Abstract
Chloride channel 3 (ClC-3), a Cl−/H+ antiporter, has been well established as a member of volume-regulated chloride channels (VRCCs). ClC-3 may be a crucial mediator for activating inflammation-associated signaling pathways by regulating protein phosphorylation. A growing number of studies have indicated that ClC-3 overexpression plays a crucial role in mediating increased plasma low-density lipoprotein levels, vascular endothelium dysfunction, pro-inflammatory activation of macrophages, hyper-proliferation and hyper-migration of vascular smooth muscle cells (VSMCs), as well as oxidative stress and foam cell formation, which are the main factors responsible for atherosclerotic plaque formation in the arterial wall. In the present review, we summarize the molecular structures and classical functions of ClC-3. We further discuss its emerging role in the atherosclerotic process. In conclusion, we explore the potential role of ClC-3 as a therapeutic target for atherosclerosis.
Keywords
Introduction
Atherosclerosis (AS), a chronic inflammatory process, is typically initiated with the retention and infiltration of cholesterol-laden circulating low-density lipoprotein (LDL) particles in the inner layer of the artery (intima), followed by endothelial dysfunction, ultimately resulting in a high inflammatory response. 1,2 Monocyte-derived macrophages transmigrate through vascular endothelial cells (VECs) into the subendothelial space of the vessel wall and generate reactive oxygen species (ROS). 3 ROS modify LDL to form oxidatively modified LDL (ox-LDL), which tends to be phagocytized by macrophages and vascular smooth muscle cells (VSMCs), thus resulting in the formation of foam cells. 4 At more advanced stages, hyper-migration and hyper-proliferation of VSMCs can cause arterial intima thickening and remodeling, which along with foam cells, contribute to the formation of atherosclerotic plaques (AP). 5 Furthermore, with the slow progression and destabilization of AP, clinical symptoms and acute cardiovascular events are likely to occur. 6
Ion channels are membrane-spanning proteins that play vital roles in physiological and pathological processes by controlling the flux of ions through cell membranes to generate electrical activity. Ca2+, K+, Na+, and Cl− transporters are widely expressed in various cell types, including VECs, VSMCs, adipocytes, and macrophages. These ion channels remain critical for regulating cell membrane potential, signal transduction, hemodynamics, and vasomotor functions. 7 Dysregulated expression of ion channels can be frequently detected in dysfunctional arterial endothelium, indicating that these ion channels are associated with the development and progression of AS. 8
ClC-3 is a key component of the volume-regulated chloride channel (VRCC), which functions as a regulatory volume decrease (RVD) to protect cells from a disproportionate increase in cellular volume due to environmental stimuli, such as hypoxia, ischemia, and osmotic perturbation. In addition, ClC-3 mediated Cl− efflux is an intracellular messenger that contributes to the activation of several signaling pathways by regulating phosphorylation. 9 -11 Accumulating evidence suggests that overexpression of ClC-3 leads to increased plasma LDL, endothelial dysfunction, pro-inflammatory activation of macrophages, hyper-proliferation and hyper-migration of VSMCs, and oxidative stress. 12,13 Furthermore, our previous studies have indicated that ox-LDL promotes foam cell formation, which is ClC-3 dependent. 14,15 Collectively, these findings suggest an important role for ClC-3 in AS development. In the present review, we summarize molecular structures and classical functions of ClC-3. Moreover, we discuss its role in the atherosclerotic process. Lastly, we assess ClC-3 as a promising therapeutic target for AS.
