Abstract
There are limited data from randomized controlled trials comparing rate control agents in atrial fibrillation. Patient-level data from the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) trial was used to compare outcomes in patients randomized to the rate control arm who were treated with a single rate control agent at baseline. The rate control agents used were beta-blockers, non-dihydropyridine calcium channel blockers, and digoxin. The independent variable for this analysis was the initial study drug used and the dependent variables were time to first hospitalization and time to death from any cause. We analyzed 1,144 out of 2,027 participants assigned to the rate control group who were on a single rate control agent at the start of the trial. There were 485 (42.5%) participants in the beta-blocker group, 344 (30%) in the calcium channel blocker group, and 315 (27.5%) in the digoxin group. All hospitalization and all-cause mortality occurred in 55.9% and 12.5% of those in the beta-blocker group, 58.4% and 16.7% in the calcium channel blocker group, and 55.2% and 21.1% in the digoxin group, respectively. After adjustment for differences in baseline characteristics, there were no significant differences in time to hospitalization or death for any group. In the AFFIRM trial, the initial rate control drug used was not associated with statistically significant differences in time to hospitalization or death after controlling for differences in baseline characteristics. There is limited data at present to guide the selection of rate control agents in patients with atrial fibrillation.
Introduction
Atrial fibrillation (AF) is the most common sustained arrhythmia in the United States. 1 Overall prevalence of the disease is noted to be between 0.5%-4% in the adult population. 2,3 AF is also the most common arrhythmia requiring hospital admission. 3 An optimal strategy to manage this abnormal rhythm continues to be a challenge for clinicians. AF is associated with stroke, heart failure, cardiomyopathy, and many other medical co-morbidities, including death. 2,3 There are generally 3 principles to managing AF: (1) anticoagulation, (2) ventricular rate control, and (3) rhythm control with the maintenance of sinus rhythm. The Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) study was a large, multicenter, randomized controlled trial that compared rate control against rhythm control strategies. 4 The AFFIRM trial suggested that rate control was an acceptable alternative to rhythm control and may have a lower risk of adverse drug toxicities without affecting overall mortality. 4 Many clinicians prefer to use rate control agents over rhythm control agents due to the findings of the study. A subsequent follow-up analysis of the AFFIRM study indicated that beta-blockers were the most effective drug for rate control. 5 This study primarily evaluated for adequate pharmacological rate control at rest and with exercise without evaluating any clinical outcomes. 5 The 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation recommends either a beta-blocker or a non-dihydropyridine calcium channel blocker as first-line agents for controlling ventricular rate in patients with AF. 6 Digoxin is not recommended as a first-line treatment. 6 Currently, there is limited data from small randomized controlled trials comparing beta-blockers, non-dihydropyridine calcium channel blockers, and digoxin for patients with chronic AF. 7 -22
This study aimed to compare the long-term effects, including the time to all-cause mortality and the first hospitalization from any cause, of pharmacological rate control agents for patients with AF using the data from the landmark, AFFIRM study. We used patient-level data to compare outcomes in patients randomized to the rate control arm who were treated with a single rate control agent at baseline, including beta-blockers, non-dihydropyridine calcium channel blockers, or digoxin.
Methods
Data Collection
The investigation assessed the clinical effectiveness of rate control agents by analyzing patient-level data from the AFFIRM trial. The AFFIRM study was approved by the AFFIRM Steering Committee, the Institutional Review Board of the University of Washington (Clinical Trial Center), and by each participating site’s Institutional Review Board. Participants of the AFFIRM study gave informed and written consent to participate in the study. Patient-level data from the AFFIRM trial was provided through the NHLBI BioLINCC biologic specimens and data repository information coordinating center. Participant treatment assignment was gathered from the enrollment data set. Baseline characteristics and initial drug used was retrieved from the baseline data set. Information on hospitalization and death were obtained from the follow-up and death data sets.
Patient Population
There were 2,027 patients in the rate control treatment arm of the AFFIRM trial. The participants in the rate control group were not randomized. Beta-blockers, non-dihydropyridine calcium channel blockers (diltiazem and verapamil), digoxin, and a combination of these drugs were used for rate control purposes. However, this analysis only included patients randomized to the rate control arm who were treated with a single rate control agent at baseline. The final data analysis included 1,144 out of 2,027 patients after excluding the combination drug groups.
