Abstract
Background:
Nitrates and nitrate-containing compounds are vasodilators used for the treatment of angina and heart failure. Phosphodiesterase type 5 inhibitors used for the treatment of erectile dysfunction are also vasodilators, and when taken together with nitrates, synergistic effects that enhance hypotensive effects may occur. Phosphodiesterase type 5 inhibitors are therefore contraindicated in patients taking organic nitrates.
Methods and Results:
A literature review was performed to provide a historical overview of different phosphodiesterase type 5 inhibitors and nitrates and their interaction. The pharmacologic characteristics of phosphodiesterase type 5 inhibitors and nitrates are reviewed, and clinical recommendations for treating cardiovascular disease in men taking phosphodiesterase type 5 inhibitors are discussed. Pharmacologic and adverse drug reactions between nitrates and phosphodiesterase type 5 inhibitors are dependent on many variables. Organic nitrates remain an absolute contraindication in men treated with phosphodiesterase type 5 inhibitors. In general, nitrates may be taken 24 hours after the last dose of short-acting phosphodiesterase type 5 inhibitors and 48 hours after the last dose of long-acting phosphodiesterase type 5 inhibitors.
Conclusions:
This literature review determined that the use of phosphodiesterase type 5 inhibitors with nitrates is a contraindication, with the duration between the last dose of phosphodiesterase inhibitor and nitrate use generally varying between short- and long-acting phosphodiesterase type 5 formulations. Patients receiving nitrates who wish to use phosphodiesterase type 5 inhibitors should be educated regarding the interaction and should be evaluated to determine whether nitrate treatment can be discontinued. Further research is needed to determine how soon phosphodiesterase type 5 inhibitors can be restarted after a patient has taken a nitrate and the effect of high and low phosphodiesterase type 5 inhibitor doses on the interaction effect.
Why Is There an Interactive Effect of Phosphodiesterase Type 5 Inhibitors and Organic Nitrates on Blood Pressure?
Phosphodiesterase type 5 (PDE5) inhibitors have transformed the treatment of erectile dysfunction (ED) and are the current first-line management approach. 1,2 Existing formulations allow for flexibility in prescribing and tailoring to patient preference. 1 A consequence of the increased use of PDE5 inhibitors is elucidating appropriate management of potential drug interactions, particularly in patients with comorbid conditions. For instance, PDE5 inhibitors indicated for the treatment of ED, including sildenafil (Viagra; Pfizer Inc, New York, New York), tadalafil (Cialis; Eli Lilly and Company, Indianapolis, Indiana), vardenafil (Levitra; Bayer HealthCare, Whippany, New Jersey), and avanafil (Stendra; Vivus, Inc, Mountain View, California), are contraindicated in patients taking organic nitrates. 3 -6
Hemodynamic effects of PDE5 inhibitors are evident. An article by Jackson and colleagues summarizing the results of early pharmacodynamic studies of sildenafil revealed that single intravenous (40-80 mg) or oral (up to 200 mg) sildenafil administration had modest (mean decrease ≤10 mm Hg) and transient effects on systolic blood pressure (SBP) and diastolic blood pressure (DBP) in healthy male volunteers and in men with stable ischemic heart disease who were not taking other antianginal medication. 7 Despite reductions in BP, there was no significant increase in heart rate. Subsequently, in a single-dose, double-blind, placebo-controlled study in 31 men with ED and coronary artery disease, Jackson and colleagues reported that mean changes from baseline in central and peripheral hemodynamic pressures with sildenafil 100 mg were greater than those with placebo but less than those with isosorbide mononitrate 40 mg. 8 In general, the hemodynamic effects of orally administered sildenafil 100 mg peaked 1 to 2 hours after dosing and returned to approximately baseline values at 6 hours. There were no significant differences between treatments in heart rate responses. Thus, systemic administration of a PDE5 inhibitor (alone) results in modest vasodilation and reductions in BP. Drug interaction studies were needed to determine the hemodynamic effects of PDE5 inhibitors when coadministered with organic nitrates.
An understanding of the biochemical effects of PDE5 inhibitors is important for the understanding of the mechanism of associated drug interactions. When sexual stimulation occurs, nitric oxide (NO) is released from the nonadrenergic–noncholinergic nerves in the corpus cavernosum, or the erectile body, of the penis as well as from the endothelial cells of the vasculature within the corpus cavernosum (Figure 1). 9 Nitric oxide activates the enzyme guanylate cyclase, which results in an increased production of cyclic guanosine monophosphate (cGMP). 10 Cyclic guanosine monophosphate induces relaxation of the smooth muscle cells in the blood vessels and sinusoids of the corpus cavernosum, which increases blood flow into the penis. 11 The sinusoids fill with blood, and as these spaces expand, they compress the veins that drain blood out of the erectile bodies against the tunica albuginea, trapping increased amounts of blood in the penis, resulting in an erection. 12 The enzyme PDE5 is found in relatively high concentrations in the blood vessels of the corpus cavernosum. 13 When PDE5 is activated, it breaks down cGMP, which can thus reduce smooth muscle relaxation and lead to a loss of an erection. Phosphodiesterase type 5 is also found in the systemic and cardiac vasculature. 14 Phosphodiesterase type 5 inhibitors impair cGMP catabolism and thus maintain vasodilation. 3 -6 For an erection to continue, there is a delicate balance between the production and the degradation of cGMP. When sexual stimulation ceases, cGMP production drops, PDE5 breaks down any remaining cGMP, and the penis returns to its flaccid state. In addition, orgasm and ejaculation are associated with the release of molecules such as noradrenaline. 15 Noradrenaline induces contraction of smooth muscles in the arterial walls and sinusoids, further decreasing blood flow to the erectile bodies and allowing the veins in the penis to drain blood away from the penis, contributing to detumescence. 16

Nitric oxide–cyclic guanosine monophosphate mechanism of corpus cavernosum smooth muscle relaxation and penile erection. Adapted with permission from Ignarro et al. 10 cGMP indicates cyclic GMP; GMP, guanosine monophosphate; GTP, guanosine triphosphate; NANC, nonadrenergic–noncholinergic nerves; NO, nitric oxide; PDE5, phosphodiesterase type 5.