Molecular Structure of ClC-3
The ClC-3 gene was first cloned from rats using PCR. Reportedly, ClC-3 yields outward Cl− currents in Xenopus oocytes and almost all vertebrate cell types. 16 Accumulated data has revealed that ClC-3 acts as a chloride channel encoding Cl−/H+ antiporters in the cytoplasm and nuclear membrane in response to hypotonic challenges. In the arteries, ClC-3 yields outward rectification of the current-voltage, resembling swelling-activated Cl− currents, presenting numerous biophysical and pharmacological characteristics of VRCC. 11,17
To date, the molecular structure of ClC-3 is relatively conserved in vertebrate cell lines. ClC-3 is a polypeptide chain composed of 760 amino acids, which is severely tilted rather than perpendicular to the membrane, forming helixes that serve the pore by extending from either side of the membrane into the central plane. 18 -20 Helixes can be divided into D1-D12. D1 connects to an N-terminal amino group. In human colon cancer epithelial cells, serine 109 in the N-terminus was identified as an active site of ClC-3. 21 D1-D8 are hydrophilic regions that possess different degrees of hydrophilicity with other chloride channel proteins. Tyrosine 284, known to occur between D5 and D6, was identified as an active site of ClC-3 in cells derived from the vascular system. 22 D9-D12 are reportedly hydrophobic regions. D12, which connects the C-terminus, demonstrates weak hydrophobicity and contains a carboxyl group. Both N- and C-terminal regions are located in the cytoplasm (Figure 1). 19 In addition, threonine 532 has been identified as the active site of ClC-3 in cell groups of the vascular system; however, the precise location remains unclear. 23 Several of these helixes do not span the width bilaterally.
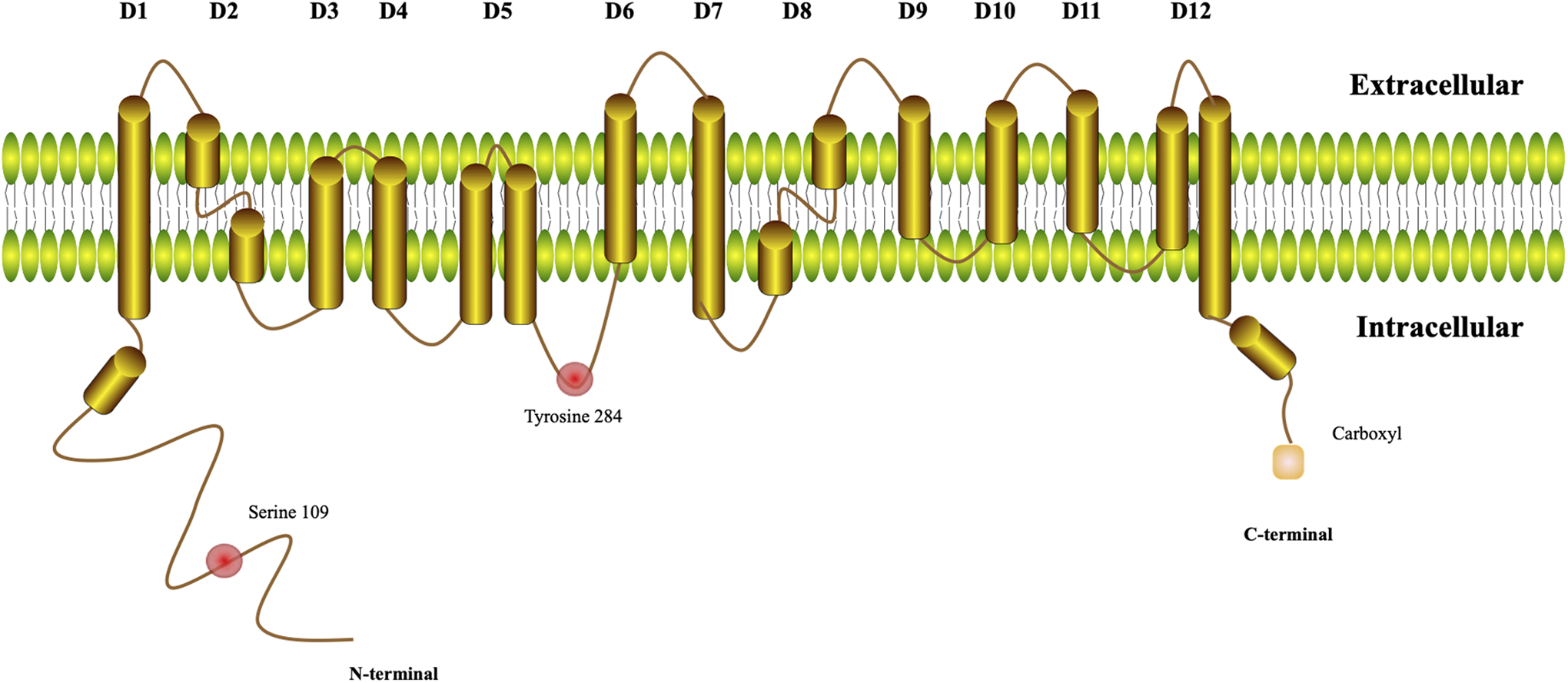
Molecular structure of chloride channel 3 (ClC-3).
