Abstract
Neonatal megakaryopoiesis and platelet turnover form a developmentally unique pattern by generating a pool of newly released reticulated platelets from the bone marrow into the circulation. Reticulated platelets are more reactive and hyperaggregable compared to mature platelets, due to their high residual mRNA content, large size, increased expression of platelet surface receptors, and degranulation. The proportion of reticulated platelets in neonates is higher compared to that in adults. Due to the emergence of an uninhibited platelet subpopulation, the newly formed reticulated platelet pool is inherently hyporesponsive to antiplatelets. An elevated population of reticulated platelets is often associated with increased platelet reactivity and is inversely related to high on-treatment platelet reactivity, which can contribute to ischemia. Measurements of the reticulated platelet subpopulation could be a useful indicator of increased tendency for platelet aggregation. Future research is anticipated to define the distinct functional properties of newly formed reticulated or immature platelets in neonates, as well as determine the impact of enhanced platelet turnover and high residual platelet reactivity on the response to antiplatelet agents.
Keywords
Introduction
Platelets are anucleated cytoplasmic discoid-shaped cells, and their physiological role is the maintenance of vascular integrity and regulation of the hemostatic process at sites of vascular injury. 1,2 Nonactivated platelets circulate without adhering to the luminal surface of the endothelium, but in response to injury become activated, causing endothelial cell activation. 2,3 Activated platelets promote fibrin formation, recruitment of inflammatory cells to the thrombus, and initiate repair of the injured vessel wall. Hemorrhage arrest depends on a set of complex regulated events; initiated by a layer of platelets adhering to a thrombogenic surface, followed by recruitment of additional platelets to the primary adhesive plug, resulting in the formation of a fibrin network of blood clot. 1
In neonates, platelet production and release into systemic circulation is regulated by an age-dependent process of maturation. 1,3 The steps in age-related regulation of platelet reactivity are an integral part of the developmentally distinct neonatal hemostatic system. 3,4 Neonatal platelet studies are hampered by challenges in obtaining samples of adequate quantity and volume to conduct functional platelet assays. 4 Platelets from umbilical cord blood, as a substitute for neonatal platelets, can partially address the problem; however, those platelets may be functionally different from circulating neonatal platelets. 1 Cord blood platelets are characterized by downregulated expression of platelet degranulation proteins, which are required for exocytosis of alpha granules (glycoprotein 1b (GPIb), GPIIb/IIIa, P-selectin/CD62p) and dense granules (adenosine diphosphate (ADP), serotonin), indicating developmental delay of platelet content secretion and an ability to increase the surface area. 5
The expression levels of platelet adhesion integrins and membrane proteins, such as GPIb and GPIIb/IIIa (integrin αIIbβ3), are described as comparable between neonatal cord blood and adult peripheral blood platelets; however, a higher concentration of platelet receptor agonists is reportedly necessary to initiate aggregation in cord blood platelets. 6 Peripheral neonatal platelets are characterized as a more immature form, tending to form fewer pseudopods, as well as exhibiting higher granular content and surface adhesion protein expression. 5 Observational studies of platelets from adults indicate that the immature platelet fraction and platelet turnover can potentially affect platelet reactivity and responsiveness to antiplatelet agents. 7,8 In neonates, the fraction of immature platelets is higher compared to that in adults. 1,4 The focus of this review is on the impact of a higher fraction of newly formed immature reticulated platelets on reactivity and the response to antiplatelets in neonates.
Megakaryopoiesis in Neonates
Platelets are released into the peripheral blood as a result of fragmentation of megakaryocytes from stem cells in the bone marrow. Neonatal megakaryocytes display a developmentally distinctive uncoupling of proliferation, polyploidization, and cytoplasmic maturation, characterized by a high proliferative rate, producing large numbers of smaller size and lower ploidy megakaryocytes. 2,9 During thrombopoiesis in neonates, polyploidization enables megakaryocytes to achieve protein synthesis with large amounts of cytoplasm, and a rapid increase in the rate of platelet production in response to hemostatic demand. 4,10 The immaturity of neonatal megakaryocytes and their progenitors indicates developmental differences between neonatal and adult megakaryopoiesis 2,3 (Figure 1).