Choice of Drugs and Study Protocol for Rate Control
The 3 atrioventricular (AV) nodal blocking agents were chosen by treating physicians, and the agent of choice was not randomized. Heart rate control was only considered if the patient was in AF at the time of evaluation. Adequate rate control was defined as a resting heart rate of 80 beats per minute or less. If a patient met the resting heart rate criteria, then a standard 6-minute walk test or a 24-hour ambulatory electrocardiographic recording (Holter monitor) was required to confirm adequate rate control. Goal heart rate during a 6-minute walk test was less than 110, and during 24-hour ambulatory Holter monitoring, it was an average of fewer than 100 beats per minute. Overall, adequate heart rate control required evidence of rate control during rest and exercise as defined above. Drug doses were titrated to achieve adequate rate control, and physicians could change drug regimens or add another drug to attempt to achieve adequate rate control. Tests needed to be repeated every time a drug dose or agent was changed. The medication was stopped if the participant developed adverse effects, such as worsening heart failure, hypotension, or bradycardia. If the pharmacological approach was not successful in achieving adequate rate control, the treating physician could proceed with AV junctional ablation, per the AFFIRM trial protocol.
Statistical Analysis
The independent variable for this analysis was the initial study drug used, which was classified as a beta-blocker alone, a calcium channel blocker alone, or digoxin alone. The dependent variables were time to the first hospitalization from any cause and time to death (cardiac and non-cardiac). Kaplan-Meier analysis, with initial therapy as the stratification variable, was performed, and the Log-rank test was reported. Cox proportional hazards regression was performed as a secondary analysis to adjust for baseline characteristics, including a history of angina pectoris, myocardial infarction (MI), coronary artery bypass grafting (CABG) or coronary artery disease, congestive heart failure or cardiomyopathy, symptomatic bradycardia, AV block or pacemaker implantation, stroke, peripheral vascular disease, diabetes, hepatic or renal disease, pulmonary disease, taking 5 or more medications, and age group.
Adverse events, such as time to (1) Torsades de pointes, sustained ventricular tachycardia or resuscitated cardiac arrest, (2) ischemic stroke, (3) major bleeding, and (4) disabling anoxic encephalopathy, systemic or pulmonary embolism, or MI, were analyzed using the Kaplan-Meier and log-rank test. Data were censored at the time of death or last contact if looking at an endpoint other than death. Baseline characteristics were analyzed using the chi-square test for categorical variables and the F-test or Kruskal-Wallis test for continuous variables, as appropriate.
Results
Patient Characteristics
The final analysis included 1,144 patients based on the initial single rate control agent at baseline. Patient characteristics are summarized in Table 1. Significant differences existed between baseline patient characteristics, including sex, history of angina pectoris, MI, CABG, or coronary artery disease, congestive heart failure, or cardiomyopathy, currently in AF or atrial flutter, left ventricular (LV) internal diastolic dimension, LV ejection fraction, and pulmonary disease. The majority of the patients were male (62.9%) and the mean age was 69.6 ± 8.0.
Baseline Clinical Characteristics of Patients.*

* Plus-minus values are means ± SD.
† Echocardiograms were obtained in 1,650 patients in the rate-controlled group. Left Ventricular ejection fraction was unknown in 279 patients. Normal ejection fraction was > 50%.
AV = atrioventricular, CABG = coronary artery bypass grafting, MI = myocardial infarction, TIA = transient ischemic attack.
Therapy
Out of the 1,144 patients included in the analysis, 485 (42.5%) were treated with a beta-blocker alone, 344 (30.0%) were treated with a non-dihydropyridine calcium channel blocker alone, and 315 (27.5%) were treated with digoxin alone. The initial AFFIRM study included 292 patients who received beta-blockers and digoxin in combination, 287 patients who received calcium channel blockers and digoxin in combination, 154 patients who received other drugs, other combinations, or no therapy, and 150 patients with missing data.
More patients were treated with beta-blockers alone compared to calcium channel blockers and digoxin. Beta-blockers were used more often for patients with angina, MI, CABG, or coronary artery disease history (43.3% versus 30.2% received calcium channel blockers and 35.6% received digoxin). Digoxin was used more often in patients with congestive heart failure or cardiomyopathy (37.8% versus 15.7% received beta-blockers and 15.4% received calcium channel blockers). Similar percentages of patients, who had congestive heart failure or cardiomyopathy, were on beta-blockers and calcium channel blockers. LV ejection fraction was lowest for the patients on digoxin alone (average was not less than 50%). Patients with pulmonary disease were more likely to be on calcium channel blockers alone and digoxin alone (19.8% and 16.2%, respectively versus 6% on beta-blockers).
