Abstract
Testosterone is an anabolic steroid hormone, which is the major circulating androgen hormone in males. Testosterone levels decreasing below the normal physiological levels lead to a status known as androgen deficiency. Androgen deficiency has been shown to be a major risk factor in the development of several disorders, including obesity, metabolic syndrome, and ischemic heart disease. In the past decades, although several studies from animal models as well as clinical studies demonstrated that testosterone exerted cardioprotection, particularly during ischemia–reperfusion (I/R) injury, other preclinical and clinical studies have shown an inverse relationship between testosterone levels and cardioprotective effects. As a result, the effects of testosterone replacement on the heart remain controversial. In this review, reports regarding the roles of testosterone replacement in the heart following I/R injury are comprehensively summarized and discussed. At present, it may be concluded that chronic testosterone replacement at a physiological dose demonstrated cardioprotective effects, whereas acute testosterone replacement can cause adverse effects in the I/R heart.
Introduction
Testosterone (or androgen) deficiency is a condition in which the concentration of testosterone hormone is lower than the normal physiological level. 1,2 The primary cause of testosterone deficiency is due to a testicular disorder or an abnormal function of the hypothalamus–pituitary axis, while a secondary cause or transient testosterone deficiency is caused by the effect of drugs; aging; acute or chronic diseases; or chronic comorbidities such as obesity, metabolic syndrome, dyslipidemia, and insulin resistance. 1,3 –5
The prevalence of symptomatic testosterone deficiency is approximately 481 000 new cases per year in US men aged between 40 and 69 years 2 and 5.6% in men aged 30 to 79 years. 6 Another study reported a higher prevalence of hypogonadism (38.7%) in men aged ≥45 years (mean age = 60 years) visiting primary care practices in the United States. 7 The prevalence of hypogonadism also increases substantially with age 6,8 in which the prevalence increased to 20% of men older than 60 years, 30% older than 70 years, and 50% older than 80 years. 9 Although this condition commonly occurs in middle- to older aged men, it can also be found in younger men. 1,2,6,8 In addition, lower levels of serum testosterone are associated with the development of several disorders including obesity, dyslipidemia, insulin resistance, metabolic syndrome, cardiovascular disease, and coronary artery disease (ischemic heart disease). 5,10 –13
Previous studies reported that men have a higher incidence, mortality, and risk of coronary artery disease than women 14 –16 and the risk of coronary artery disease increases markedly with age. 16 In addition, patients with coronary artery disease 17 –20 and congestive heart failure 21 have been shown to have lower levels of endogenous testosterone compared with healthy men, and the severity of both diseases shows a strong correlation with the degree of testosterone deficiency. 5,17,22 It has also been suggested that low serum testosterone levels could be used as a predictive marker for cardiovascular disease in men. 23
Although there are many reports indicating that testosterone provided cardioprotective effects 24 –26 such as antiarrhythmic properties, 27 reduced infarct size, 28 enhanced vasodilatation, 29 decreased atherosclerosis, 30 and improved metabolic parameters, 31 these findings are controversial at this time. 32 –36 Moreover, when it comes to the role of testosterone in the ischemic heart or cardiac ischemia–reperfusion (I/R) injury, limited data are currently available. Despite limited data, inconsistent reports exist regarding the effects of testosterone in I/R hearts. In this review, the roles of testosterone in the heart following I/R injury are comprehensively summarized and discussed. The consistent findings, as well as controversial results, are also presented, focusing on the effects of testosterone on the heart during an I/R injury.
