Abstract
Objectives:
We aimed to evaluate the association of testosterone deficiency with inflammation and how long-term testosterone therapy affects inflammation biomarkers over time.
Methods:
We conducted a 2-component study. First, we conducted a cross-sectional study using the recently released 2015-2016 National Health and Nutrition Examination Survey (NHANES) data to examine the association between testosterone deficiency and inflammation biomarkers including high sensitivity C-reactive protein (hsCRP), liver enzymes alanine aminotransferase (ALT) and aspartate aminotransferase (AST) in the US general population. Then we conducted a longitudinal study to investigate the longitudinal effect of testosterone therapy on inflammation biomarkers and the risk of cardiovascular events, using data from 776 hypogonadal men based on a registry study in Germany with up to 11 years’ follow-up.
Results:
The adjusted odds ratios (ORs) describing the associations between testosterone deficiency and hsCRP ≥ 3mg/L, ALT > 40U/L, and AST > 40U/L were 1.81 (P-value < 0.001), 1.46 (P-value = 0.009), and 0.99 (P-value = 0.971), respectively. In the control group, CRP, ALT, and AST levels increased by 0.003 (95%CI: −0.001, 0.007) mg/L, 0.157 U/L (95%CI: 0.145, 0.170), and 0.147 (95%CI: 0.136, 0.159) U/L per month, while in the treatment group, CRP, ALT, and AST levels decreased by 0.05 (95%CI: −0.055, −0.046) mg/L, 0.142 U/L (95%CI: −0.154, −0.130), and 0.148 (95%CI: −0.158, −0.137) U/L per month.
Conclusion:
Testosterone deficiency was associated with an increased level of inflammation; long-term testosterone therapy alleviated inflammation among hypogonadal men, which may contribute to the reduced cardiovascular risk. Future large trials are warranted to confirm our observational study findings.
Introduction
Testosterone is the principal male sex hormone and an important metabolic hormone for maintaining the overall physiological function, including carbohydrate, protein, and lipid metabolism in men. 1 A deficiency of testosterone secretion from the Leydig cells is called male hypogonadism, which can be classified as primary hypogonadism (testicular failure due to radiation therapy, trauma, torsion-induced ischemia, or infection; loss of testes; genetic constellations) and secondary hypogonadism (central suppression of the hypothalamus and pituitary). 2
Previous studies have suggested that testosterone deficiency in hypogonadal men may have an influence on atherosclerosis, a chronic inflammatory process and a major pathway of cardiovascular disease through which many risk factors affect cardiovascular health. 3 Animal studies have demonstrated that testosterone deficiency was associated with elevated blood-brain-barrier permeability accompanied by up-regulation of inflammatory molecules, 4 which may trigger arterial inflammation that causes arterial hyperplasia even when the traditional risk factors were absent. 5 In an experiment investigating the mechanism through which testosterone deficiency induced endothelial dysfunction in rats, the authors found a higher level of interleukin-6 (IL-6), nuclear factor kappa B (NF-κB), nicotinamide adenine dinucleotide phosphate (NADPH) oxidase-1, and NADPH oxidase-4 in castrated rats compared to castrated rats supplemented with testosterone and sham rats (P-value < 0.01), indicating elevated oxidative stress and inflammation in the rats undergoing testosterone deficiency and potential beneficial effects of testosterone therapy in attenuating this result. 6 However, to our knowledge, studies using large nationally representative samples of human subjects to evaluate the association between testosterone deficiency and inflammation are scarce.
Although low testosterone level has been associated with atherosclerosis and various cardiovascular risk factors, 2,7 controversies exist regarding the role of exogenous testosterone therapy, a treatment to improve serum testosterone levels, in preventing cardiovascular risk. A recent study reviewed 7 systematic reviews that included a total of 94 randomized controlled trials and investigated the association between exogenous testosterone and the risk of cardiovascular events. 8 Six of the reviewed studies showed no association and one showed an increased risk of cardiovascular disease associated with exogenous testosterone. In fact, no trials of testosterone therapy published to date were designed or adequately powered to assess treatment effects on cardiovascular events. A large randomized clinical trial (a planned sample size of 6,000 men, with 5-year treatment duration) that is currently recruiting will be powered to evaluate the effect of testosterone therapy on the incidence of major adverse cardiovascular events and efficacy measures in hypogonadal men (TRAVERSE trial, clinicaltrials.gov identifier NCT03518034). However, it still takes time to collect essential data before any analysis. Until then, well-designed observational studies with long follow-up periods are needed to provide real-world evidence in determining the potential beneficial effect of testosterone therapy on cardiovascular disease. Besides, whether testosterone therapy affects the cardiovascular risk by alleviating inflammation, the major pathway of cardiovascular disease, is also worth exploring.
