Abstract
Ischemic heart diseases are the major reasons for disability and mortality in elderly individuals. In this study, we tried to examine the combined effects of nicotinamide mononucleotide (NMN) preconditioning and melatonin postconditioning on cardioprotection and mitochondrial function in ischemia/reperfusion (I/R) injury of aged male rats. Sixty aged Wistar rats were randomly allocated to 5 groups, including sham, control, NMN-receiving, melatonin-receiving, and combined therapy (NMN+melatonin). Isolated hearts were mounted on Langendorff apparatus and then underwent 30-minue ligation of left anterior descending coronary artery to induce regional ischemic insult, followed by 60 minutes of reperfusion. Nicotinamide mononucleotide (100 mg/kg/d intraperitoneally) was administered for every other day for 28 days before I/R. Melatonin added to perfusion solution, 5 minutes prior to the reperfusion up to 15 minutes early reperfusion. Myocardial hemodynamic and infarct size (IS) were measured, and the left ventricles samples were obtained to evaluate cardiac mitochondrial function and oxidative stress markers. Melatonin postconditioning and NMN had significant cardioprotective effects in aged rats; they could improve hemodynamic parameters and reduce IS and lactate dehydrogenase release compared to those of control group. Moreover, pretreatment with NMN increased the cardioprotection by melatonin. All treatments reduced oxidative stress and mitochondrial reactive oxygen species (ROS) levels and improved mitochondrial membrane potential and restored NAD+/NADH ratio. The effects of combined therapy on reduction of mitochondrial ROS and oxidative status and improvement of mitochondrial membrane potential were greater than those of alone treatments. Combination of melatonin and NMN can be a promising strategy to attenuate myocardial I/R damages in aged hearts. Restoration of mitochondrial function may substantially contribute to this cardioprotection.
Keywords
Introduction
Aging is a process of gradual deterioration of physiological functions over time. 1 Cardiac aging results in increased dysfunctional mitochondria, overproduction of reactive oxygen species (ROS), elevated in mitochondrial DNA mutations, decreased mechanical and contractile efficiency of the heart and suppressed autophagy and mitophagy processes. 2 Coronary artery disease (CAD) is a dangerous multifactorial disease leading to disability and death of human in the world, and advancing age is a main risk factor for CAD. 3 Myocardial ischemia/reperfusion (I/R) injury happens after myocardial ischemia and during the restoration of coronary blood flow, reperfusion, and resulting in inflammation, overproduction of ROS, mitochondrial dysfunctions, and cardiac cell death. 4,5 It has been documented that aging is related to increment susceptibility to myocardial ischemia and also declines the recovery of myocardial function after the ischemic insults. 6,7 More importantly, aged ischemic hearts are resistant to cardioprotection via routine therapeutic strategies. 8
Melatonin (N-acetyl-5-methoxytryptamine) is recognized for its antioxidant capacity as well as anti-inflammatory properties. Melatonin has been shown to have cardioprotective impact in young animals and it owns positive impact on mitochondrial activity. The increased burden of cardiovascular diseases during aging has been related to the low levels of melatonin. 9 However, whether increasing plasma levels of melatonin can regulate ROS production or recover myocardial function in aged myocardium following I/R insults remains unknown.
Nicotinamide adenine dinucleotide (NAD+) is an essential cofactor for numerous cellular metabolic reactions and has a dominant role in energy production. 10 It regulates multiple biological functions including circadian rhythms, metabolism, calcium homeostasis, mitochondrial functions, aging, cell death, and immunity. 11,12 Studies have reported that NAD+ bioavailability decreases with age as well as ischemic insults 13,14 and is accompanied by reduced mitochondrial adenosine triphosphate (ATP) production. 15 Nicotinamide mononucleotide (NMN) is the main precursor of NAD+. Restoring NAD+ cellular content with NMN could improve age-related physiological dysfunction and various diseases. 16,17 It has been found that treatment with processors of NAD+ has cardioprotective effect against I/R injury. 18 There is no finding about the effect of NMN on myocardial and mitochondrial function in aged myocardium following I/R injury.
