Abstract
Introduction:
Data on optimal dosing of unfractionated heparin (UFH) in the presence of a direct oral anticoagulant (DOAC) to achieve and maintain an activated clotting time (ACT) of ≥300 seconds during catheter ablation of atrial fibrillation (CA-AF) are limited and prevalence of obesity adds to the unpredictable response to UFH.
Methods and Results:
One hundred seventeen consecutive patients undergoing CA-AF were prospectively administered weight-adjusted, weight-based UFH using a pre-specified detailed protocol and retrospectively analyzed. Due to lack of distribution of UFH into muscle or adipose tissue and lower degree of vascularity in the latter compartment, each patient’s ideal and actual weights were used to determine the adjusted-weight for use in all UFH doses. A UFH bolus of 200 units/kg was administered intravenously followed by an infusion of 35 units/kg/hour. The mean age was 65 years, and 85 patients (72.6%) were male. The average body mass index (BMI) was 30 (range 18-50) kg/m2. After the initial UFH bolus dose, 99 patients (84.6%) achieved ACT ≥300 sec with a mean (± SD) of 380 ± 79 sec. The mean time to reach an ACT ≥300 in all patients was 14.6 ± 12.4 minutes. Among all measured ACT values, 423 (90.8%) were ≥300 seconds. These results were consistent within all BMI categories. There were no intraprocedural thrombotic or hemorrhagic complications. Two patients (1.7%) sustained groin vascular access site hematoma without subsequent intervention and 7 patients (6%) experienced minor oozing post-procedurally.
Conclusions:
Our comprehensive weight-adjusted, weight-based UFH protocol, during CA-AF in presence of a DOAC, rapidly achieved and maintained an effective ACT irrespective of BMI.
Keywords
Introduction
CA-AF has become a mainstay of therapy in patients with symptomatic AF refractory to or intolerant of pharmacological treatment. 1,2 Within the last 2 decades, CA-AF has evolved to an electrophysiological procedure commonly performed worldwide. 3 Vigilant attention to anticoagulation during CA-AF is essential to minimize left atrial thrombus formation and systemic thromboembolic complications including stroke, TIA, or asymptomatic cerebral emboli. 1,3 Rapid and effective intraprocedural anticoagulation with UFH is a key component in reducing the risk of such events, as thrombi may form on the transseptal sheaths and catheters almost immediately after crossing the septum into the left atrium. 4 -7 The potential hemorrhagic complications include hemopericardium and tamponade. 1 In FIRE AND ICE and FREEZE trials, the incidence of thrombotic and hemorrhagic complications were comparable between the radiofrequency ablation (RFA) and cryoballoon ablation (CBA) groups. 8,9 Thus, a balance between safe and effective anticoagulation is essential during CA-AF, regardless of ablation modality.
Additionally, recent clinical trials have shown further incremental reduction in stroke complications when CA-AF is performed in the presence of an oral anticoagulant with warfarin, or a DOAC, including dabigatran, apixaban, rivaroxaban, and edoxaban. 10 -14 Concurrent dual anticoagulants with UFH plus an oral agent is a pharmacological and hematological challenge that deserves careful evaluation and analysis, with focused consideration to the UFH protocol to maintain an optimal ACT.
The 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of AF recommends administration of intravenous (IV) UFH prior to or immediately following transseptal (TS) puncture during CA procedures with adjustments to achieve and maintain an ACT of at least 300 seconds intraprocedurally. 1 Despite specific ACT goal recommendations, this expert consensus statement and the more recent updated review are silent on an UFH dosing methodology. 15 Furthermore, published data on specific UFH methods to rapidly achieve and maintain the mandatory optimal ACT intensity is currently limited in scope. 16 -18
UFH does not distribute into muscle or fat tissues and remains exclusively in the vascular circulation. 19,20 In addition, use of actual weight in weight-based UFH dosing has resulted in increased risk of bleeding in some obese patients. 21 Due to lack of distribution of UFH into muscle or fat tissue and limited vascularity in the latter compartment, adjusted-weight, rather than actual weight, was used in all weight-based UFH dose administrations in our protocol. We previously validated successful use of the weight-adjusted, weight-based approach to UFH dosing in the presence of obesity in a small number of patients and employed the same approach in the current UFH protocol. 22,23
In this communication, we describe the first comprehensive, weight-adjusted, weight-based UFH protocol in conjunction with various DOACs and report on the efficacy and safety outcomes in patients with a wide spectrum of BMI. The systematic steps in this UFH protocol are guided by incremental ACT measurements and executed by skilled nursing staff, allowing the electrophysiologist to devote his/her attention solely to the performance of the procedure.