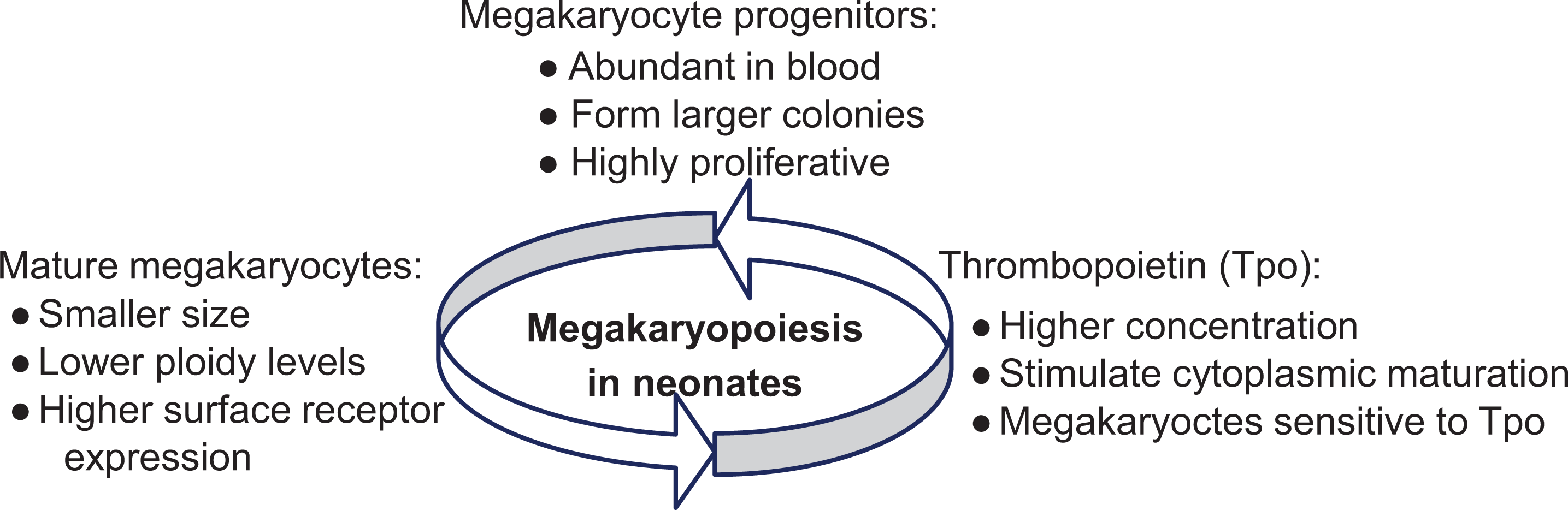
Megakaryopoiesis: In neonates, megakaryocyte (MK) progenitors are abundant in blood, form larger colonies and are highly proliferative; whereas in adults, MKs are sparse in blood, form smaller colonies and are less proliferative, indicating developmental differences between neonatal and adult megakaryopoiesis. In neonates, MKs mature at low ploidy levels, whereas in adults, MK maturation increases with ploidy, indicating the immaturity of neonatal MKs and their progenitors. The thrombopoietin concentration is higher in neonates and MK progenitor cells are more sensitive for thrombopoiesis. (Adapted from Davenport et al 9 ).