Organic nitrates, including short-acting antianginal formulations [eg, sublingual nitroglycerin (NTG) tablets, or spray], long-acting formulations (eg, transdermal NTG, isosorbide dinitrate, isosorbide mononitrate tablets, pentaerythritol tetranitrate [commonly used in Europe]), and other nitrate- or nitrite-containing compounds (eg, nicorandil), are among the approved treatments for the relief of angina. 17 Nitrates and nitrate-containing compounds are also used for the treatments of heart failure. 18 Nitrates are considered to be NO donors, thereby increasing NO bioavailability or at least some NO-containing moiety. 17 Regardless of their exact mechanism, organic nitrates increase the production of cGMP, resulting in systemic vasodilation, which may cause subsequent hypotension. If an increased production of cGMP due to organic nitrates is combined with reduced degradation of cGMP due to the action of PDE5 inhibitors, cGMP levels are further increased. 19 Thus, the additive effect of these 2 classes of vasodilators causes an augmented hypotensive response. This pharmacodynamic interaction is the reason that PDE5 inhibitors are contraindicated in patients taking organic nitrates.
Reports of the Nitrate–PDE5 Inhibitor Interaction
The major studies evaluating the interaction of PDE5 inhibitors and nitrates are listed in Table 1. The first study to demonstrate the interaction between sildenafil and nitrates was published by Webb and colleagues in 1999. 20 This article, which described the results of a randomized, double-blind, placebo-controlled, crossover study in healthy men, provided conclusive evidence that sildenafil potentiates the hypotensive effects of NTG. The men were randomized to oral sildenafil (3 times a day) or placebo for 4 days, with a single morning dose on the fifth day. On day 4 after the morning dose of sildenafil, participantsreceived graded intravenous infusions of NTG. On day 5, a NTG tablet was given via the sublingual route 1 hour after the morning dose of sildenafil. Blood pressure was monitored continuously after NTG administration on days 4 and 5. The men were instructed to remove the NTG tablet if they developed symptomatic hypotension or if their SBP decreased by >25 mm Hg. To maximize the chance of observing any interaction, the healthy volunteers were maintained at an angle of 70° on a tilt table from before their morning dose of sildenafil until after the nitrate infusion on day 4 and until 30 minutes after their sublingual nitrate on day 5. Following a ≥10-day washout phase, the participants crossed over to the opposite study drug. Two of the 12 men finished the 25-minute stepwise NTG infusion during placebo treatment and none during sildenafil treatment. During NTG infusion, the median cumulative dose of NTG was 72 μg with placebo treatment versus 33 μg with sildenafil treatment (P < .01). The median duration of the nitrate infusion was 13 minutes during placebo treatment versus only 9 minutes with sildenafil treatment (P < .01). Four of the 12 men receiving placebo removed the sublingual NTG tablet after 3 to 4 minutes, whereas 11 men removed the nitrate tablet during sildenafil treatment after 2 to 7 minutes because of symptomatic hypotension or a decrease in SBP of >25 mm Hg. During treatment with placebo, the men removed the nitrate tablet at a median time of 15 versus 4.5 minutes during sildenafil treatment (P < .01). Beginning at 3 minutes after the start of sublingual NTG, there was a greater decrease in SBP in participants receiving sildenafil compared with placebo. A >4-fold decrease in SBP from baseline was observed at 7 minutes after sublingual nitrate with sildenafil compared with placebo (Figure 2A). The changes in DBP with sildenafil or placebo plus nitrate were minimal (Figure 2B). The changes in heart rate were negligible when NTG was given with sildenafil. Headache and dizziness were the most common treatment-related adverse events during treatment with sildenafil plus NTG. These events were predominantly mild to moderate and did not result in discontinuation of treatment. Seven participants reported severe treatment-related adverse events while receiving sildenafil plus NTG, including headache, nausea, dizziness, and hypotension, but none led to treatment discontinuation. No serious adverse events were related to sildenafil or placebo alone.
Studies Investigating the PDE5 Inhibitor–Nitrate Interaction.

Abbreviations: BID, twice daily; CAD, coronary artery disease; DM, diabetes mellitus; IV, intravenous; NTG, nitroglycerin; ISMN, isosorbide mononitrate; PDE5, phosphodiesterase type 5; TID, 3 times per day.
a>50% stenosis of ≥1 coronary artery.

Mean changes from baseline in systolic blood pressure (SBP) and diastolic blood pressure (DBP) after glyceryl trinitrate tablet administration. Mean changes from baseline (±standard error [SE]) are shown for (A) SBP and (B) DBP on day 5, after sublingual administration of a 500 µg tablet of glyceryl trinitrate to healthy men who had received sildenafil (25 mg, 3 times per day) or placebo for 4 days and a 25 mg dose of sildenafil or placebo on day 5. During placebo treatment, 8 of 12 men completed the 15-minute glyceryl trinitrate challenge compared with 0 of 12 men during treatment with sildenafil. Adapted and reproduced with permission from Webb et al. 20
A second article by Webb and colleagues published in 2000 investigated the interaction of sildenafil and organic nitrates in 2 studies. 21 Unlike the 1999 study in healthy men, these 2 studies enrolled men with stable angina pectoris and support the notion that there can be a synergistic decrease in BP when the PDE5 inhibitor sildenafil is given with an organic nitrate. The first study examined men with stable angina treated with isosorbide mononitrate. Patients received isosorbide mononitrate twice daily starting 5 to 7 days before the administration of a single sildenafil or placebo dose. On the morning of the first study visit, patients received sildenafil or placebo simultaneously with isosorbide mononitrate. Patients then continued receiving isosorbide mononitrate for a minimum of 7 days after the first dose of study drug before being switched to the alternate study drug (sildenafil or placebo) simultaneously with their isosorbide mononitrate during the second study visit. The sitting mean maximum change from baseline in SBP/DBP (Figure 3) and standing mean maximal change from baseline in SBP/DBP (Figure 4) were significantly lower in patients receiving placebo plus isosorbide mononitrate compared with those receiving sildenafil plus isosorbide mononitrate. Over the 6-hour recording period, the reductions in SBP and DBP in both the sitting and standing positions were greater when isosorbide mononitrate was administered with sildenafil compared with placebo. The decreases in BP were most prominent at approximately 1 to 3 hours after administration of the drugs but were maintained at 6 hours. The modest mean maximum increases in heart rate observed over the 6-hour period in the isosorbide mononitrate plus sildenafil group (sitting: 15.8 beats/minute; standing: 18.5 beats/minute) versus the isosorbide mononitrate plus placebo group (sitting: 14.0 beats/minute; standing: 17.1 beats/minute) were not statistically significant (both P ≥ .35). The combination of isosorbide and sildenafil resulted in some reports of mild to moderate dizziness, sweating, and postural hypotension.