The Classical Role of ClC-3: Cell Volume and Signaling Pathway Regulation
Extracellular osmolarity is stabilized at approximately 300 mOsm/L. However, cells are constantly challenged by volume changes triggered by electrolyte and water movement across the cytoplasmic membrane. 24 Accordingly, the ability to regulate cell volume in response to osmotic disturbances induced by molecular exchanges is vital for maintaining structural integrity and normal cellular functions.
Cell volume regulation allows swollen cells to be restored to their original volume, partly via a transient increase in ion efflux, a process known as RVD. 25 ClC-3 acts as a cell volume sensor and is upregulated in response to cell swelling induced by stimuli such as hypoxia, ischemia, and osmotic perturbation. ClC-3 possesses an RVD function and shifts the equilibrium potential of Cl− toward a mechanism that induces membrane depolarization. ClC-3 induced membrane depolarization not only enhances the driving force for osmotically active organic molecules, such as taurine, glutamate, and myoinositol, but also promotes the activity of other ion channels to regulate the movement of electrolytes such as K+, Ca2+, and Na+/H+ exchanger, ultimately resulting in cell volume shrinkage (Figure 2). 26,27 Therefore, ClC-3-mediated RVD contributes to volume regulation, thus affording cells protection from stimulus-induced cellular edema.

Schematic diagram of the volume regulation function of ClC-3. Cell volume regulation is achieved by Cl− efflux-induced membrane depolarization. Membrane depolarization leads to the effusion of intracellular osmotically active organic molecules, such as taurine, glutamate, and myoinositol, and activation of other ion pumps, such as K+, which drives water efflux and restores cell volume to the normal level. ClC-3 indicates chloride channel 3.
Previous reports have suggested that Cl− may act as a critical intracellular second messenger, pertinent to the functions of several intracellular kinases, ion channels, and transporters. 28 For example, ClC-3 mediated Cl− efflux is likely to elicit effects on activation of the intra-endosomal signaling pathway, including BCl-2/Bax, 29 AKT/GSK3β, 30 PI3K/AKT/mTOR/p70S6 K, 31 JNK/p38 MAPK, 15 CXCR4/JAK-2, 32 ERK1/2, 33 Beclin1/Vps34, 34 and Wnt/β-catenin. 35 Moreover, ClC-3 may be a critical subunit or cooperative protein for the assembly of functional proteins or membrane receptors, such as NADPH oxidase 1 (NOX1), 36 protein kinase C (PKC), 37 calcium/calmodulin-dependent protein kinase II (CamKII), 21 matrix metalloproteinase-2 (MMP-2) receptors, 38 Sirtuin 1 (Sirt1), 39 SRY-box transcription factor 2 (SOX2), 40 and serum- and glucocorticoid-regulated kinase 1 (SGK1). 41 Notably, angiotensin II (AngII), lipopolysaccharide (LPS), interleukin (IL), endothelin 1 (ET1), transforming growth factor-β1 (TGF-β1), and tumor necrosis factor-α (TNF-α) can promote the ClC-3 expression and stimulate the activation of multiple inflammatory signaling pathways such as nuclear factor kappa B (NF-κB), 36,42 Janus kinase (JAK)-signal transducer and activator of transcription (STAT), 43 and LPS/ Toll-like receptor 4 (TLR4). 44 ClC-3 knockout or inhibition was found to suppress these inflammatory signaling pathways, alleviating the induction of inflammation both in vitro and in vivo. However, overexpression of ClC-3 or reduced intracellular Cl− results in the activation of these signaling pathways, known to be involved in inflammation. 42,44 These findings reveal that ClC-3 may be a critical link in intracellular signal conduction and may be a novel therapeutic target for inflammatory diseases.