Megakaryopoiesis is regulated by various growth factors and cytokines, among which thrombopoietin is an essential megakaryocyte growth and development factor. 3 Thrombopoietin stimulates the proliferation and differentiation of megakaryocyte progenitor cells, increasing their size and polyploidization at each stage of megakaryopoiesis. 7 Compared to adults, neonatal thrombopoietin concentrations are higher, leading to larger megakaryocyte colonies. 2,3 In a further comparison, neonatal megakaryocyte progenitor cells are more sensitive to thrombopoietin signaling and exhibit a greater proliferative response to low concentrations of thrombopoietin. 3 In neonates, the concentration of circulating thrombopoietin is inversely related to the rate of platelet production, which could enable to distinguish impaired platelet production from increased platelet destruction.
Reticulated Platelets
Reticulated platelets, also termed immature platelets, are newly released platelets from the megakaryocyte cytoplasm and contain residual amounts of messenger ribonucleic acid (mRNA) and translational machinery for protein synthesis 2,3 (Table 1). They represent immature platelets released from the bone marrow into the circulation, and reflect the activity of megakaryopoiesis. 4,10,11 Standard platelet identification via staining is inconsistent because the stains used and staining procedures are different and may not identify matching platelet populations, although reticulated and immature platelets have substantial similarities. 12,13 Reticulated platelets are identified by flow cytometry following thiazole orange staining, whereas immature platelets are detected using an automated cell counter as the percentage of newly released reticulated platelets into the circulation. Mature platelets live for 7-10 days in the circulation, whereas young reticulated platelets appear to remain in the bloodstream for 1-2 days; thereafter, the mRNA progressively degrades and their volume decreases. The number of reticulated platelets correlates with thrombopoiesis; hence, it could serve as a real-time surrogate marker of megakaryopoietic activity in the neonatal bone marrow. 12
Functional Properties of Reticulated Platelets in Neonates.

Megakaryocytes from neonates are more reticulated compared to those from adults, and elevated reticulated platelets are associated with an increase in platelet turnover rate, high megakaryocyte activity, and thrombopoiesis 10,14 (Figure 2). Reticulated platelets have a larger surface area, a greater number of dense granules, and a higher mean volume compared to non-reticulated mature circulating platelets. 3 In addition, the translationally active mRNA enables the expression of platelet surface receptors, leading to a rapid aggregation response, synthesis of more thromboxane A2 (TXA2), and release of more ADP or serotonin granules compared to older non-reticulated circulating platelets. 3,13 Reticulated platelets have a higher capacity to express platelet activation markers, as well as the cell surface fibrinogen receptor GPIIb/IIIa and von Willebrand factor (vWF) receptor GPIb, in response to ADP activation, compared with mature platelets. 15 Neonatal megakaryocytes constitutively express the adhesion protein P-selectin/CD62P at a higher level compared to adult platelets, indicating developmental upregulation of cell adhesion molecules on the surfaces of activated platelets. 1
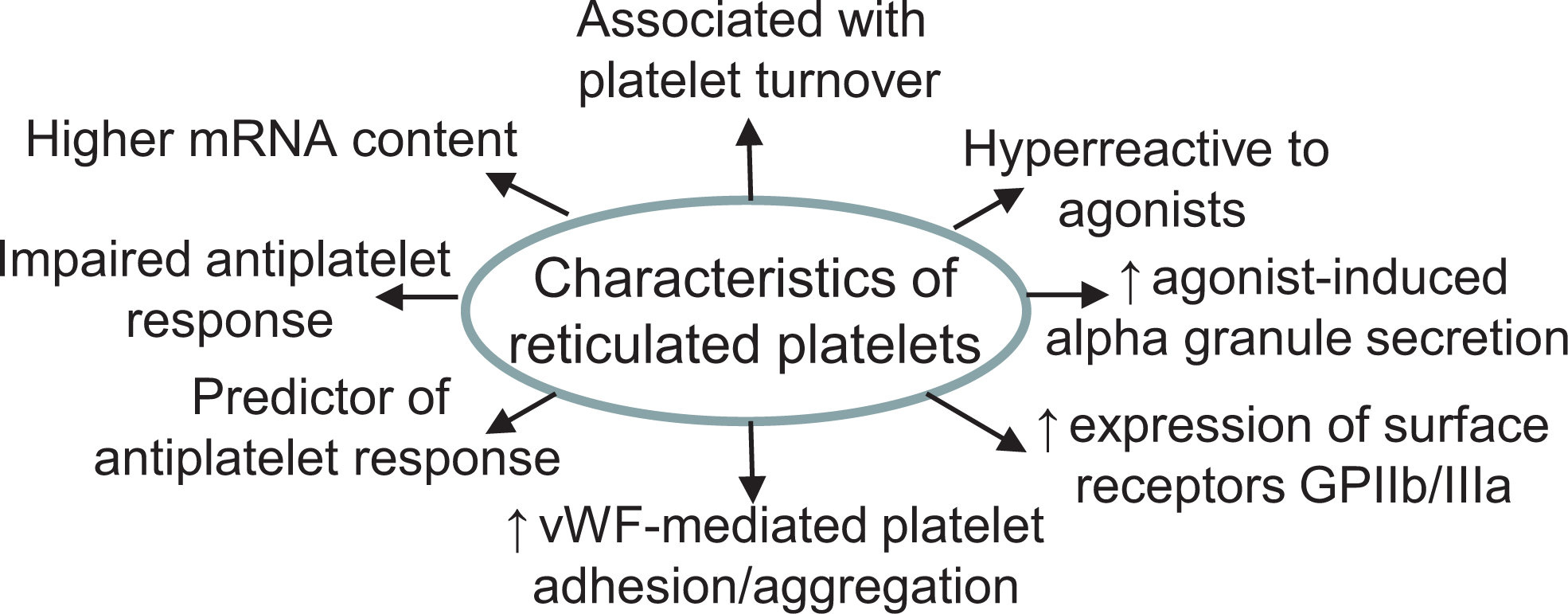