Recent evidence has confirmed that multiple inflammatory biomarkers were elevated years before the first cardiovascular event and were highly predictive of future cardiovascular risk in otherwise healthy populations. 3 Of these biomarkers, C-reactive protein (CRP), known as a systemic inflammatory biomarker, has become a standard predictor for cardiovascular risk due to its ease of measurement and abundance of clinical data. CRP is one of the best measures of the acute-phase response to inflammation, allowing for the assessment of the effect of testosterone deficiency and testosterone therapy on inflammation. Additionally, liver enzymes have recently been demonstrated as emerging risk factors for cardiovascular diseases. By using F-fluorodeoxyglucose positron emission tomography, researchers have suggested that abnormally elevated liver enzyme levels were associated with hepatic and vascular inflammation that reflects rupture-prone vulnerable atherosclerotic plaques 9 and impaired coronary flow reserve, 10 making liver enzymes, which are low-cost, sensitive, and routinely measured for patients in clinical practice, another potential marker to help assess the effect of testosterone depletion and testosterone therapy on inflammation.
In this study, we conducted a cross-sectional study using the recently released 2015-2016 National Health and Nutrition Examination Survey (NHANES) data to examine the association between testosterone deficiency and inflammation biomarkers including high sensitivity CRP (hsCRP), liver enzymes alanine aminotransferase (ALT) and aspartate aminotransferase (AST) in the US general population. We also conducted a longitudinal study to investigate the effect of long-term testosterone therapy on inflammation biomarkers and the risk of cardiovascular events using data based on a registry study in Germany. We hypothesized (1) testosterone deficiency was associated with elevated inflammation biomarkers; (2) long-term testosterone therapy decreased levels of inflammation biomarkers and the risk of cardiovascular events.
Methods and Materials
A Cross-Sectional Study Using NHANES Data
Study population
To examine the cross-sectional association between testosterone deficiency and inflammation biomarkers, we used data from the NHANES 2015-2016 cycle. NHANES is a national survey that is conducted every 2 years to assess the population’s health and nutrition status in the US through interviews, laboratory tests, and physical examinations. The sampling method for this nationwide survey is based on a stratified multistage probability design. The samples are weighted to represent the total US civilian, non-institutionalized population. 11 The NHANES 2015-2016 was approved by the Centers for Disease Control and Prevention (CDC) Institutional Review Board. Written informed consent was obtained from participants. This study was limited to male adults only because the relationship between testosterone and inflammation biomarkers may be different during puberty when hormone levels are not stable. 12 Participants who reported hepatitis (n = 103) or liver cancer (n = 2) in their medical history, or had extreme levels of testosterone, inflammation biomarkers, and total cholesterol levels (testosterone > 50 nmol/L, n = 12; hsCRP > 100 mg/L, n = 292; ALT > 200 U/L, n = 6; AST > 200 U/L, n = 2; total cholesterol > 400 mg/dL, n = 3) were excluded for the concern about the underlying disease condition or treatment status that may modify the associations of interest. A total of 2,466 male adults were included in the study.
Serum testosterone, hsCRP, ALT, and AST levels
Data on serum testosterone, hsCRP, ALT and AST levels were obtained from the NHANES Laboratory File. Details on the laboratory data collection and analysis were described elsewhere. 13 In brief, serum specimens were collected, stored, and shipped to the Collaborative Laboratory Services for analysis. Testosterone was performed via Isotope Dilution Liquid Chromatography Tandem Mass Spectrometry (ID-GC/MS) method. The lower detection limit was 0.026 nmol/L. 14 hsCRP was measured on the Beckman Coulter UniCel DxC 600 Synchron and the Beckman Coulter UniCel 660i Synchron Access chemistry analyzers. The lower detection limit for hsCRP was 0.11 mg/L. 15 The Beckman Coulter UniCel DxC 800 Synchron was used to measure ALT and AST. The DxC800 used a kinetic rate method to measure ALT activity and an enzymatic rate method to measure the AST activity in serum. The lower detection limit for both ALT and AST was 5 U/L. 16,17 In the original laboratory data files, entries below the lower detection limit were replaced by the value of the lower detection limit divided by the squared root of 2. In our study population, the testosterone, ALT and AST levels were all above the lower detection limit, and 150 participants had hsCRP below the lower detection limit.