Methods
Consecutive patients with AF undergoing CA with TS puncture in the presence of dual anticoagulation with a DOAC plus full dose IV UFH were included in the analysis. All patients took their DOAC dose the night before the CA and the morning dose on the day of the CA was held. Patients who met one or more of the exclusion criteria were omitted (Figure 1). Eligible patients were categorized into 3 groups based on their BMI: Group I (BMI <25 kg/m2), Group II (BMI 25-30 kg/m2), and Group III (BMI >30 kg/m2).

Assessed, screened, categorized, and analyzed patients. CA = catheter ablation, AF = atrial fibrillation, TS = transseptal, ACT = activated clotting time, DOAC = direct oral anticoagulant, UFH = unfractionated heparin, BMI = body mass index.
Patients undergoing RFA, CBA, or both were prospectively administered UFH with strict adherence to a pre-specified protocol and retrospectively analyzed. Ideal body weight, described by Divine as a function of sex and height decades ago, has been widely adopted in practice for drug dosing purposes and was employed in our protocol. 24 After determining the patient’s ideal body weight, the adjusted-weight was determined by adding the ideal weight to the actual weight and dividing the sum by 2 (Table 1). This adjusted-weight was used in determining all weight-based UFH doses. All patients received a weight-adjusted, weight-based initial UFH bolus of 200 units/kg intravenously, before or within 5 minutes after TS puncture, followed by a continuous infusion of 35 Units/kg/hour. All subsequent UFH dose titrations were ACT driven according to the pre-specified protocol (Table 1). All UFH doses were double checked by 2 registered nurses before administration.
Weight-Adjusted and Weight-Based UFH Protocol During Catheter Ablation of AF When the Last Dose of Apixaban, Dabigatran, or Rivaroxaban Is Taken the Night Prior to the Procedure.a

a It does not apply to edoxaban, enoxaparin, and warfarin.
The TS sheaths were continuously flushed with heparinized normal saline solution (4 units/ml) at 150 ml/hour during the entire procedure. ACT measurements were obtained and analyzed according to 2017 HRS expert consensus recommendations. 1 Abbott i-STAT Celite ACT point-of-care analyzer and i-STAT ACT cartridges were employed for all ACT measurements. The Department of Pathology provided oversight, coordination, maintenance, and quality control of ACT instruments in accordance with the regulatory and accreditation agencies. All patients received elective protamine at the end of the procedure and the femoral venous sheaths were removed when the ACT declined to <175 seconds. “Figure-of-Eight” suture, also known as the Z-stitch, was employed to achieve hemostasis after removal of 14-French caliber femoral venous sheath.
All patients were clinically assessed periprocedurally through discharge. Post-procedure, each patient’s chronic long term DOAC was resumed 6 hours after venous sheath removal and successful establishment of hemostasis. Resumption of DOAC was delayed to the next morning if the vascular access site showed evidence of oozing or hematoma. Femoral venous sheath access sites were monitored for presence of hematoma twice daily and at the time of discharge. All pre- and post-CA DOAC doses followed the United States Food and Drug Administration approved recommendations with appropriate dose adjustments for age, weight, and renal function as indicated. Neurological and physical evaluations were carried out every 4 hours the day of procedure, every 12 hours thereafter, and at the time of discharge. Additionally, all patients were evaluated daily by the EP attending, EP cardiology fellow, and EP nurse practitioner. The definition of the periprocedural period and criteria for thrombotic, hemorrhagic, and vascular access site complications were adopted from the 2017 HRS expert consensus statement on catheter and surgical ablation of AF. 1
This analysis was considered by the Institutional Review Board and authorized to be carried out as a retrospective analysis not requiring IRB approval. All data collection was conducted under a waiver of consent consistent with the requirements of 45 CFR 46.116.d. Access to medical records was authorized under a Waiver of HIPAA Authorization consistent with the requirements of 45 CFR 164.512.i.