Functional characteristics of reticulated platelets: Reticulated platelets are newly released immature platelets containing residual mRNA. They are associated with a high platelet turnover rate and exhibit increased agonist-induced responsiveness compared to adult platelets, including increased agonist stimulated aggregation, adhesion, degranulation, and activation of fibrinogen receptor GPIIb/IIIa. Neonatal platelets are also characterized by increased large multimeric vWF, which enhances the platelet-vessel wall to maintain the hemostatic system. (↑, increase; ↓, decrease; vWF, von willebrand factor).
In neonates, increased mean platelet volume serves as an index of platelet size, and indicates increased platelet reactivity because of the expression and secretions of adhesive membrane receptors, such as, fibrinogen and vWF, which mediate platelet-platelet and platelet-endothelial interactions. 2,3 Platelet volume is a useful surrogate marker to estimate platelet turnover and release of new platelets into the circulation. Reticulated platelet volume correlates with mean platelet volume, suggesting that younger reticulated platelets are larger and exhibit higher reactivity compared to mature and smaller platelets. 3 Additionally, reticulated platelets with enlarged volume are associated with high residual platelet reactivity following antiplatelet therapy. 16
The reticulated platelet fraction reflects the bone marrow’s thrombopoietic activity to compensate for peripheral destruction or consumption of platelets, which could lead to thrombocytopenia. As a result of higher demand for platelet production, the number of reticulated platelets is increased in conditions with an enhanced rate of thrombopoiesis and increased platelet turnover. 12 A larger reticulated platelet fraction is associated with a physiological response to a higher thrombopoietin concentration, as well as increased megakaryopoietic activity and platelet production rate. 4 In thrombocytopenic neonates, a low reticulated platelet fraction implies inadequate platelet production and diminished capacity to enhance the platelet turnover rate, indicating reduced megakaryopoietic activity. 10,17 By contrast, a low platelet fraction in thrombocytopenic neonates could be due to peripheral consumption (e.g., immune thrombocytopenia), leading to excessive bleeding if not compensated by an adequate increase in megakaryopoiesis. 18 An elevated reticulated platelet fraction with thrombocytopenia indicates destruction or consumption of platelets in the spleen, and a reduced fraction implies decreased compensatory megakaryopoiesis and impaired bone marrow function. 4 Thus, in thrombocytopenic neonates, knowing the reticulated platelet fraction could enable differentiation of disorders caused by reduced megakaryopoietic activity in the bone marrow from those of increased peripheral platelet destruction in. 10,11,19
Impact of Reticulated Platelets on Reactivity
Platelet reactivity results from a coexistent hemostatic balance between activation and inhibition of signaling pathways. 16,20 Previous studies using cord blood-derived platelets showed neonatal platelets to be hypofunctional and hypoaggregative in response to agonist stimulation, indicating that a developmental imbalance modifies agonist-induced platelet reactivity. 3,4 The transient hyporesponsiveness of neonatal platelets are characterized by decreased fibrinogen binding, fewer mature dense granules, and reduced intracellular calcium mobilization compared to platelets from adult peripheral blood. 3,21,22 A comparable activation response to agonists that directly activate protein kinase C or increase calcium permeability, such as phorbol esters or calcium ionophore, were reported in neonatal and adult platelets, indicating developmentally maintained intracellular signaling pathways. 9
