Abstract
Introduction
Рyridoxal-5-phosphate (PLP) is involved in various biochemical reactions that regulate cellular metabolism, being a cofactor for more than 150 enzymes. 1 It is involved in such important cellular processes as the regulation of protein, lipid, and carbohydrate metabolism, particularly the regulation of amino acid biosynthesis and catabolism, and can be an inducer of gene expression.1,2 PLP is a coenzyme for transaminase reactions that regulate protein folding and glycogen degradation, the biosynthesis of tetrapyrroles (heme, cobalamin, and chlorophyll), and neurotransmitters (epinephrine, dopamine, and serotonin), the conversion of tryptophan to niacin and homocysteine to methionine and other important processes.1,2
ATP-sensitive potassium (KATP) channels are a powerful defense system and the cell's central metabolic sensor in terms of its energy supply.3,4 Such highly ATP-sensitive molecular structures play an exceptional role in the synchronization of metabolism and electrical activity of cells, regulation of potential-dependent membrane functions, and maintenance of hormonal homeostasis, while their dysfunction may promote pathogenesis. Conversely, activation of these channels has a protective effect, including cerebro-, neuro-, and cardioprotective, which consists in the suppression of metabolism and excitability, as well as the normalization of bioenergetic processes in the cell, while maintaining a high ATP content.4–7 However, KATP channels play an essential role in the cardiovascular system, as they realize the relationship between the heart's energy resources and its electrical and contractile function, and regulate vascular tone.
Reduced expression of KATP channels and their dysfunction (including channelopathies) may be associated with pathological processes.8–13 In particular, in rats, the expression of SUR2A subunits of cardiac KATP channels decreases with age, which is accompanied by a decrease in the animals’ resistance to ischemia and exercise, while an increase in SUR2A expression increases the animals’ resistance to these factors, forming an ischemia-resistant cardiac phenotype.13–15 Arterial hypertension may be associated with decreased expression of the Kir6.1 and SUR2B subunits of vascular-type KATP channels.8,10,16,17 Even such a powerful endogenous cardioprotective phenomenon as ischemic preconditioning is thought to be associated with an increase in the density of KATP channels on the membranes of cardiomyocytes.14,18 It is likely that the search for new ways to enhance the expression of KATP channels could be a new therapeutic approach but remedies that increase the expression of KATP channels have been little studied. Systematic studies of the inducers of KATP channel expression have not been carried out. 19 There are some studies that have demonstrated an increase in the expression of certain subunits of these channels under the influence of some factors or compounds, with the most studied ones being certain models of hypoxia.14,20,21 When choosing a possible inducer of regulation of the expression of KATP channels for our study, we paid attention to hydrogen sulfide because it has cardioprotective and vasodilator effects, including through the activation of KATP channels. There is also the possibility of controlled endogenous synthesis of hydrogen sulfide using pyridoxal-5-phosphate (PLP), which is a cofactor of H2S-synthesizing enzymes. Therefore, the aim of our work was to investigate the role of KATP channels in PLP-induced cardioprotection at aging.
Methods and Materials
Laboratory Animals
Experiments were performed on adult (6 months) and old (24 months) male Wistar rats. Animals were housed in the institute's vivarium under normal conditions with standard chow and free access to water. Animals were maintained in a neutral temperature environment (22 °C±2 °C) with a standard day/night cycle. The body weight of adult and old rats was between 220–250 g and 350–450 g, respectively. The experimental rats were divided into 3 groups of 24 animals each: adult, old, and old PLP-treated rats. PLP was dissolved in distilled water each day prior to administration. The resulting solution was administered orally to the rats once a day for 14 days at a dose of 0.7 mg/kg. All procedures were performed in accordance with Directive 2010/63/EU of the European Parliament and of the Council on the protection of animals used for scientific purposes (September 22, 2010). The experimental protocols were reviewed and approved by the Biomedical Ethics Committee of the Bogomoletz Institute of Physiology National Academy of Sciences of Ukraine (No. 2/21, June 16, 2021).
Extraction of RNA, Reverse Transcription, and Real-Time Polymerase Chain Reaction Analysis
The mRNA expression of the genes KCNJ8, KCNJ11, ABCC8, and ABCC9, which encode Kir6.1, Kir6.2, SUR1, and SUR2 subunits of KATP channels, respectively, was examined. To determine the mRNA expression levels of a specific gene in the heart, we first isolated total RNA from heart tissue using the Tri reagent (Sigma-Aldrich). A NanoDrop ND1000 spectrophotometer (NanoDrop Technologies Inc.) was used to determine the purity and concentration of total RNA. Reverse transcription was carried out by dint of the RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). For the genes encoding the aforementioned subunits of KATP channels and the housekeeping gene beta-actin the real-time polymerase chain reaction (RT-PCR) amplification reaction was performed in a volume of 10 μL containing 1 μL cDNA, 5 μL SYBP-Green PCR Master Mix and 0.1 μL of forward/reverse primers. Table 1 presents primers that were used to determine gene expression in RT-PCR experiments. PCR was carried out using 7500 Fast Real-Time PCR (Applied Biosystems). PCR amplification consisted of 50 cycles of a denaturation step—95 °C for 20 s, primer annealing, and elongation of 60 °C for 30 s. The instrument software automatically calculated the threshold cycle. The expression level of each gene was standardized accordingly to the housekeeping gene, beta-actin, and was calculated as the difference between the threshold values of both genes. Analysis of the obtained data was carried out using the 7500 Fast Real-Time PCR software.
