Abstract
Introduction
The prevalence of atrial fibrillation (AF) in acute coronary syndrome (ACS) patients is increasing. Data on outcomes of anticoagulation in ACS patients with AF are lacking.
Objectives
The aim of our study was to investigate the prevalence of stroke, myocardial infarction, bleeding complications, and all-cause mortality in this population.
Patients and methods
PL-ACS and AMI-PL registries gather an all-comer population of ACS patients in Poland, exceeding half a million records. We have selected ACS survivors with concomitant AF on admission, divided them into subgroups with regard to the administered anticoagulation, and followed up with them for a 12-month period (n = 13,973). Subsequently, groups were propensity score matched for age, sex, ejection fraction, diabetes, heart failure, renal impairment, and type of ACS.
Results
The study population was divided with regard to the administration of anticoagulation. Anticoagulation was prescribed in 2,466 patients (17.6%). The (D)OAC+ patients were younger; however, comorbidities were more prevalent in this group. The 12-month follow-up showed that the (D)OAC+ patients had significantly lower rates of all-cause mortality, myocardial infarction, and ischemic stroke, with no significant increase in bleeding events. After matching, the study groups consisted of 2,194 patients each and showed no differences in baseline characteristics. The outcomes of the 12-month observation were similar to the findings before matching.
Conclusions
This all-comer national registry analysis shows that the use of guideline-recommended therapy and anticoagulation in ACS survivors with AF is associated with a lower rate of all-cause mortality, recurrent myocardial infarction, and ischemic stroke.
What's New
Patients with acute coronary syndromes (ACS) are aging. The incidence of atrial fibrillation (AF) in ACS patients is high and will increase. Data on the impact of anticoagulation in post-ACS patients with AF are scarce. Available evidence suggesting that physicians are sometimes reluctant to prescribe guideline-recommended anticoagulation for AF patients after ACS is alarming.
To the best of our knowledge, this propensity score-matched analysis from a nationwide registry indicates that AF patients after ACS clearly benefit from any kind of anticoagulation, resulting in significantly lower mortality rates and less recurrent myocardial infarctions and ischemic strokes, with no significant adverse effects in terms of significant bleeding. We need to ensure that anticoagulation in ACS patients with AF is widely adopted to improve prognosis.
Introduction
Acute coronary syndrome (ACS) is the most important and life-threatening presentation of coronary artery disease. While the introduction of reperfusion therapy, as well as aggressive antithrombotic strategies, in contemporary cardiovascular medicine has led to a significant improvement in prognosis, the mortality rate remains significant.1–3 Atrial fibrillation (AF) is the most common cardiac arrhythmia. It has been shown that 2% of the general population of United States citizens under the age of 65 years have AF and the percentage reaches 9% in the elderly. 4 In Poland, the prevalence of AF in patients over 65 years of age is estimated to be 19.2%. 5 The current European and American AF guidelines universally recommend anticoagulation in patients with moderate and severe risk of ischemic stroke assessed by the CHA2DS2VASc score.6,7
The combination of these two conditions is common. It is reported that in subjects with ACS-related hospital admissions, AF occurs in 6.8–21% of cases.8,9 In AF patients, the annual rate of ACS is roughly 2.5%. 10 Given the ACS population is aging, it should be expected that the frequency of both ACS and AF will increase. The data on AF and concomitant ACS are scarce. Still, some of the recently published data suggest that the treatment of AF and ACS even in well-organized healthcare systems may be suboptimal, especially regarding the use of anticoagulation. 11
The ST-segment elevation myocardial infarction (STEMI) guidelines adopted the indication to introduce anticoagulation for patients with new-onset peri-MI AF, based on expert consensus. The data on long-term outcomes of anticoagulation in ACS patients with AF are lacking. Does the introduction of such treatment justify the potential increase of bleeding complications? We have decided to investigate the outcomes of ACS patients with concomitant AF with regard to stroke, myocardial infarction, bleeding complications, and all-cause mortality.