While there are reports indicating hyporeactivity of neonatal platelets in response to agonists, such as ADP, epinephrine and collagen, there appears to be considerable data variability, which may be due to different sample types (cord blood verses peripheral blood), type of assay and procedures used to test platelet function. 3,23 In conditions associated with increased platelet turnover and higher levels of baseline platelet activation, such as immune thrombocytopenia patients, treatment with thrombopoietin receptor agonist resulted in a slight increase in platelet reactivity in response to thrombin, and the increase may be due to higher platelet count or higher platelet reactivity. 24 The reasons for the contrasting findings of platelet reactivity are unclear; however, neonates have higher immature platelet fraction, which are associated with lower threshold for granule exocytosis, increased expression of surface activation markers and hyperresponsiveness. 13,14 Platelets from healthy children has been reported to be hyperresponsive to various platelet agonists, such as thrombin and ADP, indicating that the increased sensitivity to platelet activation pathways and higher reticulated platelet fraction in neonates persist in infants and young children, and agonist-stimulated platelet reactivity inversely correlates with age. 25
Neonates have a well-balanced hemostatic system, with shorter bleeding time and shorter closure times as demonstrated by platelet function analyzer-100 (PFA-100) measurements in response to collagen and ADP. 3,23 This apparently discordant finding of hypercoagulability and platelet hyporeactivity has been described to be due to the presence of large vWF multimers resulting from decreased cleavage activity of the zinc metalloprotease ADAMTS-13 in neonates. 1,3 The hyperaggregable milieu, from high levels of large vWF polymers, counterbalances the hyperstimulation of inhibitory signaling that contributes to the hyporeactivity of neonatal platelets. 1 Elevated levels of vWF in neonatal plasma enhance the ability of platelets to adhere to the vessel wall and promote clotting in order to maintain hemostasis. 3,5
Umbilical cord blood-derived platelets exhibit reduced protease-activated thrombin receptor-1 (PAR-1)-mediated granule secretion, PAR-1 expression, and integrin activation relative to adult platelets, indicating loss of sensitivity to the potent platelet agonist thrombin. 26 Neonatal platelet alpha granule secretion and integrin GPIIb/IIIa activation are robust in response to ADP stimulation, indicating functional purinergic P2Y12 receptor-mediated signaling pathways. 1 Neonatal platelets display higher sensitivity to inhibition of aggregation by cyclic adenosine monophosphate (cAMP) modulators such as prostaglandin E1 (PGE1), which may be due to functional upregulation of downstream inhibitory pathways. 27,28 Enhanced P2Y12 receptor-mediated dense granule trafficking in neonatal relative to adult platelets suggests that neonatal platelets may engage an ADP-mediated positive feedback loop as a compensatory mechanism to compensate for neonatal platelet hyporeactivity. 26 A high concentration of PGE2 in cord blood platelets and hypersensitivity to PGE2-stimulated increases in cAMP levels suggest upregulation of various platelet inhibition signaling pathways in early stages of development. 28 These observations in neonates indicate that inhibitory pathways are involved in maintaining circulating platelets in a resting state.