Physiological Roles of Testosterone in the Heart
The cardiac muscle is one target tissue of the testosterone hormone. 37,38 It is well known that the level of testosterone is associated with effects on the cardiomyocytes such as hypertrophy and cardiomyocyte apoptosis 39,40 and is also associated with the contractility of cardiac muscle. 38,41 –43 Previous studies reported the beneficial effect of testosterone at physiological levels on cardiovascular function by inducing vasorelaxation of vascular smooth muscle. 39,44,45 Testosterone controlled cardiac relaxation by acting mainly through sarcoendoplasmic reticulum Ca2+-ATPase (SERCA), 46 increasing or regulating cardiac contractility, and Ca2+ handling in the heart. 38,43,47 However, conflicting evidence was that chronic administration of a synthetic testosterone (nandrolone decanoate) decreased left ventricular myocardial function and thus overall cardiac performance without altering contractility in rats. 48
The physiological effects of testosterone on cardiac electrophysiology have been reported. It has been shown that the Q-T interval was shorter in men than in women. 49 –52 Moreover, both endogenous and exogenous testosterone could shorten or decrease the dispersion of the Q-T interval, 50,53,54 whereas a contrasting study found that the testosterone level and the Q-T interval on electrocardiogram did not have any relationship with each other. 55 Furthermore, beneficial effects of testosterone on the ST-segment and atrial fibrillation have been reported. Testosterone was shown to modulate the early phase of ventricular repolarization resulting in decreasing the ST-segment. 56,57 Other studies reported that aging men with decreased serum testosterone levels had a high incidence of atrial fibrillation. 58,59 These were consistent with the study using a rat model, which reported that deficiency of testosterone was arrhythmogenic in rat atria possibly through less binding of FK506-binding protein (FKBP12.6) to ryanodine receptor type 2 (RyR2), resulting in calcium leakage from the sarcoendoplasmic reticulum. 60 Testosterone also plays an essential role in the Ca2+ regulation in male hearts in both genomic and nongenomic pathways. 46,61 –63 It has been shown that testosterone maintained or increased the expression of dihydropyridine receptor (α1c-subunit of L-type calcium channel) messenger RNA (mRNA) level and also increased the Na+/Ca2+ exchanger mRNA level, 62,64,65 improved the cardiac L-type calcium current, 62,63 increased Ca2+ released via the ryanodine receptor, 61 maintained FK506-binding protein 12.6 in ryanodine receptor type 2, 60 maintained or increased SERCA and Na+/Ca2+ exchanger activity, 46,61 and maintained phosphorylation of phospholamban Thr17, 46 thus resulting in faster Ca2+ removal out of the cytoplasm.
Testosterone and the Pathological Heart
Previous studies demonstrated that a reduction in plasma testosterone levels can be either the direct cause or the consequence of several cardiovascular diseases such as hypertension, diabetes, atherosclerosis, and heart failure. 23,66 –73 There is also a correlation between low levels of testosterone and the severity of each of these diseases. 71 –74 The incidence of chronic heart failure due to idiopathic dilated cardiomyopathy is associated with a significant decrease in testosterone concentrations, 74 in which androgen replacement therapy could improve heart failure symptoms. 75 Several lines of evidence indicate that testosterone may effectively provide protective effects in the cardiovascular system 76,77 and that decreased circulating testosterone may increase cardiovascular risk, atherosclerosis, and mortality in men. 76,77 Testosterone therapy has also been shown to improve the cardiac ischemic condition, comparable to the effects of existing antianginal drugs but has no beneficial effects in peripheral arterial and cerebrovascular diseases. 76 On the other hand, several conflicting studies have shown that testosterone replacement also increases the incidence of cardiovascular disease. 33,67,76,78 However, a recent review by Morgentaler et al clarified these controversial studies and concluded that low levels of total testosterone, bioavailable testosterone, and free testosterone are associated with an increased risk and severity of coronary artery disease, whereas high serum testosterone concentrations (endogenous or via testosterone therapy) markedly reduced cardiovascular disease and cardiovascular risk factors. 79 Furthermore, Morgentaler et al also revealed that testosterone therapy is associated with a reduction in obesity, fat mass, metabolic parameters, and inflammatory markers. 79
Testosterone and Cardiac I/R Injury
Testosterone deficiency plays a role in the development of ischemic heart disease. 19,76,77 Men with heart disease or ischemic heart disease had lower levels of testosterone than men with normal coronary angiograms 18 and that testosterone improved ischemic tolerance and quality of life in hypogonadal men with angina. 80,81 However, inconsistent reports exist regarding the beneficial effects of testosterone in the heart, 32,33 and the questions remain whether it does truly provide cardioprotective benefits. 24,25 Currently, there are only a few studies investigating the roles of testosterone in the myocardial infarction model and in the I/R heart. In spite of limited data available, the effects of testosterone on several crucial parameters in the myocardial infarction as well as the I/R heart including infarct size, lactate dehydrogenase (LDH) release, myocyte viability, left ventricular contractile function, arrhythmias, and heart rate variability (HRV) have been reported with inconsistent findings. In this review, the effects of testosterone on each of these crucial parameters in the myocardial infarction and I/R heart are comprehensively summarized and discussed.