Statistical Analysis
Continuous data were described using means
Results
A total of 233 consecutive patients undergoing CA were assessed retrospectively and 117 patients who met the predetermined inclusion criteria were evaluated, while 116 patients were excluded as outlined in Figure 1. There were 18 patients in Group I, 52 patients in Group II, and 47 patients in Group III. The average age was 65 years (range 30-87) and 85 patients (72.6%) were in the male sex category. The average BMI was 30 (Range 18-50) kg/m2. The median CHA2DS2-VASCs score was 3.0 (2.0, 5.0). Hypertension was the most common comorbidity and was present in 100 patients (85.5%). There were no patients on edoxaban and the majority of the patients were transitioned from their chronic long-term DOAC to dabigatran 3-5 days prior to the CA in support of 2017 HRS expert consensus statement with a Class-1a recommendation for the use of dabigatran. 1 Except for BMI and age, the baseline characteristics of the 3 groups—including prior AF ablation, prior CVA/TIA, CHA2DS2-VASCs score, DM, CKD-II-IV, HFrEF, baseline HCT and platelet counts, type of DOAC during AF ablation, and presence of antiplatelets—did not show any statistical difference and are summarized in Table 2.
Baseline Characteristics.
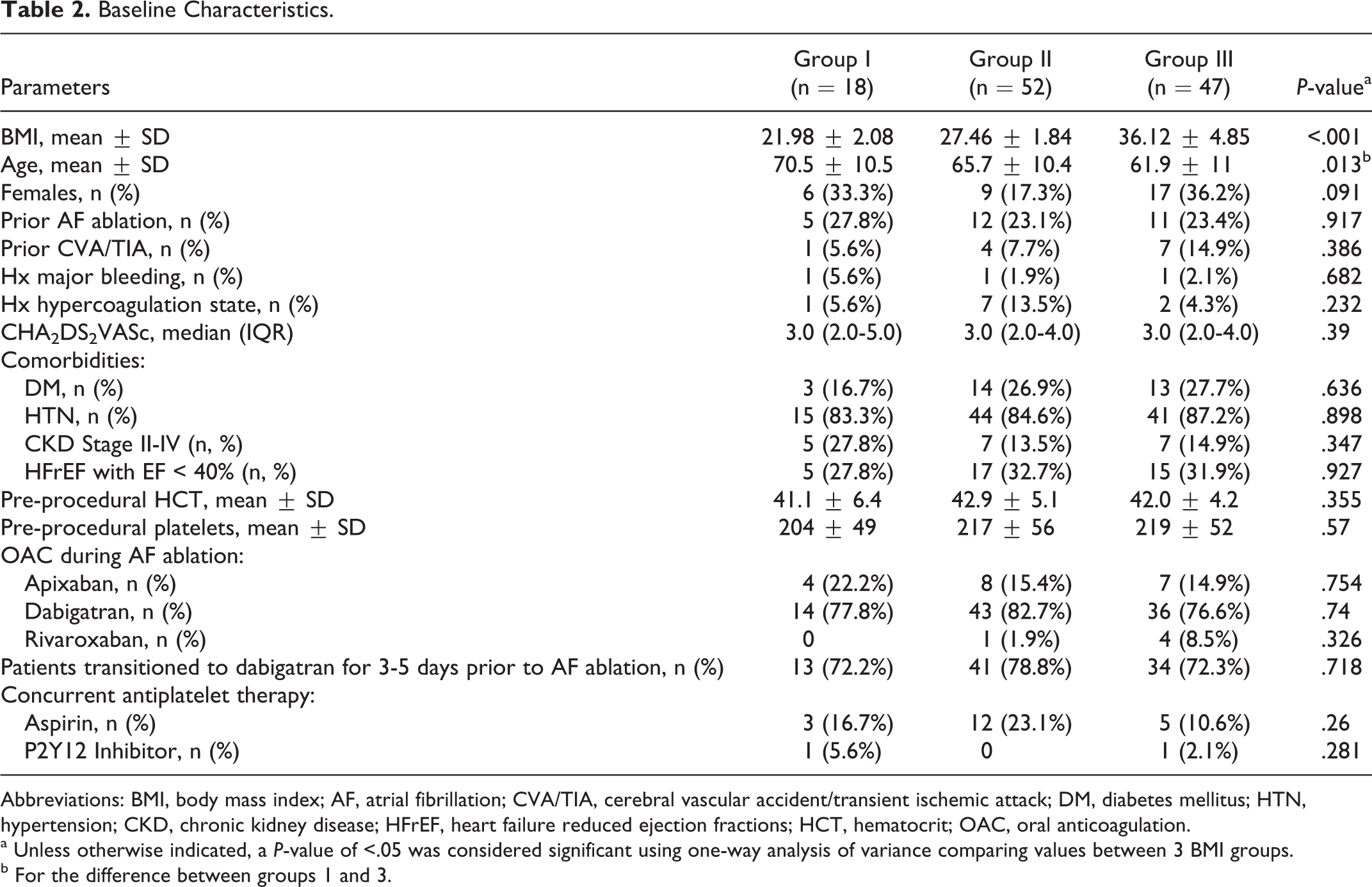
Abbreviations: BMI, body mass index; AF, atrial fibrillation; CVA/TIA, cerebral vascular accident/transient ischemic attack; DM, diabetes mellitus; HTN, hypertension; CKD, chronic kidney disease; HFrEF, heart failure reduced ejection fractions; HCT, hematocrit; OAC, oral anticoagulation.
a Unless otherwise indicated, a P-value of <.05 was considered significant using one-way analysis of variance comparing values between 3 BMI groups.
b For the difference between groups 1 and 3.
