was a single-center retrospective analysis conducted at a tertiary academic medical center. All hospitalized adults with at least one administration of sotalol or dofetilide between June 1, 2015, and August 1, 2018 were considered for inclusion. Patients were excluded if they were <18 years of age, received prior treatment with sotalol or dofetilide, or if a baseline electrocardiogram (ECG) within one month before initiation was not available. This study protocol was approved by the institutional review board of Partners HealthCare.
Patients were identified from computer-generated reports of active sotalol and dofetilide orders. Manual chart review of the electronic medical record was performed to collect patient demographics, comorbid medical conditions, baseline laboratory test results, and baseline electrocardiographic findings. Initial sotalol and dofetilide dosing strategies were defined by the FDA-approved manufacturer labeling, which indicate 2 considerations for patient eligibility and dose selection: baseline QTc interval and estimated creatinine clearance (eCrCl; Figure 1). Nonstandard dosing was defined as initial dosing that was higher than the FDA-recommended dose based on QTc (with adjustment for baseline conduction disease), eCrCl, or both. Serum creatinine on the day of medication initiation was used to calculate the eCrCl using the Cockroft-Gault equation. Baseline QTc intervals and subsequent QTc intervals after each medication dose were collected from computer-generated interval measurements uploaded in digital form (MUSE Cardiology Information System; GE Healthcare, Waukesha, Wisconsin). All computer-generated ECG findings, including QTc intervals, ventricular pacing, and abnormal rhythms such as bundle branch block, were manually confirmed and signed off on by a cardiologist. QTc intervals were collected 2 to 3 hours after each dose for at least the first 3 days of medication initiation.

Standard medication dosing algorithms.
End points of this study included the percentage of patients who received standard and nonstandard dosing, incidence of QTc prolongation, incidence of TdP, and dose reduction or medication discontinuation. QTc prolongation was defined as a QTc ≥500 milliseconds (≥550 milliseconds with ventricular conduction abnormality) or ≥15% increase from baseline. Incidence of medication dose reductions or discontinuations during the hospital encounter was collected along with the corresponding reasons determined by chart review.
Continuous data were compared using Student t test. Categorical data were compared using χ2 test and Fisher exact test as appropriate. Statistical significance was set at a level of P < .05.
Results
A total of 1033 patients were evaluated for inclusion. After exclusion, a total of 379 patients were included in this analysis (195 sotalol and 184 dofetilide patients; Figure 2). Patients were then stratified into a standard dosing group and nonstandard dosing group (Table 1). Overall, 221 (58.3%) patients received nonstandard initial dosing. There were 110 (56.4%) patients in the sotalol group and 111 (58.4%) patients in the dofetilide group that received nonstandard initial dosing. Compared to the group that received standard dosing, the nonstandard dosing group had a higher incidence of baseline heart failure (40.7% vs 29.7%) and bundle branch block (14.5% vs 4.4%). Additionally, the baseline eCrCl was significantly lower in the nonstandard dosing group than that in the standard dosing group (76 ± 25 vs 91 ± 27 mL/min). The baseline QTc interval in the nonstandard dosing group was 477 milliseconds compared to 424 milliseconds in the standard dosing group. There were no other significant differences in baseline characteristics.
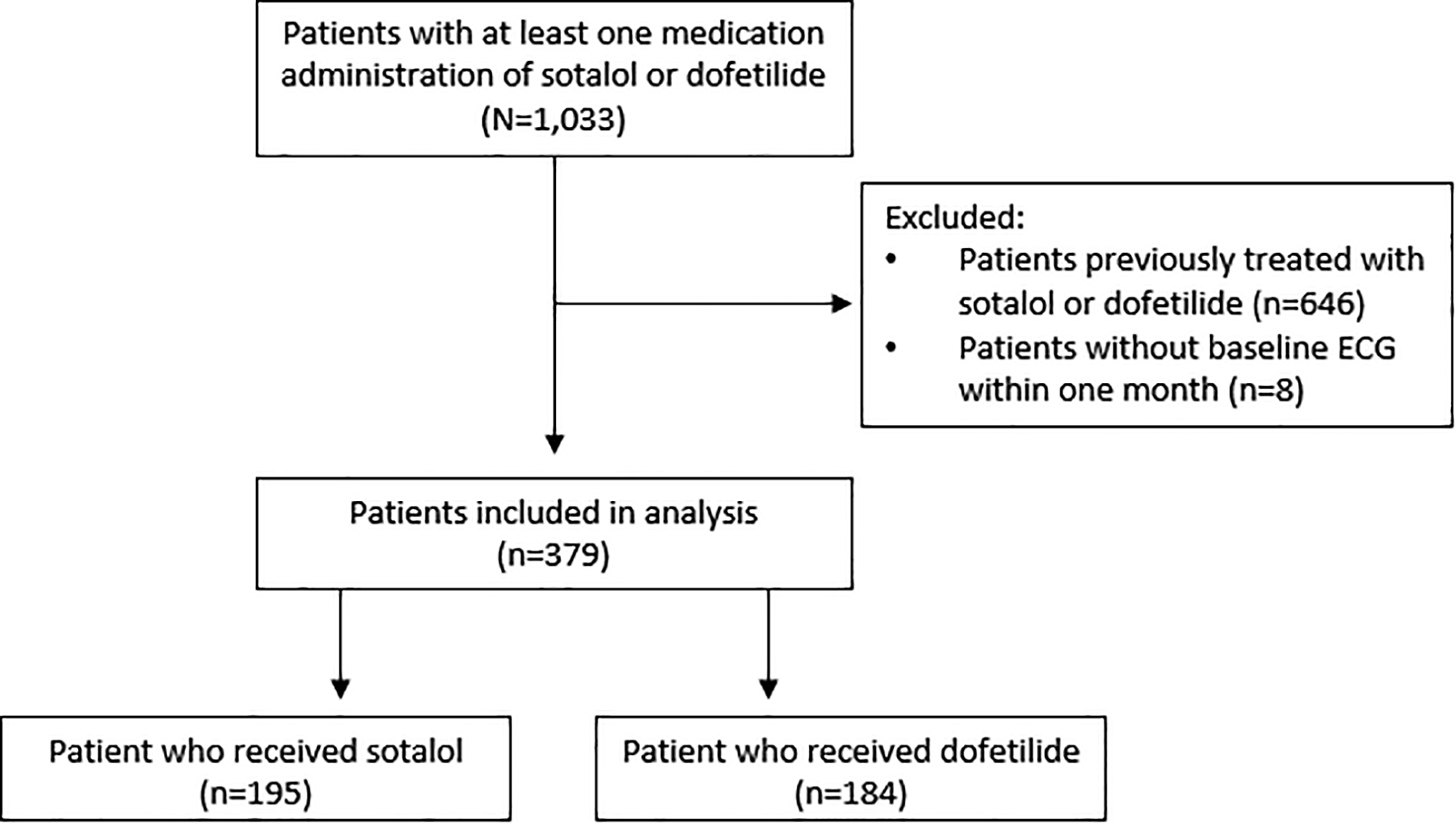
Patient inclusion.
Baseline Characteristics.

