Abstract
Background and Objectives:
Clopidogrel is widely used after the percutaneous coronary intervention (PCI) in patients with acute coronary syndrome (ACS) and requires activation by cytochrome P450 (CYP), primarily CYP2C19. Patients with CYP2C19 loss-of-function alleles are at increased risk of major adverse cardiovascular events, while more expensive novel antiplatelet agents (ticagrelor and prasugrel) are unaffected by the CYP2C19 mutations. This systematic review aims to answer the question about whether overall evidence supports the genotype-guided selection of antiplatelet therapy as a cost-effective strategy in post-PCI ACS.
Methods:
A systematic literature search of PubMed, EMBASE, EconLit, and PharmGKB was done to identify all the economic evaluations related to genotype-guided therapy compared to the universal use of antiplatelets in ACS patients. Quality of Health Economic Studies tool was used for quality assessment.
Results:
The search identified 13 articles, where genotype-guided treatment was compared to universal clopidogrel, ticagrelor, and/or prasugrel. Six studies showed that genotype-guided therapy was cost-effective compared to universal clopidogrel, while 5 studies showed that it was dominant. One study specified that genotype-guided with ticagrelor is cost-effective only in both CYP2C19 intermediate and poor metabolizers. Genotype-guided therapy was dominant when compared to universal prasugrel, ticagrelor, or both in 5, 1, and 3 studies, respectively. Only 2 studies reported that universal ticagrelor was cost-effective compared to genotype-guided treatment. All the included articles had good quality.
Conclusion:
Based on current economic evaluations in the literature, implementing CYP2C19 genotype-guided therapy is a cost-effective approach in guiding the selection of medication in patients with ACS undergoing PCI.
Keywords
Introduction
The dual antiplatelet therapy, via the use of aspirin and an P2Y12 inhibitor (ie, clopidogrel, prasugrel, or ticagrelor), has several indications, including the prevention of recurrent thrombotic events, such as myocardial infarction (MI) and stent thrombosis, in patients with acute coronary syndrome (ACS) undergoing percutaneous coronary intervention (PCI). 1 It has been estimated that in the United States alone 1.1 million ACS events occur every year, 50% of which are treated with a PCI, making the choice of antiplatelet therapy a common and important clinical decision. 2
Clopidogrel has been the mainstay of therapy after PCI for almost two decades. 2 It was reported to be the second highest-selling medication worldwide in 2010 with US$9.4 billion in sales. 3 Clopidogrel requires biotransformation to its active metabolites through cytochrome P450 (CYP), primarily CYP2C19 which demonstrated to have the most pronounced effect on clopidogrel activation. There are several variants, that is, alleles, of the loss-of-function (LOF) CYP2C19 mutation, but the most common, as reported in the different populations, are the *2 and *3 variants. Several clinical studies have demonstrated that patients with LOF CYP2C19*2 or *3 genetic variants are at increased risk of major adverse cardiovascular events (MACEs). 4,5 This is because of the impaired activation of clopidogrel into its active metabolite leading to diminished inhibition of platelet aggregation compared to clopidogrel patients without CYP2C19 mutations. 4,5 On the other hand, *17 is another common CYP2C19 genetic variant that leads to gain in the enzymatic function. Consequences of *17 on clinical outcomes (possibly increased risk of bleeding and reduced risk of MACE), however, are controversial and do not have yet a clinical implementation. 6
Alternative antiplatelets, including ticagrelor and prasugrel, demonstrated an improved efficacy in reducing the rate of MI, stroke, and cardiovascular (CV) death in patients with ACS undergoing PCI, but on the expense of a higher risk of bleeding when compared to clopidogrel. 6 -8 In addition, alternative antiplatelets are proven to be unaffected by CYP2C19 mutations, leading to less inter-individual variability. 9,10 This advantage is crucial since CYP2C19*2 genetic variant was reported to be common in approximately 15%, 15%, and 29% to 35% of the Caucasians, Africans, and Asians, respectively, while CYP2C*3 have frequencies of less than 1%, except in Asians where prevalence can range from 2% to 9% of the population. 11 According to the identified CYP2C19 genotypes, individuals are usually categorized into ultrarapid metabolizers (ie, *1/*17 and *17/*17), extensive metabolizers (ie, *1/*1), intermediate metabolizers (eg, *1/*2 and *1/*3), or poor metabolizers (eg, *2/*2 and *2/*3). 11,12 On the other hand, alternative antiplatelets can be up to 20 times more expensive than clopidogrel, in addition to the secondary cost that is associated with more frequent bleeding complications. 13
Pharmacogenetic-guided testing is used to identify patients with a potential lack of efficacy or high risk of adverse events to medications. 14,15 Preemptive identification of CYP2C19*2 or *3 carrier status might allow personalization of antiplatelet therapy so that the new, more expensive alternative antiplatelets could be selectively prescribed to patients that will not benefit from clopidogrel (those with the LOF mutations), while clopidogrel would be prescribed to the rest of the patients. 16 However, the added value of the genetic testing may not worth the value of money spent on the testing. The real cost of genetic testing goes beyond the apparent acquisition cost of the test and the value of resources utilized in running it. Since the approval of prasugrel and ticagrelor, many economic evaluation studies, specifically over the past 5 years, were conducted to determine the cost-effectiveness of applying genotype-guided antiplatelet therapy, which come from different perspectives and in relation to different interventions. The speed and volume of information production create barriers against keeping up to date with potentially contrasting information. In this light, systematic reviews are important in synthesizing a summary of the currently available evidence provided by available primary research in response to a specific question. This is needed for the better guidance for health-care professionals, managers, researchers, and patients for both decision-making and the guiding and improving of future relevant research. Here, within the context applying genotype-guided antiplatelet therapy, there are no systematic reviews in the literature that answer the question about whether the overall evidence supports the genotype-guided antiplatelet therapy as a cost-effective strategy when compared to the universal use of antiplatelet therapy after PCI in patients with ACS. Accordingly, the objective of this systematic review is to generate a summative conclusion of the published economic evaluations of genotype-guided antiplatelet therapy in patients with PCI, including a quality assessment of the published research.