All patients underwent pre-ablation transesophageal echocardiography with no evidence of left atrial or left atrial appendage thrombus before transseptal catheterization. More patients received CBA, with or without RFA, than RFA alone. The initial loading dose of UFH was administered prior to TS in 93 patients (79.5%) and the remaining 24 patients (20.5%) received it within 5 minutes of TS. None of the patients required urgent or emergent intra or post-procedural administration of reversal agents or blood products. All patients received elective protamine at the end of the procedure followed by removal of the vascular access sheaths when ACT <175 seconds. Post-procedure, 112 patients (96%) resumed their DOAC on the night of the procedure. Procedural characteristics of the 3 groups did not show any statistical difference and are summarized in Table 3.
Procedural Characteristics.

Abbreviations: TEE, trans-esophageal echocardiography; AF, atrial fibrillation; UFH, unfractionated heparin; TS, transseptal; OAC, oral anticoagulation.
a Packed rRd cells, platelets, fresh frozen plasma, or cryoprecipitate.
After the initial UFH bolus dose, 99 patients (84.6%) achieved an ACT ≥300 seconds and the mean (± SD) was 380 ± 79 seconds. The mean (± SD) time required for all patients to reach an ACT ≥300 seconds was 14.6 ± 12.4 minutes. The efficacy of the protocol after the initial UFH bolus and as a percentage of all intraprocedural ACT values, within each BMI group, is shown in Figure 2. The mean of first, second, third, and fourth ACT values were 380 ± 79, 376 ± 69, 366 ±, 56, and 377 ± 61 seconds, respectively. The distribution of aforementioned ACT values as a function of the 3 BMI groups is shown in Figure 3. There were a total of 466 intraprocedural ACT values representing all 117 patients. Among all ACT values measured, 423 were ≥300 seconds (91%), 298 were 300-400 seconds (64%), 43 were <300 seconds (9%), and 125 were >400 seconds (27%). These outcomes were consistent among the 3 groups with no statistical difference between them. All efficacy outcomes are summarized in Table 4.

Efficacy of the protocol after the initial UFH bolus and as a percentage of all intraprocedural ACT values within the 3 BMI groups.

Distribution of first, second, third, and fourth measured ACT values as a function of 3 BMI groups (sec).
Efficacy Outcomes.