The Primer Sequences That Were Used to Determine Gene Expression.

Protein Isolation and Western Blot
Isolated and washed in cold phosphate-buffered saline (PBS) aorta tissue samples were used for protein isolation. Tissues were homogenized in radioimmunoprecipitation assay buffer (RIPA buffer, Pierce, Rockford, IL, USA) supplemented with phosphatase inhibitors (PhosSTOP, Merck, Germany) and protease inhibitors (phenylmethylsulfonyl fluoride, Merck, Germany) according to the manufacturer's instructions. Protein content was determined by the Bradford assay (Bio-Rad) and 20 μg of proteins were loaded into SDS-PAGE. The following antibodies were used for Western blotting: Kir6.1 (1:1000, NBP2-59324, Novus Biologicals, USA), whereas SUR2 was detected with ABCC9 antibody (S319A-14) (1:1000, NBP2-22403, Novus Biologicals, USA). After exposure to horseradish peroxidase-conjugated secondary antibodies (antimouse, 1:10000, sc-2005, Santa Cruz Biotechnology), target proteins were visualized by enhanced chemiluminescence (Pierce, Rockford, IL, USA). Proteins were quantified by densitometry (n = 4/group) using ImageJ software. 22
Morphological Studies
Hearts were isolated, washed in cold PBS, and fixed in 4% paraformaldehyde, dehydrated, and embedded in paraffin. Cardiac tissue morphology was determined using transverse 6-μm deparaffinized sections stained with picrosirius red (PSR). Five sections from 3 rats per experimental group were analyzed. The area of fibrosis was measured using NIH ImageJ image analysis software. 23 The cross-sectional area of the heart was analyzed using an Olympus BX41 light microscope and a HUVITZ HSZ stereo microscope. PSR staining was performed in the Laboratory of Electron Microscopy, Nencki Institute of Experimental Biology, Warsaw, Poland.
Measurement of Contraction–Relaxation Responses of Isolated Rat Aortic Rings
The contraction–relaxation reactions of isolated vascular preparations were studied in experiments on isolated aortic rings of rats, perfused at the temperature of 37 °С normal Krebs solution. Measurements of contraction–relaxation reactions were made in the isometric mode, at the initially set tension of the vascular preparation, when it developed maximum contractions in response to the introduction of noradrenaline (10 μmol/L). The temperature of the working solution in the perfusion system and the experimental chamber was maintained using a KISS 208B automatic thermostat (Huber, Germany). The working solution was saturated with oxygen using a gas mixture containing 95% oxygen and 5% carbon dioxide. Before the experimental measurements, the aortic rings fixed and stretched as indicated above were kept in the working chamber until their stabilization. Krebs solution contained (in mmol/L): NaCl, 120.4; NaHCO3, 15.5; NaH2PO4, 1.2; KCl, 5.9; glucose, 11.5; CaCl2, 2.5; MgCl2, 1.2. Studies of vasodilatory effects were performed with a prerequisite of the enhanced tone of preparations of the aorta, which was obtained by perfusion of norepinephrine (10 μmol/L). For activation of KATP channels, the pharmacological KATP channel flocalin opener was used in doses of 0.1–100 μmol/L.24,25 The character of vasorelaxation responses was determined by an affection of the specific inhibitor of KATP channels glibenclamide, which was administrated into a perfusion solution in a dose of 100 μmol/L 10 min before the administration of flocalin; and its administration was continued during the perfusion of vascular rings with flocalin.
Ischemia-Reperfusion of an Isolated Rat Heart
The thoracic cavity of anesthetized animals was opened at the level of the junction of the ribs and the sternum, and the heart was removed and immediately placed in an ice-cold Krebs-Henseleit solution containing (in mmol/L) NaCl, 118.2; NaHCO3, 25; KH2PO4, 1.2; KCl, 4.8; glucose, 12; CaCl2, 1.7; MgSO4, 1.2. Coronary vessels were retrogradely perfused with Krebs-Henseleit solution at a stable perfusion pressure of 75–80 mmHg. The solution was saturated with oxygen using a gas mixture containing 95% of oxygen and 5% of carbon dioxide. The temperature of the system was maintained at the level of 37 °С with an accuracy of ±0.5 °С by means of an automatic thermostat. The change in the contractile activity of the heart was determined by the change of the pressure in the latex balloon injected into the left ventricle and by the first derivative of the pressure in the left ventricle—dP/dt. Registration was performed using a strain gauge (Elema, Sweden) and recorded using Global Lab software. Coronary flow (CF) was determined by the volume of perfusing solution that passed through the heart in 1 min. The cardioprotective effects of course administration of PLP to old rats were studied in experiments simulating ischemia (20 min) and reperfusion (40 min) of the heart. Ischemia-reperfusion of the isolated heart of untreated old rats was used as a control. The nature of cardioprotection was identified using a specific KATP channels inhibitor—glibenclamide, administered intraperitoneally at 15 mg/kg 20 min before decapitation.