Materials and Methods
The Polish Registry of Acute Coronary Syndromes (PL-ACS) is a registry collecting comprehensive information on in-hospital MI treatment. Patients’ data are entered into a dedicated web-based form by attending physicians. The registry was created by the Silesian Center for Heart Diseases in Zabrze and the Polish Ministry of Health. Currently, over 720,000 hospitalizations for acute coronary syndromes are listed in the PL-ACS registry (2006–2019). Its design has been described previously. 12
The Acute Myocardial Infarction in Poland (AMI-PL) is a registry of administrative data on ACS from the national healthcare provider. The database currently gathers data from over 550,000 hospitalizations since 2009. The detailed design of AMI-PL has been presented previously. 13 AMI-PL data allow full access to cardiovascular events after hospital discharge. The SILesian CARDiovascular (SILCARD) database also integrates complete administrative data from the national healthcare provider on a regional basis, covering a population of 4.6 million patients and adding further follow-up information. Its structure has been previously described. 2
The combination of PL-ACS, AMI-PL, and SILCARD registries forms an all-comer population of ACS patients in Poland with complete in-hospital and post-discharge data.
For this study, we have selected a joint subpopulation of all-comer patients hospitalized for ACS with concomitant AF on admission. The patients included in the analysis were screened for the diagnosis of AF 5 years prior to inclusion. If such a diagnosis was present, they were excluded from further analysis. The joint analysis was performed on a per-patient basis and incorporated the data from 2006 to 2019. As 12-month endpoints were assessed, only survivors of the index hospitalization were included in the study.
All data were anonymized. Matching information concerning the individual patient was possible through the encoded national personal identification number. Subsequently, the patients were divided into subgroups reflecting the status of administered anticoagulation—(D)OAC+ group (n = 2,466) and (D)OAC− group (n = 10,611). The patients’ outcomes were analyzed in a 12-month follow-up period with regard to all-cause mortality, myocardial infarction, stroke, and bleeding complications.
As significant differences were observed in the group characteristics, a propensity score matching was performed. For the matching procedure, we have selected variables that created significant differences among the two study groups, as well as those that had previously proven prognostically significant in our analyses of the PL-ACS registry. The study groups were matched for age, sex, left ventricular ejection fraction, type of ACS, invasive strategy, history of heart failure, renal impairment, and diabetes. The 12-month outcomes were reanalyzed after matching. The protocols of analyzed registries were approved by the ethics committee.
Statistical analysis
Continuous variables are expressed as mean and standard deviation or median and interquartile range. Comparisons between groups were calculated with the use of the Student's t-test. The normality of data was checked with the Shapiro-Wilk test. Categorical variables were presented as frequencies and percentages, and the between-group differences for these variables were assessed using the χ2 test or Fisher exact test when appropriate. The association between the use of anticoagulant drugs and the 12-month all-cause mortality or myocardial infarction was analyzed by the Kaplan-Meier method with log-rank testing.
Moreover, propensity score matching was used to control for confounding bias. The groups were matched on a 1:1 ratio according to parameters/variables listed in the supplementary material, Figure S1. The propensity scores were calculated using a multiple logistic regression model. The greedy data matching procedure with Mahalanobis distance within the caliper radius set to 0.25×sigma was used. A P value < .05 was considered statistically significant. All analyses were performed with SAS version 9.4 (SAS Institute Inc., Cary, North Carolina, USA)
Results
Of the general PL-ACS registry population from the years 2006–2019, 7.1% of subjects had AF on admission. Vital status was available for the whole study cohort. Completeness of clinical follow-up (data on myocardial infarction, stroke, bleeding, or the administered antithrombotic regimen) was the main limitation of further analyses. After the exclusion of patients with incomplete follow-up, the sample cohort was formed (n = 13,077 patients).
Subsequently, the group was divided with regard to the administration of oral anticoagulation. Anticoagulation was prescribed on discharge in 2466 patients—the (D)OAC+ group (18.9%). The (D)OAC+ group consisted mostly of patients treated with vitamin K antagonists (87.48%).