Abbreviations: AV, atrioventricular; BMI, body mass index; eCrCl, estimated creatinine clearance; K, potassium; Mg, magnesium; non-DHP, non-dihydropyridine; QTc, corrected QT interval; SCr, serum creatinine; SD, standard deviation.
a Values signify mean ± SD.
b Statistically significant difference between groups.
c Some patients received more than one QTc-prolonging medication.
Initial medication dosing is described in Table 2. Of the 58.3% of total patients who received nonstandard dosing, 54.6% received nonstandard dosing according to baseline QTc interval, 11.6% of patients received nonstandard dosing according to baseline eCrCl, and 7.9% of patients received nonstandard dosing according to both parameters.
Initial Medication Dosing.

Abbreviations: BID, twice daily; eCrCl, estimated creatinine clearance; QTc, corrected QT.
a Some patients received initial overdosing according to both baseline QTc and eCrCl.
Corrected QT prolongation occurred in 57.5% in the nonstandard dosing group compared to 43.0% in the standard dosing group (P = .005; Table 3). The mean QTc interval after each dose was higher in the nonstandard dosing group throughout the duration of the in-hospital initiation period (Figure 3). The mean maximal QTc during initiation in the nonstandard dosing group was 517 milliseconds compared to 471 milliseconds in the standard dosing group (P < .001). Patients initiated on nonstandard dosing required dose reductions or therapy discontinuation more frequently than the standard dosing group (37.6% vs 23.4%, P = .003). The most common reasons for dose reduction or therapy discontinuation were QTc prolongation and therapy ineffectiveness (Table 4).
Treatment Outcomes by Medication Dosing Strategy.

Abbreviations: IQR, interquartile range; QTc, corrected QT; SD, standard deviation.
a Defined as QTc ≥500 milliseconds (or ≥550 milliseconds with ventricular conduction abnormality) or QTc ≥15% from baseline.
b Values signify mean ± SD.