Covariates
Testosterone deficiency as the main variable of interest was defined as serum testosterone level ≤12.1 nmol/L. The threshold of 12.1 nmol/L was selected based on clinical experience and confirmed by Bhasin et al. 18 Age, race, comorbidity condition, and smoking and drinking status that are related to the inflammation biomarkers were included in the model as confounders. Age (years), race/ethnicity (Non-Hispanic White, Non-Hispanic Black, Hispanic, Other), ever diagnosed with coronary heart disease (yes/no), diabetes (yes/no), and hypertension (yes/no) were self-reported during the NHANES interview and extracted from the NHANES Questionnaire File. Body mass index (BMI) was categorized into 4 categories (Underweight: <18.5 kg/m2; Normal: 18.5-<25 kg/m2; Overweight: 25-<30 kg/m2: Obese: ≥30 kg/m2), using the BMI data from the NHANES Examination File. Alcohol consumption was categorized into 2 categories (≥5 drinks every day or not), using the alcohol use data from the NHANES Questionnaire File. Smoking status was categorized into 2 categories (active smoker (serum cotinine > 10 ng/mL 19 ) or not), using the serum cotinine level data from the NHANES Laboratory File. Total cholesterol was categorized into 3 categories (Normal: <200 mg/dL; Borderline High: 200-239 mg/dL; High: ≥240 mg/dL), 20 using the cholesterol data from the NHANES Laboratory File.
Statistical analysis
Descriptive analyses comparing the characteristics in the testosterone deficiency group and non-testosterone deficiency group using t-tests and Chi-Square tests were conducted. For continuous variables, means and standard deviations were presented. For categorical variables, counts and proportions were presented.
Multiple linear regression models were fitted to the data to assess the association between testosterone deficiency and inflammation biomarker levels. Changes of inflammation biomarkers associated with testosterone deficiency and 95% confidence intervals (CIs) were presented. Multiple logistic regression models were also fitted to the data to examine the association between testosterone deficiency and the odds of having elevated inflammation biomarkers. Dichotomization cutoff points for hsCRP (High: hsCRP ≥3 mg/L; Normal: hsCRP <3 mg/L), ALT (High: ALT > 40 U/L; Normal: ALT ≤40 U/L), and AST (High: AST > 40 U/L; Normal: AST ≤40 U/L) were based on prior literature and clinical experience. 11,21 Odds ratios (ORs) for elevated inflammation biomarkers associated with testosterone deficiency and 95%CIs were presented.
All analyses were performed with Stata/MP 14.0.
A Longitudinal Study Using Data Based on a Registry Study in Germany
Study population
To investigate the longitudinal effect of testosterone therapy on inflammation, we used de-identified data from a registry study in Germany that contain participants’ treatment status and repeated measurement of testosterone and inflammation biomarkers. Seven hundred and seventy-six hypogonadal men were recruited from one urology center in Bremerhaven, Germany from 2004 to 2016. Hypogonadism diagnosis was confirmed if they had total testosterone level ≤12.1 nmol/L and symptoms such as decreased libido and erectile dysfunction, as assessed by the Aging Males’ Symptoms scale (AMS). The threshold of 12.1 nmol/L was selected based on clinical experience and confirmed by Bhasin et al. 18 Ethical guidelines formulated by the German Ärztekammer (German Medical Association) for observational studies in patients receiving standard treatment were followed. After receiving an explanation about the nature and the purpose of the study, all patients provided written consent to be included in the registry and have their data analyzed. Participants were followed semi-annually for updates in serum testosterone level (nmol/L), CRP (mg/L), ALT (U/L), AST (U/L), and several other physical, laboratory, and imaging test results. We did not dichotomize the outcome variables, as in that case we were not able to capture the potential treatment effect on alleviating inflammation if inflammation biomarker levels had not yet been reduced to normal range.
Cardiovascular outcome ascertainment
Cardiovascular events (i.e., myocardial infarction and stroke) occurring during follow-up were recorded. Cardiovascular events were partly reported in the form of “physician letters” from the hospital or the cardiologist/neurologist/family physician, and partly by patients themselves or relatives. The latter usually occurred when already scheduled patient visits had to be postponed due to an event.
Treatment assignment
Patients with prostate-specific antigen (PSA) levels less than 4 ng/mL were given the option of testosterone therapy at the beginning of the study. However, the final decision on whether or not to receive testosterone therapy was based on the patients’ own choice. Patients who decided to take testosterone therapy were classified as the treatment group (n = 400), and those who opted against testosterone therapy were classified as the control group (n = 376). As described previously, 1,22 patients on testosterone therapy received injections of 1,000 mg of testosterone undecanoate with the second injection 6 weeks after the first injection, followed by injections at 12-week intervals throughout the observation time. Since every injection was administered and documented in the urology office, the adherence to testosterone therapy was 100%. No participants changed their treatment status during the study period.