Methods
Data Sources and Search
A systematic literature review was performed using 4 search databases: PubMed, EMBASE, PharmGKB, and EconLit, to identify relevant articles. The search strategy followed the PICO format. As an example, within the PubMed database, the population was ACS and PCI; the intervention/comparator was genotype-guided, genetic guided, genetic diagnostic test, genomic diagnostic test, ticagrelor, prasugrel, clopidogrel, and platelet aggregation inhibitors; the outcome was cost-effectiveness, cost-benefit, costs and cost analysis, and economic evaluation. For other databases, a similar search approach was utilized. Keywords were customized to database-specific indexing terms, for example, the use of Medical Subject Heading terms. As appropriate, the terms and their substitutes were combined using Boolean connectors (AND/OR/NOT). PubMed and EMBASE search terms can be seen in Supplement 1. In addition to the electronic search, a manual search of bibliographies and references of identified articles was conducted. The grey literature was also searched using advanced and technical reports, conference proceeding, theses, conference proceeding, and guidelines, as well as searching Google Scholar. A search protocol of the systematic review was developed and registered in PROSPERO (ID#CRD42019133599).
Study Population
Included patients were those having ACS and receiving clopidogrel, ticagrelor, or prasugrel after PCI. Whereas the excluded patients were those receiving clopidogrel for other indications, such as stroke prevention, peripheral artery disease, or ACS without PCI.
Eligibility Criteria
Eligible studies for this review were economic evaluation studies comparing genetic testing for ACS patients with PCI, followed by targeted administration of the alternative antiplatelets in CYP2C19 mutation carriers compared to the universal use of antiplatelets. Included comparative studies were restricted to the English language of human species and in journal articles with full-text availability. Exclusion criteria included reviews and non-comparative studies. The inclusion and exclusion of articles were conducted by two independent reviewers via the initial screening of titles/abstracts of articles, before a follow-up screening of the full text. When disagreements occurred, articles were discussed with a third reviewer author until consensus. The inclusion of comparative studies was from inception until August 2019.
Outcome Measures
The outcome measure of interest is the observed trends in relation to the comparative economic outcomes of the CYP2C19 genotype-guided antiplatelet therapy versus universal use of antiplatelet, including total cost, cost avoidance, and the incremental cost-effectiveness ratio (ICER).
Data Extraction and Synthesis
A data extraction tool was developed, and pilot testing was performed using a sample of the eligible studies (n = 3). The extracted data were the genetic-guided antiplatelet therapy, antiplatelet therapy used, country, disease states, time horizon, primary and secondary outcomes, funding, the comparative model used, type of cost-effectiveness analysis, the source of clinical and cost data, perspective, type of cost, uncertainty tests, time adjustment, and summary of findings. Two reviewers independently extracted the data from the included articles, ensuring data reliability and trustworthiness. A consensus was reached whenever differences occurred.
Assessment of Quality of Studies
The quality of articles was assessed by two independent reviewers, who critically appraised the included articles to assess the risk of bias and methodological quality. For this purpose, the Quality of Health Economic Studies (QHES) tool was used. 17 The QHES tool includes 16 questions; each question is associated with a potential score ranging from “0” to “9,” with the total of all questions’ scores adding to 100 points. The interpretation of a QHES score was as per 1 of the 4 categories of quality, that is, good, fair, poor, and extremely poor, associated with the scores 75 to 100, 50 to 74, 25 to 49, and 0 to 24, respectively. 18,19 A third independent reviewer would contribute whenever a disagreement occurs.
Only articles with fair or good methodological quality were included in this review. The systematic review followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guideline, including a 27-item checklist of the essential items to be reported in a systematic review, as relevant (Supplement 2). 20
Results
Study Selection
Of 204 retrieved articles, 13 articles met the inclusion criteria. The search results are shown in Figure1.