The (D)OAC+ patients were significantly younger and more frequently men (61.3% vs 57.4%, P = .004). Notwithstanding, comorbidities such as diabetes (40.2% vs 36.9%, P = .002), heart failure (25.3% vs 16.5%, P < .001), chronic kidney disease (14.1% vs 11.6%, P < .001), and history of prior myocardial infarction (24.7% vs 22.1%, P = .005) were more prevalent in this group. The left ventricular ejection fraction (LVEF) was similar in both groups (43.1 ± 12.0 vs 43.4 ± 12.0, P = .278; Table 1). The 12-month follow-up showed that the (D)OAC+ patients had better prognosis with significantly lower rates of all-cause mortality (11.8% vs 19.4%, P < .001) MI (5.9% vs 7.5%, P = .003), and ischemic stroke (2.2% vs 4.0%, P < .001). However, there was no significant increase in intracranial bleeding (0.4% vs 0.3%, P = .159) and bleeding requiring transfusion and/or hospitalization (1.9% vs 1.7%, P = .537; Figure 1, Table 2).

Kaplan-Meier 12-month survival (A); myocardial infarction (B); ischemic stroke (C) and cumulative endpoint of death, stroke, or myocardial infarction (D) before propensity score matching and 12-month survival (E); myocardial infarction (F); ischemic stroke (G) and cumulative endpoint of death, stroke, or myocardial infarction (H) after propensity score matching. The mean survival times are available in the supplementary material (Table S1).
Patient Characteristics.
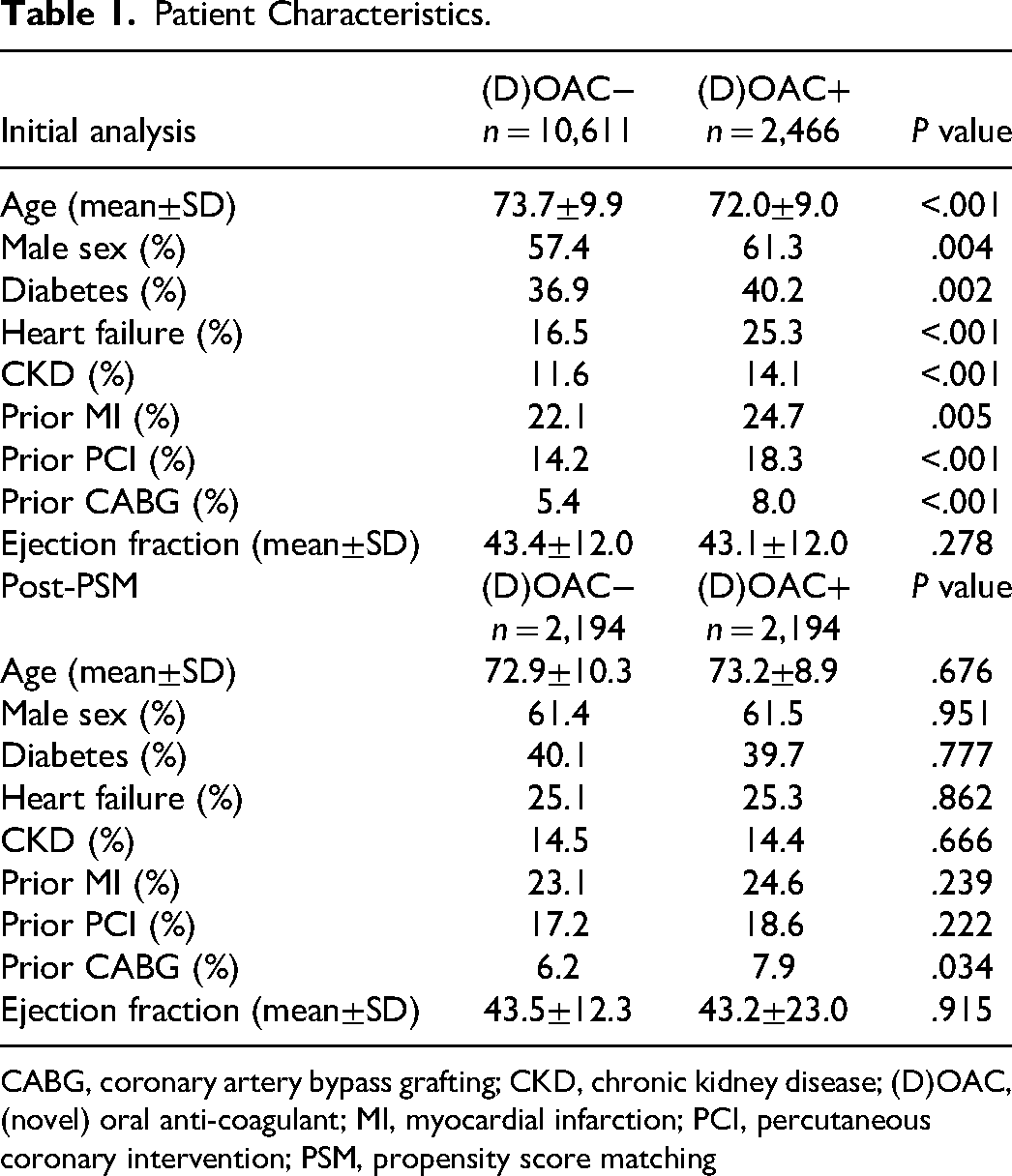
CABG, coronary artery bypass grafting; CKD, chronic kidney disease; (D)OAC, (novel) oral anti-coagulant; MI, myocardial infarction; PCI, percutaneous coronary intervention; PSM, propensity score matching