Effect of testosterone on the infarct size in I/R hearts
Testosterone has been shown to have an infarct limiting effect in ex vivo and in vivo models of orchiectomized (ORX) rats with I/R injury. These reports are summarized in Table 1. In the Langendorff isolated heart, a physiological dose of testosterone 2 mg/kg body weight has been shown to decrease the infarct size in I/R heart by either the reduction in myocardial apoptosis through α1-adrenoceptor stimulation 28 or the promotion of neovascularization of heart tissues in myocardial infarction model through increased synthesis of 70-kDa heat shock proteins (Hsp70s) 25 and activated κ-opioid receptor agonist. 82 Consistently, a recent in vivo model study in ORX rat’s heart demonstrated that acute injection of testosterone prior to the reperfusion period could reduce the infarct size. 83 However, the supraphysiological dose of testosterone replacement (500 and 50 mg/kg) in the myocardial infarction and I/R models exerted neither adverse effect nor infarct size reduction benefits. 84,85
Summary of the Effect of Testosterone on the Infarct Size in I/R Heart.

Abbreviations: ORX rat, orchiectomized rat; MI, myocardial infarction; LAD, left anterior descending coronary artery; I/R, ischemic and reperfusion; IV, intravenous; MRI, magnetic resonance imaging; IM, intramuscular; SC, subcutaneous; IEI, infarct expansion index.
It is of interest to note from these findings that physiological levels or low doses of testosterone may provide a cardioprotective effect against I/R injury. Most of the positive effects of testosterone on the infarct size in I/R models also occurred in the combination of testosterone supplement and conditional treatments, including I/R preconditioning and adrenergic receptor stimulation. The underlying mechanisms responsible for these findings need to be investigated in future studies.
Effect of testosterone on cardiac function in I/R hearts
Testosterone exerts a beneficial effect on cardiac function in ex vivo models of ORX rats with I/R injury and in vivo models of ORX rats with myocardial infarction (Table 2). The beneficial effects of testosterone in the ex vivo model using the Langendorff isolated heart preparation technique demonstrates that a physiological dose of testosterone (2 mg/kg) combined with I/R preconditioning, 82 and adrenergic receptor stimulation 28 improves contractile recovery during the I/R period. The postulated mechanism of this effect was through α1-adrenoceptor stimulation 28 and increased synthesis of Hsp70 via a κ-pioid receptor agonist. 82 Interestingly, supraphysiological doses of both testosterone and its metabolite (5α-dihydrotestosterone [DHT]) also exhibited a beneficial effect on cardiac function during I/R injury by attenuating intracellular Ca2+ overload during the reperfusion period, 24,86 whereas the supraphysiological dose of testosterone replacement (50 mg/kg) in another I/R model exerted neither adverse nor beneficial effect on cardiac function. 85 This suggests that testosterone plays a cardioprotective role by reducing intracellular acidification resulting in the maintenance of physiological cytosolic Ca2+ levels during ischemic stress. 86 In addition, echocardiographic study also demonstrated that testosterone replacement therapy (2 mg/kg) could improve myocardial performance in a myocardial infarction model. 25
Summary of the Beneficial Effect of Testosterone on Cardiac Function in I/R Heart.

Abbreviations: ORX rat, orchiectomized rat; LAD, left anterior descending coronary artery; SC, subcutaneous; I/R, ischemic and reperfusion; IV, intravenous; IM, intramuscular; LVDP, left ventricular develop pressure; LVEDP, left ventricular end diastolic pressure; MI, myocardial infarction; ARs, androgen receptors; EF, ejection fraction; FS, fractional shortening.