Statistical analysis
In the descriptive analysis, baseline testosterone, CRP, AST, ALT levels, and other characteristics were compared in the treatment and control groups using t-tests or Chi-square tests. Mean testosterone, CRP, ALT, AST levels were plotted to visually compare the changes in the treatment and control groups.
Cardiovascular event (myocardial infarction and stroke) incidence rates in the treatment and control groups were compared using an incidence rate difference and its 95%CI.
Linear mixed-effect models with a random intercept, a random slope for time (month), and fixed effects of treatment, time (month), and an interaction between treatment and time (month) were fitted to the data to investigate the longitudinal effect of testosterone therapy on CRP, ALT, and AST. Covariates that are closely related to CRP, ALT, and AST including age, BMI, smoking/drinking status, comorbidities (type 2 diabetes, hypertension, dyslipidemia), and coronary heart disease were included in the model.
All analyses were performed with Stata/MP 14.0.
Results
Results for the Cross-Sectional Study Using NHANES Data
As showed in Table 1, characteristics were different between the testosterone deficiency group and the non-testosterone deficiency group.
Characteristics of Participants by Testosterone Deficiency Status, NHANES 2015-2016.a

Abbreviations: NHANES, National Health and Nutrition Examination Survey; TD, testosterone deficiency; SD, standard deviation; BMI, body mass index.
aAge, race/ethnicity, alcohol consumption, hypertension, diabetes, and coronary heart disease were self-reported by the participants during the survey. BMI was calculated based on height and weight measured in the mobile examination center during the survey. Total cholesterol levels and serum cotinine levels were measured during the survey.
b Numbers may not add up to total because of missing values.
c P-value for t-test if the variable is continuous; P-value for Chi-Square test if the variable is categorical.
d Active smoker: serum cotinine level >10 ng/mL.
As compared to the participants without testosterone deficiency, those with testosterone deficiency were older, more likely to be obese, none activate smokers, have diabetes and hypertension (P-value < 0.05). There was no difference in race/ethnicity, drinking status, cholesterol levels, and coronary heart disease status between the two groups (P-value > 0.05).
The multiple linear regression results were presented in Table 2. Because of very few observations in the underweight BMI category (n = 33), the underweight BMI category and normal BMI category were collapsed in the analyses. As compared to men without testosterone deficiency, those with testosterone deficiency had 1.53 mg/L higher hsCRP (P-value < 0.001), 2.17 U/L higher ALT (P-value = 0.004), and 0.05 U/L lower AST (P-value = 0.926), after adjustment.
Multiple Linear Regression Results by Outcome, NHANES 2015-2016.a
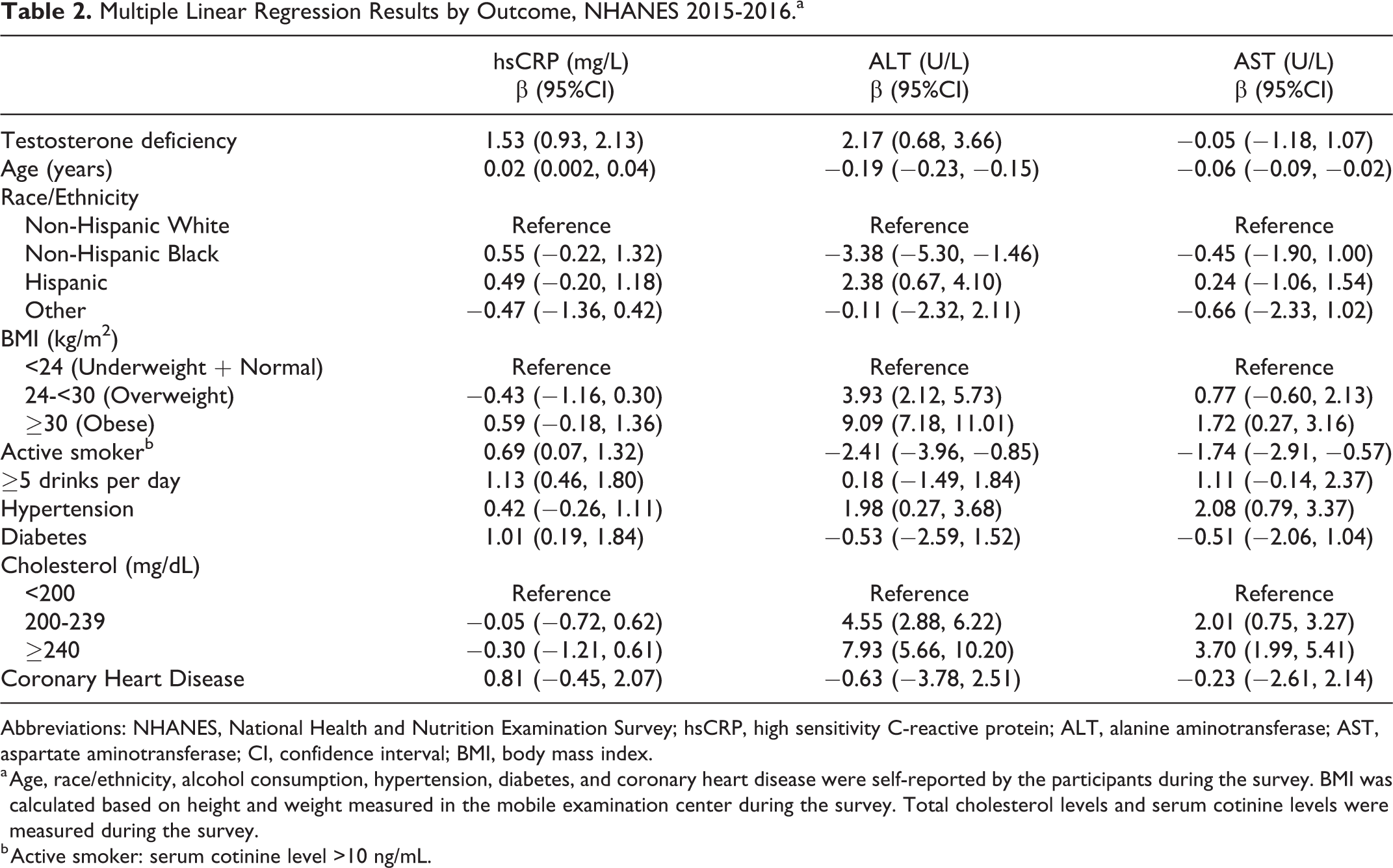
Abbreviations: NHANES, National Health and Nutrition Examination Survey; hsCRP, high sensitivity C-reactive protein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; BMI, body mass index.