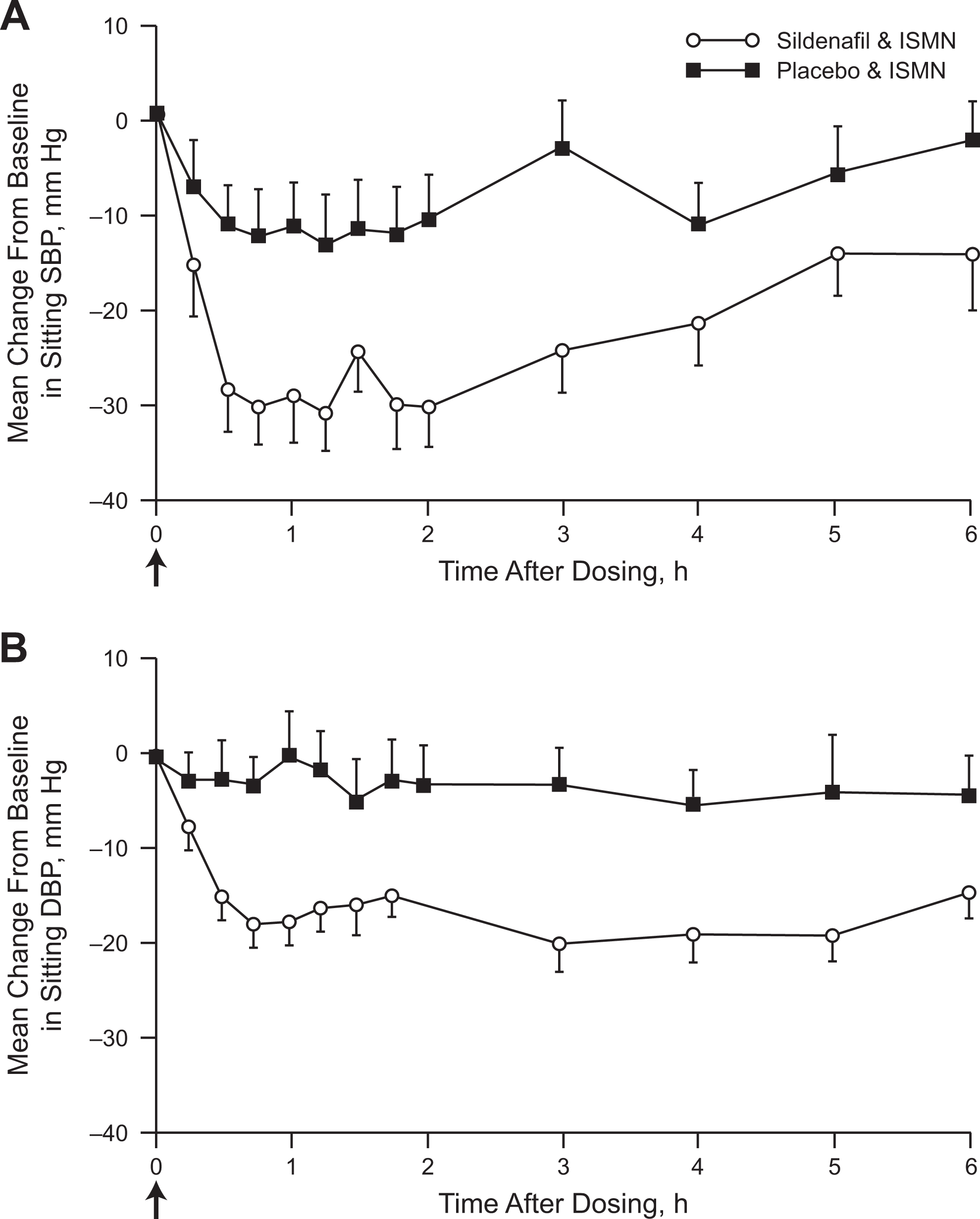
Mean changes from baseline in sitting systolic and diastolic blood pressure after isosorbide mononitrate (ISMN) administration. Mean changes from baseline are shown for sitting systolic (A) and diastolic (B) blood pressure after ISMN administration in men with a history of stable angina. Sildenafil plus ISMN caused significant decreases in systolic and diastolic blood pressure compared with placebo plus ISMN (mean maximum decreases −41/−26 mm Hg versus −22/−13 mm Hg, respectively; P < .01 vs placebo). Oral ISMN (20 mg) and oral sildenafil (50 mg) or placebo were administered at time 0, as indicated by the arrows. Baseline (time 0) is the average of the measurements taken immediately before sildenafil or placebo administration. Values represent mean ± standard error of the mean (SEM); n = 16. Adapted and reproduced with permission from Webb et al. 21
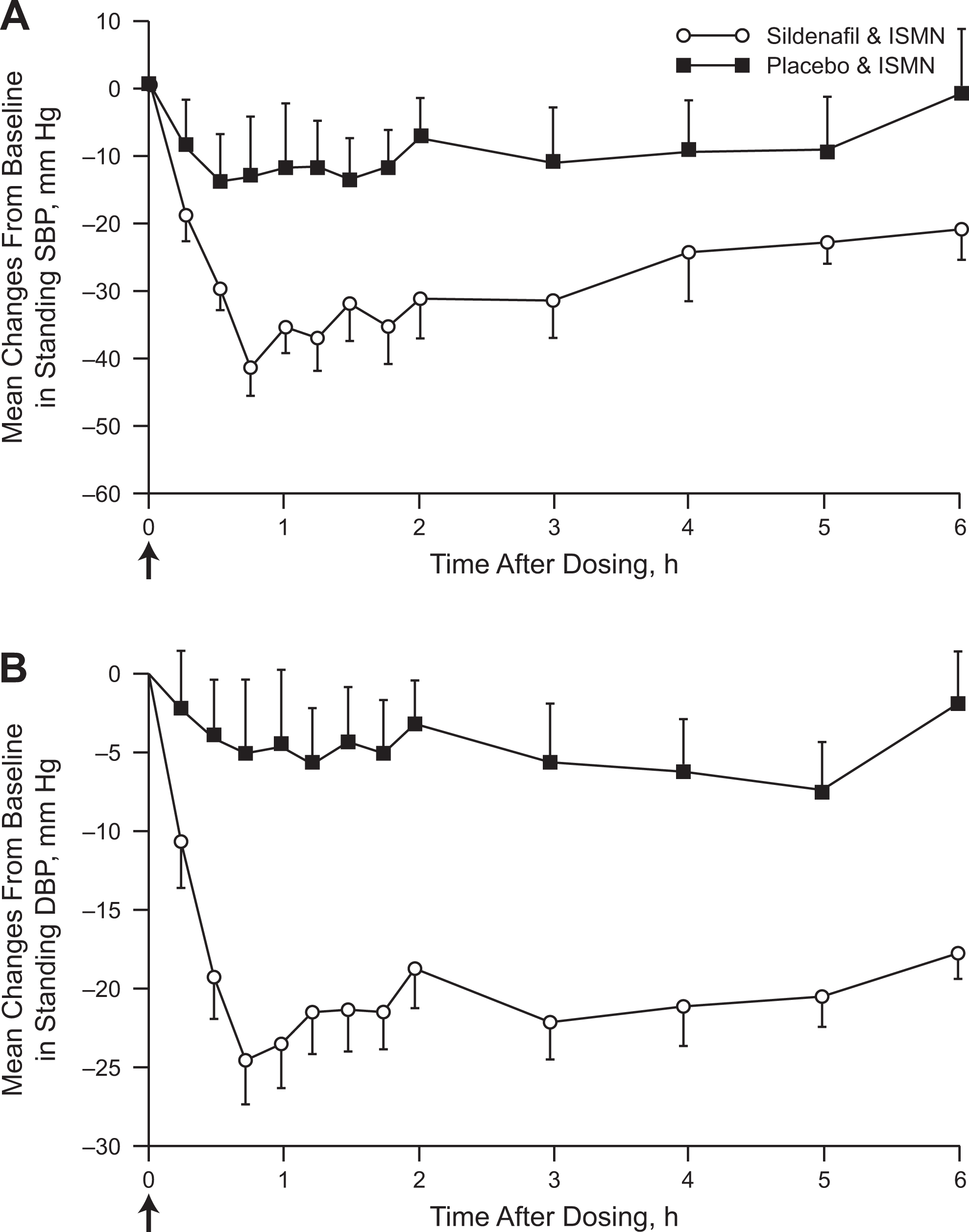
Mean changes from baseline in standing systolic and diastolic blood pressure after isosorbide mononitrate (ISMN) administration. Mean changes from baseline are shown for standing systolic (A) and diastolic (B) blood pressure after ISMN administration in men with a history of stable angina. Sildenafil plus ISMN caused significant decreases in systolic and diastolic blood pressure compared with placebo plus ISMN (mean maximum decreases −52/−29 vs −25/−15 mm Hg, respectively; P < .001 vs placebo). Oral ISMN (20 mg) and oral sildenafil (50 mg) or placebo were administered at time 0, as indicated by the arrows. Baseline (time 0) is the average of the measurements taken immediately before sildenafil or placebo administration. Values represent mean ± standard error of the mean (SEM); n=16. Adapted and reproduced with permission from Webb et al. 21
In a second study reported in the same 2000 article, Webb and colleagues studied men with stable angina using a randomized, double-blind, placebo-controlled, crossover protocol. 21 On the day of the clinic visit, patients were given sildenafil or placebo, followed 1 hour later by sublingual NTG. At a minimum of 7 days later, patients returned and received the alternate study drug (sildenafil or placebo) followed by the nitrate. Heart rate and BP were measured in a sitting position before sildenafil or placebo (baseline) and at various intervals up to 6 hours after the study drug. The mean maximal change from baseline in sitting SBP/DBP was −26/−11 mm Hg in patients who received placebo plus sublingual NTG and −36/−21 mm Hg in patients receiving sildenafil plus sublingual NTG (P < .01 vs placebo). The greatest BP differences between the 2 treatment groups occurred at approximately 1 hour after NTG administration, and BP differences persisted for up to 6 hours. There also were transient increases in heart rate following NTG plus sildenafil compared with NTG plus placebo. Overall, the mean maximum increase in heart rate with NTG plus sildenafil (15.8 beats/min) versus NTG plus placebo (10.2 beats/min) was statistically significant (P < .01). Headache (1 patient) and dizziness (2 patients) were reported as treatment-related adverse events during sildenafil treatment. No serious adverse events were reported in either group. One patient in the sildenafil group was withdrawn owing to feeling unwell and reporting a mild headache after NTG administration; the patient experienced transient hypotension (SBP/DBP 6 and 12 minutes after NTG: 86/47 and 70/39 mm Hg, respectively).