Inconsistent findings of the effect of testosterone on cardiac contractile function have been reported in ex vivo models of ORX rats with I/R injury. These reports are summarized in Table 3. The study using Langendorff isolated hearts reported that chronic exogenous testosterone (subcutaneous implantation of DHT 200 mg) neither improved nor worsened myocardial functional recovery after I/R injury. 87 Acute infusion of 17β-hydroxy-4-androsterone at a physiological level (10 ng/mL/min) in isolated hearts could depress the recovery of myocardial function during I/R injury 36 by inducing hypertrophic response in the heart via androgen receptors, resulting in an increase in ventricular stiffness. 36 There is a report that the androgen receptor blocker flutamide could improve cardiac contractile function during cardiac I/R injury. 88 However, this study was carried out in intact animals without testosterone deprivation. These findings suggest that endogenous testosterone may also act via other mechanisms instead of just the genomic effect on cardiac function during I/R injury. 38,89 This could be due to the nongenomic effects that are similar to other steroids, 89 which could bind to specific membrane binding sites and stimulate the secondary messenger signaling cascades, protein kinases (A and C), mitogen-activated protein kinase (MAPK), adenylyl cyclase, and intracellular Ca2+. 39,89 –91 These alterations in the nongenomic effect could induce various cellular outcomes including a rapid rises in the intracellular Ca2+ concentration, 44,45,92 neuromuscular or junctional signal transduction, 39,44 and vasorelaxation 39,45 and could be responsible for cardioprotection.
Summary of the Adverse or Neutral Effects of Testosterone on Cardiac Function in I/R Heart.

Abbreviations: ORX rat, orchiectomized rat; MI, myocardial infarction; LAD, left anterior descending coronary artery; LV, left ventricular; I/R, ischemic and reperfusion; SC, subcutaneous; EF, ejection fraction; LVDP, left ventricular develop pressure; LVEDP, left ventricular end diastolic pressure.
The importance of endogenous testosterone is emphasized by many reports of androgen deprivation therapy in patients with prostate cancer, which demonstrated that androgen deprivation therapy could increase cardiovascular events such as increased risk of ischemic heart disease, 93,94 induction of metabolic syndrome, 93 –96 increased fat mass and decreased lean mass, 97,98 and increased atherosclerosis in animal models. 30 However, there are a few clinical studies in patients with prostate cancer, demonstrating that androgen deprivation therapy did not have any correlation with cardiovascular disease and mortality. 99 –102 The discrepancies in these clinical findings could be due to differences in study design and populations, selection bias in men treated with Androgen suppression therapy (AST), and confounding factors such as comorbidity or lifestyle. However, on the basis of the current data, lacking of endogenous testosterone or androgen suppression may be associated with increased risks of cardiovascular diseases.
The discrepancies in these findings regarding the role of testosterone in cardiac function during I/R could be due to differences in the experimental model, an addition of conditional treatments, and dosages of testosterone treatment (physiological or supraphysiological dosage). Moreover, it is interesting that most reports showing the positive effects of testosterone used similar durations of treatment (chronic treatment for 2-8 weeks), whereas acute treatment of testosterone (infusion 5 minutes before ischemia) in myocardial infarction model resulted in decreased cardiac function. Further studies are needed to elucidate the mechanisms responsible for these effects on cardiac function.
Effect of testosterone on LDH release in I/R hearts
Testosterone has been shown to exert beneficial effects in ex vivo models of ORX rats with I/R injury (Table 4). These studies using the Langendorff isolated hearts demonstrated that both a physiological dose (2 mg/kg) or supraphysiological dose of testosterone could decrease LDH release in the I/R heart, 24,28,82 and the proposed mechanisms could be via either α1-adrenoceptor stimulation 28 or increased synthesis of Hsp70s. 82
Summary of the Effect of Testosterone on LDH Released in I/R Heart.

Abbreviations: ORX rat, orchiectomized rat; I/R, ischemic and reperfusion; SC, subcutaneous; LDH, lactate dehydrogenase; LV, left ventricular; ARs, androgen receptors; LAD, left anterior descending coronary artery.
Effect of testosterone on myocyte viability in I/R hearts
Testosterone has been shown to have beneficial effects on cell viability in ORX rats with I/R injury and ORX rats with the myocardial infarction model (Table 5). Recent in vitro studies using the isolated ventricular myocytes demonstrated that a physiological concentration of testosterone combined with I/R preconditioning 82 and adrenergic stimulation 28 could increase ventricular myocyte viability in the I/R model. Testosterone could work through the angiogenesis pathway 82 and α1-adrenoceptor stimulation, which are mediated by both androgen receptors and nongenomic actions. 28 A physiological dose of testosterone (2 mg/kg) has also been shown to decrease the apoptotic cells in the myocardial infarction model, and these beneficial effects are thought to occur through the nongenomic pathway. 25
Summary of the Effect of Testosterone on Cardiomyocyte Viability in I/R Heart.