The majority of preclinical studies usually perform on the young animal models and use just one treatment and focus on very limited aspects of the disease. Human patients with myocardial I/R injuries commonly have other background comorbidities and cardiovascular risk factors; therefore, cardioprotective treatments usually fail to reduce infarct size (IS) and patients outcomes in clinical settings of myocardial I/R injury. One strategy to overcome the failure of individual treatments and to achieve optimized cardioprotection would be the administration of combined therapies. 19 Therefore, it is necessary to design targeted and multidimensional studies with 2 or more therapeutic approaches instead of monotherapy. Because aging interferes with I/R injury and the monotherapies with NMN or melatonin have limited effect on aging, 20,21 it is clinically important to study the combined effects of these 2 protective agents on aging heart. Thus, the main aim of this research was to investigate the individual and combined effects of melatonin and NMN on myocardial function, mitochondrial activity, and oxidation status following myocardial I/R injury in aging rats.
Materials and Methods
Animals and Materials
Sixty male Wistar rats (22-24 months old) weighting 400 to 450 g were prepared from animal laboratory of University of Medical Sciences. Three animals were housed in each standard cage and all animals were kept at 20°C to 24°C with a 12-h/12-h light/dark cycle, and unlimited access to tap water and standard pellet food. All of the experimental protocols were approved by the ethics committee of the animal research of the University (ethical code: IR.TBZMED.REC.1396.395) and conducted under the National Institutes of Health (NIH) guidelines (publication no. 85-23, revised 1985). Melatonin and NMN were bought from Sigma-Aldrich (St. Louis, Missouri) and all materials of Krebs-Henseleit (K-H) solution from Merck Company (Munchen, Germany).
Experimental Design
First, the aged rats were randomly allocated to 5 experimental groups (n = 12 in each group): (1) sham group (without I/R), (2) control group (receiving I/R), (3) I/R+meltonin (Mela), (4) NMN+I/R (NMN), and (5) NMN+I/R+Mela (NMN+Mela). In NMN-receiving groups, NMN (100 mg/kg/body weight) was intraperitoneally (IP) administered to aged rats every other day for 28 days. 22 In addition, in melatonin-receiving groups, during the I/R protocol (see next sections and Figure 1), the isolated beating hearts were nourished with a K-H perfusion solution containing 50 µM melatonin for 20 minutes, from 5 minutes before the start of reperfusion up to 15 minutes of reperfusion. 23

Langendorff-isolated perfused heart experimental protocol. Sham group received 105 minutes of persistent perfusion; control group received 30 minutes of regional ischemia and 45 or 60 minutes of reperfusion; Mela group received 30 minutes of regional ischemia and 45 or 60 minutes of reperfusion plus adding melatonin to perfusion solution from 5 minutes before the onset of reperfusion up to 15 minutes of reperfusion; NMN group received 30 minutes of regional ischemia and 45 or 60 minutes of reperfusion; and the rats in this group had already been treated with NMN for 28 days; NMN+Mela group received 30 minutes of regional ischemia and 45 or 60 minutes of reperfusion plus adding melatonin to perfusion solution and pretreatment of rats with NMN. All I/R-receiving groups experienced 15 minutes of stabilization period at the beginning. Biochemical and mitochondrial analysis was performed from tissue sampling after 45 minutes of reperfusion (n = 6), and infarct sizes were evaluated after 60 minutes of reperfusion (n = 6). I/R indicates ischemia/reperfusion; Mela, melatonin; NMN, nicotinamide mononucleotide.