12-Month Follow-Up.

Legends. aFisher exact test.
In the initial analysis, significant differences in the type of ACS were observed—there were fewer STEMI in the (D)OAC− group (21.3% bs 28.9%, P < .001), while the frequency of coronary angiography and percutaneous coronary interventions was higher (92.4% vs 83.9%, P < .001). The differences in coronary angiography rates were seen in all types of ACS, while the differences in the rate of PCI were driven by minor discrepancies in all subgroups (Table 3).
Type of ACS and Diagnostic and Therapeutic Modalities.
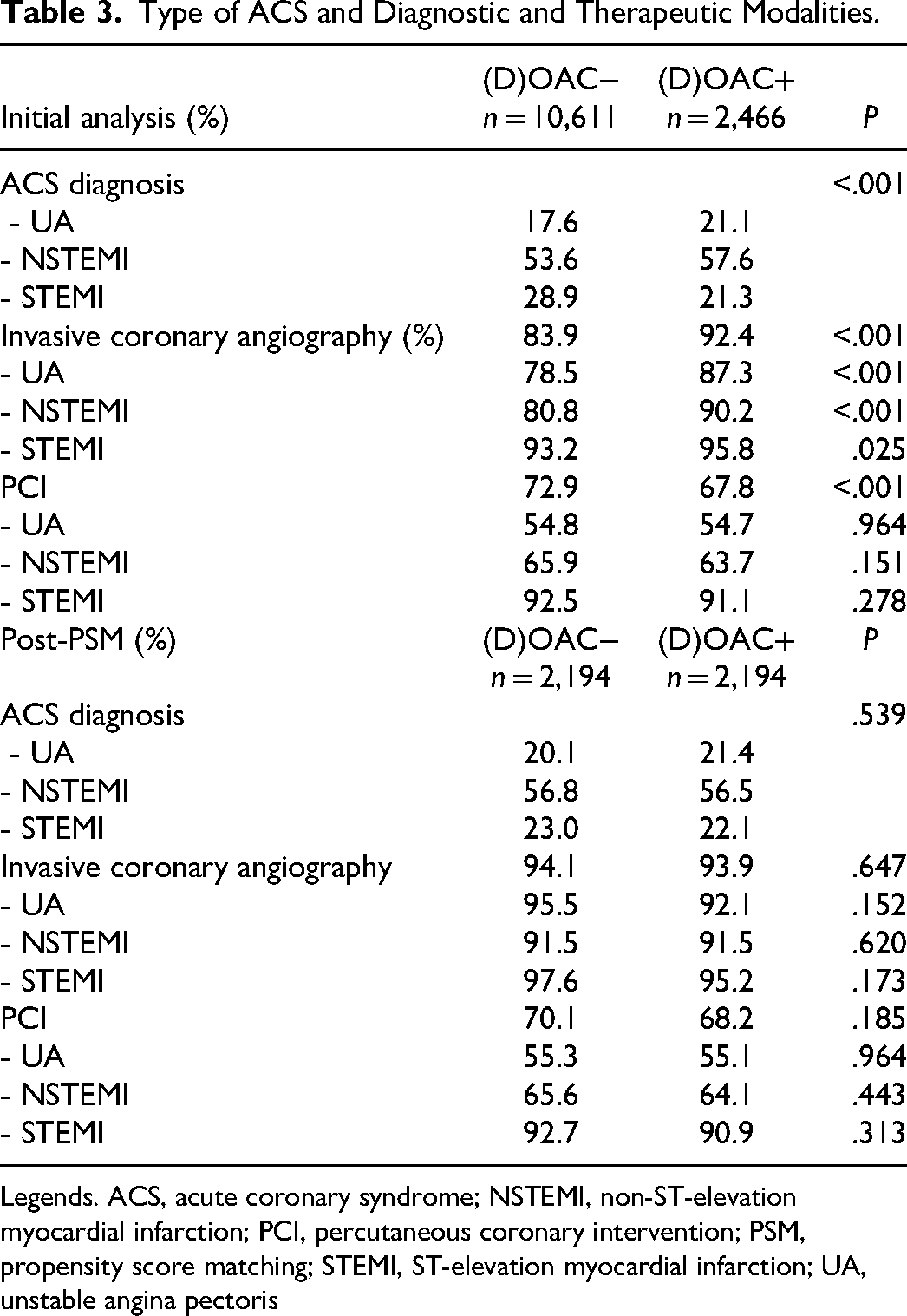
Legends. ACS, acute coronary syndrome; NSTEMI, non-ST-elevation myocardial infarction; PCI, percutaneous coronary intervention; PSM, propensity score matching; STEMI, ST-elevation myocardial infarction; UA, unstable angina pectoris
Moreover, apart from the analysis of prescribed anticoagulation, we have analyzed the prevalence of guideline-recommended antiplatelet therapy (Table 4). Dual antiplatelet therapy was administered on discharge in over 72% of cases (72.5% vs 73.9%, P = .155), respectively, with or without anticoagulation.
Prescribed Antiplatelet Treatment.

ASA, acetylsalicylic acid; DAPT, dual antiplatelet therapy; (D)OAC, (novel) oral anti-coagulant; P2Y12, inhibitors of P2Y12 receptor; PSM, propensity score matching.
Subsequently, the groups were propensity score-matched (PSM) for age, sex, ejection fraction, diabetes, heart failure, renal impairment, type of ACS, and coronary angiography. After matching, the study groups consisted of 2,194 patients each and showed