Abbreviations: ORX rat, orchiectomized rat; MI, myocardial infarction; LAD, left anterior descending coronary artery; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; I/R, ischemic and reperfusion; SC, subcutaneous.
Effect of testosterone on molecular signaling in I/R hearts
Most of the molecular studies on testosterone replacement demonstrated adverse effects in an ex vivo model of ORX rats with I/R injury (Table 6). In Langendorff isolated hearts of ORX rats, acute exogenous testosterone replacement at a physiological level had damaging effects on the myocardium after I/R injury by the inhibition of signal transducers and activators of transcription 3 (STAT-3) and the suppression of the cytokine signaling 3 (SOCS-3) antiapoptotic pathway. 32 In addition, acute testosterone infusion to physiological level in an isolated heart also increased the activation of the p38 MAPK and c-Jun N-terminal kinase pathways. These molecular signalings were involved with myocardial inflammation, 36 increased apoptotic protein caspase 3, 36 decreased antiapoptotic protein BcI-2, 36,103 and Forkhead box O3a phosphorylation (p-FOXO3a), 103 and downregulation of PI3K/Akt antiapoptotic pathways, resulting in reduced phosphorylation of Bcl-2-associated death promoter protein (Bad) and unable to inhibit Bax-triggered apoptosis. 103 Consistent with these results, blocking of androgen receptor in an I/R injury model using the Langendorff isolated heart resulted in decreased proinflammatory cytokine production, decreased p38-MAPK and caspase 1, decreasing the expression of apoptotic protein caspases 3 and 11, and increasing the antiapoptotic protein Bcl-2. 88
Summary of the effect of Testosterone on Molecular Parameters in I/R Heart.

Abbreviation: ORX rat, orchiectomized rat; MI, myocardial infarction; LAD, left anterior descending coronary artery; SC, subcutaneous; I/R. ischemic and reperfusion; HIF-1α, hypoxia-inducible factor 1-α; SDF-1α, stromal cell-derived factor 1α; VEGF, vascular endothelial growth factor; STAT-3, signal transducer and activator of transcription 3; SOCS-3, suppressor of cytokine signaling 3; mRNA, messenger RNA; p38 MAPK, p38 mitogen-activated protein kinase; JNK, c-Jun N-terminal kinases; Bcl-2, B-cell lymphoma 2; TNF-α, tumor necrosis factor α; IL-1β, interleukin 1β; IL-6, interleukin 6; Akt, protein kinase B; p-Bad, The Bcl-2-associated death promoter; FOXO3a, Forkhead box O3; Bax, bcl-2-like protein 4; Fas-L, Fas ligand.
In contrast to findings following acute testosterone administration, a study using chronic exogenous testosterone replacement demonstrated improved cardiac function and restored the activation of the myocardial PI3K/Akt pathway including decreased proapoptotic (Bax and Fas-L protein) and increased antiapoptotic proteins (Bcl-2, p-Bad, and p-FOXO3a) in this isolated heart investigation. 103 The beneficial effect of testosterone on molecular parameters has also been demonstrated in ORX rats with an myocardial infarction model. Testosterone administration at a physiological dose level (2 mg/kg) could promote angiogenesis after myocardial infarction by increasing hypoxia-inducible factor 1α (HIF-1α) and stromal cell-derived factor 1α (SDF-1α) signaling cytokines, which are generated at low oxygen concentration to promote neovascularization. 25 Moreover, testosterone could also increase the number of CD-34 progenitor stem cells that can differentiate into hematopoietic and endothelial progenitor cells in the ischemic myocardium. 25
Regarding the effects of testosterone in an I/R model, it may be proposed that either acute exogenous testosterone replacement or endogenous testosterone could provide deleterious effects on the heart by accelerating the apoptotic signaling process after I/R injury. However, chronic exogenous testosterone replacement may exert a beneficial effect in both I/R and myocardial infarction models.