Circulating reticulated platelets, compared to mature platelets, have lower thresholds for release of dense granular contents and are hyperactive, with a tendency for increased aggregation. 3,14 The hyperactivity in response to agonists is due to increased receptor expression and receptor-mediated signal transduction, as well as a higher granule content. Following activation by ADP, reticulated platelets exhibit a higher capacity for increased expression of proaggregatory surface proteins such as GPIIb/IIIa and P-selectin, compared to mature non-reticulated platelets, which may be due to protein synthesis by the residual mRNA. 14,15 In neonates, a higher proportion of circulating reticulated platelets implies a greater potential for platelet activation and reactivity. 3,8
Reticulated Platelets in Response to Antiplatelets
Elevated circulating levels of reticulated platelets are associated with an attenuated response to antiplatelets. 3,8,14 The mechanism underlying the attenuated response of reticulated platelets to antiplatelet drugs is related to an enhanced rate of turnover of platelets containing residual mRNA, which increases expression of platelet surface adhesion receptors, cyclooxygenase-1 (COX-1) activity and degranulation, promoting platelet activation and aggregation. 7,12,29 To overcome the reduced antiaggregatory response of reticulated platelets, higher antiplatelet dosages are needed to effectively suppress their increased reactivity. 30 Thus, circulating levels of reticulated platelet fractions could serve as an indicator of residual platelet reactivity and a predictor of antiplatelet effectiveness. 20
The pharmacokinetic characteristics of an antiplatelet drug could influence its efficacy in effectively inhibiting reticulated platelets. 16 The association of an increased rate of platelet turnover with high on-treatment platelet reactivity varies with the exposure duration needed for the pharmacologically active drug metabolite to inhibit newly formed reticulated platelets. 8 The appearance of an uninhibited and newly released reticulated platelet subpopulation during treatment with antiplatelet drugs is more apparent for drugs with shorter plasma half-lives. 30 In the presence of elevated proportions of reticulated platelets, antiplatelet drugs with active metabolites of shorter half-lives may not be effective inhibitors of the newly formed platelets; whereas drugs with longer half-lives or multiple daily dosing could provide effective inhibition of a high platelet turnover rate. 14,16,30 Thus, the ability of antiplatelet drugs to effectively inhibit newly released circulating reticulated platelets depends on the half-life of the active drug in the plasma, and on covalent/noncovalent receptor binding.
Antiplatelet Monitoring Based on Targeted Range of Platelet Inhibition
Infants and young children with certain types of congenital and acquired heart disease receive antiplatelet dosages extrapolated from adults. Dose selection using allometric scaling and simulation analysis on the basis of body weight may not account for all variables related to the elevated proportion of reticulated platelets, and therefore may not reliably predict an effective dosage for the neonatal age group. Reticulated platelets are associated with an impaired response to antiplatelets because they are characterized by a higher mRNA content, increased mean volume, greater number of dense granules, and hyperactivity. 3,13 The fraction of circulating reticulated platelets and high platelet turnover have gained interest as a plausible explanation for hyporesponsiveness to antiplatelets and the higher threshold for platelet inhibition 4,13 (Figure 3). Dose titration experiments might enable the determination of a targeted range of platelet inhibition better than would a linear extrapolation of dosages from adults to neonates based on body weight. In addition, measurement of reticulated platelet levels and periodic platelet reactivity testing could facilitate to achieve effective platelet inhibition.

