Abstract
Introduction
Atrial fibrillation (AF) represents the most prevalent type of arrhythmia and is associated with high mortality and morbidity. 1 AF is caused by structural, electrical, and autonomic remodeling of the atrium 2 and appears to be facilitated by both structural and electrical remodeling, although the specific mechanism is not understood.
Electrical remodeling in AF occurs as the result of increases in Ca2+ concentrations, together with reductions in L-type calcium currents (ICaLs). Decreased ICaL densities can reduce the action potential duration (APD) of the atrium, and reduced numbers of atrial effective refractory periods (AERP) can lead to AF. 3 It has been reported that levels of Cav1.2 are downregulated by the calcineurin/nuclear factor of activated T cells (CaN/NFAT) pathway, resulting in reduced ICaL densities and APDs, ultimately leading to the development of AF. These effects can be prevented by the CaN inhibitor FK-506. 4 Activation of Ca2+/CaN/NFAT signaling by transient receptor potential channel 6 (TRPC6) has also been found to promote cardiomyocyte hypertrophy 5 which is negatively regulated by the cyclic guanosine monophosphate (cGMP)/protein kinase G (PKG) axis. 6 It is likely that electrical remodeling is dependent on TRPC6/CaN/NFAT signaling. The features of structural remodeling are hypertrophy and fibrosis of the atria, with significant upregulation of collagen I, collagen III, and ST2 seen in fibrosis. 7 Targeting the TRPC6/CaN/NFAT pathway may thus be a way of reducing and preventing electrical remodeling in AF.
Vericiguat is a soluble guanylate cyclase agonist that acts independently of nitric oxide (NO) to increase the sensitivity of the enzyme to NO and promote cGMP signaling. 8 This has been found to enhance both vascular and myocardial function and reverse hypertrophy and fibrosis of the left ventricle, as well as reduce ventricular remodeling and afterload by the modulation of both pulmonary and systemic vasodilation, and has been used for treating HF. 9 The trials found that vericiguat was better than placebo in lowering the incidence of both first hospitalization and death in HF patients with deteriorating HF and those with reduced ejection fraction (EF) (HFrEF), as well as improving the Kansas City Cardiomyopathy Questionnaire physical limitation assessment scores (KCCQ PLS) in patients with preserved EF (HFpEF).9,10 Vericiguat has been shown to be both safe and effective and is included in the 2021 European Society of Cardiology (ESC) guidelines and the 2022 American College of Cardiology (ACC)/American Heart Association (AHA)/Heart Failure Society of America (HFSA) guidelines for HF. Reports have indicated that vericiguat decreases ventricular electrical and structural remodeling in HF.11–13 Here, we investigated whether vericiguat can reduce atrial electrical and structural remodeling in AF.
Materials and Methods
Rabbit Rapid Atrial Pacing Model
All procedures related to animal experiments conformed with the Guide for the Care and Use of Laboratory Animals (eighth edition, NIH Guide 2011 edition) and were approved by the Animal Experimentation Ethics Committee of Harbin Medical University (Harbin, China); the approving authority was the First Affiliated Hospital of Harbin Medical University and the approval number of the application was 201551. Thirty-six New Zealand white rabbits (male, 2.5‒3.0 kg) were purchased and maintained in the Experimental Animal Center of the First Affiliated Hospital of Harbin Medical University. The rabbits were housed in separate cages in a specific pathogen-free environment at 23 °C and 12-h light-dark cycle and received filtered water ad libitum and a standard laboratory diet.
The AF model was created as previously described. 14 Rabbits were anesthetized with 30–35 mg/kg ketamine and 5 mg/kg xylazine (both Sigma-Aldrich). The procedure was conducted using mechanical ventilation and sterile thoracotomy. A pacemaker (Harbin University of Science and Technology) was implanted subcutaneously on the dorsal surface with the electrode stitched to the right atrium. The rabbits were allowed to recover for one week following surgery.
The 36 rabbits were randomly allocated to four groups (n = 9 per group), with three animals in each group used for the atrial myocyte isolation and patch clamp detection. The groups were: (1) the sham-operated group (sham group), who received only electrode suturing electrodes without pacing; (2) the rapid atrial pacing (RAP) group, with electrode suturing and rapid right atrial pacing for three weeks at 600 beats/min; (3) vericiguat-treated group (RAP+vericiguat group), daily oral administration of 1.5 mg/kg vericiguat (MedChemExpress, USA) for three weeks 15 ; (4) vericiguat-only group (vericiguat group) with electrode suturing without pacing and only daily oral administration of 1.5 mg/kg vericiguat for three weeks.
Cell High-frequency Electrical Stimulation Model
The HL-1 cell line (of AT-1 mouse atrial cardiomyocyte tumor lineage) was grown in DMEM with low glucose (HyClone, USA) containing 10% fetal calf serum (BI, USA) and 1% penicillin-streptomycin solution (HyClone). Cells (≥1 × 106) were grown in six-pore plates and were stimulated by high-frequency 10-Hz square pulses (5 ms duration, 1.5 V/cm) using a stimulator (YC-2), as described previously. 14 The required capture efficiency was approximately 90% of the cells as monitored microscopically during stimulation.