Effect of testosterone on cardiac arrhythmias in I/R hearts
In Langendorff perfused hearts, a physiological dose of testosterone (2 mg/kg) combined with adrenergic stimulation has been shown to reduce reperfusion arrhythmia during I/R injury by reducing the incidence of premature ventricular beats, 28 whereas the supraphysiological dose of testosterone replacement (50 mg/kg) demonstrated antiarrhythmic effect in I/R model. 85 The mechanism responsible for the protective effect of testosterone on arrhythmia reduction was thought to be through α1a-adrenoceptor stimulation 28 and shortened the QT interval. 85 Since this positive effect of testosterone on the cardiac arrhythmia during I/R occurred together with the use of adrenergic receptor stimulation (Table 7), it is still unclear whether testosterone alone could provide a similar benefit. This needs to be investigated further.
Summary of the Effect of Testosterone on Arrhythmia Parameters and Heart Rate Variability (HRV) in I/R Heart.

Abbreviations: ORX rat, orchiectomized rat; I/R, ischemic and reperfusion; LAD, left anterior descending coronary artery; SC, subcutaneous; PVB, premature ventricular beat; MI, myocardial infarction; SDNN, the standard deviation of NN intervals; rMSSD, root mean square of the successive differences; pNN50, the proportion of NN50; IM, intramuscular.
Effect of testosterone on HRV in myocardial infarction model
Currently, there is no basic study on the effects of testosterone on the HRV, and there is only 1 clinical study investigating the effect of testosterone on HRV in patients with coronary artery disease. This cross-sectional study in men with stable coronary artery disease (age 36-73 years, mean age: 56 years) reported that a high level of blood testosterone was associated with increased HRV (Table 7). 104 These findings suggested that testosterone might have a positive effect on cardiac autonomic regulation in men after myocardial infarction by promoting parasympathetic dominance. Future basic and clinical studies are needed to investigate the roles of testosterone in the cardiac autonomic balance in both I/R injury and myocardial infarction models.
The Relationship Between Testosterone Levels and Coronary Artery Disease in Clinical Studies
Clinical studies show that low levels of testosterone were linked to increased blood pressure, dyslipidemia, atherosclerosis, arrhythmia, thrombosis, and endothelial dysfunction, as well as to impaired left ventricular function. 105 Haring and colleagues demonstrated that serum testosterone levels were inversely related to mortality rate due to cardiovascular disease or cancer, whereas low serum testosterone was associated with an increased risk of all-cause mortality 23 and that plasma testosterone level could potentially be used as a predictive tool for mortality from cardiovascular disease. 106 Regarding the relationship between plasma testosterone levels and coronary artery disease, a previous clinical study has shown that the plasma level of total testosterone, free testosterone, and bioavailability testosterone was significantly lower in men with coronary artery disease than healthy men. 18 However, several reports found that only free testosterone was significantly lower in men with coronary artery disease. 107 –110 It has also been shown that a low testosterone level was associated with atherosclerosis 111 and myocardial infarction 112 in men, supporting the previous findings that low levels of testosterone could increase the risk and contribute to a high prevalence of coronary artery disease in men. 22,110,113 –116 In addition, the level of testosterone shows an inverse correlation with the severity of coronary artery stenosis. 17,107,108,117,118 These findings suggested that endogenous testosterone may play a preventive role in the development of coronary arteriosclerosis. 117,119 Despite these beneficial reports on testosterone, inconsistent findings exist. A recent clinical report demonstrated that both low and high levels of total testosterone were associated with an increased ischemic arterial disease risk ratio, suggesting that only an optimal range of plasma testosterone could provide cardiovascular protection. 120 A summary of clinical reports regarding the association between the plasma testosterone levels and coronary artery disease is shown in Table 8.
Summary of Clinical Studies Reported on the Association Between Testosterone Levels and Coronary Artery Disease.

Abbreviations: CAD, coronary artery disease; TT, total testosterone; FT, free testosterone; DHEA, dehydroepiandrosterone; SHBG, sex hormone binding globulin; AR, androgen receptor; MI, myocardial infarction; DHEA, dehydroepiandrosterone; MVO, microvascular obstruction; STEMI, ST-elevation myocardial infarction; SA, stable angina; ECG-MVO, electrocardiographic microvascular obstruction; angio-MVO, angiographic microvascular obstruction; IAD, ischemic arterial disease.