Determination of Hydrogen Sulfide Levels and Oxidative Stress Markers
H2S levels were determined in rat blood plasma using the method previously described with modifications. 26 We measured the rate of superoxide (•O2−) formation and the levels of hydrogen peroxide (H2O2), malondialdehyde (MDA), and diene conjugates (DC) as markers of oxidative stress and lipid peroxidation. Measurements were taken in the blood plasma of adult, old, and old PLP-treated rats. The methods for measuring these indicators have been described in detail in our previous work. 26 In brief, to determine the formation of the superoxide anion radical, we used the reduction reaction of cytochrome c.26,27 H2O2 definition consisted of the indirect registration of its utilization during the oxidation of iodide (I–) to iodine (I3–) under conditions of excess lactoperoxidase. 28 DC definition consisted of lipid extraction from the samples using organic solvent (isopropanol/heptane, 1:1) and measurement of absorbance at 232 nm. 29 MDA was determined by its reaction with 2′-thiobarbituric acid, which results in the formation of thiobarbituric acid reactive substances that can be measured spectrophotometrically. 30
Statistical Analysis
The normality of the data distribution in each group was checked using the Shapiro-Wilk test. Comparisons between groups were made using parametric one-way analysis of variance (ANOVA) followed by post hoc Tukey HSD test and nonparametric tests were used, namely Mann–Whitney U-test and Kruskal–Wallis test for multiple independent samples with post hoc test by Conover's method. In experiments made to determine biochemical indicators and protein expression by a Western blot, as well as in those made on isolated vessels, P values were calculated by one-way ANOVA followed by post hoc Tukey HSD test. In experiments on an isolated heart, P values were calculated by the Mann–Whitney U-test. When determining gene expression, P values were calculated by the Kruskal–Wallis test with a post hoc test by Conover's method. In the morphological studies, the differences between the groups were assessed using a one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for multiple comparisons. Statistical analyses were performed using Prism software (GraphPad, San Diego, CA, USA). P < .05 values were considered statistically significant. Data are presented as mean ± SEM.
Results
Effect of PLP Treatment on the Function of the Ischemic Heart of Old Rats During Reperfusion
PLP significantly improved the recovery of cardiac function in old rats during reperfusion of the ischemic heart compared to control animals. In particular, myocardial contractile activity and CF were significantly better (Figure 1). Namely, reperfusion pressure in the left ventricle (LVP) in PLP-treated animals was significantly higher than in old untreated rats, with a maximum increase of 45% (P = .006), 38% (P = .007) and by 39% (P = .02) at the 5th, 20th, and 40th minutes of reperfusion, respectively (Figure 1a). Left ventricular developed pressure (LVDP) in treated animals was higher than in the group of control rats (not treated with PLP) throughout reperfusion, with a maximum of almost twice (P = .003) of the normal level at the fifth minute of reperfusion and by 74% (P = .005) and 75% (P = .001) at 10th and 20th minutes of reperfusion, respectively (Figure 1b). Indicators of myocardial relaxation rate (dP/dtmin) and myocardial contraction rate (dP/dtmax) were also significantly better in PLP-treated rats compared to control animals (Figure 1c). CF was also significantly better with the administration of PLP (Figure 1d). The most positive changes in CF in PLP-treated rats were observed in the first 10 min of reperfusion, with a 34% (P = .001) increase in flow at 5 min and a 31% (P = .018) increase at 10 min of reperfusion.

Effect of course administration of PLP and glibenclamide to PLP-treated rats on the function of the isolated rat heart of old rats in ischemia-reperfusion. (A) LVP; (B) LVDP; (C) dP/dtmax, dP/dtmin, the maximum and minimum rates of pressure development in the left ventricle; (D) CF rate. Data are represented as mean ± SEM. For old rats, n = 9; for old PLP-treated rats, n = 8; for rats treated with PLP and glibenclamide, n = 8. *Р<0.05 versus old rats. P values were calculated by the Mann–Whitney U-test.
Administration of a specific inhibitor of KATP channels, glibenclamide, to PLP-treated animals, significantly reduced the cardioprotective effect of PLP on cardiac contractile function, particularly on indicators such as LVP, LVDP, and dP/dt (Figure 1). However, glibenclamide only partially reversed the increase in CF in PLP-treated old rats. This may indicate that the increase in CF with the introduction of PLP does not only occur through KATP channels. It is possible that hydrogen sulfide, which has increased in the treated animals, is involved in this process.
Effect of Course Administration of PLP on the Structure of the Myocardium of Old Rats
Since cardiac remodeling, namely myocardial fibrosis, is one of the hallmarks of aging, we analyzed cardiac morphology in experimental groups of animals using PSR staining. As expected, we observed an increase in interstitial and perivascular fibrosis in the hearts of old animals (Figure 2). However, in the hearts of PLP-treated old rats, quantitative analysis showed a 2-fold reduction in the area of fibrosis compared to untreated old animals (P = .0011). Although the level of fibrosis in the hearts of this group of animals remained significantly higher than in young animals (Figure 2d), our data indicate that 2 weeks of PLP treatment significantly reduces the level of fibrosis in the hearts of 2-year-old animals.

Representative PSR staining of rat hearts of different groups. (A) whole heart, bar, 1 сm; (B) and (C) representative staining of heart tissues (transverse sections, ×10) interstitial (B) and perivascular (C) fibrosis; (D) quantitative analysis of fibrosis area. Data are represented as mean ± SEM; n = 5/group. Indicated P values were calculated by ANOVA test followed by Tukey's multiple comparisons test.