Three groups of cells were established for verification. These were the control group (grown for 24 h with no other treatment), the pacing group (high-frequency field stimulation for 24 h), and the pacing and vericiguat group (high-frequency field stimulation for 24 h plus treatment with 10 µM vericiguat. 15
Biochemical Evaluation
Blood samples were taken from the rabbits, centrifuged (3000 rpm, 20 min), and the plasma was retained. Plasma samples were stored at −80 °C before analysis with avoidance of repeated freeze-thaw cycles. ST2 concentration was measured using kit (Jiangsu Meimian Industrial Co., Ltd, China), following the provided directions, and absorbances were measured in a microplate reader (BioTek Instruments, USA).
Echocardiographic Evaluation
Surface echocardiography (Philips CX50, USA) was used for the evaluation of cardiac structure and function after 3 weeks of treatment. Structural and functional parameters including the left atrial anteroposterior, axial, and transverse diameters, left atrial maximal volume, right atrial axial and transverse diameters, end-diastolic diameter of left and right ventricle, left ventricular ejection fraction (LVEF), and fractional shortening (FS) were determined.
Electrophysiological Analysis
Electrophysiological analysis was conducted using a previously published method. 16 Programmed simulation was conducted using an electrophysiological instrument (Henan Huanan Medical Technology Co., Ltd, China), with eight basic stimuli (S1) followed by an extra stimulus (S2) with the S1-S2 cycle having a basic cycle length of 150 ms with decreases in the interval in 2 ms after being increased by 5-ms steps until S2 no longer captured the depolarization (the AERP). Three AERP determinations were made at the basic cycle length of 150 ms (AERP150) and the mean value was calculated. An induction was considered successful if the AF duration was over 1000 ms. A train of 10-Hz stimuli at 2-ms intervals was induced using an intracardiac electrode 10 times to the left atria of the rabbits for 10 s at four times the threshold current, and the AF induction rate (AF times/induction times) were calculated for each group (Figure S1).
Pathological and Immunohistochemical Analyses
Histological analyses, including hematoxylin-eosin (HE) and Masson staining, were performed as described previously. 14 Left atrial tissue specimens from the rabbits were fixed with 10% neutral formalin and sectioned (4 μm) after embedding in paraffin. After staining with HE or Masson stain, the sections were dehydrated, cleared, and sealed before examination under a microscope (Eclipse Ci-L, Nikon Instrument Co., Ltd, Japan). Morphological changes in the myocardial cells were assessed by HE staining; normally, cardiomyocytes stain a reddish color and are closely arranged while the nuclei are ordered and stained blue. With Masson staining, the cardiomyocytes stain red and collagen blue. The sections were analyzed with ImageJ, specifically, the myocardial collagen volume fraction (CVF), defined as the proportion of collagen in a test field of myocardial tissue.
Immunohistochemistry was performed as previously described. 14 Five-micrometer paraffin-embedded sections were dewaxed before antigen retrieval, incubation with hydrogen peroxide for 15 min, and blocking with serum for 15 min. The sections were then incubated with primary antibodies against TRPC6 (1:100; Bioss, bs-2393R), CaN (1:100; Bioss, bs-0030M), p-NFAT4 (1:100; Bioss, bs-19787R), NFAT4 (1:100; Bioss, bs-2952R), and Cav1.2 (1:300; Millipore, USA, #MAB13170) overnight at 4 °C. The sections were then incubated with appropriate secondary antibodies (1:5000; ThermoFisher, USA, #31460 and #31430), and DAB chromogenic solution (Maixin reagent, China, DAB-1031) was used for color development. After counterstaining with hematoxylin, the sections were dehydrated, clarified, sealed, and analyzed under a microscope. A positive reaction was visible as dark-brown staining of the target proteins, and the average optical density (integrated optical density/area sum) was calculated by ImageJ software.
Western Blotting
Western blotting was performed following a previously published method. 17 The total protein content of atrial tissue samples and HL-1 cells was isolated and concentrations were measured using a BCA kit (Beyotime, China). Proteins were denatured in 5x SDS-PAGE sample loading buffer (Beyotime), electrophoresed at 120V, and transferred to PVDF membranes (Millipore) at 180V. After blocking, the blots were incubated with the primary antibodies used for immunohistochemistry described above, namely, against TRPC6 (1:1000), CaN (1:1000), p-NFAT4 (1:1000), NFAT4 (1:1000), and Cav1.2 (1:500), as well as against collagen Ⅰ (1:500; Wanleibio, China, WL0088), collagen Ⅲ (1:300; Wanleibio, WL03186), GAPDH (1:2000; Bioss, bs-2188R), and β-actin (1:1500; ZSGB-BIO, China, TA-09). The membranes were then incubated with goat anti-rabbit (1:10 000; ZSGB-BIO, ZB-2301) or goat anti-mouse IgG (1:10 000; ZSGB-BIO, ZB-2305) and developed using a hypersensitive ECL chemiluminescence kit (Beyotime) and quantified using a gel scanning and image analysis system (Bio-Rad, USA).