a Age, race/ethnicity, alcohol consumption, hypertension, diabetes, and coronary heart disease were self-reported by the participants during the survey. BMI was calculated based on height and weight measured in the mobile examination center during the survey. Total cholesterol levels and serum cotinine levels were measured during the survey.
b Active smoker: serum cotinine level >10 ng/mL.
The multiple logistic regression results were presented in Table 3. After adjustment, the odds of having hsCRP ≥3mg/L in participants with testosterone deficiency was 1.81 times that in those without testosterone deficiency (P-value < 0.001); the odds of having ALT >40U/L in participants with testosterone deficiency was 1.46 times that in those without testosterone deficiency (P-value = 0.009); the odds of having AST >40U/L in participants with testosterone deficiency was 0.99 times that in those without testosterone deficiency (P-value = 0.971).
Multiple Logistic Regression Results by Outcome, NHANES 2015-2016.a

Abbreviations: NHANES, National Health and Nutrition Examination Survey; hsCRP, high sensitivity C-reactive protein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; OR, odds ratio; CI, confidence interval; BMI, body mass index.
a Age, race/ethnicity, alcohol consumption, hypertension, diabetes, and coronary heart disease were self-reported by the participants during the survey. BMI was calculated based on height and weight measured in the mobile examination center during the survey. Total cholesterol levels and serum cotinine levels were measured during the survey.
b Active smoker: serum cotinine level >10 ng/mL.
Results for the Longitudinal Study Using Data Based on a Registry Study in Germany
As shown in Table 4, the total follow-up duration for the control group was between 2 and 11 years, and 1.25 to 10.75 years for the treatment group. There was no difference in the mean follow-up duration between the treatment and control groups (7.25 vs 7.01, P-value = 0.067). The baseline characteristics were in general different in the 2 groups. As compared to the control group, men in the treatment group had higher BMI, CRP, ALT, and AST, were more likely to have hypertension, and dyslipidemia, but on average were younger, had a lower proportion of alcohol users, diabetic patients, and patients with prior cardiovascular events (P-value < 0.001). There was no difference in baseline testosterone levels and the proportions of smokers in the 2 groups.
Characteristics of Participants, by Treatment Status, German Registry Study.

Abbreviations: SD, standard deviation; BMI, body mass index; CRP, C-reactive protein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CV, cardiovascular.
a P-value for t-test if the variable is continuous; P-value for Chi-Square test if the variable is categorical.
b As the distribution of CRP levels are right-skewed, median and interquartile range (IQR) are provided as follows: treatment group-median 2.10, IQR 1.30-6.25; control group-median 0.90, IQR 0.50-1.65.