Preparation of Langendorff-Isolated Perfused Hearts
After injection of heparin (500 IU/kg of body weight), the male rats were anesthetized by injection of ketamine/xylazine (60/10 mg/kg, IP, respectively). Then, the chest was opened and the heart was quickly isolated and transferred to a Langendorff-isolated hearts apparatus (ML176-V; AD Instruments Co, New South Wales, Australia). Therein, the heart was perfused retrogradely through the aortic cannula with a K-H solution at pH 7.4, containing (in mmol/L: NaCl, 118; KCl, 4.7; KH2PO4, 1.2; MgSO4, 1.2; CaCl2, 2.5; NaHCO3, 25; and glucose, 11). The perfusion pressure was set at 80 mm Hg and the perfusion solution was gassed with carbogen containing 5% CO2 and 95% O2, at 37°C ± 0.5°C at all times. During the stabilization, the coronary flow (CF) rate was at 12 to 14 mL/min. To maintain temperature near to 37°C, a homeothermic glass cover surrounded the isolated heart. 24
Myocardial I/R Protocol
At the beginning of the I/R experiments, the beating isolated hearts were perfused with the K-H perfusion solution for 15 minutes as stabilization period. Subsequently, the left anterior descending (LAD) coronary artery was ligated to induce regional ischemia for 30 minutes and then reperfusion was performed by reopening of LAD (for 45 or 60 minutes). Sham-operated group was subjected to the same procedures, except that the ligature was left untied. The CF was used as an indicator of sufficient perfusion and the efficiency of coronary ligation. A decline of at least 25% in CF during obstruction was considered satisfactory. 25 Biochemical and mitochondrial analysis were performed from tissue sampling after 45 minutes of reperfusion (n = 6), and ISs were evaluated after 60 minutes of reperfusion (n = 6). Figure 1 shows the experimental protocol of the study. 26
Measurement of Heart Hemodynamics Parameters
After attaching the hearts to the Langendorff apparatus, a water-filled latex balloon (Harvard Apparatus Ltd, Eden Bridge, the United Kingdom) was inserted into the left ventricle (LV) after the removal of the left auricle. The left ventricular end-diastolic pressures (LVEDPs) were adjusted between 5 and 10 mm Hg by inflating LV balloon. The heart rates (HRs), LV-developed pressures (LVDPs), LVEDP, and LV contractility (±dP/dt) were recorded using a Power Lab data acquisition system and analyzed with Chart 7.3 software (AD Instruments, Newcastle, NSW, Australia). All hemodynamic variables recorded continuously in all groups throughout the experiment. The coronary effluent was timely collected over the experiment to quantify the CFs. 24 In each group, the data of 10 hearts (n = 10) were recorded and reported for hemodynamic parameters.
Measurement of IS
After finishing the reperfusion period for 60 minutes, the LAD coronary artery was retightened and approximately 3 to 4 mL of 0.25% Evans blue dye was gradually infused through the aortic cannula into the coronary circulation. This separates the area at risk (AAR) as unstained from the blue, living myocardial tissue. Then, the hearts were frozen, underwent slicing into 2-mm-thick cross sections, and incubated for 15 minutes in 1% 2,3,5-triphenyltetrazolium-chloride phosphate-buffered solution (PBS), pH 7.4 at 37°C. Then, the slices were placed in 10% formalin for 24 hours to increase the staining contrast. The infarct volume was determined and analyzed using Image-J software (NIH, Bethesda, Maryland). Area at risk and ISs were reported as the percentages of the LV and AAR volumes, respectively. 24
Lactate Dehydrogenase Release
Measurement of lactate dehydrogenase (LDH) release into coronary effluent (during the reperfusion) was used in order to evaluate myocardial damage. The level of LDH enzyme was determined spectrophotometrically using an assay kit (Pars-Azmoon Co, Tehran, Iran). The LDH solution absorbance was read at 492 nm. The results were presented in U/L. 25
Measurement of NAD+/NADH Ratio
Intracellular levels of NAD+ and NADH were assayed using the NAD/NADH Quantification Kit (Sigma-Aldrich), according to the manufacturer’s instructions, as described previously. 14 Total NADH and NAD+ were quantified in a colorimetric assay (450 nm). 14