Measurement of Ca2+ Concentrations in HL-1 Cells
The fluorescent dye Fluo-4 AM (Beyotime, S1060) penetrates the cell membrane to label Ca2+ with green fluorescence. After entry into the cell, Fluo-4 AM is cleaved by esterases to Fluo-4 which then reacts with Ca2+ producing strong fluorescence. Fluorescence labeling was conducted according to the product specifications. After warming in a water bath at 20–25 °C, the Fluo-4 AM mother liquor (2 mM) was diluted to a 4-µM working solution with phosphate-buffered saline (PBS) and incubated with cells at 20–37 °C in the dark for 40 min. The cells were rinsed three times in PBS and incubated for a further 20 min to ensure full conversion of Fluo-4 AM to Fluo-4.
DAPI (Beyotime, c1005) was used to counterstain the nuclei. Cells were incubated with DAPI at room temperature for 4 min, rinsed three times in PBS, and examined under fluorescence microscopy (Zeiss, Germany). The excitation and emission wavelengths were 488 nm and 530 nm, respectively, and relative Ca2+ concentrations were calculated by the fluorescence intensity. ImageJ software was used to determine the mean density, as described above.
Detection of L-type Cav Channel Currents in the Left Atria of Rabbits
L-type Cav channel currents in myocytes isolated from the left atria were measured as described previously.14,18 Specifically, a Langendorff apparatus was flushed with Buffer A (100 ml Ca2+-free Tyrode) to drain the bubbles, followed by perfusion for 2 min with Buffer C (100 ml Ca2+-free Tyrode, CaCl2 containing 1 mM/L Ca2+ and 1 mg/mL BSA). The hearts were removed from anesthetized rabbits and fixed on the Langendorff apparatus before perfusion with Buffer C for 3 min until the blood was completely discharged from the heart before perfusion with Buffer A for 3 min. Finally, Buffer E (150 ml Ca2+-free Tyrode, 1 mg/mL BSA, and 0.9 mg/mL collagenase II) was used for perfusion until the heart became transparent and soft. The left atria were then harvested, minced, and centrifuged. Varying concentrations of calcium-replacement solutions (0.25 mM: 1 ml Buffer C + 3 ml Buffer B [100 ml Ca2+-free Tyrode, CaCl2 containing 50 µM/L Ca2+ and 1 mg/mL BSA]; 0.33 mM: 1 ml Buffer C + 2 ml Buffer B; 0.5 mM: 1 ml Buffer C + 1 ml Buffer B; 1 mM: buffer C can be added as needed) were used in turn. The cells were then inoculated into poly-L-lysine (PLL)-coated Petri dishes and incubated at 4 °C for subsequent patch-clamp experiments. The intracellular solution (120 mM CsCl, 1 mM MgCl2, 0.3 mM Na2-GTP, 4 mM Mg-ATP, 10 mM HEPES, 10 mM EGTA, pH 7.20 with CsOH) and extracellular solution (140 mM TEA-Cl, 10 mM CaCl2, 2 mM MgCl2, 5 mM D-glucose, 10 mM HEPES, pH 7.4 with TEAOH) were used for ICaL detection.
The Narishige capillary glass (BF150-86-10, Sutter Instruments, USA) was pulled into a recording electrode by a microforge (P97, Sutter Instruments). The recording electrodes were then used for vacuum absorption of the cells and whole-cell patch-clamp recordings were conducted under an inverted microscope (IX71, Olympus, Japan). The dingle current was normalized to the membrane capacity and expressed as the Ca current density (PA/PF). Data were collected with an amplifier (EPC-10, HEKA, Germany) and stored in PatchMaster software (HEKA).
Statistical Analyses
All quantitative data apart from the AF induction rates expressed as percentages are represented as the mean ± standard error of the mean. Differences between groups were determined using t-tests or one-way analysis of variance, and P < .05 was considered statistically significant.
Results
Electrophysiological Parameters
There was a significantly greater incidence of AF in rabbits in the RAP group than in the sham rabbits (Figure 1A and B). AF was, however, significantly reduced after treatment with vericiguat (Figure 1B). The AERP 150 ms was dramatically lower in rabbits in the RAP group while this reduction was reversed after vericiguat treatment (Figure 1C).

Electrophysiological changes in rabbit models. (A) Electrocardiogram of AF induced by rapid pacing. (B) Inducibility of AF. (C) AERP 150 ms. *P < .05 vs. sham group, #P < .05 vs. RAP group, n = 6 per group.
Echocardiographic Characteristics
Enlargement of both right and left atria was visible in rabbits in the RAP group was enlarged, with increased volume of the left atrium and decreases in both the ejection fraction (EF) and fractional shortening (FS), although no remarkable changes were apparent in the left and right ventricular end-diastolic diameters. Vericiguat treatment delayed atrial enlargement and dysfunction caused by rapid pacing (Table 1).