Primary Endpoints
The primary endpoints of time to all-cause mortality and all hospitalization are summarized in Figure 1 and Figure 2, respectively. Table 2 summarizes the primary endpoints and secondary adverse events. Regarding all-cause mortality, 59 (12.5%) patients in the beta-blocker group, 56 (16.7%) patients in the calcium channel group, and 66 (21.1%) patients in the digoxin group alone died at the end of the 5 years follow up. However, secondary analysis accounting for the baseline covariates did not show significant differences in mortality. The results of the secondary analysis are summarized in Figure 3. Hazard ratio (HR) for beta-blockers compared to digoxin was 0.78 [95% confidence interval (CI) 0.54-1.14], HR for calcium channel blockers compared to digoxin was 0.95 [95% CI 0.65-1.37], and HR for beta-blockers compared to calcium channel blockers was 0.83 [95% CI 0.57-1.21].

Probability of death from any cause in the beta-blocker group alone, the non-dihydropyridine calcium channel blocker group alone, and the digoxin group alone. * Represents unadjusted data. Time zero is the day of randomization. Data truncated at 5 years or 1825 days. BB = beta-blocker, CCB = calcium channel blockers.
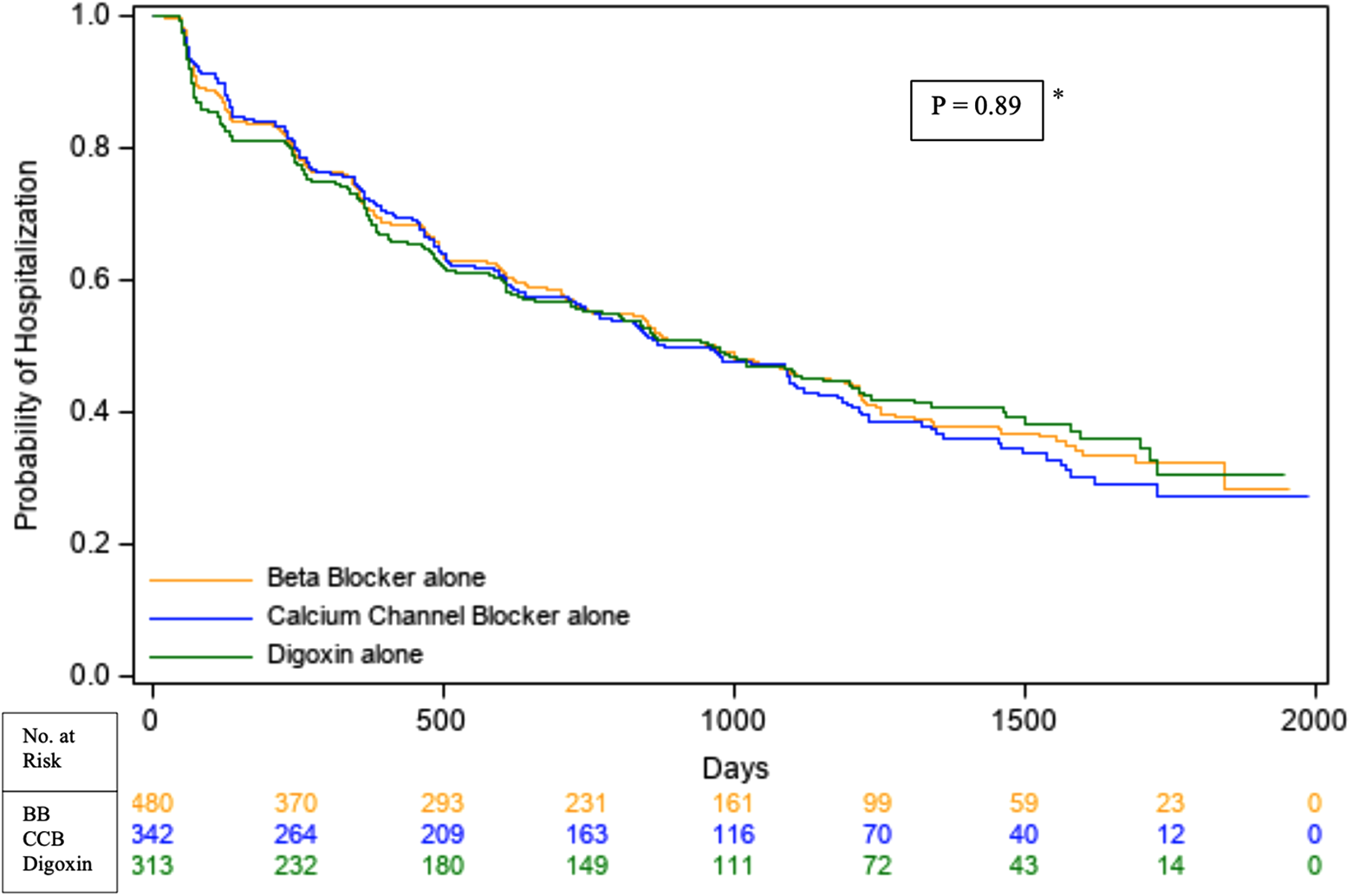
Probability of hospitalization from any cause in the beta-blocker group alone, non-dihydropyridine calcium channel blocker group alone, and the digoxin group alone. * Represents unadjusted data. Time zero is the day of randomization. Data truncated at 5 years or 1825 days. BB = beta-blocker, CCB = calcium channel blockers.