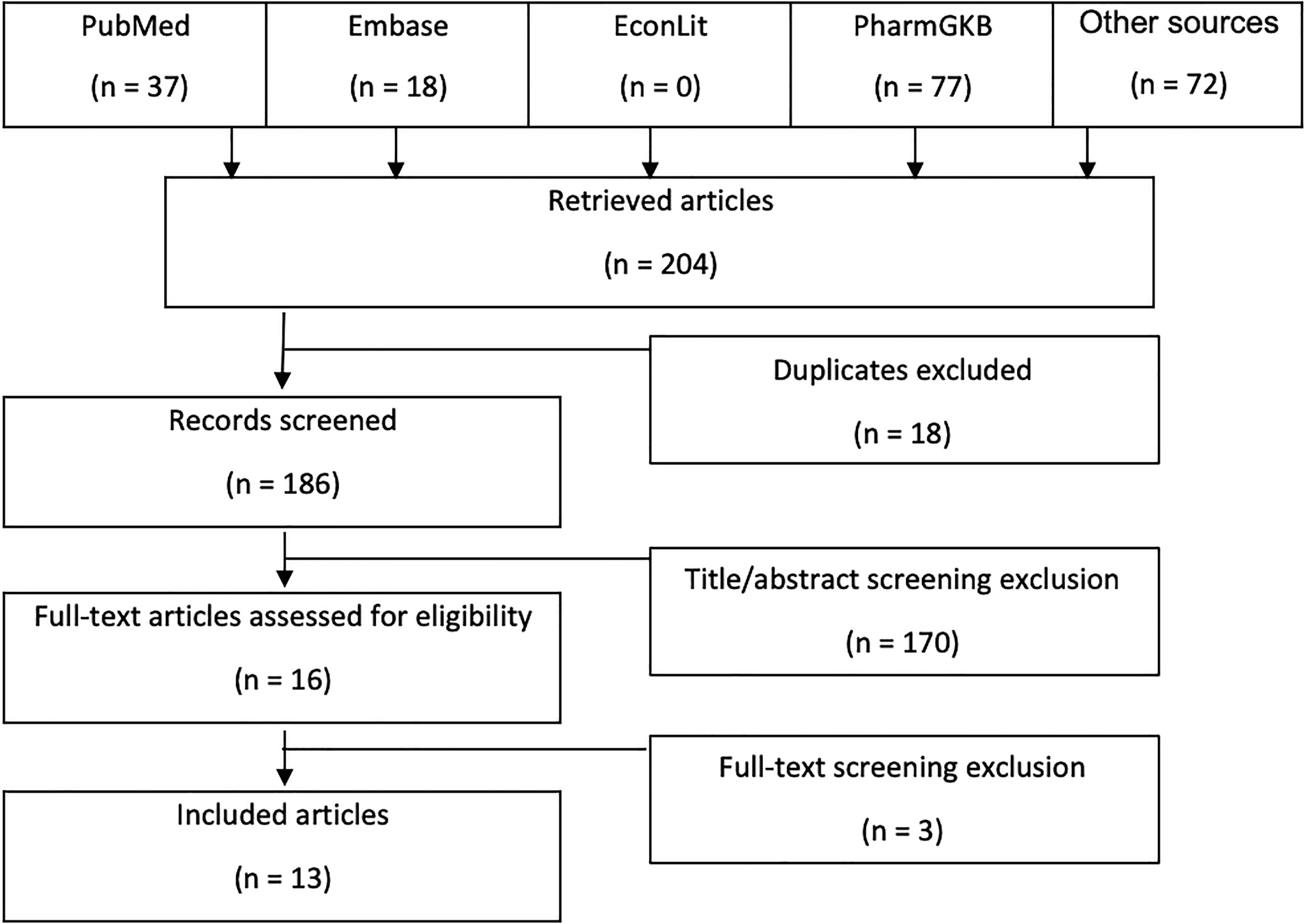
Flow diagram of the literature search and selection.
Characteristics of the Included Articles
In patients with ACS undergoing PCI, genotype-guided therapy with prasugrel was compared to universal clopidogrel or prasugrel in five articles. 21 -25 Genotype-guided therapy with ticagrelor was compared to universal clopidogrel or ticagrelor in four articles. 26 -29 Four articles evaluated the genotype-guided use of the P2Y12 inhibitors ticagrelor or prasugrel as compared against the universal use of P2Y12 inhibitors, that is, clopidogrel, ticagrelor, or prasugrel. 30 -33 In general, key clinical effectiveness data in the majority of the studied comparative models were derived from the 2 landmark clinical trials of antiplatelet therapy in patients with ACS; the TRITION-TIMI 38 clinical trial (prasugrel vs clopidogrel) and the PLATO clinical trial (ticagrelor vs clopidogrel). 6 ,7 The most commonly studied LOF allele was CYP2C19*2, with others being *3, *4, *5, *6, *7, and *8. All the studies considered the use of an alternative to clopidogrel in the genotype-guided arm whenever there was LOF allele, regardless of whether it was homozygote (two copies of the LOF allele) or heterozygote (one copy of the LOF allele). Among the 13 included studies, seven were conducted in the United States, 21,22,24 -27,33 four in China, 29 -32 one in Australia, 28 and one in New Zeeland. 23 Characteristics of the included studies are summarized in Table 1.
Characteristics of the Included Articles.