Table 5 presents the incidence rates and incidence rate difference in the treatment and control groups. All 50 myocardial infarctions and 46 strokes occurred in the control group, resulting in a lower incidence rate in the treatment group as compared to the control group (P-value < 0.001).
Incidence Rate and Incidence Rate Difference for Myocardial Infarction and Stroke, German Registry Study.

Abbreviation: CI, confidence interval.
Table 6 presents the longitudinal data analysis results using mixed-effect models. After adjustment, CRP levels in the control group increased by 0.003 (95%CI: −0.001, 0.007) mg/L per month, while CRP levels in the treatment group decreased by 0.05 (the sum of coefficients for month and interaction term month*treatment) (95%CI: −0.055, −0.046) mg/L per month; the variance of residual was 1.53, the variance of the random intercept was 11.77, the variance of month (slope) was 0.0015, and the covariance between the random intercept and month (slope) was −0.13; ALT levels in the control group increased by 0.157 U/L (95%CI: 0.145, 0.170) per month, while the ALT levels in the treatment group decreased by 0.142 U/L (the sum of coefficients for month and interaction term month*treatment) (95%CI: −0.154, −0.130) per month; the variance of residual was 43.18, the variance of the random intercept was 71.57, the variance of month (slope) was 0.01, and the covariance between the random intercept and month (slope) was −0.72; AST levels in the control group increased by 0.147 (95%CI: 0.136, 0.159) U/L per month, while the AST levels in the treatment group decreased by 0.148 (the sum of coefficients for month and interaction term month*treatment) (95%CI: −0.158, −0.137) U/L per month; the variance of residual was 30.13, the variance of the random intercept was 62.23, the variance of month (slope) was 0.009, and the covariance between the random intercept and month (slope) was −0.62.
Linear Mixed Effect Model Results, by Outcome, German Registry Study.

Abbreviations: CRP, C-reactive protein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; BMI, body mass index; CV, cardiovascular.
Figure 1 depicts the changes of mean testosterone, CRP, ALT, and AST levels in the 2 groups during the follow-up period with more fluctuations in the later years due to fewer number of observations. Over time, in the treatment group, testosterone level increased, and 3 inflammation biomarkers decreased, whereas in the control group, testosterone level decreased, and 3 inflammation biomarkers increased.
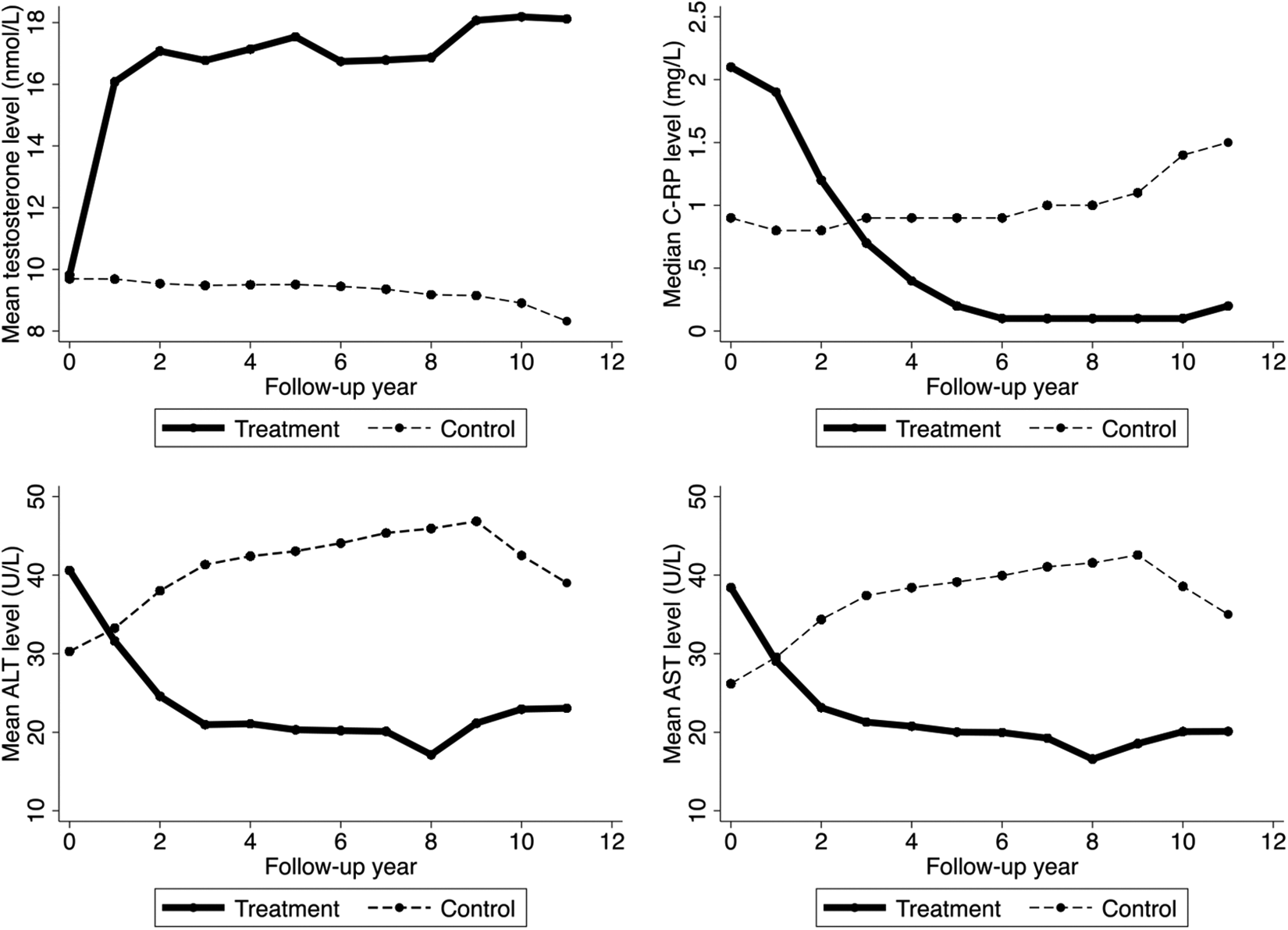