Effect of PLP Administration on Vasodilatory Responses of Isolated Aortic Rings
Perfusion of thoracic aortic rings from old and adult rats with the pharmacological opener of KATP channels flocalin at doses of 0.1–100 μmol/L led to dose-dependent vasodilatory responses of vascular strips (Figure 3). Preperfusion of aortic rings for 10 min with a specific KATP channel inhibitor, glibenclamide, at a dose of 100 μmol/L, prevented the vasodilatory effects of flocalin, indicating that these vasodilator responses are due to activation of cell membranes KATP channels (Figure 3a). The vasodilatory responses of aorta rings from old rats in response to different doses of flocalin were significantly different in control and PLP-treated animals (Figure 3a). PLP administration to old rats significantly increased the amplitude of the vasodilatory effects of the pharmacological opener of KATP channels flocalin, namely 2.7 times (P = .005), 2.1 times (P = .018), 1.4 times, and 1.25 times at doses of flocalin of 0.1, 1, 10 and 100 μmol/L, respectively.

Vasodilatory responses of aortic rings on the administration of opener of KATP channels flocalin, effects a specific inhibitor of KATP channels glibenclamide, and levels of mRNA expression of Kir6.1 subunits of vascular-type KATP channels in PLP-treated and untreated old (A) and adult (B) rats. Data are represented as mean ± SEM; n = 8–11. *P < .02 versus old rats; #P < .001 versus old PLP-treated rats. P values for vasodilatory responses were calculated by ANOVA test. P values for Kir6.1 expression were calculated by the Kruskal–Wallis test.
Effects of PLP Treatment on the Expression of Different Subunits of KATP Channels
Administration of PLP to old rats resulted in a 3.64-fold (P = .00011) increase in mRNA expression of pore-forming Kir6.1 subunits of sarcolemmal KATP channels, predominantly of the vascular type (Figure 4a). At the same time, mRNA expression of pore-forming Kir6.2 subunits of cardiac-type sarcolemmal KATP channels did not change significantly in old rats under the influence of PLP (Figure 4b). The difference in mRNA levels of SUR1 subunits of the mitochondrial KATP channels in control and PLP-treated old rats was also not statistically significant (Figure 4c). However, measurement of mRNA levels of the regulatory SUR2 subunit of sarcolemmal KATP channels of both vascular and cardiac type in old rats showed a 2.38-fold increase (P = .016) with PLP administration (Figure 4d).

Levels of mRNA expression of Kir6.1 (A), Kir6.2 (B), SUR1 (C), and SUR2 (D) subunits of KATP channels in the heart of adult, old, and PLP-treated old rats. Data are represented as mean ± SEM; n = 7–11. Indicated P values were calculated by the Kruskal–Wallis test.
Comparison of the expression levels of SUR1, SUR2, Kir6.1, and Kir6.2 subunits of KATP channels in adult and old untreated rats showed that Kir6.2 expression was significantly increased in old animals (3.2-fold, P = .0002), whereas the expression levels of SUR1, SUR2, and Kir6.1 were not significantly different in adult and old untreated rats (Figure 4).
Kir6.1 and SUR2 Protein Levels in Aortic Lysates From PLP-Treated Rats
Since the results of determining the mRNA expression of KATP channels subunits after course administration of PLP showed a significant increase only in Kir6.1 and SUR2 subunits, we decided to check the change of these subunits also at the protein expression level. The expression of Kir6.1 protein, which forms the pore of KATP channels in the aorta of adult and old rats, was not significantly different, whereas PLP treatment in old rats increased Kir6.1 content by 40% (P = .03) (Figure 5a). The expression of SUR2, the regulatory protein of KATP channels, was 2.1 times higher (P = .01) in the aorta of old rats than in adult animals (Figure 5b). In PLP-treated old rats, SUR2 protein levels were 20% higher than in untreated old rats (Figure 5b). Thus, PLP treatment of old rats not only significantly increases the mRNA expression of the pore-forming subunit Kir6.1 and regulatory subunit SUR2, but also significantly increases the protein expression of these subunits of KATP channels.

(A) Western blot of Kir6.1 in aorta lysates of adult, old, and PLP-treated old rats and densitometry of Kir6.1 protein normalized to GAPDH. (B) Western blot of SUR2 in aorta lysates of adult, old, and PLP-treated old rats and densitometry of SUR2 protein normalized to GAPDH. Data are represented as mean ± SEM; n = 4/group. Indicated P values were calculated by ANOVA test.
Effect of PLP on H2S Levels and Markers of Oxidative Stress in the Blood Plasma of Old Animals
In our experiments, H2S levels in the blood plasma of old animals were significantly lower (1.69-fold, P < .05) compared to its level in adult rats (Table 2). Administration of PLP not only restored H2S levels in aged rats to adult levels but significantly increased them, by 2.54-fold (P < .05). At the same time, indicators of oxidative stress in the blood plasma of old rats were significantly higher than in adult rats, indicating an increase in free radical processes (Table 2). In particular, the rate of generation of •O2− in the blood plasma of old rats was almost 3 times higher (P < .05) than in adult animals. PLP treatment of aged rats significantly reduced the rate of superoxide radical generation by 3.87-fold (P < .05), which was even lower than in adult rats (Table 2). A 3.65-fold (P < .05) significant increase in H2O2 levels was observed in old rats (Table 2). However, PLP treatment reduced these indicators by 4.5 times (P < .05). Changes in the levels of DC and MDA, which are products of nonenzymatic lipid peroxidation, may also indicate the limitation of oxidative stress in aged animals treated with PLP. The levels of DC and MDA in the blood plasma of old animals were significantly higher (by 6.53 and 2 times, respectively) compared to their levels in adult rats. At the same time, PLP treatment of old rats reduced the blood plasma levels of both the final and intermediate products of lipid peroxidation, MDA and DC, to their levels in adult animals.