Isolation of Cardiac Mitochondria
The LV of the myocardium was rapidly harvested at the end of reperfusion for mitochondria isolation. First, 100 mg ventricular tissue from AAR was minced and homogenized in isolation buffer at 4°C and pH 7.4, containing: 200 mM mannitol, 70 mM sucrose, 10 mM HEPES, and 2 mM ethylenediaminetetraacetic acid (EDTA). The homogenate was centrifuged at 1300 g for 10 minutes. Then the supernatant was gathered and recentrifuged at 12 000 g for 10 minutes. Finally, 100 µL storage buffer, pH 7.4, was used to suspend the mitochondrial pellet. The composition of this storage buffer was 250 mM sucrose, 1 mM ATP, 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 5 mM sodium succinate, 0.08 mM adenosine diphosphate, 1 mM dithiothreitol, and 2 mM K2HPO4. The mitochondrial protein concentrations were measured using Nanodrop (Thermo Fisher Scientific, Wilmington, USA). 27
Assessment of Cardiac Mitochondrial ROS Generation
To evaluate ROS production in mitochondria, isolated mitochondria were incubated with 40 μL dichlorodihydrofluorescein diacetate dye for 30 minutes in the dark. Dichlorodihydrofluorescein diacetate (DCFDA) was oxidized in the existence of ROS to 2′,7′-dichlorofluorescein. The fluorescence intensities (FIs) were measured at λex 485 nm and λem 530 nm in a fluorescence plate reader and ROS level was expressed as FI/mg of protein. 28
Measurement of Cardiac Mitochondrial Membrane Potential Changes (ΔΨm)
The mitochondria staining JC-1 dye (Sigma-Aldrich) was employed to measure the changes of mitochondrial membrane potential in heart. Isolated myocardial mitochondria were gently suspended in JC-1 stain (2.5 μg/mL) in the dark at 37°C for 30 minutes. The red fluorescence of JC-1-aggregates (in normal condition) was excited at 525 nm and then the emission was detected at 590 nm. The green fluorescence of JC-1 monomer (low ΔΨm, ie, depolarized state) was excited at 485 nm and its emission was recorded at 530 nm. The reduction in the red/green FI ratio was indicated as cardiac mitochondrial membrane depolarization. 28
Measurement of Oxidative Stress Markers
The myocardial contents of superoxide dismutase (SOD), glutathione peroxidase (GPX), and malondialdehyde (MDA) were measured to indicate the levels of intracellular oxidative stress. For measuring SOD activity, the cardiac samples from the LVs were homogenized with PBS (pH 7.4). Then, the homogenates were centrifuged at 10 000 g for 15 minutes at 4°C. The supernatant and kit reagents were added to plate wells and SOD activity was measured at 505 nm by a spectrophotometer. The results were expressed as U/mg of protein. 29
The GPX activity was determined with a commercial kit (ZellBio GmbH, Ulm, Germany). Briefly, frozen left ventricular tissues were homogenized in 50 mM phosphate buffer containing 0.5 mM EDTA (pH 7.5). The homogenates were centrifuged at 10 000g for 15 minutes at 4°C. The reduction in absorbance of supernatant at 340 nm was measured at 37°C using a spectrophotometer. The activity of GPX in the heart tissue was expressed as U/mg of protein.
Malondialdehyde is the major product of lipid peroxidation. Accordingly, Draper and Hadley’s method has been employed as a standard protocol for analyzing the lipid peroxidation by measuring thiobarbituric acid-reactive substances in tissue’s homogenate. The MDA levels were expressed in nmol/mg of protein. 30
Statistical Analysis
Descriptive data were presented as mean ± standard errors of means. Hemodynamic data were analyzed using of repeated-measures analysis of variance (ANOVA) and other parameters were analyzed by 1-way ANOVA. The post hoc Tukey test was used to determine the statistical significance between groups. All analyses were performed using Graph Pad Prism (version 6.01) and the statistical significance was considered when the P values were less than .05.
Results
Hemodynamic Parameters in Baseline
At preischemic period, the significant differences were not seen in hemodynamic parameters, including HR, CF, LVEDP, LVDP, +dP/dt, and −dP/dt among the animal groups. These baseline parameters were summarized in Table 1.