Average baseline and initial 4 corrected QT (QTc) intervals by dosing strategy.
Reasons for Medication Discontinuations and Dose Reductions.a,b

Abbreviation: QTc, corrected QT.
a Values signify n (%).
b Some patients had more than one reason for dose reduction or discontinuation.
c Defined by physician assessment obtained from the electronic medical record. In all cases, physicians ruled the antiarrhythmic therapy as ineffective if atrial fibrillation recurred despite adequate medication loading.
One patient in the sotalol group receiving a nonstandard dose developed TdP in the setting of prolonged QTc and acute kidney injury, which subsequently returned to normal sinus rhythm without intervention. There were no cases of in-hospital mortality in either group.
Discussion
In this retrospective study, assessing dosing strategies of sotalol and dofetilide, we found that more than half of patients received initial doses that were higher than recommended based on their baseline QTc interval and kidney function. To our knowledge, this is the first report of safety outcomes in this specific population.
Overall, the prescribing patterns of sotalol and dofetilide at our institution do not reveal any preference for one antiarrhythmic medication over the other. Interestingly, the dofetilide group had fewer patients who received nonstandard dosing based on eCrCl (Table 2). This may be attributed to physician recall of the prior FDA-mandated REMS Program, recently discontinued in 2016. Conversely, fewer patients prescribed sotalol received the maximum FDA-recommended dose which is likely due to differences in its dosing algorithm compared to dofetilide. Rather than starting at the highest dose with dose reductions as indicated based on QTc, sotalol is started at a more moderate dose and titrated up based on clinical effect.
It is important to note that the majority of patients received nonstandard dosing based on a prolonged QTc interval at baseline, while FDA-approved manufacturer labeling recommends against using the medication completely. 11,12 Manufacturers define baseline QTc prolongation as 450 milliseconds or longer for sotalol and 440 milliseconds or longer for dofetilide. Our findings show that many patients encountered in clinical practice do not present with baseline QTc intervals below this cutoff. There may be several reasons for this observation. There was a significantly greater number of patients in the nonstandard dosing group who had bundle branch block at baseline, in which a widening of the QRS interval causes a false prolongation in the QTc interval. Additionally, nearly half of our patients were in atrial fibrillation at the time of medication initiation, which makes true QTc interval estimation difficult. 15
Approximately 58% of patients in the nonstandard dosing group and 43% of patients in the standard dosing group experienced QTc prolongation. These rates are somewhat higher compared to those of similar single-center studies. 16,17 The mean maximal QTc interval during medication initiation was also higher in the nonstandard group. Additionally, it was greater than the maximum recommended QTc interval of 500 ms. The patients included in our study did have significant cardiac comorbidities that may have heightened drug-induced QTc prolongation (Table 1). Additionally, one-third of patients received at least one other QTc-prolonging medication during their hospital encounter, which is not reported in all studies.
Clinical trials of sotalol and dofetilide have previously demonstrated a dose–response relationship with TdP, especially at excessive doses. 4,18 In one prior study by Benditt et al, sotalol patients did not develop TdP if QT intervals were monitored and doses were adjusted according to renal function. 19 This was a double-blinded, randomized controlled trial that excluded patients with a baseline QTc >450 milliseconds or CrCl <40 mL/min, utilized continuous transtelephonic monitoring, and routinely discontinued treatment if the QTc increased to 520 milliseconds or greater. These carefully selected patients and resource-intensive study settings may have offered more protection against TdP than a real-world clinical practice. Nevertheless, in our study, there was only one occurrence of TdP despite a greater incidence of QTc prolongation in the nonstandard dosing group. This is a similar incidence as previous reports. 7 -11
The finding of more frequent medication dose adjustments or discontinuation in patients who received nonstandard dosing is also important to discuss. In studies regarding dofetilide, dose reductions and medication discontinuation due to increased QTc interval occurred in 5% to 7% and around 2% of patients, respectively. 7,8 However, our study mirrors the results of other real-world experiences which have reported dose reductions and discontinuations rates up to 17% to 31% and 10% to 21%, respectively. 20 -22
Our study has several limitations. First, due to the single-center and retrospective design, there were no prespecified selection criteria for medication initiation with dosing strategies reflecting individual provider discretion. Second, patients with a longer QTc interval at baseline are more likely to hit the threshold for QTc prolongation of ≥500 ms; however, this remains a clinically meaningful measure since it is well known that most arrhythmias occur when the QTc interval is prolonged to 500 milliseconds or greater. 23,24 Corrected QT prolongation ≥500 milliseconds during sotalol or dofetilide initiation is also the point where manufacturers clearly recommend that the dose be reduced, the dosing interval be lengthened, or the medication be discontinued. 11,12 Finally, given the retrospective nature of our study, we did not assess for long-term efficacy or safety outcomes past the index hospitalization.
Further studies may seek to prospectively evaluate the use of sotalol and dofetilide in patients with prolonged QTc intervals at baseline. Based on the current evidence, antiarrhythmic medication options should be considered on a patient-by-patient basis, considering relevant comorbidities, concomitant medications, and baseline laboratory test results and electrocardiographic findings.
Conclusion
In our single-center experience, receipt of sotalol or dofetilide at doses higher than recommended was associated with a higher incidence of QTc prolongation during initiation and more frequent drug therapy modification.
Footnotes
Author Contributions
Clara Ting contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Rhynn Malloy and Danielle Knowles contributed to conception and design, contributed to acquisition, analysis, and interpretation, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