Effects of Testosterone Replacement on Myocardial Infarction and Heart Failure in Clinical Studies
Previous studies demonstrated that testosterone replacement therapy exerts protective effect against myocardial ischemia such as delayed time to ischemia, 80,81,121 reduced exercise-induced myocardial ischemia, 80,121 and decreased severity of coronary stenosis by increased coronary artery diameter and coronary blood flow. 122,123 This could be partly due to its effects of reducing cholesterol and triglycerides 81,122 and decreasing serum tumor necrotic factor α. 81 It has also been shown that testosterone therapy was associated with a reduced risk of myocardial infarction. 124 However, as discussed earlier, inconsistent findings from clinical reports exist. Several studies found that testosterone therapy could increase the risk of myocardial infarction, especially in older men. 33,67 Several complications have been reported with supraphysiologic doses of testosterone replacement in athletes including hypertension, cardiomyopathy, pulmonary embolism, cardiac arrhythmias, and myocardial infarction, 125 suggesting that unnecessary use of testosterone without medical supervision is likely to cause undesirable effects. A summary of clinical reports regarding the effects of testosterone replacement on myocardial infarction and heart failure is shown in Table 9.
Summary of Clinical Studies Reported on the Effect of Testosterone Replacement on Myocardial Infarction and Heart Failure.
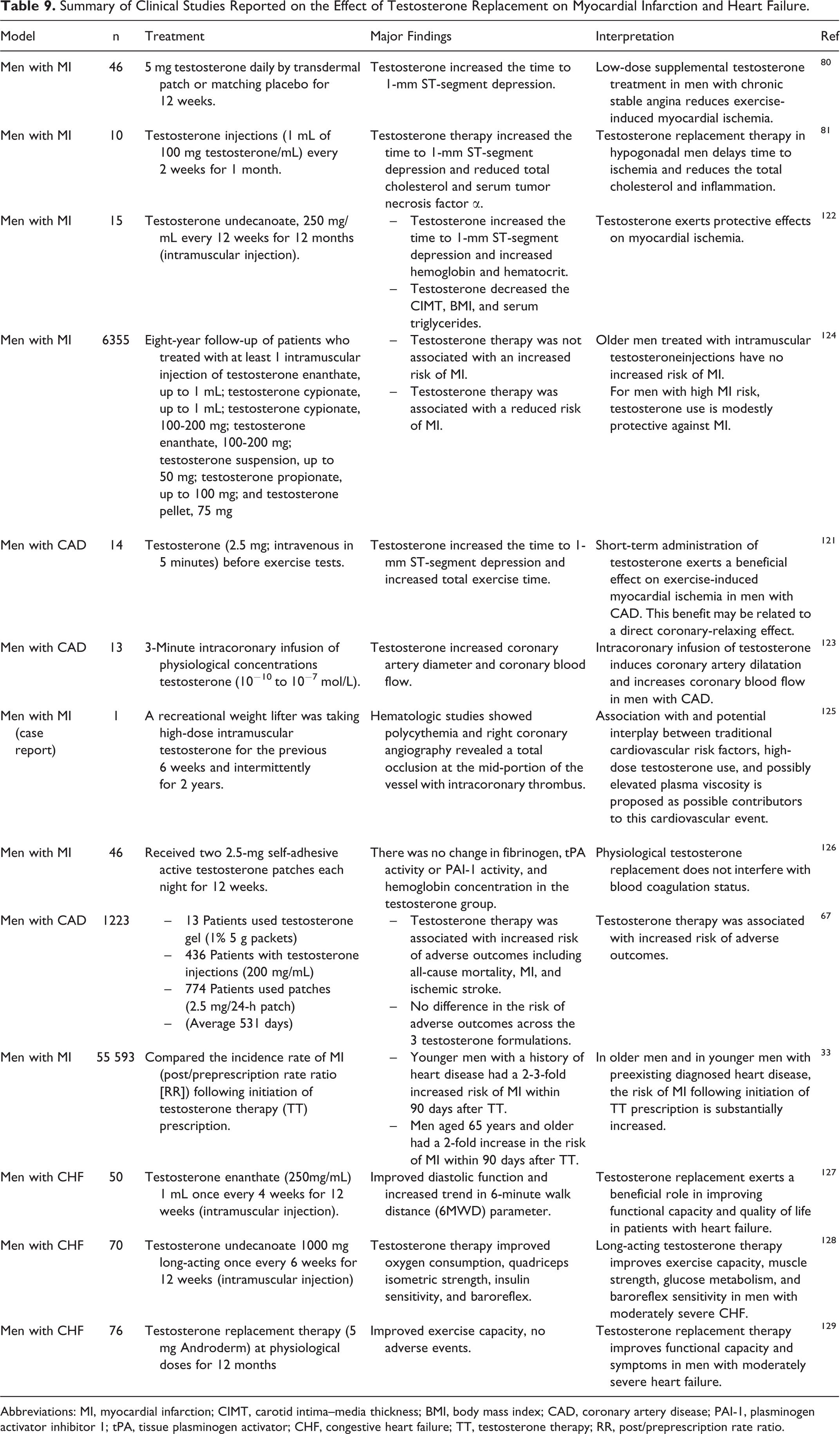
Abbreviations: MI, myocardial infarction; CIMT, carotid intima–media thickness; BMI, body mass index; CAD, coronary artery disease; PAI-1, plasminogen activator inhibitor 1; tPA, tissue plasminogen activator; CHF, congestive heart failure; TT, testosterone therapy; RR, post/preprescription rate ratio.
Conclusion
The cardioprotective effects of chronic testosterone replacement to the physiological dose have been demonstrated in an I/R heart. This technique is seen to be effective by reducing the infarct size, improving contractile functions, decreasing LDH release, increasing ventricular myocytes viability, reducing arrhythmias, and improving autonomic regulation of the heart after I/R. These beneficial effects of testosterone are thought to occur via the stimulation and expression of α1a-adrenoceptor, increased synthesis of Hsp70, increased neovascularization of CD34+ stem