Effects of Vericiguat Treatment on Cardiac Ultrasonic Parameters in Rabbits.

Data represent means ± standard error of mean (SEM). *P < .05 vs. sham, #P < .05 vs. RAP, n = 6 per group.
Vericiguat Inhibited the RAP-Induced Pathological Changes
Morphological changes in the atrial myocytes were assessed after HE and Masson staining (Figure 2A-C). HE staining showed an orderly and tight arrangement of the cardiomyocytes in the sham group with neatly arranged nuclei. In contrast, the cells in the RAP group appeared disordered and loose, even broken, with ectopic nuclei. These changes were alleviated by treatment with vericiguat (Figure 2A). Masson staining showed clear increases in collagen in RAP rabbits, together with significantly greater CVF, both of which were reversed by vericiguat (Figure 2B and C).
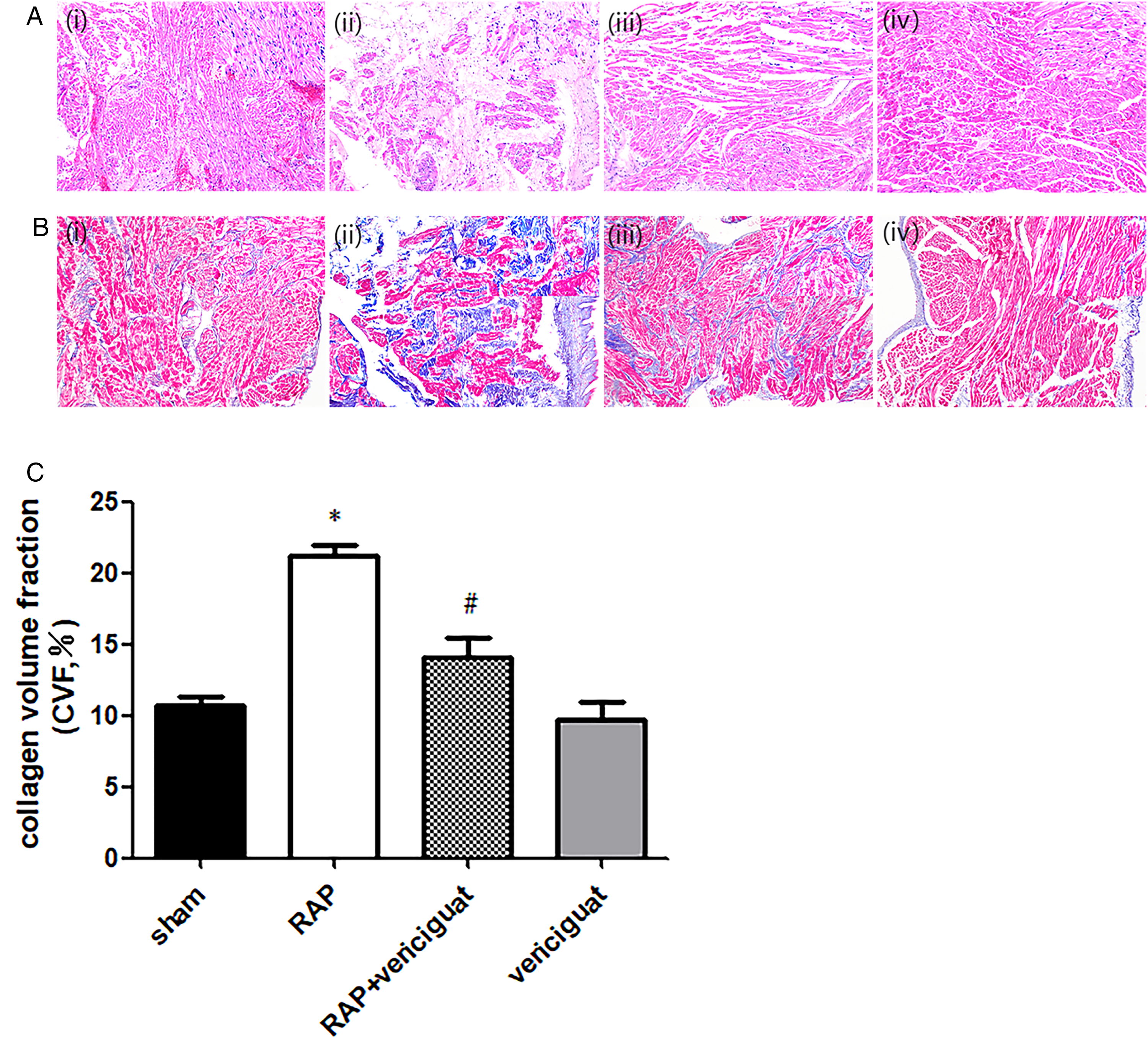
Histological changes in rabbit myocardia. (A) HE staining of left atrial myocardia of rabbits in the different groups. (i) sham, (ii) RAP, (iii) RAP+ vericiguat, (iv) vericiguat. (B) Masson staining of left atrial myocardia of rabbits in the different groups. (i) Sham, (ii) RAP, (iii) RAP+ vericiguat, (iv) vericiguat. (C) Collagen volume fractions (CVF%) in the left atria of rabbits. Magnification, 10× and 40×. *P < .05 vs. sham; #P < .05 vs. RAP; n = 6 per group.