Primary Endpoints and Secondary Adverse Events.*

* Number of patients (%) reported.
† Percentages were derived from Kaplan-Meier analysis. P-values were derived from the log-rank statistic. After adjustment for baseline covariates, no significant differences were noted.
MI = myocardial infarction, VT = ventricular tachycardia, NS = not significant.

Secondary analysis accounting for baseline covariates for primary outcomes (time to all-cause mortality and all hospitalization) represented as hazard ratios. All results were statistically non-significant. BB = beta-blocker, CCB = calcium channel blocker, vs. = versus.
There were no significant differences between the 3 groups regarding hospitalization at 5 years. When accounting for baseline covariates, HR for beta-blockers compared to digoxin was 1.12 [95% CI 0.91-1.37], HR for calcium channel blockers compared to digoxin was 1.08 [95% CI 0.88-1.34], and HR for beta-blockers compared to calcium channel blockers was 1.03 [95% CI 0.85-1.25] regarding hospitalization.
Other Adverse Events
Secondary adverse events are listed in Table 2 along with primary endpoints. There were no significant differences between the 3 rate control agents for Torsades de pointes, sustained ventricular tachycardia, resuscitated cardiac arrest, ischemic stroke, major bleeding, disabling anoxic encephalopathy, system or pulmonary embolism, and MI.
Other Observations From the AFFIRM Trial
Changes in treatment were frequent in the AFFIRM trial. Within the first year, 23% of patients were switched from calcium channel blocker to beta-blocker, 19% were switched from beta-blocker to calcium channel blocker, and 34% began either beta-blocker or calcium channel blocker after only having taken digoxin. The AFFIRM trial reported only 34.6% of rate-controlled patients were in sinus rhythm at 5 years, and this study did not analyze the rhythm status of patients on rate-controlled agents. A total of 108 patients underwent radiofrequency ablation of the AV junction to eliminate AV conduction. Finally, during the trial, 248 patients crossed over from the rate-control group to the rhythm-control group. However, 86 of those patients crossed back over to the rate-control group by the end of the study.
Discussion
In this study, rate control agents in patients with AF using the patient-level data from the landmark AFFIRM trial were compared. The analysis included initial single-agent therapy at baseline, and the agents were beta-blockers, non-dihydropyridine calcium channel blockers, and digoxin after excluding any combination therapy. The AFFIRM trial was randomized between rate-control and rhythm-control groups; however, the rate control group itself was not randomized. Patients received different rate control agents based on the preferences of their treating physicians. Significant differences existed between baseline clinical characteristics, including sex, history of angina pectoris, MI, CABG, or coronary artery disease, congestive heart failure or cardiomyopathy, currently in AF or atrial flutter, LV internal diastolic dimension, LV ejection fraction, and pulmonary disease, likely secondary to patients being non-randomized and the aforementioned physician preferences. All of the baseline clinical characteristics, except LV internal diastolic dimension and currently in AF or atrial flutter, were included in the secondary analysis as covariates. Most of the patients in the study were males and elderly (greater than 65 years old) similar to current epidemiology of AF. 23,24
Again, likely due to lack of randomization, treatment groups were not equal across the 3 arms. Significantly more participants were on the beta-blocker group compared to the calcium channel blocker and the digoxin groups. However, the calcium channel blocker and the digoxin groups were similar in number. As expected, beta-blocker was used more frequently in patients with angina, MI, CABG, or coronary artery disease history, and much less frequently in patients with pulmonary disease. Surprisingly, similar percentages of patients with congestive heart failure or cardiomyopathy were in the beta-blocker and non-dihydropyridine calcium channel blocker groups. However, significantly more patients with congestive heart failure or cardiomyopathy were in the digoxin group. Similarly, patients in the digoxin group had the lowest LV ejection fraction.