The authors of these studies made several important points. The change in SBP/DBP with isosorbide mononitrate alone or NTG alone (decreases of 22-26/11-15 mm Hg) is greater than the BP-lowering effect of sildenafil alone (decrease of 10/7 mm Hg). 7,21,26 The additional drop in SBP/DBP observed when sildenafil was added to isosorbide mononitrate, especially in the standing position (mean maximum decrease: 52/29 mm Hg), is at least an additive effect and may involve a synergistic interaction. Presumably by attenuating the cGMP breakdown, sildenafil amplifies NO-related activity provided by exogenously administered isosorbide mononitrate or NTG. The authors concluded that sildenafil should not be given to patients taking organic nitrates. Similar hemodynamic interactions have also been demonstrated with PDE5 inhibitors other than sildenafil. 4,23,24
What Should a Patient With Cardiovascular Disease Do If He Develops Angina After Using a PDE5 Inhibitor?
A patient with cardiovascular disease and taking a PDE5 inhibitor who develops angina during physical activity must discontinue the activity immediately and relax for 5 to 10 minutes. 27 He should sit on the edge of a bed/chair (to reduce risk of postural hypertension) and then stand up to help reduce the preload, which may relieve angina symptoms. If the angina pain does not resolve after 20 to 30 minutes, or if other concerning symptoms such as lightheadedness, profuse sweating, weakness, or palpitations occur, emergency services should be sought and the physical activity should not be attempted again before the patient has undergone a thorough cardiovascular reassessment. 27
When Is It Safe to Restart Nitrate Therapy in a Patient Who Has Taken a PDE5 Inhibitor?
There are 2 main groups of drugs in the PDE5 inhibitor category: short-acting and long-acting compounds. At the maximum dose (100 mg), sildenafil has a time of onset of approximately 30 minutes, a tmax of 30 to 120 minutes, and a half-life of approximately 4 hours. 3 Vardenafil and avanafil have similar pharmacokinetic profiles. 4,5 In contrast, tadalafil is a long-acting compound with an onset of action of 30 minutes, a tmax of approximately 2 hours, and a half-life of 17.5 hours. 6 In an early consensus paper, it was suggested that nitrates not be restarted until approximately 6 half-lives after the last PDE5 inhibitor dose. 28 Based on a half-life of approximately 4 hours for sildenafil, the initial recommendation was that nitrates not be given until at least 24 hours after the last sildenafil dose. Vardenafil and tadalafil were not yet approved or available on the market when these initial guidelines were released.