Hydrogen Sulfide Levels and Oxidative Stress Markers in the Blood Plasma of Old Rats After a Course Administration of PLP (Mean ± SEM).
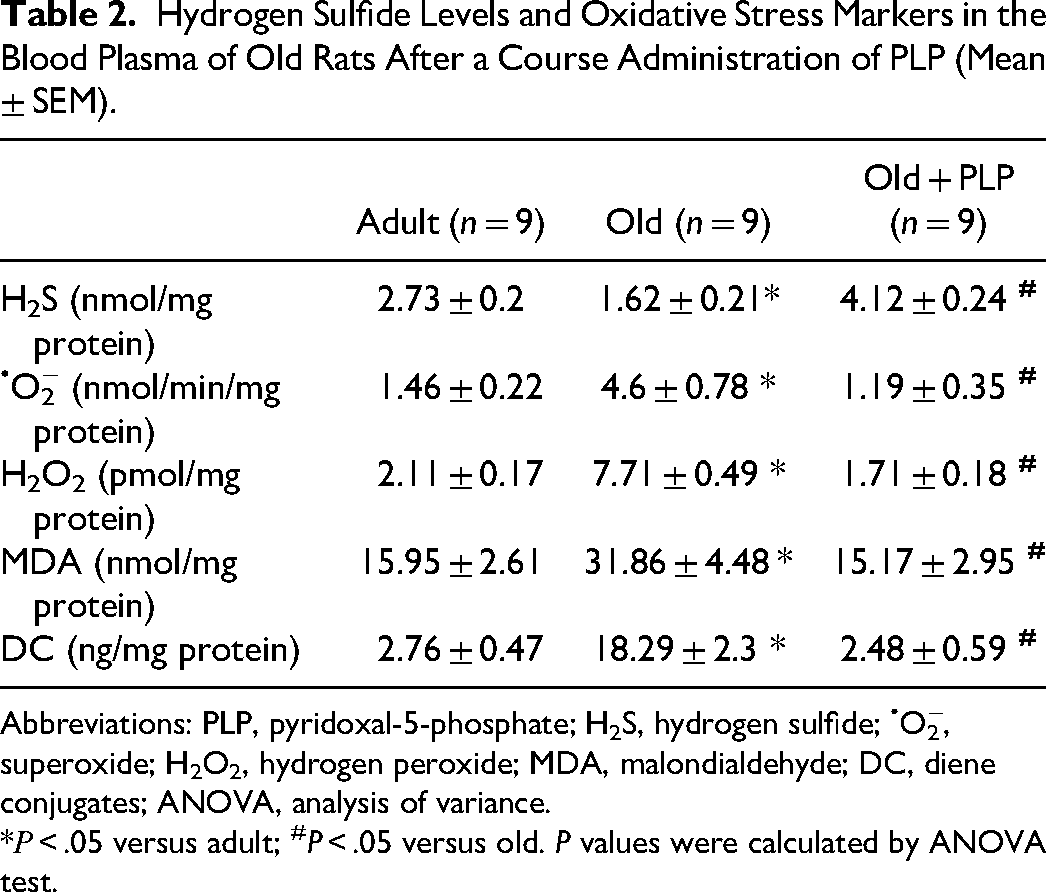
Abbreviations: PLP, pyridoxal-5-phosphate; H2S, hydrogen sulfide; •O2−, superoxide; H2O2, hydrogen peroxide; MDA, malondialdehyde; DC, diene conjugates; ANOVA, analysis of variance.
*Р < .05 versus adult; #P < .05 versus old. P values were calculated by ANOVA test.