no significant differences in baseline characteristics (Table 1). The CHA2DS2VASc score was calculated for all patients. The median score was similar in both cohorts [median 3 (interquartile range 2–4), P = .803]. The differences in 12-month outcomes after PSM were similar to the findings in the unmatched cohort. The 12-month all-cause mortality was significantly lower in the (D)OAC+ group (11.8% vs 17.3%, P < .001). The incidence of myocardial infarction in the (D)OAC+ group was significantly lower (5.7% vs 7.4%, P = .026). The incidence of stroke (both ischemic and hemorrhagic) was also lower in the (D)OAC+ group (3.3% vs 4.6%, P = .02). The differences in ischemic stroke frequency were more evident (2.3% vs 4.1% vs P < .001). The incidence of intracranial bleeding was very low in both study groups (0.5% vs 0.3%, P = .34). There were also no differences regarding bleeding requiring transfusion and/or hospitalization (2.1% vs 1.7%, P = .37). The frequency of a composite endpoint of death, stroke, or myocardial infarction was lower by 7.5% in the (D)OAC+ group (18.0% vs 25.5%, P < .001). Detailed data on follow-up before and after propensity score matching are presented in Figure 1 and Table 2. Of note, after PSM, there were no differences with regard to the diagnosed type of ACS as well as no differences in the invasive diagnostics and PCI, both for the whole study cohort (coronary angiography 93.9% vs 94.1%, P = .647; PCI 68.2% vs. 70.1%, P = .185) and in per ASC type analysis (Table 3). The comparison of the administered antiplatelet treatment was similar to the pre-PSM analysis (Table 4).