The Emerging Role of ClC-3 in Atherosclerosis
ClC-3 has been associated with the development of AS, a progressive inflammatory disorder. Our previous research has demonstrated that ClC-3 ablation, which induces high intracellular Cl− levels, inhibits foam cell formation, and represents a modest reduction in atherosclerotic lesions. 14,15 More recently, several studies have identified that ClC-3 is associated with increased plasma LDL, excessive oxidative stress, endothelial dysfunction, pro-inflammatory activation of macrophages, and hyper-proliferation and hyper-migration of VSMCs, which have been identified as contributing factors of AS. 45,46 These findings indicate the important role of ClC-3 in AS progression.
ClC-3 and Increased Plasma LDL
Typically, the main blood lipids include cholesterol, triglycerides, phospholipids, and glycolipids. Among them, cholesterol binds to various lipoprotein particles possessing biological and physiological functions. LDL is the main carrier of endogenous cholesterol and accounts for approximately 30% of plasma lipoproteins. Increased LDL tends to be retained and accumulated in the innermost layer of the artery (intima), the leading risk factor for AS induction and progression. 47
ClC-3 overexpression was correlated with adipocyte proliferation and apoptosis. Adipocytes, as energy storage units, reportedly participate in LDL secretion. Growing evidence has indicated that homeostatic changes in adipocyte proliferation and apoptosis can influence total cholesterol levels and plasma LDL levels associated with dyslipidemia and AS. 48 ClC-3 overexpression significantly enhanced the proliferation of adipocytes and plasma LDL levels. ClC-3 blockade using tamoxifen or 4,4’-diisothiocyanato-2,2’-stilbenedisulfonic acid (DIDS) inhibited adipocyte proliferation and plasma LDL levels. 49 Moreover, compared with wild-type mice, ClC-3−/− mice fed a high-fat diet displayed improved lipid metabolism, resulting in a reduction in the adipose tissue area and overall body weight. 50 Furthermore, ClC-3 deletion prevents adipocyte apoptosis induced by endoplasmic reticulum stress and palmitate in vitro, as well as in mice with type 2 diabetes. 51 These results suggest that ClC-3 may play a vital role in adipocyte-induced LDL elevation and AS induction.
Furthermore, ClC-3 is involved in insulin secretion via the exocytosis function of pancreatic β-cells. Insulin levels positively correlate with total cholesterol and LDL levels, and hyperinsulinemia or insulin resistance critically increases the risk of developing AS. 52 Insulin exocytosis from pancreatic β-cells is dependent on the maturation and acidification of large dense-core vesicles (LDCVs), which are necessary for prohormone convertase cleavage of proinsulin. ClC-3 is expressed on LDCVs, and ClC-3 deletion and inhibition of trans-granular Cl− fluxes reduces insulin secretion by directly reducing LDCV acidification and exocytosis. 53 -55 In animal experiments, pancreatic β-cells revealed defective insulin exocytosis in ClC-3−/− mice. 54 These findings suggest a crucial role for ClC-3 in insulin secretion in pancreatic β-cells by employing a mechanism that mediates Cl−/H+ transport to promote LDCV acidification. Inhibition of ClC-3 may play a significant role in treating insulin-induced elevations in total cholesterol and LDL levels.