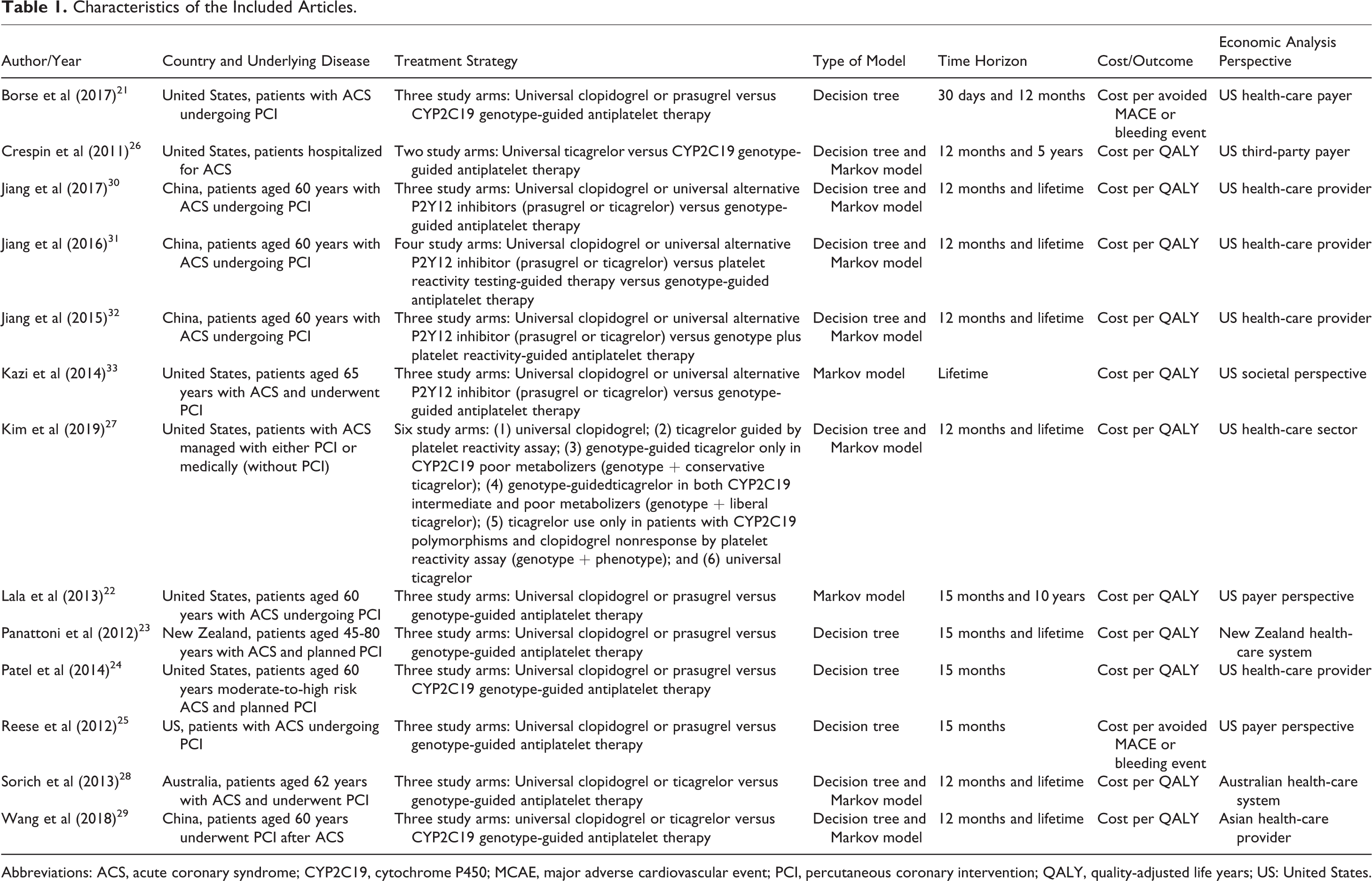
Abbreviations: ACS, acute coronary syndrome; CYP2C19, cytochrome P450; MCAE, major adverse cardiovascular event; PCI, percutaneous coronary intervention; QALY, quality-adjusted life years; US: United States.
All included articles applied decision-analytic models (basic decision tree and/or the Markov model) that followed a hypothetical cohorts of patients over follow-up periods of 12 months, 15 months, and/or lifetime of patients. All economic evaluation studies analyzed direct medical costs. A cost-utility analysis was performed in 11 of 13 studies, and the outcome measure used in these studies was cost/quality-adjusted life-year (QALY). 22 -24,26 -33 The other two studies conducted a cost-effectiveness analysis, and the outcome measure was cost/avoided CV or bleeding event. 21,25 All studies evaluated the generic clopidogrel and branded ticagrelor or prasugrel at the base-case of their analyses. Only four articles reported the genotyping methods, in which Wang et al and Crespin et al utilized conventional genotyping, 26,29 whereas Kazi et al and Kim et al utilized point-of-care genotyping. 27,33
Genotype-Guided Therapy With Prasugrel Versus Universal Use of Prasugrel or Clopidogrel
Clinical and cost outcomes
In the genotype-guided arm, patients with LOF alleles received prasugrel, while patients without LOF alleles received clopidogrel. 21 -25 All studies reported MACE (ie, composite of nonfatal stroke, nonfatal MI, and CV death), as well as major bleeding as the primary clinical outcome measures. Only Borse et al and Panattoni et al reported stent thrombosis in addition to MACE and major bleeding. 21,23 The modeled clinical events in the included cost-effectiveness studies were consistent, as all five studies used TRITON-TIMI 38 and genetic sub-studies to obtain the base-case probabilities. Only Panattoni et al used New Zealand’s public hospitals’ reports in addition to the TRITON-TIMI 38 trial to obtain the clinical effectiveness data. 23
Generally, multiple sources of cost data were used to describe the cost of medications, genetic testing, hospitalization, outpatient visits, and adverse events. Medicare reimbursement rates and local published data were commonly used among the five articles. 21 -25 For the purpose of cost adjustment according to time, Lala et al and Panattoni et al discounted cost and utilities, 22,23 while Patel et al and Reese et al discounted cost only. 24,25
Cost-effectiveness results
Base-case results by Borse et al, Patel et al, Panattoni et al, and Reese et al showed that genotype-guided treatment was cost-effective compared to universal clopidogrel (ICER/MACE or bleeding event avoided of US$50 308; ICER/QALY gained of US$4200; NZ$24 617; and US$2300, respectively) and dominant when compared to universal prasugrel. 21,23 -25 Lala et al stated that genotype-guided treatment was dominant compared to both universal prasugrel and clopidogrel. 22 As illustrated in Table 2, all studies performed sensitivity analyses to assess the robustness of the results.