Discussion
The current MI and AF guidelines consistently support the notion of anticoagulation in AF patients with medium-to-high ischemic risk as assessed by the CHA2DS2-VASc score.6,7 As MI is part of this classification, in everyday clinical practice, almost all AF patients after MI should receive anticoagulation. However, reports in this area suggest that this guideline is not strictly followed in clinical practice. A recently published analysis from the Norwegian NORMI database revealed that among CHA2DS2-VASc 2 and more MI survivors, 56.3% were prescribed (D)OACs. 11 The 2017 ESC STEMI guidelines became the first to recommend anticoagulation for new-onset peri-MI AF patients; however, it was supported by expert consensus rather than definite clinical data. 14
The risk of bleeding complications has become an essential issue in contemporary cardiovascular medicine. Consecutive studies prove that the shortening of dual or triple antithrombotic therapy time is safe.15,16 Novel devices are created to reduce the necessity of antithrombotic regimens.
Nonetheless, the percentage of MI survivors with diagnosed AF receiving anticoagulation in our analysis is low. The most plausible justification for this phenomenon is the timeframe of the analysis, reaching a period preceding current guidelines that introduced stringent anticoagulation in AF. This is also observed in other available data, such as the SWEDEHEART registry, where a rapid growth of anticoagulation in similar populations is apparent only after 2011. 16 Moreover, the same SWEDEHEART data show that the distribution of specific antithrombotic regimens (single, dual, or triple antithrombotic therapy with or without anticoagulation) was similar to this observed in our population.
In addition, the insufficient use of anticoagulation may be caused by the concept that new-onset peri-ACS AF results from reversible ischemia. Sometimes physicians believe that the resolution of ischemia resolves at the same time the problem of arrhythmias. As such, similar results were published by Batra et al., showing that only 23% of all myocardial infarction AF patients received anticoagulation. 17 As mentioned above, only recently have the STEMI guidelines adopted the indication to introduce long-term anticoagulation in patients with new-onset peri-MI AF with class IIa C recommendation. 14 This expert consensus was driven by data that new-onset AF in STEMI patients significantly impairs prognosis, while the benefit of anticoagulation in this scenario is yet to be proven.18,19 Only a small proportion of our study population was treated according to these guidelines—both given the timeframe and the percentage of STEMI patients.
With all these considerations in mind, the main goal of our analysis was to assess the impact of anticoagulation on the 12-month outcomes in ACS survivors with AF. The initial analysis showed significant differences in prognosis with a nearly twofold lower all-cause mortality, significant reduction in myocardial infarction, and all cerebrovascular incidents—driven by the lower occurrence of ischemic stroke—in the (D)OAC+ cohort. However, there were no significant differences in major bleeding and intracranial bleeding, with low frequencies observed in both study groups. The occurrence of bleeding in our study was very similar to that presented in randomized trials and registries.18,20–23
The major differences in the baseline characteristics of the study groups led to a decision to perform propensity score matching. After propensity score matching, the analyzed cohorts were comparable with regard to the clinical characteristics and types of ACS, as well as the frequency of invasive strategy and coronary angioplasty procedures. The comparison of outcomes again showed very similar results, with a clear benefit of anticoagulation. The all-cause mortality remained significantly lower in the (D)OAC+ group as was the recurrence of MI. The number of all cerebrovascular incidents was again significantly lower due to the lower incidence of ischemic stroke—the frequency of which was similar to other reports. 24 Post-PSM an anticipated trend toward more intracranial bleeding was observed in the (D)OAC+ group. The number of these events was very low in both cohorts. In our population, the majority of subjects received VKA. Although the predominant use of VKA in our study may be considered a limitation, we believe that with even safer novel anticoagulants, the results may be extended to the contemporary patient population. The results of ROCKET AF, RELY, and ARISTOTLE trials suggest that these results might have been better in the (D)OAC+ group if the direct oral anticoagulants (DOACs) were used more frequently.20–23
The most significant finding of our analysis is the difference in the prevalence of myocardial infarction during the 12-month follow-up. The Kaplan-Meier curves show that the influence of anticoagulation on this outcome became significant after roughly 100 days and the difference increased over time. As the administration of anticoagulation blocks additional pathways of the coagulation system, a reduction of ischemic incidents in the long term should be expected. The composite endpoint of death, myocardial infarction, or stroke showed abundantly better prognosis in patients on anticoagulation. In recent years, the benefit of additional anticoagulation in patients with coronary artery disease was shown in the COMPASS trial. 25 Perhaps there is still potential for further studies on additional anticoagulation in patients after ACS.