Abbreviations: ACT, activated clotting time; SD, standard deviation.
a A P-value of <.05 is considered significant using the one-way analysis of variance (ANOVA) to compare ACT values between the 3 BMI groups. Bonferroni adjustment was performed and reported only when one-way ANOVA was significant.
There were no intraprocedural thrombotic or hemorrhagic events and postprocedural complications were related to vascular access site. One patient in Group 1 developed a transient migraine accompanied by blurred vision in the right eye post-procedurally. A stat head CT was unremarkable and a neurology consult evaluation did not reveal any gross motor or cognitive neurological deficit. The patient’s migraine symptoms resolved with administration of a minor analgesic. Two patients (1.7%) sustained moderate groin hematoma 8-14 hours after removal of vascular access sheath. The same 2 patients experienced a >20% HCT decline the next day and met the HRS criteria for a postprocedural hemorrhagic event. 1 Neither patient required an intervention and both patients were discharged home without any additional sequalae. Seven additional patients (6%) experienced minor oozing after removal of vascular access sheath and required temporary application of external pressure to establish hemostasis. The overall safety profile of the UFH protocol between the 3 groups is detailed in Table 5.
Safety Outcomes.

Abbreviations: CVA/TIA, cerebral vascular accident/transient ischemic attack; HRS, Heart Rhythm Society.
(a) P value of <.05 is considered significant using one-way analysis of variance comparing values between 3 BMI groups.
(b) Per HRS criteria defined as ≥20% decline in hematocrit including hydration.
(c) Same patient.
(d) Same patient.
(e) Packed Red Cells, platelets, fresh frozen plasma, or cryoprecipitate.
Discussion
Earlier studies have identified intraprocedural ACT as an independent predictor of silent cerebral ischemia (SCI). 25,26 In their well-designed and executed trial, Di Biase and colleagues demonstrated the relevance of dual anticoagulation and validated the role of intraprocedural ACT in prevention of SCI. 27 Their study provided insight into the correlation between SCI and absence of therapeutic INR with warfarin, failure to administer UFH prior to TS, and two or more consecutive ACT measurements <300 seconds; each serving as significant predictor of SCI. In their study, male and female patients received 10,000 or 8,000 units of UFH respectively prior to TS regardless of body weight. Subsequent unspecified UFH doses were administered to maintain an ACT >300 seconds. Patients older than 80 years of age were excluded and time to reach an effective ACT ≥300 seconds is not reported. In our analysis, patients up to age 87 were included and the mean (± SD) time required for all patients to reach an ACT ≥300 seconds was 14.6 ± 12.4 minutes.
Uninterrupted warfarin (COMPARE trial) and uninterrupted DOACs including apixaban (AXAFA-AFNET5 trial), dabigatran (RE-CIRCUIT trial), edoxaban (ELIMINATE-AF trial), and rivaroxaban (VENTURE-AF trial) have shown similar favorable reduction in thrombotic outcomes accompanied by a decline in bleeding complications. 10 -14 In the COMPARE trial, the initial UFH dose was fixed regardless of the patients’ weight, and patients’ BMI were not reported. In the 4 DOAC trials, the UFH dose is not reported and the maximum BMI was close to 35 kg/m2. In the ELIMINATE-AF trial only 66.7% of the cases achieved an ACT >300 seconds during the entire procedure. In our report we offer a single, rapid, and detailed UFH protocol applicable in the presence of apixaban, dabigatran, and rivaroxaban in patients with BMI ranging from 18 to 50 kg/m2.
Konduru, et al reported the differences in intraprocedural ACTs with standardized UFH dosing during CA-AF in patients whose dabigatran was held for 2 doses versus those with uninterrupted warfarin. 28 In this study, the initial UFH bolus was 70 units/kg followed by an infusion rate of 12 units/kg/hr with subsequent heparin dose adjustments at the discretion of the operating electrophysiologist. The time to reach an ACT ≥300 seconds was 45 minutes in the dabigatran group. The UFH dose was based on actual body weight and the baseline characteristics did not include the patients’ weight in either group. In contrast, the time to reach an effective ACT ≥300 seconds was about one third shorter with our UFH protocol regardless of the type of DOAC used or the patient’s BMI.