Base-Case and Sensitivity Analysis Results for Genotype-Guided Therapy With Prasugrel Versus Universal Use of Prasugrel or Clopidogrel.a
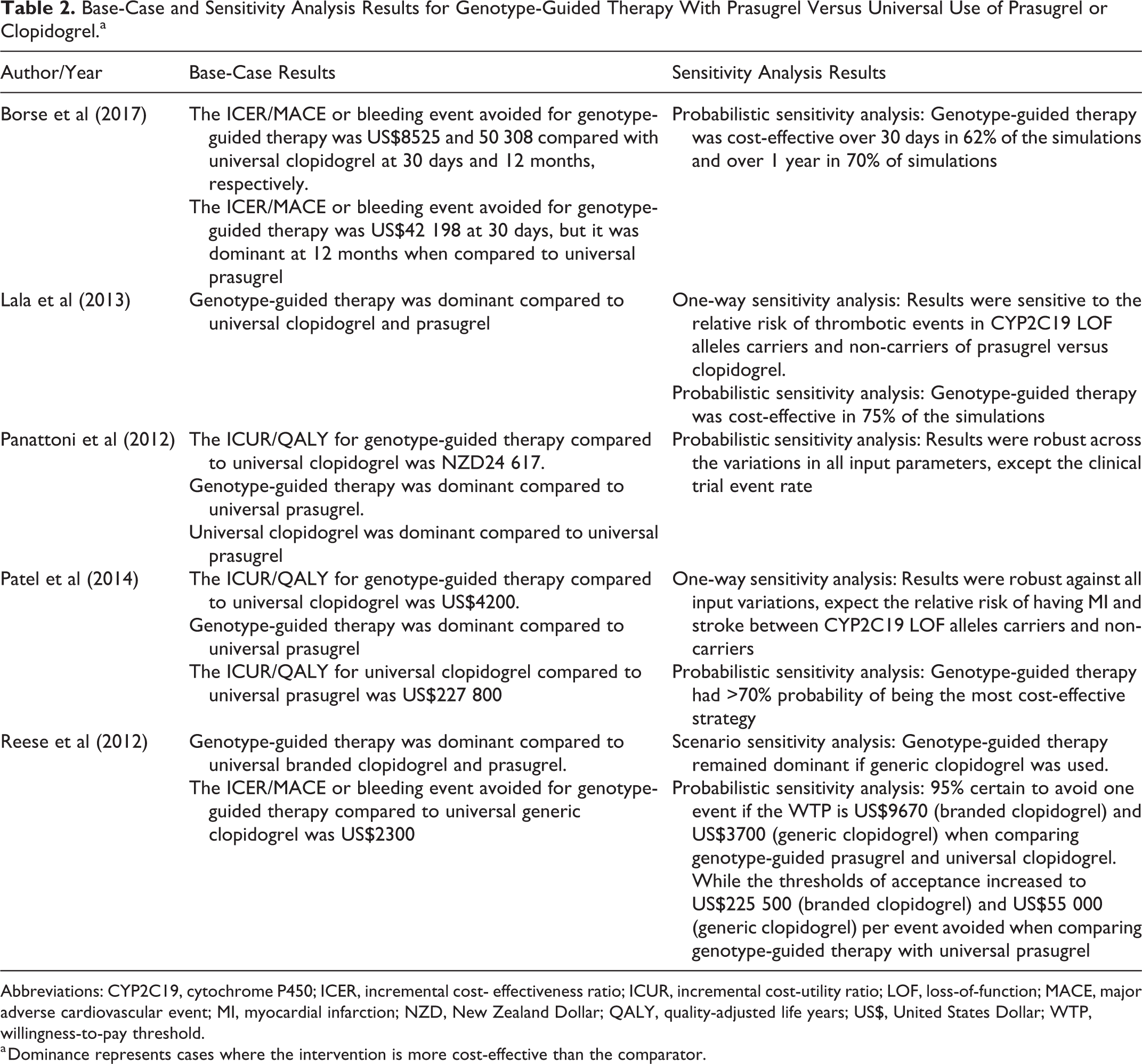
Abbreviations: CYP2C19, cytochrome P450; ICER, incremental cost- effectiveness ratio; ICUR, incremental cost-utility ratio; LOF, loss-of-function; MACE, major adverse cardiovascular event; MI, myocardial infarction; NZD, New Zealand Dollar; QALY, quality-adjusted life years; US$, United States Dollar; WTP, willingness-to-pay threshold.
a Dominance represents cases where the intervention is more cost-effective than the comparator.