Mean testosterone, CRP, ALT, and AST levels over time, by treatment status.
Discussion
In our cross-sectional study using the recently released NHANES 2015-2016 cycle data, we found participants with testosterone deficiency had higher levels of inflammation biomarkers including hsCRP and ALT as compared to the participants without testosterone deficiency. In this longitudinal study using data from 776 hypogonadal men in a registry study in Germany, we found long-term testosterone therapy was associated with decreased CRP, ALT, AST, and cardiovascular event incidence during the study period among hypogonadal men.
By using the nationally representative samples, we examined the association of testosterone deficiency with hsCRP and liver enzymes in the US general population, which has not been investigated before. We found a positive association between testosterone deficiency and hsCRP, which was in line with previous studies investigating the associations between tertiles/quantiles of testosterone level and CRP. 11,12 As for liver enzymes, we only identified a positive association of testosterone deficiency with elevated ALT, but not with AST. Studies have shown that testicular feminized mice with very low testosterone levels had increased lipid deposition in the liver, 23 and low testosterone level was independently associated with non-alcoholic fatty liver disease, 24 the hepatic component of the metabolic syndrome that is a strong predictor for cardiovascular disease. 10,25 Non-alcoholic fatty liver disease, which affects cardiovascular health in many ways including increased liver and chronic low-grade inflammation and reduced coronary flow reserve due to liver fibrosis, is characterized by hepatic steatosis and injured hepatocyte that may result in increased permeability of hepatic cell membrane and release of liver enzymes. 10,26 Liver enzymes such as aminotransferases ALT and AST are produced in the liver and used to synthesize glycogen, a stored form of glucose that provides energy for the body. Most glycogens are stored in the liver, and a few are distributed to other organs. Unlike ALT that is mainly found in the liver, AST is “scattered” in various organs including the liver, cardiac muscle, and skeletal muscles. 27 Damage of any of these tissues may lead to increased AST levels. Thus, AST may not be as specific to the liver as ALT; this could be one potential explanation that we did not observe a direct association between AST and testosterone deficiency that may lead to the pathological changes on the liver.