An article by Oliver and colleagues suggested that it might be safe to administer nitrates earlier than 24 hours after the last dose of sildenafil, 22 but this article did not prompt a review of existing guidelines. These investigators reported the results of 2 randomized, double-blind, placebo-controlled, crossover studies designed to determine the time course of the interaction between sildenafil and sublingual NTG in healthy male volunteers and in men with angina (Table 1). The first study enrolled 33 healthy men who received either sildenafil or placebo and then received sublingual NTG at 1, 4, 6, and 8 hours and at other time points up to 48 hours after the sildenafil or placebo dose. There was a statistically significant greater reduction in sitting mean maximum SBP/DBP with sildenafil plus NTG when the nitrate was given 1 hour after sildenafil versus placebo, but not at any other time point (Figure 5A). In the second study of 20 men with angina, the nitroglycerin challenge was given at 1, 4, 6, and 8 hours after a dose of sildenafil or placebo. In this study, there was a significantly greater reduction in the absolute mean BP with sildenafil plus NTG than with placebo plus NTG that lasted at least 8 hours after the sildenafil 100 mg dose (Figure 5B). The mean maximum difference (sildenafil minus placebo) in sitting BP was −16 mm Hg at 1 hour, −12 mm Hg at 4 hours, −6 mm Hg at 6 hours, and −9 mm Hg at 8 hours. The mean maximum differences in sitting DBP were −9, −9, −5, and −7 mm Hg, respectively. The authors noted that at 6 and 8 hours the interaction between sildenafil and the nitrate was additive and not synergistic. However, a limitation of this study is that in men with angina, the investigators did not follow the interaction between sildenafil and the nitrate until there was no longer any interaction demonstrated, so it is not clear when the interaction ended. At the 1-hour time point, there was a statistically significant increase in heart rate when sildenafil was combined with the nitrate (4.7 beats/minute) compared with placebo plus the nitrate (0.5 beats/minute; P < .01), but this interaction was not significant at later time points. Headache, dizziness, and flushing were the most common adverse events reported in healthy volunteers. There were no serious adverse events and no patient withdrew from the study owing to adverse events. Adverse events most commonly occurred with NTG administration occurring 1 hour after sildenafil dosing. In those with angina, symptomatic hypotension occurred in 6 patients when the nitrate was given 1 hour after sildenafil and in 3 patients when the nitrate was given 4 hours after sildenafil. Symptomatic hypotension was not reported when the nitrate was given 6 or 8 hours after sildenafil. These data imply that not all patients who have taken sildenafil must wait 24 hours before receiving nitrates; however, they do not absolutely prove the safety of giving a nitrate earlier because the sample size was small and additive drug interactions in men taking concomitant vasoactive medications were not considered.

Mean changes in sitting blood pressure in sildenafil- and NTG-treated participants. A, Mean changes in sitting blood pressure of NTG with sildenafil and with placebo in angina patients. Baseline values are those taken before sildenafil or placebo administration. For each time point, baseline values are followed by values measured every 3 minutes after NTG (preNTG values are not shown). B, Mean changes in sitting blood pressure (BP) with NTG given after sildenafil and placebo in angina patients. Changes are from prenitroglycerin recordings. Adapted and reproduced with permission from Oliver et al. 22
A study by Swearingen and colleagues that was published in 2013 reported the results of a randomized, double-blind, 3-way crossover study of the hemodynamic interaction between avanafil and NTG sublingual tablet compared with that with sildenafil and placebo in healthy men. 23 Each participant was treated on separate days with a single dose of avanafil, sildenafil, or placebo, which was followed by a single nitro glycerin tablet at 12, 8, 4, 1, or 0.5 hours (5 subgroups) after study drug. A hemodynamic interaction between both PDE5 inhibitors and NTG was observed up to the 4-hour interval, but no significant differences versus placebo in BP or HR were observed after 8 hours with avanafil and after 12 hours with sildenafil. The most common treatment-related adverse events after NTG administration were headache, dizziness, and nausea, with most cases of headache and dizziness reported in the ≤4-hour subgroups. No serious adverse events were observed in any subgroup.
Sildenafil, vardenafil, and avanafil have half-lives of approximately 3 to 5 hours, whereas tadalafil has a half-life of 17.5 hours. 3 -6 Therefore, if a patient takes tadalafil, the interaction between tadalafil and organic nitrates would theoretically last considerably longer than with sildenafil or tadalafil. In a study by Kloner and colleagues, the hemodynamic interaction between tadalafil and sublingual NTG lasted 24 hours but was not observed at or beyond 48 hours after tadalafil. 24 Therefore, if a patient is experiencing angina pain after taking tadalafil, nitrates should not be administered until at least 48 hours after the last dose of tadalafil, which is a recommendation included in an American Heart Association consensus document. 29 The study by Kloner and colleagues was a randomized, placebo-controlled, double-blind, crossover study of 134 healthy men and 32 men with diabetes that examined the time course of the interaction between tadalafil and nitrates. 24 Participants received a daily dose of either tadalafil or placebo for 7 days. On the seventh day, participants received repeated sublingual NTG tablets at 2, 4, 8, 24, 48, 72, and 96 hours after the final dose of either tadalafil or placebo. Following a 10- to 21-day washout period, the men received the alternate treatment (tadalafil or placebo) with the nitrate challenge on day 7. Heart rate and BP were monitored at the 2-hour and subsequent time points, with the primary end point being the percentage of men with a minimum standing SBP of <85 mm Hg after each of the NTG challenges. Mean standing BP assessments over the course of 2 hours following administration of sublingual NTG showed that decreases in SBP/DBP in response to NTG were greater with tadalafil versus placebo at 4, 8, and 24 hours, but not at 48, 72, or 96 hours (Figure 6). The heart rate changes related to NTG were generally similar with tadalafil and placebo. Subpopulation analysis showed that tadalafil did not interact with NTG at or beyond 48 hours either in men with diabetes or men aged >60 years. The most common adverse events were headache, myalgia, back pain, dizziness, dyspepsia, and flushing, with most characterized as mild in severity. The incidence of dizziness was similar with tadalafil and placebo, with most episodes occurring during NTG administration. One patient receiving placebo had a serious sinus arrest following NTG administration (24 hours after final placebo dose) that spontaneously resolved; no serious adverse events occurred with tadalafil.