Vericiguat Alters the Level of ST2 in the Plasma of RAP Rabbits
Elevated levels of ST2 were seen in the plasma of rabbits in the RAP group. The increases, however, were reversed after treatment with vericiguat (Figure 3A).
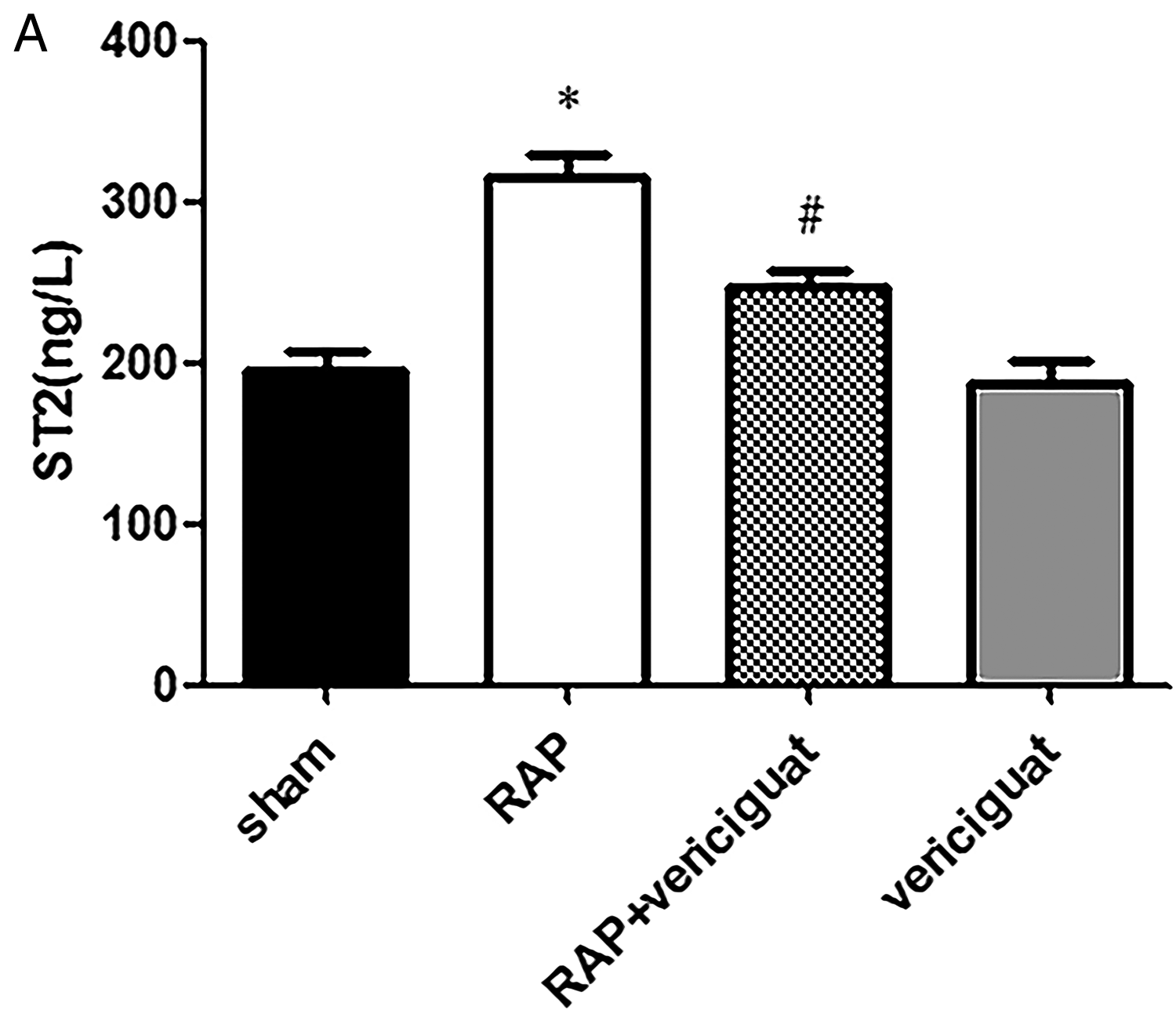
Changes in ST2 levels in rabbit plasma. (A) ST2 levels. *P < .05 vs. sham; #P < .05 vs. RAP; n = 6 per group.
Vericiguat Inhibits the Ca2+ Overload and the Reduction of ICaL in AF Models
A clear increase in intracellular calcium concentrations, measured by the intensity of the green fluorescence, was seen in the HL-1 cell models of high-frequency pacing. These concentrations, however, were reduced after treatment with vericiguat (Figure 4A and B). There was also a marked decrease in the ICaL density in rabbits in the RAP group, which was reversed by vericiguat (Figure 4C).