Therapeutic window of reticulated platelets for antiplatelet therapy: On-treatment platelet reactivity is targeted to maintain antiplatelet therapy within an optimal range to balance the risk of bleeding and ischemic events. Elevated levels of reticulated platelets and a high platelet turnover correlate with hyporesponsiveness to antiplatelets and high on-treatment platelet reactivity, which are predictive of ischemic events. Monitoring the effectiveness of antiplatelets based on a targeted range of platelet inhibition guided by the reticulated platelet count and platelet reactivity testing may enable achievement of optimal on-treatment platelet reactivity.
Aspirin (acetylsalicylic acid) is the most widely used antiplatelet agent in the pediatric age group for a variety of conditions, except during episodes of viral infection, where the drug has been associated with a low incidence of developing Reye’s syndrome, which causes swelling in the liver and brain. Aspirin mediates its antiplatelet effects by irreversible acetylation of COX-1 and subsequent blockade of TXA2 formation; however, the half-life of aspirin is on the order of minutes, and may not inhibit newly formed platelets that resynthesize COX-1. In neonates, elevated platelet turnover is a major contributing factor to increased reactivity because newly formed reticulated platelets contain mRNA, which resynthesizes COX-1 enzyme; and aspirin, because of its very short half-life, has been described as an inadequate platelet inhibitor. 30
Pediatric dosages of aspirin are based on empirical body weight-adjusted doses extrapolated from adults, which, as previously observed, may not be optimal to provide adequate inhibition of platelet aggregation. 20 Children with heart disease given a standard dosage range for aspirin (1-5 mg/kg/day) reported increased urinary 11-dehydro-thromboxane B2 (11-dehydro-TXB2) and thromboembolism, indicating suboptimal inhibition of platelets and high on-aspirin platelet reactivity. 20 Higher levels of urinary 11-dehydro-TXB2 have been reported in young children with congenital heart defects, which required increases in aspirin dose (6-10 mg/kg) to inhibit the residual thromboxane production. 31 In addition, other reports indicated that a pragmatic approach of higher dosages of aspirin (>7 mg/kg/day) may not be sufficient to reduce shunt-related adverse events in infants following modified Blalock-Taussig shunt replacement. 32 These studies suggest the need for platelet function testing and measurement of serum TXB2 and urinary 11-dehydro-TXB2 concentrations to determine the appropriate dosing for effective platelet inhibition.
Reticulated platelets are associated with reduced antiplatelet effects of aspirin and enhanced aspirin resistance, due to higher reactivity and uninhibited COX-1 activity. 23 As a result, an elevated count of immature reticulated platelets is inversely related to aspirin responsiveness. In addition, an elevated platelet turnover is associated with a greater-than-expected reduction in the antiplatelet effect, and consequently, increased platelet turnover could be a useful predictor of on-treatment platelet reactivity. 12
Previous studies in infants and young children have shown that lower dosages of clopidogrel, an irreversible thienopyridine derivative P2Y12 receptor antagonist, are required to achieve similar inhibition of ADP-induced platelet aggregation compared to those in adults. 33 However, in those studies, most patients given clopidogrel were also treated with aspirin, which may explain the lower clopidogrel dose per kilogram body weight to achieve a similar level of inhibition of platelet aggregation.
In addition, there is an inconsistency between the observations of lower dosages of clopidogrel and the notion that elevated reticulated platelets require higher dosages of antiplatelet agents. This is because a higher proportion of new platelets is released into the circulation, during which time the active drug metabolites with short half-lives are largely cleared. Clopidogrel is an inactive prodrug that undergoes oxidative activation by several cytochrome P450 (CYP) isoforms to convert into an active metabolite containing a thiol group, which may complicate interpretation of clopidogrel studies. The expression of CYP2C19 is reported to be reduced in neonates, indicating that the neonate’s ability to activate clopidogrel could be lower than that of adults; thus, neonates may be at a higher risk for ischemic events. 34 This suggests a need for CYP2C19 genotyping to differentiate non-responsiveness due to factors other than loss of activity resulting from metabolism of the prodrug.