Mean standing systolic and diastolic blood pressure in tadalafil- and placebo-treated participants. Sublingual NTG (0.4 mg) is indicated by the dashed vertical line. The 4- to 96-hour time intervals represent time of NTG administration after the last dose of tadalafil or placebo. In each graph, the upper set of curves represent systolic blood pressure and the lower set of curves represent diastolic blood pressure. Standard deviation for systolic and diastolic blood pressure ranged from 12 to 19 mm Hg and 9 to 13 mm Hg, respectively. A significantly greater percentage of patients experienced a standing systolic blood pressure <85 mm Hg when NTG was given 4, 8, and 24 hours after tadalafil compared with placebo (46% vs 31% [P < .001], 31% vs 21% [P = .008], and 41% vs 32% [P = .028], respectively). The interaction with tadalafil and the nitrate was not observed at 48, 72, or 96 hours after the final dose of tadalafil. Adapted and reproduced with permission from Kloner et al. 24
Overall, the interaction studies mentioned above in which men received sildenafil or tadalafil in combination with an organic nitrate demonstrated BP reductions, heart rate increases, and some hypotensive adverse events. However, all hypotensive events resolved spontaneously with simple supportive measures (ie, no need for intravenous fluids or vasoconstrictors). Several studies were designed to specifically characterize the length and severity of the interaction, usually using a worst-case scenario (eg, timing the drug administration so that peak concentrations of the nitrate and the PDE5 inhibitor occur at a similar time). It should be stressed that certain aspects of the nitrate-PDE5 inhibitor interaction depend on many factors, not all of which have been completely studied. The nature of the interaction may vary from drug to drug, depending on the dose and half-life of the PDE5 inhibitor; when the PDE5 inhibitor was taken; the half-life, formulation, and pharmacodynamic characteristics of the nitrate; and when the nitrate was taken. A review of the literature suggests that if a patient has taken a PDE5 inhibitor, then nitrates should not be started until 24 hours after (possibly earlier, but not definitive) the last dose of sildenafil, vardenafil, or avanafil and not until 48 hours after the last dose of tadalafil to avoid the drug interaction of excessive vasodilation and hypotension.
US Food and Drug Administration Labeling
Phosphodiesterase type 5 inhibitors remain absolutely contraindicated if patients are taking nitrates, according to US Food and Drug Administration requirements for the labeling of all PDE5 inhibitors. This contraindication is true for short-acting nitrates, such as sublingual NTG preparations, as well as for long-acting nitrates, such as isosorbide dinitrate, isosorbide mononitrate, and transdermal NTG preparations. The official wording on the package insert for sildenafil is “CONTRAINDICATIONS: Administration of VIAGRA to patients using nitric oxide donors, such as organic nitrates or organic nitrites in any form. VIAGRA was shown to potentiate the hypotensive effect of nitrates.” 3 Similar wording is included in the package inserts for vardenafil and avanafil. 4,5
Are There Exceptions?
Parker and colleagues published an article in 2007 suggesting there might be exceptions to the hard and fast rule of not combining PDE5 inhibitors with nitrates. 25 In this study, the authors make the point that despite the contraindication, there are situations when a patient who has ingested a PDE5 inhibitor might need intravenous NTG, for example, a patient developing severe angina pain following sexual activity. Parker and colleagues explored whether the contraindication was absolute or whether certain dosing regimens might be safe for some patients. In a randomized, double-blind, placebo-controlled, crossover study, 34 men who had a history of angina and known coronary artery disease received sildenafil or placebo followed by ascending doses of intravenous NTG. Both BP and heart rate were monitored. After a 3- to 7-day washout period, patients were crossed over to the alternate therapy (sildenafil or placebo) and rechallenged with the NTG infusion. The primary end point was the maximum tolerated NTG infusion rate (rate immediately prior to that associated with discontinuation of the infusion). Infusion of NTG was stopped based on a decrease in SBP either of >35 mm Hg or to an absolute pressure of <100 mm Hg sustained for 1 minute, an increase in heart rate of >25 beats/minute or to >100 beats/minute sustained for 1 minute, or outright symptomatic hypotension. Following administration of sildenafil but before NTG, the mean change in SBP/DBP was −12/−8 mm Hg versus −5/−4 mm Hg with placebo, with a small increase in heart rate with sildenafil (2 beats/minute). During infusion of NTG, sildenafil was associated with a 4- to 6-mm Hg decrease in SBP compared with placebo. With infusion of NTG, a decrease in BP to prespecified limits occurred in 43 instances, most of which were asymptomatic: 26 during the sildenafil period and 17 during the placebo period. Recovery of BP took <10 minutes in 25 instances and >1 hour in 4; there were no cases of syncope. The median maximum tolerated dose of NTG was 80 μg/min during the sildenafil period versus 160 μg/min during the placebo period. During sildenafil administration, 59% of men completed the NTG 80 μg/min infusion and 25% completed the 160 μg/min infusion compared with rates of 88% and 59%, respectively, with placebo. Treatment-related adverse events were mainly mild to moderate, including hypotension (symptomatic or BP below prespecified limits), headache, and dizziness, which were often associated with NTG alone. The authors concluded that if other therapies to manage acute myocardial ischemia are not available, “it may be possible to safely administer intravenous NTG to men with coronary artery disease if a careful titration regimen is used.”