Changes in Ca2+ concentrations in HL-1 cells and the ICaL in rabbits. (A and B). Images and quantification of Ca2+ concentrations in HL-1 cells, n = 6 in both the control and pacing groups; n = 7 in the pacing + vericiguat group. (C) ICaL densities in rabbits, n = 6 in the sham group; n = 5 in the RAP group; n = 4 in both the vericiguat group and RAP+ vericiguat group. *P < .05 vs. control/sham; #P < .05 vs. pacing/RAP.
Immunohistochemical Analysis of Atrial Tissue from RAP Rabbits
Vericiguat treatment reduced the increases in TRPC6, CaN, and NFAT4 observed in RAP rabbits, together with reversing the reductions in p-NFAT4 and Cav1.2. RAP increased the levels of TRPC6, CaN, and NFAT4, while reducing the levels of p-NFAT4 and Cav1.2, and vericiguat reversed these changes (Figure 5A-F).
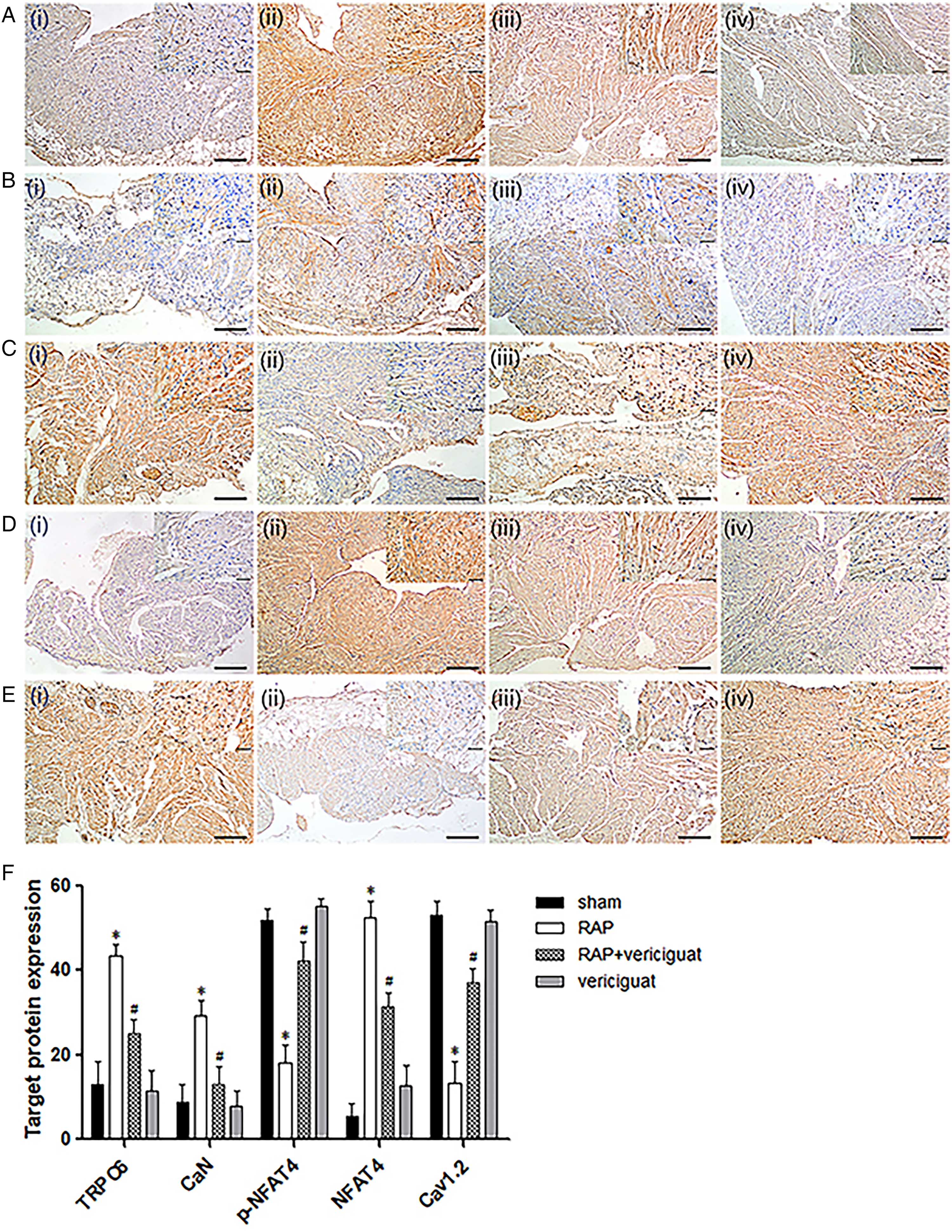
Alterations in the levels of TRPC6, CaN, p-NFAT4, NFAT4, and Cav1.2 rabbit atrial tissue, shown by immunohistochemical analysis. (A–E) Representative images of immunohistochemical staining of TRPC6, CaN, p-NFAT4, NFAT4, and Cav1.2 proteins in the left atrial tissue from different rabbit groups. (i) sham, (ii) RAP, (iii) RAP+ vericiguat, (iv) vericiguat. (F) Quantification of TRPC6, CaN, p-NFAT4, NFAT4, and Cav1.2 levels. Magnification, 10× and 40×. *P < .05 vs. sham, #P < .05 vs. RAP, n = 6 per group.