Study Limitations
This analysis of combined data from two nationwide registries and one regional registry has typical limitations of retrospective analyses of prospective data. Not all ACS cases were registered in the PL-ACS registry. Pre-existing AF may not have been properly diagnosed and reported to the healthcare provider. The follow-up was based on administrative data with no additional checks performed; thus, some human mistakes were possible. Selecting only survivors of the index hospitalization may have some impact on results. During the follow-up, the same patients could permanently migrate and be lost to follow-up. However, we consider these effects to be small. These limitations do not apply to mortality status, available for all cases, independently of the place of residence. No data were available on the further course of AF during hospitalization and after discharge. The analyzed differences in antithrombotic regimens were analyzed as prescribed at discharge, but data on compliance were unavailable. The long timeframe of study inclusion was another study limitation. Moreover, with a limited population of patients on novel anticoagulants, the authors were unable to compare the impact of vitamin K antagonists and DOACs in this analysis. The study thus concentrated on the impact of anticoagulation in general, not specific treatment regimens. Although we aimed to present comparable patient populations, there are limitations to PSM. PSM is effective, yet not a perfect solution for causal inference and has drawbacks. It discards some subjects who are not matched, reducing the statistical power of the analysis. Additionally, PSM relies on the assumption of conditional independence, which may not always be valid, particularly when there are unobserved confounders that influence both the treatment and the outcome.
Conclusion
To the best of our knowledge, this all-comer registry analysis shows for the first time that the use of guideline-recommended anticoagulation in ACS survivors with concomitant AF is related to improved outcomes—lower all-cause mortality, lower occurrence of ischemic stroke, myocardial infarction, and the composite endpoint of death, myocardial infarction, and stroke. In our analysis, the use of such treatment did not increase the risk of bleeding, especially with regard to the most feared complication of such treatment—intracranial bleeding. We strongly believe that these findings advocate the clinical benefit and safety of anticoagulation in the analyzed clinical scenario.
Supplemental Material
sj-pdf-1-cpt-10.1177_10742484231212106 - Supplemental material for Anticoagulation for Atrial Fibrillation in Acute Coronary Syndrome Survivors Reduces Major Cardiovascular Events and Mortality
Supplemental material, sj-pdf-1-cpt-10.1177_10742484231212106 for Anticoagulation for Atrial Fibrillation in Acute Coronary Syndrome Survivors Reduces Major Cardiovascular Events and Mortality by Łukasz Pyka, Bartosz Hudzik, Stanisław Bartuś, Paweł Buszman, Marek Gierlotka, Wojciech Wojakowski, Jarosław Hiczkiewicz, Andrzej Kleinrok, Michał Skrzypek, Wiktor Kuliczkowski and Mariusz Gąsior in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
sj-docx-2-cpt-10.1177_10742484231212106 - Supplemental material for Anticoagulation for Atrial Fibrillation in Acute Coronary Syndrome Survivors Reduces Major Cardiovascular Events and Mortality
Supplemental material, sj-docx-2-cpt-10.1177_10742484231212106 for Anticoagulation for Atrial Fibrillation in Acute Coronary Syndrome Survivors Reduces Major Cardiovascular Events and Mortality by Łukasz Pyka, Bartosz Hudzik, Stanisław Bartuś, Paweł Buszman, Marek Gierlotka, Wojciech Wojakowski, Jarosław Hiczkiewicz, Andrzej Kleinrok, Michał Skrzypek, Wiktor Kuliczkowski and Mariusz Gąsior in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