A few studies suggest that the combination of PDE5 inhibitors plus nitrates may have a therapeutic role in patients with treatment-resistant systemic hypertension 30 and in patients with pulmonary artery hypertension secondary to heart failure. 31 Both of these reports are based on a small number of patients, so larger studies will be needed to determine whether there is a therapeutic role for the combination of PDE5 inhibitors and nitrates in certain niche areas. There are also other investigations being carried out to assess the utility of combining PDE5 inhibitors with inhaled NO to gain selective action on the pulmonary circulation in pediatric as well as in adult patients with pulmonary hypertension. 32,33
Pharmacologic Characteristics of Nitrates
Nitrates and PDE5 inhibitors are not a homogeneous group of drugs. In this section, we examine the nitrate compounds that are available, their pharmacokinetic profiles, and how they are used. Angina pectoris is a manifestation of an imbalance between the supply of oxygen to the heart and the demand for oxygen by the heart. Nitrates help restore the imbalance between oxygen supply and demand by acting on vascular smooth muscle to produce arterial and venous vasodilation. Their effect on capacitance veins reduces preload, cardiac dimensions leading to reductions in wall stress, and myocardial oxygen demand. To a lesser extent, vasodilation of systemic conduit arteries reduces afterload, which also lowers oxygen demand. In addition, nitrates dilate epicardial coronary arteries and may, in the setting of stenotic coronary disease, improve coronary blood flow, hence improving oxygen supply. The organic nitrates have been a mainstay in the treatment of angina for more than 100 years. When NTG is used acutely via either sublingual or intravenous formulation, it is highly effective in relieving episodes of angina. For patients with stable angina, long-acting oral or transdermal preparations can be used for the prevention of angina symptoms, improving exercise capacity. Of note, although nitrates are useful for treating the symptoms of angina, they have never been shown to reduce major adverse cardiovascular events, such as death or myocardial infarction, in patients with stable coronary artery disease. 34 One significant disadvantage of nitrates is the development of tolerance. Tolerance to the hemodynamic and symptomatic benefits of the organic nitrates develops within 24 hours if they are administered in a manner that leads to continuous nitrate concentrations throughout the day. Nitrate tolerance can be prevented if a low or nitrate-free interval is provided. An article by Hashimoto and Kobayashi published in 2003 provides a thorough review of the results of studies of the pharmacokinetics and pharmacodynamics of NTG during the previous 20 years, together with the association between these results and the development of nitrate tolerance. 35
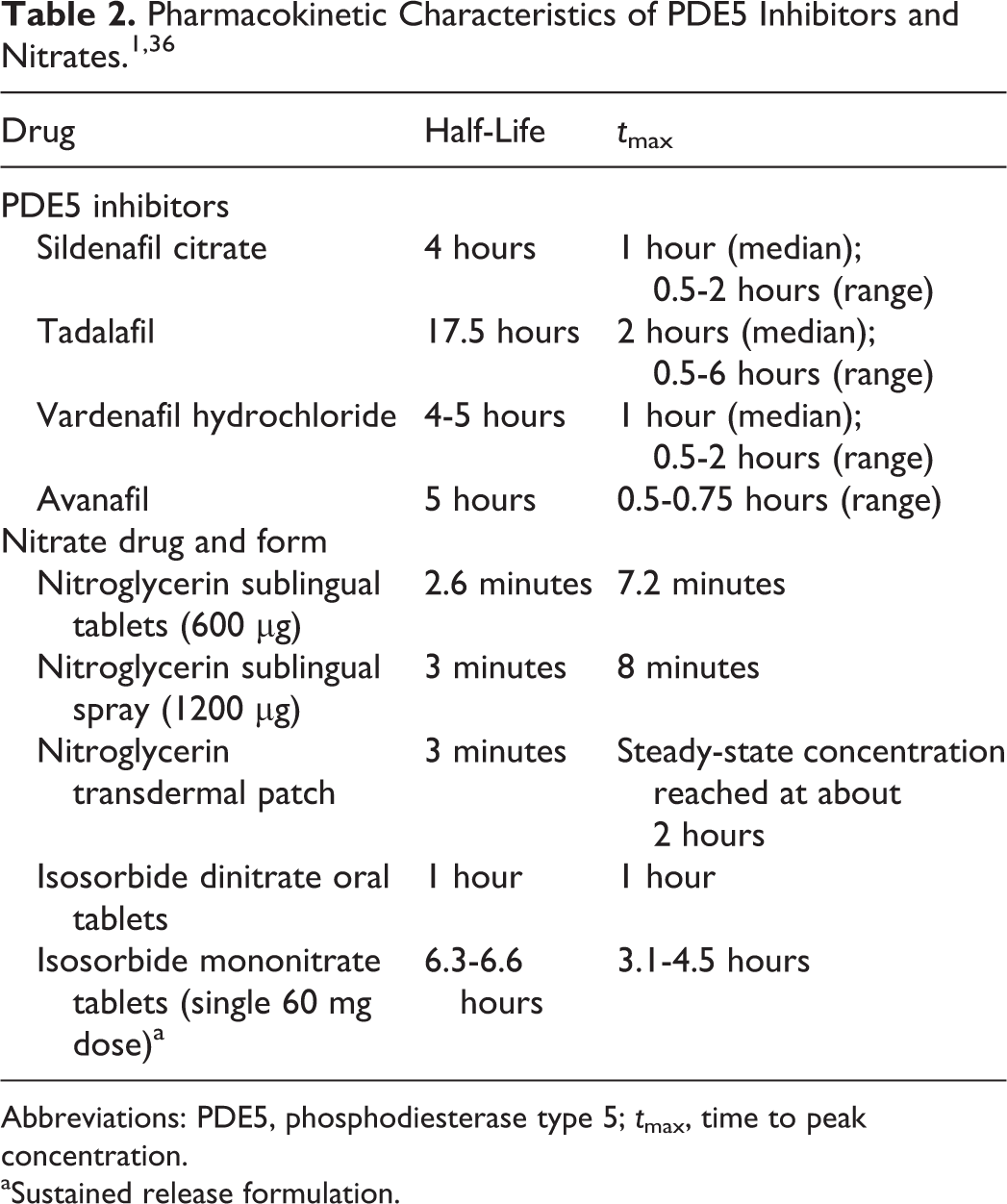
The pharmacokinetic characteristics for various formulations of nitrate preparations used in the treatment of angina and the PDE5 inhibitors are shown in Table 2. 1,36 Sublingual NTG has a rapid onset of action and can relieve symptoms of angina within 1 to 3 minutes of administration. Nitroglycerin has a short half-life (about 3 minutes) and is rapidly eliminated from the circulation. However, NTG does have 2 active dinitrate metabolites that have longer half-lives; therefore, the hemodynamic effects of NTG are longer than the parent compound itself. Isosorbide dinitrate, the second class of commonly used organic nitrate preparations, is available as an oral tablet and as a sublingual tablet. Oral isosorbide dinitrate has a longer half-life and time to peak concentration (tmax) than NTG (approximately 60 minutes). It has a major metabolite (isosorbide-5-mononitrate) with a half-life of approximately 5 hours. Isosorbide mononitrate is supplied in a variety of extended-release formulations, with a tmax ranging from 1 to 4 hours and a half-life in excess of 5 hours. It has no active major metabolites and is widely used in patients with angina and compromised coronary artery circulation.
Abbreviations: PDE5, phosphodiesterase type 5; tmax, time to peak concentration.
aSustained release formulation.
Of note, differences in cardiovascular risk of PDE5 inhibitors in combination with short- versus long-acting nitrates remain unknown. Logically, men treated only occasionally with a short-acting nitrate may be at less cardiovascular risk compared with men who are treated with long-acting nitrates. This supposition is purely speculative since there is no published information regarding this topic.
Are Nitrates Always Necessary?
Many users of short-acting nitrates, which have been prescribed for the symptomatic relief of angina, are waiting for or have received a surgical intervention or percutaneous coronary artery intervention to ameliorate their symptoms. These patients often are prescribed a number of other drugs to help manage their condition and may carry the nitrate spray or sublingual tablet as a “just in case” rescue therapy if their symptoms occur. It is likely that not all patients who are carrying short-acting NTG, especially after they have had successful revascularization, will need to stay on the drug. However, some physicians are hesitant to discontinue sublingual NTG therapy for fear that the patient will be unprotected if angina reappears. Given the range of pharmacological agents currently available to manage angina, each patient should be regularly reviewed with respect to disease progression. Consequently, in a patient with stable coronary artery disease, not experiencing angina, there is no strong rationale and no prognostic benefit for that patient to continue to carry sublingual NTG and there should be consideration of terminating the NTG prescription. 8 While the package inserts for the PDE5 inhibitors make it clear that the use of NTG is a contraindication for prescribing PDE5 inhibitors, if a health-care provider makes the off-label clinical decision to give a patient a PDE5 inhibitor who is taking nitrates, then at the very least the health-care provider should thoroughly educate the patient about the nitrate–PDE5 inhibitor interaction and tell the patient not to use sublingual NTG or other NTG preparations if they develop chest pain after taking a PDE5 inhibitor.