Genotype-Guided Therapy With Ticagrelor Versus Universal Use of Ticagrelor or Clopidogrel
Clinical and cost outcomes
In the genotype-guided arm, patients with LOF alleles received ticagrelor, whereas patients without LOF alleles received clopidogrel. 26 -29 Sorich et al examined MACE; Wang et al examined MACE as well as stent thrombosis and fatal bleeding; Crespin et al evaluated MI, bleeding, dyspnea, and death; and Kim et al examined MI, ischemic stroke, intracranial hemorrhage, major bleeding, and death. 26 -29 Three articles used similar sources of clinical effectiveness events including the PLATO trial and sub-studies, 26 -28 whereas Crespin et al and Kim et al used the Medicare program and a meta-analysis by Mega et al, 4 respectively, in addition to the PLATO trial. 26,27 On the other hand, Wang et al relied on local observational studies done in Asian populations. 29
Wang et al described the costs of medications, inpatient, outpatient, and CYP2C19 genotype testing that were based on public hospital formulary, locally published data and international experience. 29 Crespin et al and Kim et al presented the cost of resources used after ACS diagnosis, including the cost of the genetic test, medications, ACS events, hospitalization, and subsequent monthly ACS care. 26,27 Crespin et al derived the cost data from the Medicare program and sources available in the literature, whereas Kim et al calculated the cost from the wholesale price available from RedBook, national statistics, and previous reports in the literature. Costs and health outcomes were discounted in all articles. 26 -29
Cos-effectiveness results
Base-case analyses by Wang et al and Sorich et al illustrated that genotype-guided treatment with ticagrelor was cost-effective compared to universal clopidogrel (ICER/QALY of US$2560 and AU$6000, respectively). 28,29 Wang et al concluded that the genotype-guided therapy was dominant compared to universal ticagrelor. 29 On the contrary, Sorich et al and Crespin et al demonstrated that universal ticagrelor was cost-effective compared to genotype-guided treatment with an ICER/QALY of AU$23 000 and US$10 059, respectively. 26,28 Kim et al showed that genotype-guided treatment with ticagrelor in both CYP2C19 intermediate and poor metabolizers (genotype + liberal ticagrelor) was a cost-effective strategy with an ICER/QALY of US$29 412, whereas genotype-guided treatment with ticagrelor only in CYP2C19 poor metabolizers (genotype + conservative ticagrelor) was not a cost-effective strategy. 27 All studies performed sensitivity analyses, with the results presented in Table 3.
Base-Case and Sensitivity Analysis Results for Genotype-Guided Therapy With Ticagrelor Versus Universal Use of Ticagrelor or Clopidogrel.

Abbreviations: AUD: Australian Dollar; CYP2C19, cytochrome P450; ICER: incremental cost-effectiveness ratio; QALY: quality-adjusted life years; US$: United States Dollar; WTP: willingness-to-pay threshold.
Dominance represents cases where the intervention is more cost-effective than the comparator.
Genotype-Guided Therapy With Alternative P2Y12 Inhibitors (Prasugrel and Ticagrelor) Versus Universal Use of Alternative P2Y12 Inhibitors or Clopidogrel
Clinical and cost outcomes
Unlike the studies mentioned above where either prasugrel or ticagrelor is evaluated, four studies assessed both medications in a single study group. 30 -33 In the genotype-guided group, patients with LOF alleles received either prasugrel or ticagrelor, whereas patients without LOF alleles received clopidogrel. Only one article used both CYP2C19 genotype plus platelet reactivity-guided antiplatelet therapy as the intervention. 32 Jiang et al (2017), Jiang et al (2016), and Jiang et al (2015) evaluated MACE, major bleeding and stent thrombosis, while Kazi et al assessed only MACE and bleeding. 30 -32 Generally, TRITON-TIMI 38 trial, PLATO trial, and meta-analyses were the main sources of clinical effectiveness data in the four articles. 30 -33
Cost of clinical outcomes, adverse drug reactions, medications, and genetic testing were estimated from inpatient diagnosis-related group data, retail prices, claims databases, and published economic data. 30 -33 Costs and health outcomes were discounted in all articles. 30 -33
Cost-effectiveness results
Base-case results illustrated that the genotype-guided treatment was the dominant strategy compared to both universal clopidogrel and P2Y12 inhibitors. 30 -32 Kazi et al concluded that genotype-guided therapy with ticagrelor was the most cost-effective strategy. 33 Jiang et al (2017), Jiang et al (2016), and Jiang et al (2015) showed that universal use of alternative P2Y12 inhibitors was cost-effective compared to universal clopidogrel with an ICER/QALY of US$28 542, US$43 683, and US$139 588, respectively. 30 -32 In contrast, Kazi et al explained that universal clopidogrel was dominant compared to universal prasugrel, while universal ticagrelor was cost-effective compared to the universal clopidogrel with an ICER/QALY of US$40 300. 33 The four articles performed sensitivity analyses and the results are summarized in Table 4.
Base-Case and Sensitivity Analysis Results for Genotype-Guided Therapy With Alternative P2Y12 Inhibitors (Prasugrel and Ticagrelor) Versus Universal Use of Alternative P2Y12 Inhibitors or Clopidogrel.a

Abbreviations: ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life years; US$, United States Dollar.
a Dominance represents cases where the intervention is more cost-effective than the comparator.
Cost outcomes of all studies, adjusted to the US$ and the 2019/2020 financial year, can be seen in Supplement 3.