Baseline Values of Hemodynamic Parameters in Hearts of Experimental Groups.a
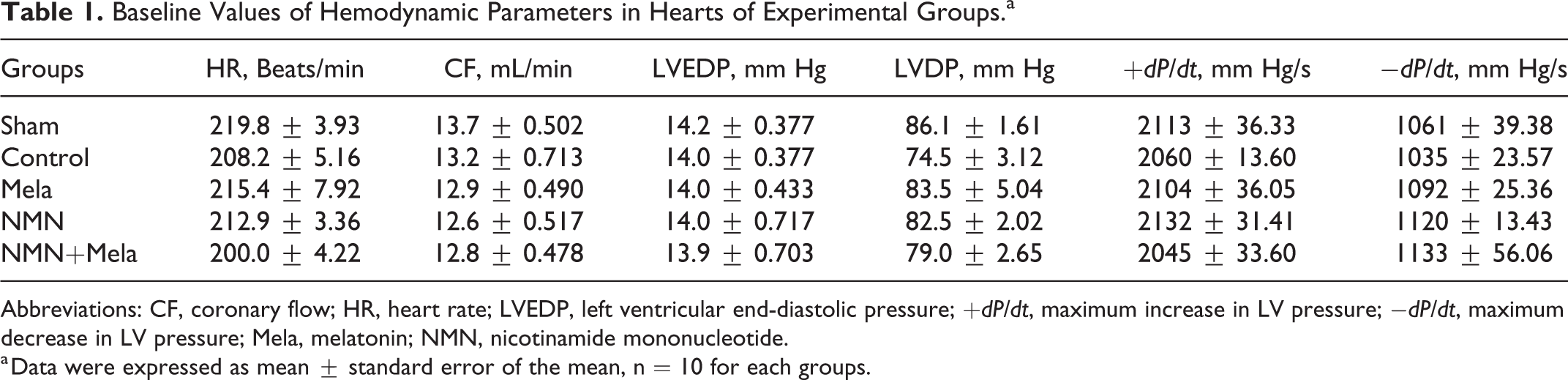
Abbreviations: CF, coronary flow; HR, heart rate; LVEDP, left ventricular end-diastolic pressure; +dP/dt, maximum increase in LV pressure; −dP/dt, maximum decrease in LV pressure; Mela, melatonin; NMN, nicotinamide mononucleotide.
a Data were expressed as mean ± standard error of the mean, n = 10 for each groups.
Hemodynamic Parameters During I/R Experiments
Induction of ischemia significantly lowered the CFs from their baseline values in all groups with I/R injury. Restoration of CF was observed in reperfusion phase of all groups, but this increment in CFs was not significantly varied between treated and untreated groups (Figure 2A). The HRs at ischemic phase were significantly lower in all I/R groups in comparison to sham group (P < .001 at 15 and 30 minutes of ischemia). At reperfusion phase in all groups, the HR was increased as compared to the respective ischemic phases. There were no significant differences in HRs before I/R and during the reperfusion phase between groups (Figure 2B).

Assessment of coronary flow and heart rates in aged hearts subjected to 30-minute ischemia and 60-minute reperfusion. A, Coronary flow (CF, mL/min). B, Heart rates (HR; beat/min). The data were expressed as mean ± standard error of the mean (n = 10). *P < .05, **P < .01, and ***P < .001 control group versus sham group. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
Analysis of the data showed that LVEDPs were obviously elevated in I/R hearts as compared to the baselines (P < .001; Figure 3A). Melatonin and NMN significantly prevented the sharp increase in LVEDP at different time points of reperfusion as compared with control group values (P < .01, P < .00, respectively). At 30 minutes of ischemic period, LVEDP was significantly lower in NMN+Mela group than control group (P < .05). Furthermore, the combination of melatonin and NMN remarkably reduced LVEDP in treated hearts at reperfusion times compared to control group and this effect was stronger than individual treatments (P < .001 and P < .05). Figure 3B shows the changes in LVDP during ischemia and at various time points of reperfusion among all groups. Both NMN and NMN+Mela treatments significantly improved LVDP (P < .001) compared to control group. Also, melatonin presented better LVDP recovery throughout the reperfusion period (P < .01). Moreover, NMN+Mela group showed significant lower LVDP than melatonin group (P < .05 at times 45 and 60 minutes of reperfusion).

A, Left ventricular end-diastolic pressure (LVEDP) and (B) left ventricular developed pressure (LVDP) in aged hearts subjected to 30-minute ischemia and 60-minute reperfusion. The data were expressed as mean ± standard error of the mean (n = 10). ***P < .001 control group versus sham group, #P < .05 and ## #P < .001: NMN or NMN+Mela groups versus control group; +P < .05 and ++P < .01 Mela group versus control group; $P < .05 NMN+Mela group versus Mela group. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
In addition, dP/dt is useful index for evaluating ventricular contractility. Administration of melatonin could significantly recover the ±dP/dt in reperfusion phase compared to controls (P < .01), Moreover, NMN+Mela and NMN also remarkably recovered the ±dP/dt compared to the control hearts in reperfusion phase and their significance levels were greater than those of melatonin alone (P < .001; Figure 4A and B).