Inflammation is a pathophysiological process as well as a pathway to the development of many chronic inflammatory diseases such as diabetes and coronary artery disease. 28 It was implied that interventions that suppress the inflammation responses might be considered as a strategy to prevent cardiovascular events. 29 Testosterone has been reported to elicit anti-inflammatory effects in tissues and organs by regulating the function of the cellular components of the immune system (e.g., macrophages, neutrophils, mast cells) and reducing the release of pro-inflammatory cytokines (e.g., tumor necrosis factor-alpha (TNF-α), IL-6, IL-1β) that are linked to endothelial injury in vessels. 28,30 In an animal study investigating the effect of testosterone depletion and administration on abdominal aortic aneurysm formation (a pathological phenotype of vascular aging) in male mice, the authors found that for male mice under depletion of testosterone (castrated), exacerbated aortic aneurysm characterized with enhanced aortic diameter was formed as compared to sham-operated mice. They also conducted histological analyses that showed macrophages infiltrating in the destroyed aorta and excessive expression of IL-6 and IL-1β, indicating that the formation of aortic aneurysm in the castrated male mice was probably a result of pronounced inflammation. They found after 9-week testosterone administration, the aortic expression of IL-6 and IL-1β was decreased, and the dilated aortic diameter was also ameliorated, indicating the anti-inflammatory actions of testosterone administration on vascular pathologies. 31 In another animal study, the authors reported testosterone therapy improved lipid accumulation in rabbits that were fed fat-rich diets. A decrease in liver and plasma TNF-α was also observed in these rabbits. 32 This implies that testosterone therapy may ameliorate the liver inflammation resulting from abnormal lipid profiles due to fat-rich diet and eventually reduces cardiovascular events. Prior human studies also reported positive associations between low testosterone level and abnormal lipid/glucose metabolism. 33 Our previous study investigating the longitudinal treatment effect of testosterone therapy on the Framingham Risk Score found continuously increasing testosterone level and improved lipid profiles in the treatment group.
To our knowledge, this is the first study investigating the long-term treatment effect of testosterone therapy on liver enzymes and inflammation biomarkers using longitudinal data with up to 11 year’s follow-up. Unlike prior trials with relatively short follow-up periods, 34,35 the longer duration of treatment in our study, repeated measurement of inflammation biomarkers, and a larger sample size increase the power to detect the potential longitudinal effect of testosterone therapy on inflammation and cardiovascular risk. By using mixed-effect models, we were able to evaluate the testosterone treatment effect on predictors of cardiovascular disease with taking into account individual’s variabilities. Besides, the cross-sectional study investigating the association between testosterone deficiency and inflammation biomarkers was conducted using large nationally representative samples. This increases the study power to detect this potential association.
Limitations
First, the study results are specific to the population studied and may not be generalizable. Second, the treatment assignment was not randomized and may therefore be subject to the placement of generally healthier participants in the treatment group. Because there were zero cases in the treatment group, we were not able to get adjusted HR associated with treatment therapy using Cox proportional hazards models. Third, there were 16 patients who had originally decided against testosterone therapy and were then convinced by their cardiologist to receive treatment. Some of these patients had a prior cardiovascular event and/or a cardiovascular event while they were not on testosterone, but there were no more events after they switched to testosterone therapy. These patients were not included in the analysis, which might have potentially impacted our estimates. Fourth, though there were no CVD events in the treatment group, our previous study found that the all-cause mortality was not zero in the treatment group. Nevertheless, the control group had a significantly higher all-cause mortality as compared to the treatment group, after propensity score matching. 36 Risk factors associated with cardiovascular disease were significantly improved in the treatment as well, which, at least in part, explains the reduced cardiovascular risk in the treatment group, though further large longitudinal studies (e.g., TRAVERSE trial) is warranted to confirm our study findings. Fifth, due to the nature of observational study design, residual confounding may still exist. However, after adjusting for major comorbidities and lifestyle characteristics, residual confounding alone is unlikely to explain the study findings. Lastly, due to the availability of data, we were not able to assess the association based on other key inflammation biomarkers such as IL-1, IL-6, or serum amyloid A. Future studies with rich data in these biomarkers are needed to verify our study findings.
Conclusion
By using nationally representative samples, we found a positive association between testosterone deficiency and inflammation biomarkers hsCRP and ALT; by using clinical data based on a registry study in Germany, we found long-term testosterone therapy alleviated inflammation, the major pathway of cardiovascular disease, which may explain in part how testosterone therapy reduced the risk of cardiovascular disease in our study population.
Footnotes
Author Contributions
Xiao Zhang: Conceptualization, Methodology, Formal analysis, Writing-Original draft preparation. Hongwei Zhao: Conceptualization, Methodology, Formal analysis, Visualization, Writing-Reviewing and Editing. Jennifer Horney: Conceptualization, Writing-Reviewing and Editing. Natalie Johnson: Conceptualization, Writing-Reviewing and Editing. Farid Saad: Writing-Reviewing and Editing. Karim Sultan Haider: Writing-Reviewing and Editing. Ahmad Haider: Writing-Reviewing and Editing. Xiaohui Xu: Resources, Supervision, Conceptualization, Visualization, Writing-Reviewing and Editing, Project administration.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Farid Saad is a senior consultant for Bayer AG, Berlin. Dr. Ahmad Haider has received research support, lecture honoraria and travel grants from Bayer AG. Karim Sultan Haider has received lecture honoraria and travel grants from Bayer AG.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