Discussion
Our data show that PLP treatment of aged rats reduces cardiac fibrosis and improves cardiac function during ischemia-reperfusion, promotes vasorelaxation responses, and reduces oxidative stress. At the same time, the expression levels of the SUR2 and Kir6.1 subunits of KATP channels and H2S production increase in these animals. The administration of an inhibitor of KATP channels—glibenclamide, prevented the increase in cardioprotective and vasorelaxation effects in these animals, demonstrating the association of these effects with the raised up-expression of KATP channels. In particular, measurement of relaxation responses of aortic rings from old rats in response to KATP channels opening by flocalin showed that the amplitude of these responses was significantly greater in PLP-treated rats than in untreated rats, which was associated with increased expression levels of both mRNA and protein of Kir6.1 pore-forming subunits of vascular-type KATP channels in PLP-treated old rats (Figures 3a and 5a). At the same time, in another series of experiments, we showed that neither the expression of Kir6.1 nor the vasodilatory responses were significantly different in PLP-treated and untreated adult animals (Figure 3b). Therefore, such differences in the vasodilatory responses of adults and old PLP-treated rats in response to the action of the pharmacological opener of KATP channels flocalin are associated with a level divergence in the expression of Kir6.1 in these animals. The increased vasodilator function of old PLP-treated rats may be explained not only by the increased expression of Kir6.1, but also by the high expression of SUR2, since their high levels in the aorta create the conditions for increased formation of vascular Kir6.1/SUR2B complexes. However, it is currently unknown whether PLP increases directly or indirectly the up-regulation of the expression of these KATP channel subunits, as it is a cofactor of H2S-synthesizing enzymes and increases H2S production. At the same time, as shown by Sun et al, 16 H2S donors can increase the expression of SUR2B and Kir6.1 subunits at their reduced expression levels in spontaneously hypertensive rats, which lowered blood pressure in these animals and was accompanied by an enhancement in the vasodilatory effects of the pinacidil, KATP channel opener, in vitro experiments, similar to our experiments. In general, the vascular effects of KATP channel opening during ischemia-reperfusion make a significant contribution to cardioprotection, associated with both the prevention of reperfusion-induced increases in coronary vascular resistance and a moderate reduction in blood pressure in in vivo experiments. 31
In general, the enhancement of vasodilator responses in PLP-treated animals may occur via multiple signaling pathways, including known gas transmitters such as H2S and nitric oxide (NO). In particular, hydrogen sulfide can directly open the KATP channel by sulfhydration of the channel and can interact with the NO system, significantly enhancing NO signaling, as well as is probably able to act as an endothelium-derived hyperpolarizing factor.32–34 A schematic representation of the H2S signaling pathways with possible involvement of the NO system and KATP channels leading to vasorelaxation and cardioprotection upon administration of PLP is shown in Figure 6.

Schematic representation of the H2S signaling pathways with possible involvement of NO, cGMP-dependent protein kinase (PKG), and KATP channels leading to cardioprotection and vasorelaxation upon administration of PLP in vivo.
There are several pathways by which H2S affects the NO system. 34 First of all, H2S can stimulate the release of NO from its stable pools, in particular nitrite. A powerful mechanism of H2S action is an increase in NO synthesis by endothelial NO synthase (eNOS). 34 The activity of eNOS can be modulated by acylation, nitrosylation, phosphorylation, acetylation, glycosylation, and glutathionylation of the enzyme. 35 H2S donors increased NO production by eNOS through mobilization of Ca2+ from the endoplasmic reticulum (ER), activation of the Ca2+–calmodulin system, phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) signaling pathway, and Akt-, AMP-stimulated protein kinase (AMPK) or p38 mitogen-activated protein kinase (p38 MAPK)-dependent phosphorylation of Ser1177 of the eNOS molecule.34,36,37 Consequently, these signaling pathways may increase vascular NO generation by hydrogen sulfide and enhance vascular relaxation. In the case of inhibition of eNOS activity, the cardioprotective effects of H2S are significantly reduced, indicating the importance of the interaction between H2S and NO, as well as their common signaling pathways. Another level of interaction between H2S and the NO system occurs through direct sulfhydration of eNOS cysteines by H2S, which stabilizes the enzyme in its physiological, dimeric state and optimizes NO production. 34 In smooth muscle cells, NO activates soluble guanylate cyclase (sGC), increases 3′,5′-cyclic guanosine monophosphate (cGMP) levels, and activates cGMP-dependent protein kinase (PKG). It is believed that NO signaling is mainly mediated by cGMP, which is synthesized by NO-activated guanylyl cyclases and is cleaved by cyclic nucleotide phosphodiesterases. 38 PKG activated by cGMP phosphorylates serines and threonines on cellular proteins, which can induce changes in their activity, function, or cytoplasmic localization. 38 Typically, these modified PKGs regulate the calcium homeostasis or calcium sensitivity of cellular proteins, the function of vascular smooth muscle and the heart, and more. 38 PKG has a broad spectrum of actions and can induce vasorelaxation and cardioprotection through multiple signaling pathways. In particular, by direct phosphorylation and opening of the KATP channels, and as a result, a decrease in Ca2+ entry into the cell due to the inhibition of Ca2+ channels L-type. In general, PKG can reduce cytoplasmic Ca2+ and reduce protein sensitivity to it through a number of signaling pathways, both by modulating the activity of Ca2+ channels and receptors. In particular, PKG phosphorylates inositol triphosphate receptor, which decreases Ca2+ release from IP3-receptive storehouses, inhibits Ca2+ channels L-type, and phosphorylates transient receptor potential Ca2+ channel isoforms 3 and 6, which decreases Ca2+ influx into cells. In cardiac muscle, phospholamban signaling, which increases Ca2+ uptake by Serca (sarco(endo)plasmic reticulum Ca2+-ATPase), troponin I signaling that desensitizes protein to Ca2+ and some other ways, can be added to these mechanisms of action of PKG. 38 Moreover, H2S can stabilize sGC in a physiological, NO-sensitive, reduced form and can inhibit vascular cGMP phosphodiesterase (PDE5), thus increasing the period of existence of cGMP and therefore enhancing vasodilatation. 34 Polysulfides formed from H2S can also directly activate PKG. 34 All these signaling pathways of H2S and NO are also related to cardioprotection. Regarding the H2S/KATP channel signaling pathway, as already mentioned, H2S can directly activate KATP channels by sulfhydration of the channel and can cause activation of KATP channels via NO signaling at the cGMP site via PKG. In addition, polysulfides formed from H2S can also directly activate PKG with subsequent activation of KATP channels. Thus, H2S is an important endogenous enhancer of NO signaling and both of them can activate KATP channels contributing to both vasorelaxation and cardioprotection. In our previous work, 39 in aged PLP-treated animals, H2S levels in aortic tissue were significantly increased compared to untreated animals and were associated with increased eNOS activity and NO levels in these animals. This may be additional evidence of an increase in H2S-induced NO production by eNOS in old animals after the administration of PLP. At the same time, in old PLP-treated animals compared to old untreated rats, the amplitude of relaxation of isolated aortic preparations in response to acetylcholine has significantly increased. Endothelium-dependent responses of vascular relaxation to acetylcholine in old animals were restored by PLP to the level in adult rats and prevented by NO inhibition with N-nitro-l-arginine methyl ester hydrochloride. 39 This indicates the participation of NO in these processes of vasorelaxation.