Quality Assessment Results
All studies were good in quality and none performed poorly. Table 5 presents the results of the quality assessment of the included articles using the QHES tool. Detailed QHES results can be seen in Supplement 4.
Quality Assessment; QHES Tool.

Abbreviation: QHES: Quality of Health Economic Studies.
Discussion
The present study is the first systematic review that summarizes the cost-effectiveness studies of genotype-guided antiplatelet therapy compared to the universal use of antiplatelet therapy in patients with ACS undergoing PCI. To the best of our knowledge, there are no systematic reviews with similar objectives, comprehensiveness, and scope, specifically focusing on CYP2C19 genetic testing. In 2016, a systematic review of economic evaluations of pharmacogenetic testing for guiding therapies was reported, but it was not drug specific, nor disease/gene specific, and was focusing primarily on the prevention of adverse drug reactions of therapies. Importantly, the review only included four articles that evaluated clopidogrel, with the most recent being in 2013. 34 The systematic review concluded that high-quality evidence supported the cost-effectiveness of CYP2C19 genotype-guided antiplatelet therapy. 34 Another review of relevance was a 2015 one. 35 However, it was narrative in nature and included only seven studies published between 2000 and 2014, where authors did not assess the quality of the retrieved studies. 35 The review illustrated that while the genotype-guided use of prasugrel compared to the universal use of prasugrel or clopidogrel was a cost-effective strategy, results in support of the genotype-guided use of ticagrelor compared to the universal use of ticagrelor or clopidogrel were inconsistent. 35
The choice of antiplatelet therapy after PCI in patients with ACS is complicated. This choice has radical clinical and economic implications and entails a marked trade-off between differences in drug costs, thrombotic events, and major bleeding. The randomized clinical trial by Claassens et al demonstrated a clinical benefit of CYP2C19 genotype-guided antiplatelet therapy. After 12 months, CYP2C19 genotype-guided antiplatelet strategy was non-inferior to universal administration of oral P2Y12 inhibitor in relation to thrombotic events and was associated with a reduced incidence of bleeding. 36 Unlike other studies included in this systematic review, the study done by Crespin et al estimated the costs and outcomes for a cohort of ACS patients, without specifying if they underwent PCI or not. However, because PCI is an integral part of the management of ACS, 37,38 and just in case it took place to a large extent in the ACS population, the study was included in the current review to increase the comprehensiveness of our findings. For the same reason, we included the study by Kim et al, although it considered patients with ACS managed with PCI and those managed medically without PCI.
Six of the included studies showed that genotype-guided antiplatelet therapy was cost-effective when compared to universal clopidogrel, 21,23 -25,28,29,33 whereas five studies showed that it was dominant. 22,30 -32 On top of that, genotype-guided antiplatelet therapy was dominant when compared to universal prasugrel, ticagrelor, or both in five, 21 -25 one, 29 and three studies 30 -32 , respectively. Kim et al specified that genotype-guided antiplatelet with ticagrelor is cost-effective in both CYP2C19 intermediate and poor metabolizers but not in poor metabolizers only. 27 Surprisingly, Sorich et al and Crespin et al concluded that universal ticagrelor was cost-effective compared to genotype-guided therapy, which was not in line with our findings from the other studies. 26,28 Such an inconsistency can only be explained by the variability in the residual benefit and cost of the intervention among the different health-care systems as demonstrated by Sorich et al and Crespin et al. Generally speaking, based on the sensitivity analyses results, the variability of outcomes of economic decision models in studies are mostly driven by variability among studies in the relative risk of the thrombotic events between CYP2C19 mutation carriers and noncarriers, 22,23,24,27,28,30,33 in addition to how different are the acquisition costs of the medications. 27,33 In relation to why the conclusions of the studies by Sorich et al and Crespin et al were particularly in contrast to other studies, a more specific reason would be the variations in the clinical events included in the decision-analytic models used, whereby Sorich et al evaluated MACE only, excluding stent thrombosis or adverse drug reactions (eg, bleeding and dyspnea), and Crespin et al studied MI, death, bleeding, and dyspnea, excluding stent thrombosis and stroke. Such excluded outcomes are particularly associated with high management costs. 26,28 Also important is that the willingness-to-pay thresholds that were used by Sorich et al and Crespin et al (US$20 475-US$34 125 and US$50 000, respectively) are at the lower end of the range of what is universality considered acceptable, which may explain the cost-effectiveness of universal ticagrelor.
Here, it is worth noting that whether an ICER indicated cost-effectiveness in a study was based on the local interpretation of the ICER as reported in the study. This is as cost-effectiveness thresholds, based on which ICERs are judged, can vary widely from a country to another, depending on methods and assumptions used in calculations. Cost-effectiveness thresholds can vary from as little as US$4419 per QALY in Thailand to US$180 653 per QALY in Belgium. 39 In the United States, over 77% of cost-effectiveness research refers to US$50 000 or US$100 000 per QALY as a suitable threshold. Those values, however, are not well justified, with the former, as an example, stemming from the cost of dialysis in the 1980s. 40,41 Currently, however, the range of acceptable threshold value in the United States is increasingly up to US$150 000 per QALY. 42
Although generic ticagrelor and prasugrel are going to be commercially available sometime in the future, we cannot anticipate that the genetic-guided antiplatelet therapy will become less economically attractive, as the return to its cost is multifactorial in nature. First, it has been proposed that the cost of genetic testing will be reduced in the future, just like the costs of generic ticagrelor and prasugrel, and it will become part of the routine practice. 43,44 In addition, ticagrelor and prasugrel are associated with an increased risk of major bleeding, which has a high cost of management and a significant reduction in patient’s quality of life. 33 The secondary costs of therapies are not less important for consideration than the acquisition costs of the therapies. Thus, for an accurate assessment of the impact of introducing generic ticagrelor or clopidogrel to the market, evaluations under future circumstances would ideally be needed in future research.