A, Maximum increase in left ventricular pressure (+dP/dt) alterations and (B) maximum decrease in left ventricular pressure (−dP/dt) alterations in aged hearts subjected to 30-minute ischemia and 60-minute reperfusion. The data were expressed as mean ± standard error of the mean (n = 10). ***P < .001 control group versus sham group, ###P < .001: NMN or NMN+Mela groups versus control group. ++P < .01: Mela group versus control group. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
Level of LDH Enzyme Released From I/R Hearts
The LDH release is considered an indicator of myocardial damage. The release of LDH was significantly increased in control hearts versus sham group (P < .001). Pretreatment of NMN in I/R hearts significantly reduced the activity of LDH in comparison with hearts of control group (P < .001). Administration of melatonin at the end of ischemia caused an effect almost similar to NMN effect (P < .001). Furthermore, the combination treatment markedly lessened the level of this enzyme toward control values (P < .001; Figure 5).

The levels of lactate dehydrogenase (LDH) release into the coronary effluent in aged hearts subjected to 30-minute ischemia and 45-minute reperfusion. The results are expressed as mean ± standard error of the mean (n = 6). ***P < .001 versus sham. ###P < .001 versus control. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
Infarct Size
The volumes of AAR were similar among all groups (Figure 6A). Both melatonin alone (P < .01) and NMN-pretreatment alone (P < .001) significantly decreased the ISs of aged animals to 28.5% + 2.4% and 22% + 3.1% from 44.5% + 2.9% of control hearts. The combination of melatonin and NMN also decreased the ISs significantly in comparison to control group (17.1% + 2.4% vs 44.5% + 2.9%; P < .001) and this IS-reducing effect was greater than the effect of melatonin individual treatment (P < .05; Figure 6B).

Area at risk (AAR; A) and infarct sizes (B) percentages in aged hearts subjected to 30-minute ischemia and 60-minute reperfusion. The data were expressed as mean ± standard error of the mean (n = 6). ##P < .01 and ###P < .001 versus control group, $P < .05 versus Mela group. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
NAD+/NADH Ratio
The NAD+/NADH ratio was reduced following I/R injury, while NMN treatment markedly restored the NAD+/NADH ratio compared to control group (P < .001). Application of melatonin could also increase this ratio (P < .05). Moreover, the NAD+/NADH ratio in groups pretreated with NMN was higher than in melatonin group (P < .01; Figure 7).

Effects of NMN and melatonin on heart NAD+/NADH ratio. The results are expressed as mean ± standard error of the mean (n = 6). ***P < .001 versus sham. ###P < .001 and #P < .05 versus control, and $$P < .01 versus Mela. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
Mitochondrial Membrane Potential (ΔΨm)
Myocardial I/R injury caused dissipation of ΔΨm (more depolarization) versus sham hearts (P < .001; Figure 8). Analysis of data showed that the red/green ratio (indicating the membrane polarization near to resting level) was significantly decreased in control group versus sham group (P < .001). Administration of melatonin alone, NMN alone and combination of melatonin with NMN treatments, induced a significant increase in the red/green ratio compared to controls (P < .01, P < .001, respectively). Moreover, mitochondrial membrane potential in NMN (P < .001) and combined treatments (P < .01) was significantly less depolarized than in melatonin group (Figure 8A).

A, Cardiac mitochondrial membrane potential change (red/green JC-1 ratio) and (B) mitochondrial ROS production level (DCFDA intensity) in aged hearts subjected to 30-minute ischemia and 45-minute reperfusion. The results are expressed as mean ± standard error of the mean (n = 6). ***P < .001 versus sham. ###P < .001 and ##P < .01 versus control, and $$P < .01 and $$$P < .001 versus Mela. Mela indicates melatonin; DCFDA, dicholorofluorescin diacetate; NMN, nicotinamide mononucleotide; ROS, reactive oxygen species.
Cardiac Mitochondrial ROS Assessment
We observed a high production of ROS in control hearts in comparison to sham hearts (P < .001). Figure 8B shows that comparing to control group, all treatments decreased ROS levels significantly (P < .001). Moreover, the level of ROS generation in NMN + Mela-treated hearts was lower than melatonin hearts (P < .01), so combination therapy was more efficient in increasing the efficiency of melatonin in the maintenance of mitochondrial function.