The primary endpoints of the study were time to all-cause mortality and hospitalization from any cause. The digoxin group had a higher all-cause mortality rate compared to the non-dihydropyridine calcium channel blocker and beta-blocker groups. The beta-blocker group had the lowest risk of all-cause mortality at 5 years. Kaplan-Meier analysis showed a difference between the 3 groups regarding time to all-cause mortality; however, secondary analysis with proportional hazard regression did not show differences between the 3 groups. This finding is likely due to non-randomized patients along with physician selection bias. There were no significant differences between the 3 groups regarding time to all hospitalization at 5 years with primary and secondary analysis. No other significant differences were noted between the 3 groups regarding Torsades de pointes, sustained ventricular tachycardia, resuscitated cardiac arrest, ischemic stroke, major bleeding, disabling anoxic encephalopathy, systemic or pulmonary embolism, and MI. A large part of the participants crossed over to the different groups of rate control therapy during the AFFIRM trial.
Another analysis of rate control agents from the AFFIRM trial was performed by Olshansky et al. 5 In this study, the primary objective was to investigate the effectiveness of rate control agents and switches from one class of drug to another as it was very frequent in the AFFIRM trial. This analysis included beta-blocker alone, non-dihydropyridine calcium channel blocker alone, digoxin alone similar to our analysis but also, included a combination of beta-blocker and digoxin along with calcium channel blocker and digoxin. The overall finding of the study showed that rate control could be achieved in the majority of patients and that beta-blockers were the most effective rate-controlling drug. This study did not evaluate different clinical outcomes such as mortality and hospitalization when comparing different rate-controlling agents as our analysis of the AFFIRM trial did.
Digoxin is one of the least used rate control agents in patients with AF. Digoxin seems to be the least effective, but maybe a good choice for elderly patients, with difficult to control heart rate and patients with heart failure. 25 Several studies have shown increased overall mortality with digoxin in patients with AF. 26 -28 However, after accounting for patient characteristics, such as older age and frailty, one study demonstrated that digoxin use had a neutral effect on mortality and major cardiovascular events in patients with AF. 29 In one analysis of the AFFIRM trial by Whitbeck et al., digoxin was associated with increased mortality even after accounting for baseline characteristics and comorbidities. 30 However, this study included patient populations from both rate and rhythm control groups. Another post-hoc propensity-matched analysis of the AFFIRM trial indicated no evidence of increased mortality or hospitalization in patients taking digoxin at baseline. 31 Another topic to consider regarding mortality is that the AFFIRM trial had a fairly strict heart rate control with a goal resting heart rate less than 80. Studies performed after the AFFIRM trial such as RACE II indicated similar outcomes in patients with strict (less than 80) and lenient (less than 110) heart rate control with increasing morbidity and mortality toward the strict rate control group. 32 Perhaps, a mortality and hospitalization benefit would be observed in this analysis if patients had lenient heart rate control at rest. One subset of the population that should get special attention is patients with congestive heart failure as treatment options for them differ. Non-dihydropyridine calcium channel blockers are usually avoided in patients with heart failure with reduced ejection fraction. Certain beta-blockers have been shown to improve mortality in patients with heart failure with reduced ejection fraction. Digoxin can also be used in this patient population. One study comparing rate and rhythm control in patients with heart failure with reduced ejection fraction (less than 35%) showed no significant differences between the 2 strategies. 33 However, again, in this study, a strict heart rate control of 80 beats per minute was used. More prospective studies are needed with heart failure patients with lenient heart rate control in the treatment of AF.
Conclusion
This analysis of the landmark AFFIRM trial did not show a clear best rate control treatment group for patients with AF. Primary analysis favored beta-blockers over non-dihydropyridine calcium channel blockers and digoxin; however, this favorability faded with adjustment of baseline covariates. The major limitations of this study included a lack of randomization within the rate control group in the AFFIRM trial along with physician selection bias. There were numerous crossovers between different therapies. Finally, patients in the rate-control group of the AFFIRM trial were under strict heart rate control. Further randomized prospective comparative studies are needed to determine the best rate-controlling therapy for patients with AF.
Footnotes
Author Contributions
All authors contributed equally to the design of the investigation, analysis and interpretation of data, drafting and revising the manuscript, and approving the version to be published.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Gerald Naccarelli, MD (Consultant and Research funding: Janssen; Consultant: Milestone, Sanofi, Omeicos, Acesion).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant disclosure: This research project was funded by a research grant provided by the Department of Medicine at the Pennsylvania State College of Medicine, Hershey, Pennsylvania, United States of America.