cell and cytokines HIF-1α, SDF-1α, and vascular endothelial growth factor in the early stages of myocardial infarction. In addition, testosterone may play a beneficial role by maintaining intracellular Ca2+ homeostasis during ischemic stress (Figure 1). However, acute testosterone replacement might cause adverse effects in the I/R heart by reducing cardioprotective signaling proteins such as STAT-3 and SOCS-3 after I/R and also decreasing myocardial Akt activation, leading to reduced Bad phosphorylation and decreased Bcl-2 levels, which consequently results in worsened I/R-depressed myocardial function.

The potential roles of testosterone replacement in cardiac ischemia–reperfusion (I/R) and myocardial infarction models. During cardiac I/R, testosterone administration has been shown to reduce infarct size, decrease lactate dehydrogenase (LDH) release, and increase cell viability via increased neovascularization, increased heat shock protein 70 syntheses, α1a-adrenoceptor stimulation, and decreased myocardial apoptosis. The reduction in sympathovagal imbalance may occur due to decreased oxidative stress. The reduction in cardiac arrhythmia could occur due to decreased intracellular Ca2+ accumulation and α1a-adrenoceptor stimulation. Furthermore, testosterone could decrease intracellular Ca2+ accumulation, increase an expression of α1a- and β1-adrenoceptors, decrease LVEDP, increase LVDP, and ejection fraction, thus resulting in increased contractility and improved cardiac function. HIF-1α indicates hypoxia-inducible factor 1-α; SDF-1α, stromal cell-derived factor 1α; VEGF, vascular endothelial growth factor; CD34, hematopoietic progenitor cell antigen CD34; LVDP, left ventricular develop pressure; LVEDP, left ventricular end diastolic pressure.
Footnotes
Author Contribution
W. Pongkan and N. Chattipakorn contributed to conception and design; acquisition, analysis, and interpretation; drafted the article; critically revised the article; gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. S. Chattipakorn contributed to conception and design, analysis and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Thailand Research Fund Royal Golden Jubilee PhD program (WP and NC), the Thailand Research Fund BRG5780016 (SC), a NSTDA Research Chair Grant from the National Science and Technology Development Agency Thailand (NC), and the Chiang Mai University Center of Excellence Award (NC).