Oxidative Stress Markers Assessment in Heart
As shown in Figure 9A to C, the effects of NMN and melatonin on oxidative stress markers in aged hearts were examined with measuring MDA level as indicator of peroxidation and SOD and GPX levels as enzymatic antioxidants. Myocardial I/R injury markedly attenuated the activities of GPX and SOD and increased the content of MDA (P < .001). Nevertheless, NMN and/or melatonin significantly decreased the level of MDA and enhanced the enzymatic antioxidant defense capacity by the upregulation of SOD and GPX activities (P < .001). Results showed that co-application of NMN and melatonin could increase GPX activity as compared to NMN group (P < .05). Further, combined treatment led to a significant increase in SOD activity in comparison to melatonin group (P < .05).

A, Superoxide dismutase (SOD) activity, (B) glutathione peroxidase (GPX) activity, and (C) malondialdehyde (MDA) content. The results are expressed as mean ± standard error of the mean (n = 5). ***P < .001 versus sham. ###P < .001 versus control, $P < .05 versus Mela and ψP < .05 versus NMN. Mela indicates melatonin; NMN, nicotinamide mononucleotide.
Discussion
This study revealed the therapeutic potentials of NMN and melatonin following myocardial I/R injury in aged rats, specifically when they used in combination. We found that NMN and melatonin had protective effects against I/R injury in aged rats by decreasing the IS and improving myocardial function. Besides, NMN treatment and melatonin replenished heart NAD+ levels. Co-application of NMN and melatonin could provide stronger cardioprotection. These positive effects of interventions may be through the improvement of mitochondrial function due to the reduced mitochondrial ROS generation and membrane depolarization as well as reduced oxidative stress and enhanced enzymatic antioxidant defense system.
The previous experimental studies on the cardioprotective effect of therapeutics such as melatonin or NMN or other agents have been carried out on the young healthy animal and focus on very limited aspects of the disease. 14,31 Aging results in structural and functional alterations in heart, leading to myocardial dysfunction and cardiovascular diseases. 32 Growing evidence indicates that aging and I/R injury induce free radicals overproduction, reduction in antioxidant enzyme activity such as SOD and GPX, and increase lipid peroxidation products and ROS generation, leading to mitochondrial dysfunction. 33,34 The increased formation of ROS noticeably enhances susceptibility to Ca2+-induced opening of mitochondrial permeability transition pores (MPTP), which finally causes the loss of membrane potential, further oxidative stress and cell death. 35,36 Due to these alterations and age-related loss of antioxidant defense, improvement of I/R injury in old patients is reduced. 37
In the present study, the I/R insult developed myocardial infarction, cardiomyocytes damage, cardiac dysfunction, and increased the level of LDH release in aging hearts, while aged rats pretreated with NMN showed lesser myocardial injury by restoring abovementioned indexes. This cardioprotective effect of NMN in aged hearts was accompanied with its positive influences on mitochondrial function and ROS production. Our data indicated that administration of NMN before ischemia could prevent the ischemia-induced decrement in NAD+/NADH ratio in the heart, suggesting that NMN enters the heart and boosts cellular NAD+ levels. In the current study, SOD and GPX activities were reduced and MDA content was increased markedly following I/R injury in heart tissues, indicating an increased oxidative stress level. However, NMN pretreatment could prevent the myocardial I/R-induced increase in MDA levels and decrease in the GPX and SOD activities. In line with our study, Zhang et al 38 reported that administration of NAD+ in young animals can alleviate myocardial I/R injury via decreasing apoptotic damage and increasing the antioxidant capacity. Moreover, Yamamoto et al have reported that NMN has protective effects in both ischemic and reperfusion phases of young hearts, which mediated by reduction in acetylation of forkhead transcription factor 1 and activation of Sirtuin 1 as well stimulation of autophagy flux. 14 NAD+ cellular levels decline with aging process as well as ischemia, which can limit NADH generation and thereby decrease oxidative phosphorylation. 39 Since NAD+ is a key cofactor in many biological processes, so we suggested that use of NMN before ischemia can improve cellular negative alterations in aging and cause less damage following I/R injury. It has been noted that NMN supplementation can be considered an effective therapy protecting against vascular dysfunction and oxidative stress in aged mice. 40 Conforming to our study, Naia et al 42 demonstrated that nicotinamide and NMN can ameliorate mitochondrial function through improvement in mitochondrial Δψm and oxygen consumption in Alzheimer disease model 41 and both central and peripheral Huntington disease in vitro models.