Previous studies in adults using prasugrel, another thienopyridine prodrug P2Y12 receptor antagonist, reported that elevated levels of circulating reticulated platelets are associated with a hyperactive platelet subpopulation, resulting in higher-than-expected platelet reactivity. 35 Prasugrel exhibits a nonlinear relationship between exposure and body weight, making it challenging to consistently extrapolate dosages of the prodrug from adults to infants. 36,37 In children with sickle cell disease, dose-ranging titration to achieve a prespecified target range of 30% to 50% platelet inhibition was able to balance the safety and efficacy of prasugrel. 36 Weight-based methods for determining pediatric doses of P2Y12 receptor antagonists may not account all variables related to the different stages of maturation and/or forecast the optimal dose for neonates. This indicates that individualization approaches of dosing titration using real-time pharmacodynamic assays may enable dose adjustments to achieve a steady-state inhibition of platelet without a higher bleeding risk. 37 Thus, infants and young children may need a dosing model to achieve effective platelet inhibition rather than body weight-based linear extrapolation of the adult dose.
An example of an active parent drug is ticagrelor. It is a reversible and direct-acting P2Y12 receptor inhibitor that does not need metabolic activation and shows no significant differences in antiplatelet potency in blood samples among children of various ages compared to adults. 38 This indicates that similar pharmacodynamic effects can be expected using ticagrelor, which has a longer half-life, because it provides more consistent levels of inhibition of ADP-induced platelet aggregation than an inactive prodrug that needs to be metabolized to an active form. However, the dosages of ticagrelor needed to achieve optimal inhibition of newly released reticulated platelets in neonates have not yet been evaluated.
Another reversible P2Y12 receptor inhibitor is cangrelor, an adenosine triphosphate (ATP) analogue administered intravenously, which was reported to achieve marked reduction of platelet aggregation in neonates requiring systemic-to-pulmonary artery shunts. 39 Using a dose titration model, the dosages at a steady state plasma level of cangrelor in the neonates were approximately seven-fold lower than that reported for adults, emphasizing the importance of pharmacodynamic assays to estimate dosages for optimal thromboprophylaxis in neonates. 39 Cangrelor is a fast-acting P2Y12 receptor inhibitor characterized by rapid onset and offset of action, and hence is suitable for short-term use. Collectively, these examples underscore the importance of investigating the impact of higher platelet turnover, elevated proportion of circulating levels of reticulated platelets, and the neonatal response to antiplatelets.
Conclusions
The available data indicate that newly formed reticulated platelets are hyperreactive and hyperaggregable compared to mature platelets, due to the presence of residual mRNA and their capacity to express platelet surface receptors. Observational studies in adults have shown that high platelet turnover and elevated levels of reticulated platelets are associated with high on-treatment platelet reactivity due to enhanced platelet turnover and the emergence of uninhibited reticulated platelets. An increased circulating reticulated platelet subpopulation may serve as an important parameter to predict the antiplatelet response and the optimal magnitude of platelet inhibition to minimize the risk for bleeding.
Neonates have a higher proportion of the reticulated platelet fraction, an elevated platelet turnover rate, larger mean platelet volume, and abundant dense granules, all of these indicating that neonatal platelets are hyperactive. In particular, measurement of newly released immature or reticulated platelet subpopulations, as well as real-time pharmacodynamics with a platelet function guide to adjust antiplatelet dosing, may help achieve a prespecified target range of platelet inhibition. Therefore, further studies are needed to characterize the reactivity of circulating reticulated platelets in neonatal peripheral blood to develop strategies for monitoring the response to antiplatelets. Finally, efforts are needed to evaluate the contribution of age-related developmental regulation of reticulated platelets to on-treatment platelet reactivity and the response to antiplatelets in neonates.
Footnotes
Author’s Note
This article reflects the views of the author and should not be construed to represent FDA’s views or policies.
Acknowledgments
The author would like to thank Joanne Berger (FDA library) and Dr. Graeme O’May for editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