Although the mRNA expression of Kir6.2 pore-forming subunits of cardiac-type sarcolemmal KATP channels did not change significantly in PLP-treated old rats compared to untreated animals (Figure 4b), they had a strong cardioprotective effect in experiments with ischemia-reperfusion of isolated Langendorff-perfused hearts (Figure 1). However, it is known that increased expression of the SUR2A subunit of the cardiac-specific KATP channels is critical for enhanced anti-ischemic protection, whereas expression levels of Kir6.2 subunits may remain unchanged.14,20,21 In our experiments, PLP administration significantly increased SUR2 expression levels, whereas Kir6.2 expression levels were already elevated in old rats (Figure 4b and d). It is likely that increased production of H2S and NO (generated by eNOS), increased signaling between them, and activation of KATP channels as one of the end effects of these gas transmitters may contribute to the enhancement of cardioprotection in PLP-treated old animals. The reperfusion recovery of the contractile function of the ischemic heart in PLP-treated old animals may be significantly influenced by significantly less cardiac fibrosis (Figure 2). In general, fibrosis is a universal age-related disease that affects almost all organs. 40 It is possible that H2S, NO, and KATP channels may be involved in the reduction of cardiac fibrosis in PLP-treated rats. In particular, the reduction of cardiac fibrosis after administration of exogenous H2S can occur via various signaling pathways, including glycogen synthase kinase-3β (GSK-3β)/β-catenin pathway, cGMP/PKG pathway, nuclear factor E2-related factor 2 (Nrf2) signaling pathway, adiponectin-AMPK signaling, an increase of NO bioavailability, opening of KATP channels and decrease of oxidative stress, inhibition of mitochondrial permeability transition pore (mPTP) opening in the aging cardiomyocytes, ER stress via sirtuin 1, AMPK-mediated Akt/GSK-3β/hypoxia-inducible factor-1α activation, and some others. 41
The degree of anti-ischemic protection of the myocardium depends on the type of activated KATP channels in the plasma membrane (sarcKATP) and mitochondrial (mitoKATP) membranes of cardiomyocytes, smooth muscle, and endothelial vascular cells. The protective mechanisms induced by the opening of sarcKATP and mitoKATP channels complement each other and can interact through a number of signaling pathways (in particular protein kinases and reactive oxygen species [ROS]). The essence of the anti-ischemic protection of sarcKATP opening is that the shift of the membrane potential toward hyperpolarization reduces the duration of the cardiomyocyte action potential, during which Ca2+ influx the cell, in particular through L-type Ca2+ channels, and consequently reduces the intracellular level of calcium, which is one of the main secondary mediators of numerous metabolic pathways and an activator of many enzymes. This should slow down the metabolic processes in the cell and reduce its oxygen demand, as well as lead to the saving of energy materials, which is very important in ischemia. The inhibition of Ca2+ entry into the cell should reduce the activity of enzymes and prevent the degradation of membrane phospholipids by phospholipases, which is a membrane-stabilizing effect of opening these channels. 42 Preventing cardiomyocyte overload with calcium ions averts necrotic damage to the myocardium due to contractures of myofibrillar. 43 Activation of sarcKATP channels in coronary smooth muscle and endothelial cells dilates these vessels increasing the flow of blood, oxygen, and energy resources to the myocardium and preventing reperfusion vasoconstriction in ischemic myocardium. The protective consequences of opening these channels can also be considered a moderate decrease in blood pressure, which reduces the load on the ischemic heart and the relative preservation of myocardial contractility during reperfusion. 6
An excessive increase of Ca2+ ions and ROS in the mitochondrial matrix during ischemia and especially during reperfusion of the ischemic myocardium leads to an increase in the permeability of mitochondrial membranes and the formation of mPTP. An increase in this permeability leads to impaired oxidative phosphorylation and decreased ATP synthesis, as well as the release of pro-apoptotic compounds (in particular cytochrome c, BID, BAK, BAX, and BAD) from the mitochondria and a decrease in anti-apoptotic compounds (Bcl-2 and Bcl-xl), which, together with caspase signaling cascades, cause cardiomyocyte apoptosis. 44 The opening of mitoKATP channels is accompanied by the entry of potassium into the matrix, depolarization of the inner mitochondrial membrane, and inhibition of the potential-dependent calcium uniporter. This prevents excessive accumulation of calcium in the mitochondria, enhances their resistance to this ion, and prevents mPTP opening.45–47 Signaling pathway: mitoKATP channels opening/increasing ROS/activation of protein kinase Cε2 also prevents mPTP opening and apoptosis. 48 The entry of potassium ions into the mitochondrion at the opening of mitoKATP channels is accompanied by the entry of inorganic phosphate and water (which leads to moderate osmotic swelling of the mitochondrial matrix), changes in respiration and phosphorylation processes, and alkalosis of the mitochondrial matrix.45,49 Activation of these channels inhibits ATP hydrolysis, stimulates respiration and potential reduction on NAD-dependent substrates, and slightly inhibits respiration–phosphorylation coupling.42,45 Reduced ATP hydrolysis during ischemia protects adenine nucleotides by delaying their degradation, thereby conserving ADP for a normal reperfusion response. 49 The compensatory increase in proton pumping and oxygen consumption (respiration), when mitoKATP channels are opened, is caused by energy dissipation not related to ATP production (uncoupling). However, experiments with various openers of these channels showed that mitochondrial respiration depends on the experimental conditions and the energy state of the cell.40,50 It is likely that the cardioprotective role of moderate swelling during ischemia is due to the preservation of contact sites between the outer and inner membranes in the optimal orientation for ADP entry into the mitochondrial matrix and the preservation of the interaction of transport systems. In particular, nucleotide transport into the mitochondrial matrix is maintained through the outer membrane potential-dependent anion channel, the intermembrane space (Mi-CK), and ADP/ATP translocases located on the inner membrane. 42 During ischemia, activation of mitoKATP channels supports efficient energy transfer between mitochondria and cellular ATPases and protects ADP from deamination. 51 Thus, mitoKATP channels opening contributes to the preservation of mitochondrial function during ischemia-reperfusion of the heart and prevents cardiomyocyte apoptosis and necrosis.