Dussault, et al reported on the comparison of initial UFH 200 units/kg bolus followed by an infusion of 20 Units/kg/hour compared to 100 Units/kg bolus followed by an infusion of 10 units/kg/hour. 29 The mean times to reach an ACT > 300 seconds were 15.2 min (95% CI 12.9-17.0) and 51.2 min (95% CI 40.6-61.8) respectively. While our protocol utilized the same initial UFH bolus of 200 units/kg, there are several distinct differences in the application of preprocedural DOACs, patient populations, and methodology in the 2 articles. In their study, DOACs were held for 24 hours before the procedure, patients were younger, had leaner BMI obviating the potential for excessively elevated ACT in absence of body weight adjustment in the morbidly obese, subsequent periprocedural UFH bolus doses were not pre-specified, and 23% of the patients did not have AF or flutter. More importantly, while withholding DOACs for 24 hours was standard of care during their study period of 2012-2013, it is no longer supported by the contemporary evidence-based practices.
An extensive meta-analysis and meta-regression conducted by Briceno, et al., included 7,150 patients from 19 studies. 16 For the primary endpoint, patients with ACT >300 seconds had fewer thrombotic events (OR, 0.51; 95% CI 0.35–0.74) and less bleeding (OR, 0.70; 95% CI 0.60–0.83) compared to those with ACT <300 seconds, when using any type of oral anticoagulant. For the secondary endpoint, 7 studies reported data for the total heparin dose required to achieve target ACT (N = 3,256), and 5 studies reported time to achieve target ACT (N = 1,798). Among DOACs users in the 5 studies, the mean time to achieve the target ACT was 49 minutes. In our report, the mean time to achieve an ACT ≥ 300 seconds was 3-fold shorter without any intraprocedural thrombotic or hemorrhagic complications.
Martin, et al reported on periprocedural management of anticoagulation during CA-AF in patients treated with apixaban, dabigatran or rivaroxaban. 30 Patients received fixed doses of UFH ranging from 4,000 to 10,000 units corresponding to doses ranging from 38 to 110 units/kg based on actual body weight. Additional UFH doses to maintain an ACT ≥ 300 seconds were left to the discretion of the electrophysiologist. After the initial UFH bolus, only 18% of their patients reached an ACT ≥300 seconds and 42% never reached an ACT ≥300 seconds during the entire procedure. They concluded that the weight-adjusted UFH dose and subsequent ACT value had a poor correlation and intraprocedural UFH monitoring was complex in the presence of the DOACs studied. Contrary to their findings, the analysis of our weight-adjusted, weight-based UFH protocol achieved an ACT ≥300 seconds in 84.6% of the patients after the initial UFH bolus and maintained it in 91% of all measured ACT values intraprocedurally in the presence of same DOACs and in background of a diverse BMI range.
In a trial by Kishima et al, a protocol for an initial UFH bolus administration during CA-AF was proposed in patients taking direct oral anticoagulants including apixaban, dabigatran, edoxaban, and rivaroxaban. 17 The initial UFH dose was “100 units/kg + 5000 units” in patients with baseline ACT <130 seconds and “100 units/kg + 3000 units” in patients with baseline ACT >130 seconds. Their protocol achieved an ACT ≥300 seconds in 80.5% of their patients in 30 minutes after the initial UFH administration. Their protocol did not include an initial UFH infusion rate, and there is no information on how well their protocol maintained an effective ACT ≥300 seconds during the procedure. Additionally, this study was conducted in a homogenous population and in patients with a uniform BMI of 24 ± 3 kg/m2. While we applaud their efforts in paving the path toward discovery of an optimal UFH protocol during CA-AF, their protocol warrants further testing in a heterogeneous population with greater BMI variation.