Western Blotting Analysis of Atrial Tissue of RAP Rabbits and HL-1 Cells
Vericiguat attenuated the up-regulation of TRPC6, CaN, NFAT4, collagen Ⅰ, and collagen Ⅲ, together with the down-regulation of p-NFAT4 and Cav1.2 in AF models. Vericiguat was also found to slow remodeling, both structural and electrical, in the HL-1 cell model of AF through the TRPC6/CaN/NFAT pathway. Figure 6 illustrates the increased protein levels of TRPC6, CaN, NFAT4, collagen Ⅰ, and collagen Ⅲ, and the reduced levels of p-NFAT4 and Cav1.2 in rabbits in the RAP group. These changes were reversed by vericiguat (Figure 6A-D). Likewise, the levels of TRPC6, CaN, and NFAT4 were markedly raised while those of Cav1.2 and p-NFAT4 were reduced, with all changes counteracted by vericiguat treatment in HL-1 cells (Figure 6E and F).

Western blots showing TRPC6, CaN, p-NFAT4, NFAT4, Cav1.2, collagen Ⅰ, and collagen Ⅲ protein levels in AF animal and cellular models. (A-D) Protein levels of TRPC6, CaN, p-NFAT4, NFAT4, Cav1.2, collagen Ⅰ, and collagen Ⅲ in rabbit atria. *P < .05 vs. sham; #P < .05 vs. RAP; n = 6 per group. (E–F) Western blots and quantitative analysis of TRPC6, CaN, p-NFAT4, NFAT4, and Cav1.2 levels in HL-1 cells. *P < .05 vs. control; #P < .05 vs. pacing; n = 4 per group.
Discussion
The principal findings of this investigation were that: (1) Vericiguat slowed the electrical remodeling of the atrium in AF by reducing calcium concentrations and ICaL current density, thus preventing AERP reduction and reducing AF induction, and that these effects might be mediated by the TRPC6/CaN/NFAT pathway; (2) Vericiguat alleviated structural remodeling of the atrium in AF by reducing myocardial fibrosis, mediated by reductions in the levels of collagen Ⅰ, collagen Ⅲ, and ST2.
Electrical remodeling is usually apparent in the early stages of atrial remodeling in AF 19 and is seen in the shortening of AERP and APD, resulting from alterations in calcium currents caused by dysregulated expression of calcium channels. 14 The primary pathway for calcium influx is via L-type calcium channels (LTCCs). 20 The LTCC subtype expressed in the myocardium is Cav1.2, which is responsible for reducing the ICaL4,21 to prevent cytotoxic calcium overload. 22 Our findings are consistent with those of earlier studies while also showing, for the first time, that vericiguat slowed the AF pathology.
CaN, a Ca2+/calmodulin-dependent phosphatase, is responsible for the dephosphorylation of numerous cytoplasmic proteins, including NFAT, which down-regulates Cav1.2 transcription. 14 TRPC6 is a non-selective calcium channel, and increased TRPC6 expression elevates intracellular calcium concentrations in myocardial cells. Cardiomyocyte hypertrophy is then completed through the Ca2+-CaN-NFAT pathway.5,6,23,24 Activation of TRPC6 can promote Ca2+ influx of atrial myocytes, trigger electrical remodeling, and further the occurrence of AF, 25 however, TRPC6 channel blockers can significantly inhibit the above changes. 26 As a second messenger, cGMP regulates many downstream effector molecules, including cGMP-dependent protein kinase G, and participates in the physiological modulation of cardiovascular systems. 27 The cGMP/PKG pathway is reported to be closely associated with the cardioprotective mechanism of ischemic preconditioning and postprocessing. 28 Studies have confirmed the elevation of cGMP levels in chronic AF with decreases seen after successful cardioversion in non-valvular AF. 29 Raised cGMP levels have also been observed after AF onset and measurements of plasma cGMP in patients with paroxysmal AF indicated significantly raised levels compared with patients with sinus rhythm.30,31 It is reported that activation of PKG inhibits TRPC6 channel activity and associated CaN/NFAT signaling, as well as reducing cardiac hypertrophy. 32 Therefore, we assessed the expression of TRPC6, CaN, p-NFAT4, NFAT4, and Cav1.2, as well as the ICaL density and the Ca2+ concentration. Consistent with these findings, we found that 3 weeks of rapid atrial pacing in rabbits raised levels of TRPC6, CaN, and NFAT4 while reducing those of p-NFAT4 and Cav1.2. These results were confirmed in the HL-1 cell models. Our study also showed that high-frequency electrical stimulation resulted in Ca2+ overload in HL-1 cells while rapid atrial pacing in rabbits leads to decreased ICaL densities, resulting in shortened AERP and added AF induction. However, these changes were reversed by vericiguat.