What about patients with stable coronary artery disease who are prescribed long-acting organic nitrates? In these patients, organic nitrates are taken daily, creating a contraindication to PDE5 inhibitor therapy. Many of these patients can safely discontinue their long-acting nitrates and alternative pharmacologic antianginal therapy can be considered, as demonstrated by George et al in a study of hemodynamically stable patients free of angina. 37 Other studies have suggested the safety of stopping nitrate therapy in some patients wishing to be placed on a PDE5 inhibitor for the treatment of ED. 8,38 Furthermore, there are new antianginal agents such as ranolazine, trimetazidine, and ivabradine that might be used instead of long-term nitrate therapy. 39 -41
Unanswered Questions About the Nitrate–PED5 Inhibitor Interaction
Several unanswered questions remain about the nitrate–PDE5 inhibitor interaction, such as (1) timing of restarting PDE5 inhibitors after nitrates and (2) the effect of different PDE5 inhibitor doses on the interaction. The question of how long to wait before safely taking a PDE5 inhibitor after use of a nitrate drug has not been adequately studied in the literature, so one can only speculate on the correct answer. Although nitroglycerin has a short half-life (approximately 3 minutes), its dinitrate metabolites are vasoactive and have longer half-lives (approximately 40 minutes). As such, it is not clear how long an interaction between short-acting NTG and PDE5 inhibition might be present. Similarly, long-acting nitrates such as isosorbide dinitrate and isosorbide-5-mononitrate have half-lives of several hours, and the time of potential interaction with a PDE5 inhibitor is not clear and has never been studied. Therefore, at the moment there is no clear guidance to suggest when a PDE5 inhibitor may be safely given following administration of any organic nitrate. 42
Another question is the effect on the nitrate–PDE5 inhibitor interaction when higher-than-recommended doses of PDE5 inhibitors are used. A published report found that sildenafil at up to twice the maximum recommended daily dose was effective in approximately one quarter of patients who did not respond to the maximum recommended dose, 43 indicating high doses may be considered for some patients. Conversely, clinical studies of tadalafil for the treatment of urinary symptoms associated with benign prostatic hyperplasia commonly evaluated daily doses of 2.5 to 5 mg, 44 which is well below the dose used for ED. It is possible that higher or lower doses may affect the magnitude of the nitrate interaction. Further research is needed to evaluate these questions.
Summary
The use of organic nitrates remains a contraindication to the use of PDE5 inhibitors. Studies indicate that in some men the combination of PDE5 inhibitors plus nitrates leads to symptomatic hypotension. The nature of the nitrate–PDE5 inhibitor interaction is dependent on many variables, with some studies indicating that nitrates may be taken 24 hours (possibly earlier) after the last dose of short-acting PDE5 inhibitors and 48 hours after the last dose of long-acting PDE5 inhibitors. Patients receiving nitrates who wish to use PDE5 inhibitors should be educated regarding the interaction and evaluated to determine whether nitrate treatment can be discontinued. Further research is needed to determine the timing of when PDE5 inhibitors can be restarted after a patient has taken a nitrate and the effect of high or low PDE5 inhibitor doses on the interaction effect.
Dedication
Dr Graham Jackson (1947-2016)
Graham Jackson left us far too early, in April 2016, after a long debilitating illness. I was fortunate to know him as a good friend from my medical school days at Kings, London, in the ’70s. He continued to write and review papers until the day of his death. Graham worked as consultant cardiologist for nearly 40 years at Kings, Guy’s, and London Bridge hospitals. He was the first to describe the strong predictive links between erectile dysfunction and subsequent cardiovascular events, especially in younger men. He was the first to set up a cardiac sexual dysfunction clinic and conducted many of the early sildenafil trials. He was a founding member of the Princeton Consensus Panels, which comprehensively summarized the links between cardiovascular disease, erectile dysfunction (ED), and low testosterone. 45,46
Graham realized that cardiologists rarely asked men about their erectile function, resulting in excessive prescribing of nitrates, which deprived them of access to phosphodiesterase type 5 (PDE5) inhibitors. He conducted the first study to show that 80% of patients could successfully stop nitrates to facilitate oral ED therapy.
Graham was editor of the International Journal of Clinical Practice for many years and entertained us all with his lectures and inspirational editorials. He taught us that ED (erectile dysfunction) = ED (endothelial dysfunction), the “way to a man’s heart is through his penis,” and ED diagnosis offered a “window of opportunity for cardiac prevention.” In 2005, a meeting had been arranged to discuss ED guidelines with the National Institute for Health and Care Excellence when a letter was received requesting him not to attend as he knew “too much.” From that date, Graham was known to his friends as “the man who knew too much”! 45
Dr Geoffrey Hackett
Footnotes
Authors’ Note
Guest editor on this manuscript was Dr Harin Padma-Nathan.
Authors' Contributions
R. A. Kloner, P. Goggin, I. Goldstein, G. Hackett, M. G. Kirby, I. Osterloh, J. D. Parker, and R. Sadovsky contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R. Kloner is a consultant for Pfizer, Bayer, and Sanofi. P. Goggin is an employee of Pfizer Ltd, Sandwich, UK. I. Goldstein is a consultant for Dornier, has served as a speaker for Coloplast and Dornier, has served on advisory boards for Aytu, Bayer, Lipocine, and Pfizer, and has received research grants from Auxilium, Dornier, Ixchelsis, Lipocine, and Tissue Genesis. G. Hackett is an occasional speaker for Bayer and Besins and advisory board member for Pfizer. M.G. Kirby has received funding for research, conference attendance, lecturing and advice from Astellas, Pfizer, Takeda, Bayer, Merck Sharp & Dohme, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, AstraZeneca, and Menarini, is an editor for the Primary Care Cardiovascular Journal, and has served on several NHS advisory boards, including the Prostate Cancer Risk Management Programme and the Prostate Cancer Advisory Group. I. Osterloh is a former employee of Pfizer, serves as a consultant for Pfizer, and owns shares of Pfizer stock. J.D. Parker has consulting relationships with Bayer, Servier, Novartis and Theracos. R. Sadovsky has served as a consultant for Pfizer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was sponsored by Pfizer Limited. Editorial assistance was provided by Patricia B. Leinen, PhD, of Complete Healthcare Communications, LLC (West Chester, Pennsylvania), a CHC Group company, and funded by Pfizer Limited.