The results of the included articles were primarily based on clinical outcomes derived from retrospective analyses of the published clinical trials TRITON-TIMI 38 and PLATO, except for the study by Wang et al, in which small-scale local observational studies were utilized. 29 Although the models relied on the best available sources, none of the articles used an outcome-driven prospective randomized controlled trial. The utilized literature clinical trials, however, are large international in design, with the smallest of which including over 700 sites from 30 countries. This means that while economic studies in the current review are from different countries, differences in reported outcomes are mostly due to variability in the cost and resource utilization aspects of the therapies, and it is nothing to do with variability in comparative effectiveness or underlying demographic variabilities, such as the level of sickness and the ethnicity of cohorts. All articles used local estimates for cost data such as hospital reports, Medicare reimbursement rates and drug retail pricing are appropriate as cost data should be based on the local setting. However, relying on the local health-care setting might not necessarily reflect the cost-effectiveness of genotype-guided therapy in other countries. The 13 studies used only direct medical costs associated with clinical and adverse outcomes and none used indirect medical cost or intangible cost, and thus cost-effectiveness results might be underestimated. All the studies used an appropriate horizon of analysis (≥ 12 months) that allows time for all the relevant and significant outcomes to be detected. 45 Five articles examined the CYP2C19*2 LOF allele only, 22,23,26,28,29 which might limit the generalizability of the results, as it is known that *3 is also a common allele. 6
The results of the quality assessment reflected that all the studies had a good quality which means that they adhered to most of the QHES items. However, a common element that was not reported in all the articles was discussing the direction and magnitude of potential biases. In addition, the methodology for data abstraction including the value of health status and other benefits were not completely stated in six articles. 21 -23,28,30,33 Moreover, the measurement of costs and the methodology for the estimation of quantities and unit costs were not clearly described. 21 -23,25,32,33 All these poorly executed methodological items are important for the journal editors and researchers to note, not just for the benefit of enhanced quality in future research but to also enable replication of studies.
This review included only English language articles which may have excluded possible relevant studies; however, no resources to translate non-English articles were available to authors. Despite the comprehensive search that was done, additional studies could have been identified in the literature with the use of additional search engines and/or combinations of search terms. Nevertheless, it is noted that the PubMed and EMBASE databases encompass almost 80% of the literature, and with the use of EconLit and PharmGKB as well, we believe to have covered a representative sample of literature. 46 In addition, the quality assessment did not account for the quality of the journals and their editorial requirements. An additional limitation of the study relates to the fact that of the 13 included studies, no economic evaluations of genotype-guided antiplatelet therapy have been undertaken in Europe, Africa, and the Middle East. Thus, one might assume that the results of the current review might be biased toward the US and Chinese populations. It is important to note, however, that this can only be relevant to changes in the cost of resources among countries and is not in relevance to differences in the clinical performance of interventions. As already discussed above, clinical outcomes in included studies were all, except in one study, primarily extracted from the same literature clinical trials as data sources.
Conclusion
Implementing genotype-guided antiplatelet therapy, followed by a targeted administration of ticagrelor or prasugrel in CYP2C19 LOF mutation carriers and clopidogrel in noncarriers, is the most cost-effective approach compared to the universal use of antiplatelets in patients with ACS undergoing PCI.
Recommendations
Future studies may need to focus on comparing the cost-effectiveness of conventional genotyping and point-of-care genotyping, as well as performing economic evaluations of CYP2C19 genotype-guided antiplatelet therapy in Europe, Africa, and the Middle East.
Supplemental Material
Supplemental Material, Supplement_1_AlMukdad_et_al - Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review
Supplemental Material, Supplement_1_AlMukdad_et_al for Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review by Sawsan AlMukdad, Hazem Elewa and Daoud Al-Badriyeh in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplement_2_AlMukdad_et_al - Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review
Supplemental Material, Supplement_2_AlMukdad_et_al for Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review by Sawsan AlMukdad, Hazem Elewa and Daoud Al-Badriyeh in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplement_3_AlMukdad_et_al - Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review
Supplemental Material, Supplement_3_AlMukdad_et_al for Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review by Sawsan AlMukdad, Hazem Elewa and Daoud Al-Badriyeh in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplement_4_ALMukdad_et_al - Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review
Supplemental Material, Supplement_4_ALMukdad_et_al for Economic Evaluations of CYP2C19 Genotype-Guided Antiplatelet Therapy Compared to the Universal Use of Antiplatelets in Patients With Acute Coronary Syndrome: A Systematic Review by Sawsan AlMukdad, Hazem Elewa and Daoud Al-Badriyeh in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
All authors contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by GSRA award (GSRA5-2-0521-18060) from Qatar National Research Fund and local student Qatar University Grant (QUST-1-CPH-2019-16).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