In our experiments, the levels of oxidative stress indicators were significantly raised in old animals compared to adults, which may indicate an age-related increase in free radical processes. However, in PLP-treated old animals, markers of oxidative stress were reduced to the levels of adult rats. Of course, on the basis of the data obtained, 2 antioxidant mechanisms can be distinguished in our case, in particular, the increased expression of KATP channels and the increased production of H2S, since their antioxidant properties are well known.52,53 Finally, the H2S molecule has strong reducing properties, so it can have antioxidant properties through the quenching of ROS and active forms of nitrogen, as well as protein sulfhydration. Hydrogen sulfide can also modulate cellular levels of a powerful antioxidant such as glutathione and upregulate the expression of antioxidant enzymes. 54 Several mechanisms are involved in the reduction of oxidative stress during the activation of KATP channels, but the most obvious is the reduction of calcium entry into the cell and the suppression of metabolic processes, particularly calcium-dependent ones.53,55 Therefore, another powerful mechanism of PLP administration to old rats is a significant reduction in oxidative stress due to an increase in hydrogen sulfide production and up-expression of KATP channels.
Therefore, PLP treatment of old animals significantly increases the mRNA and protein expression of Kir6.1 subunits of vascular-type KATP channels, which is associated with an increase in vasodilatory responses to the pharmacological opening of KATP channels, and significantly increases the mRNA and protein expression of regulatory SUR2 subunits that may be associated with better recovery of function of the ischemic heart during reperfusion. The improved cardiac function in PLP-treated old animals compared to untreated rats could be due to significantly less cardiac fibrosis and significantly reduced indicators of oxidative stress, which could probably be contributed by both increased expression of KATP channels and H2S production. Therefore, the aforementioned mechanisms, induced by PLP in old rats, may be used for therapeutic protection in the future, especially considering that under physiological conditions KATP channels remain mostly in a closed state. Optimism for the development of such a promising cardioprotective strategy is further boosted by data showing that overexpression of SUR2A has no adverse effects, at least on heart rate during exercise and on diurnal heart rate variation. 56
Conclusions
Thus, PLP administration reduces cardiac fibrosis and improves heart function during ischemia-reperfusion, promotes vasorelaxation responses, and decreases oxidative stress in old rats. We suppose this potential therapeutical effect may be a result of increased expression of KATP channels and H2S production.
Footnotes
Acknowledgments
The figures were partly generated using Servier Medical Art, provided by Servier, licensed under a Creative Commons Attribution 3.0 unported license.
Author Contributions
RBS, NAS, OOP, PD and VFS conceptualized the study, planned the experiments, and discussed the results. RBS, NAS, OOP, PD and VFS manuscript writing. RBS, NAS, OOP, LAM, YVG, RAF, IYO, and VRS carried out the experiments. RBS, NAS, OOP, LAM and RAF formal analysis and figures preparation. RBS, OOP, PD and VFS resources, funding acquisitions and project administration. All authors participated in the design of the study and interpretation and analysis of the data and reviewed of the manuscript.
Data Availability Statement
Data supporting the findings of this study are available in the Methods and Results sections of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Ukraine, the National Science Centre of Poland, and Bogomoletz Institute of Physiology, the NAS of Ukraine. Ruslan B. Strutynskyi was supported by the National Research Foundation of Ukraine (grant number 2020.01/0204); Vadym F. Sagach was supported by the National Research Foundation of Ukraine (grant number 2021.01/0210); Oksana O. Piven was supported by the National Science Centre, Poland (grant number UMO-2022/01/3/NZ4/00010) and FEBS Ukrainian Short-Term Fellowship; Pawel Dobrzyn was supported by grants from the National Science Centre, Poland (grant numbers UMO-2014/13/B/NZ4/00199 and UMO-2016/22/E/NZ4/00650).