In addition to electrical remodeling, structural remodeling is also present in AF. This involves both enlargement of the atria and fibrosis, adversely affecting atrial conduction and resulting in AF.7,33 The left atrium is critically involved in both the recurrence and continuation of AF. 34 AF can result in left atrial dilation, which is thought to play a part in structural remodeling. 34 A study has also shown that patients tend to develop AF if their left atria are enlarged. 35 In terms of AF-associated structural remodeling, enlargement of the left atrium and fibrosis have been reported in both human postmortem histological analyses and animal models. 34 Structural remodeling is also closely linked with fibrosis of the atrium, interfering with normal electrophysiological activity of the myocardium, preventing normal atrial activation, slowing atrial conduction, and leading to disorganized or asynchronous conduction. 7 Fibrosis is associated with increased collagen accumulation, 36 and it has been reported in both animals and humans that such accumulation is caused by AF. 33 A study comparing structural remodeling between AF-affected and healthy horses found elevated collagen deposition in horses with AF, with more marked differences in the left, rather than the right atrium. 33 The main collagen species in the myocardium are collagens I and III.37,38 There are several proteins in the circulation that function as biomarkers of cardiac remodeling and fibrosis in AF. 39 One such biomarker is ST2 which is expressed by cardiomyocytes when the myocardium is stretched and is involved in myocardial remodeling and fibrosis. 39 Levels of ST2 are known to increase significantly in both heart failure and AF.40,41 Here, marked enlargement of the atria of rabbits in the RAP group was seen on echocardiography, a clear indication of structural remodeling, and the LVEF was also reduced, although it did not reach the degree of HFrEF and worsening of HF. CVF levels also increased significantly in the RAP group, shown by, and western blotting and biochemical assays demonstrated raised levels of collagens I and III and ST2 in the RAP group, an indication that atrial fibrosis was a consequence of rapid pacing. Our research confirmed that vericiguat attenuated the above changes.
Plasma cGMP levels in dogs were found to increase 15 min after the start of pacing, 31 and increased significantly within 30 and 60 min. 42 Compared with patients with sinus rhythm, plasma cGMP concentrations during AF were elevated to a greater extent than in patients with paroxysmal AF, 42 almost four times those of sinus rhythm patients. 31 The authors believed that these high cGMP concentrations might be a protective compensatory reaction to AF.29,43 Treatment of adult rats cardiac fibroblasts with a cGMP analog (8-bromo-cGMP) was found to significantly reduced collagen synthesis induced by angiotensin II.44,45 Raising the levels of cGMP assists in reducing collagen levels and the prevention of myocardial fibrosis. 46 Several drugs used to treat HF that increase cGMP, such as B-type natriuretic peptide and sacubitril/valsartan, can delay structural and electrical remodeling in AF.14,18,47,48 Our study found that vericiguat treatment can slow and prevent both types of remodeling. We have demonstrated the effectiveness of vericiguat both in vitro and in vivo in treating AF and have clarified the possible mechanism behind it. We found that vericiguat inhibited the up-regulation of protein expression of TRPC6, CaN, and NFAT4, as well as the down-regulation of p-NFAT4 and Cav1.2, induced by AF. Our study also found that vericiguat can ameliorate atrial structural remodeling by reducing atrial fibrosis.
Vericiguat is known to be effective for treating chronic HFrEF and worsening HF, slows and prevents myocardial hypertrophy and fibrosis, consistent with our results. The present study is the first to demonstrate that vericiguat can delay structural and electrical remodeling in AF. These findings suggest a new direction for AF treatment and it is feasible that vericiguat may represent a new clinical treatment for AF.
Conclusions
It was found that vericiguat attenuated both electrical and structural remodeling of the atrium in AF models and that these actions might be mediated by the TRPC6/CaN/NFAT pathway. These findings suggest that vericiguat may be a novel treatment for AF.
Supplemental Material
sj-tif-1-cpt-10.1177_10742484231185252 - Supplemental material for Vericiguat reduces electrical and structural remodeling in a rabbit model of atrial fibrillation
Supplemental material, sj-tif-1-cpt-10.1177_10742484231185252 for Vericiguat reduces electrical and structural remodeling in a rabbit model of atrial fibrillation by Qi Lou, Luyifei Li, Guangzhong Liu, Tiankai Li, Li Zhang, Yanxiang Zang, Chengchuang Zhan, Hong Wang and Weimin Li in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgements
Authors are thankful to the lab for providing facilities.
Authors Contributions
QL: conceptualization, methodology, project administration, the resource of the work, the data curation and analysis, writing, reviewing and editing; LL: project administration, validation, investigation and software; GL, TL and LZ: funding, conceptualization and methodology, revision of the work; YZ, CZ and HW: project administration; WL: funding, visualization, and supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Nature Scientific Foundation of Heilongjiang Province (grant_number LH2021H042).
Statement of Data
The datasets available from the corresponding author.
Study Limitations
The species used in the in vivo and in vitro experiment in this study are different.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