It has also found that the plasma level of melatonin has been decreased in elderly individuals and patients with coronary heart disease. 43,44 Previous evidence in young animals shows that melatonin has positive effects in myocardial I/R injury, mediated by antioxidative effects and decrease in lipid peroxidation. 45 Animal studies have revealed that melatonin is protective in myocardial I/R young rat model, especially when administered during the reperfusion period. 23,46 In this study, administration of melatonin conferred a cardioprotective effect in aged rat hearts, as confirmed by improved postischemic cardiac hemodynamic and mitochondrial function and decreased myocardial IS. Our results support that melatonin acts as a potent free radical scavenger and antioxidant during I/R-induced cardiac damage. The previous studies have demonstrated that melatonin has ameliorated LV function 47 and alleviated oxidative stress levels in rats following I/R. 48,49
Interestingly, our results showed that NMN treatment prior to I/R insult and prior to application of melatonin in reperfusion time could induce more cardioprotective effect and tend to markedly decrease the IS and improve cardiac function. Some previous studies have also shown greater cardioprotective effects of combined therapeutic strategies in aged hearts. 50 According to our findings, the combination effect was significant in comparison to the melatonin alone, not in comparison to NMN alone. This may indicate that combined therapy enhanced the efficacy of melatonin in aged I/R hearts. Given that both NMN and melatonin alone have antioxidant effects, and their combination therapy increased the activity of antioxidant enzymes SOD and GPX along with reducing MDA content, this situation has more likely resulted in decreased ROS production and improved mitochondrial function. The pathophysiological conditions of aging may reduce the effects of individual interventions (especially melatonin) against I/R injury. Restoring NAD+ levels toward control values through preconditioning of rats with NMN have increased the effects of melatonin. Therefore, in the presence of NAD+, intracellular structures such as mitochondria function in almost normal condition so that the melatonin can activates its protective signaling and cellular targets more potently in aged cardiomyocytes, and this is why combined therapy has better effects than melatonin alone on IS, mitochondrial ROS, and other parameters.
It has been shown that mitochondrial NAD+ reduction is an index of MPTP opening and cytochrome c release. 51 Our study and previous researches showed that boosting cellular NAD+ levels could improve mitochondrial function. The effects of NAD+ restoration may be due to decrease in ATP consumption used for NAD+ synthesis, prevention of MPTP opening, and reactivation of sirtuins and antioxidative effects. In addition, melatonin inhibits MPTP opening and cardiolipin peroxidation. 52 Thus, combining melatonin and NMN may enhance their cardioprotective effects partly through potent inhibition of MPTP opening and subsequent antioxidative effects, which warrants further investigation. Furthermore, the cardioprotective effects of this combined therapy may be achieved via the activation of similar or distinct signaling pathways by NMN and melatonin, such as JAK2/STAT3 and AMPK-PGC-1α-SIRT3 autophagy and mitophagy signaling, 47,53 which need to be clarified in aged hearts in future.
Conclusion
These results disclosed that combination of melatonin and NMN in aged rat hearts may be a promising candidate for alleviating myocardial I/R injury and boosting cardioprotection during aging by restoring oxidative stress, mitochondrial function, and cardiac contractile activities. Nevertheless, further studies are essential to illuminate the role of each of the multiple mechanisms contributing to cardioprotection, especially in the in vivo models of I/R injury during aging.
Footnotes
Author Contributions
Leila Hosseini performed the experiments and analyzed and interpreted the data. Reza Badalzadeh and Manouchehr Seyedi Vafaee designed the experiments and edited the article. Reza Badalzadeh contributed to interpretation of the data. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported in part by a grant from Aging Research Institute and Drug Applied Research Centre, Tabriz University of Medical Sciences, Tabriz-Iran.