In another trial Payne et al. provided evidence that patients on DOACs require higher initial weight-based UFH than those on warfarin and suggested an initial UFH dose of at least 150 units/kg, particularly in those with a weight >90 kg. 18 In their prospective cohort of 25 patients who received a DOAC prior to CA-AF and were administered an initial UFH dose of 160 ± 12.5 units/kg, 11 patients (44%) achieved an ACT ≥300 seconds at 30 minutes and only 49% of all intraprocedural ACT values were ≥300 seconds. Their report did not include the time of TS with respect to the initial UFH dose, the last DOAC dose was held for a wide range of 12-24 hours before the procedure and did not offer a specific UFH infusion rate or subsequent titration doses. Although their conclusion is a meaningful step toward optimizing intraprocedural ACT, over one half of their patients were at risk for thrombotic events in the first 30 minutes and 50% of all measured ACT values were <300 seconds.
False elevation of routine coagulation tests have been reported during DOAC use. 31 In patients who had taken the last dose of DOAC within the previous 12 hours, the activated thromboplastin time (aPTT) and international normalized ratio (INR) were reported to be falsely elevated in 45% and over 65% of them respectively. Due to this interference, we did not correlate the baseline aPPT or INR to the intraprocedural ACT results in our analysis.
Lastly, while the hematological response to UFH may be influenced by the intensity of fibrin clot formation driven by the underlying thrombotic conditions at the time of TS puncture, the complex pharmacological properties of UFH deserve a clear understanding to allow for a meaningful clinical outcome assessment. UFH is a heterogeneous molecule with respect to its size, anticoagulant activity, and pharmacokinetic properties. 19,20,32,33 It requires binding to antithrombin to inactivate factors IIa (thrombin), Xa, IXa, XIa, and XIIa. Unlike other pharmacological agents, the molecular size of the UFH varies widely ranging from 3,000-30,000 kDa with the larger moieties possessing a shorter duration of action. The anticoagulant response to UFH is nonlinear with respect to its intensity and duration and both increase disproportionately at higher doses. UFH resistance is multifactorial including antithrombin-III deficiency, accelerated clearance, elevations in factor VIII, and increased UFH-binding proteins. These hematological and pharmacokinetic complexities account for the variability in response to UFH. Despite significant variation in response to UFH among individual patients, its rapid onset of action, ease of point-of-care monitoring, relatively short duration of action, and immediate reversibility with protamine, make it a useful and at times unpredictable parenteral anticoagulant during CA-AF.
Limitations
While the UFH protocol was carried out prospectively, the data analysis was performed retrospectively. Due to retrospective observational nature of our analysis, power calculations were not considered leading to potential Type II error arising from possible lack of sufficient sample size to detect significance. HAS-BLED risk score was not assessed directly, although many of its independent variables—including hypertension, stroke, bleeding predisposition, age, concurrent use of anti-platelet agents—were evaluated individually. More patients were taking dabigatran than apixaban or rivaroxaban, and there were no patients taking edoxaban. All stroke/TIA assessments were clinical and neuroimaging diagnostics were not utilized. The follow up period was limited to inpatient length of stay.
Future Directions
We plan to carry out a prospective analysis of the same protocol in a larger group of patients representing a more equitable distribution of DOACs prior CA-AF.
Conclusions
Our comprehensive, weight-adjusted, weight-based UFH protocol, during CA-AF in presence of different DOACs, rapidly achieved and maintained an effective initial ACT ≥300 seconds in the majority of the patients. There were no major intraprocedural thrombotic or hemorrhagic complications. The most common complications were related to post-procedure vascular access sites. All outcomes were consistent in patients with a wide range of BMI. With strict adherence to its elements, this protocol provided wide-ranging efficacy and safety. We did not include patients who were taking warfarin or enoxaparin, and there were no patients on edoxaban, prior to CA-AF and our UHF protocol may not be suitable in such settings.
Footnotes
Acknowledgments
The authors would like to acknowledge the support of Dr. Jennifer Le for her statistical analyses, Grace Lee, PharmD, and Taryn Ng, PharmD, for partial contributions with data collection and interim analysis during their internship from School of Pharmacy, University of California, San Francisco, CA, Christina Benton for graphical skills, Matt Franko for proofreading, and the entire catheter ablation team including cardiac anesthesiologists, nursing, and fluoroscopy technicians for their outstanding patient care during the procedure.
Author Contributions
All authors made substantial contributions in varying degrees to the design, analysis, write up, and revisions and provided final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