ClC-3 and Endothelial Dysfunction
VECs, which form a single cell layer of the endothelium in the interior surface of blood vessels, are subjected to hemodynamic stress with pulsatile blood flow and high-pressure amplitude. VECs at arterial branching and curvatures are susceptible to LDL accumulation and deposition. LDL induces constant injury to VECs, causing VEC atrophy or damage or desquamation of the arterial wall. Furthermore, VEC dysfunction can increase the arterial intimal permeability to facilitate LDL deposition in the subendothelial space and alter endothelial barrier properties. 56
Reportedly, ClC-3 mediated RVD can increase arterial intimal permeability and lead to endothelial injury. ClC-3 is extensively expressed on the surface of VECs throughout the endothelium. 57 Although ClC-3 appears to protect VECs from an excessive increase in cell volume, LDL stimulation or high-fat diet feeding can induce ClC-3 expression, resulting in RVD function, causing VEC atrophy. Moreover, ClC-3 mediated RVD can induce VEC apoptosis, resulting in increased arterial intimal permeability and endothelial injury. 58 Previous reports have revealed that upregulation of ClC-3 induces VEC atrophy, as well as delocalization of LDL into the subintima. 15 These findings indicate that the ClC-3 mediated RVD function may be one mechanism responsible for the progressive accumulation of LDL in the subintima.
Additionally, ClC-3 may contribute to endothelial precursor cell (EPC)-associated endothelial dysfunction. EPCs can migrate to injured areas of the vascellum and differentiate into the vascellum composition, which plays a vital role in neointimal hyperplasia and re-endothelialization. In hypertensive–hypercholesterolemic hamsters, inhibition of EPC activity has been suggested to prevent endothelial dysfunction and AS development under certain inflammatory conditions. 59 ClC-3 can mediate the adhesion and migration of EPCs by regulating CXCR4/JAK-2 signaling, contributing to EPC-mediated neovascularization in ischemic tissues. 32 However, ClC-3 deficiency reportedly reverses Ang II-induced EPC apoptosis. 29 ClC-3 may be involved in the healing process of vascular intimal injury, and inhibition of ClC-3 may suppress EPC-associated endothelial dysfunction and AP formation. The detailed mechanisms are poorly understood and warrant further investigations.
ClC-3 and Oxidative Stress
ROS is a group of small reactive molecules that play important roles in regulating various cellular functions and biological processes. However, ROS overproduction can lead to oxidative stress. In the endothelial intima, ROS may promote endothelial nitric oxide synthase uncoupling, decrease endothelial nitric oxide synthase activation, reduce nitric oxide generation, as well as participate in LDL oxidative modification, finally forming ox-LDL. High levels of ox-LDL reportedly mediate AS pathogenesis. 60 Moreover, due to their high reactivity, ROS induce tissue and cell damage, thus impairing cellular functions and resulting in dysfunction of endothelial cells, macrophages, and VSMCs, which contribute to AS development. 61,62
ClC-3 is known to be involved in oxidative stress through ROS generation. NOX, a membrane-bound enzyme family located in the cytoplasmic membrane, is a major source of ROS. 63 Notably, ROS production paralleled ClC-3 upregulation and the presence of Cl− currents; this phenomenon is implicated in mediating the beneficial effects of ClC-3 in NOX activation. 64 NOX activity is markedly reduced in ClC-3 knockout VECs, VSMCs, and macrophages. 65 Mechanistically, ClC-3 promotes the membrane translocation of key cytosolic regulatory subunits of NOX1, including p22phox, p47phox, and p67phox, and then increases the formation of the NOX1 complex. 66 Furthermore, LPS, IL, AngII, AT1, TNFα, and TGFβ1, all primary triggers for NOX activation and ROS overproduction, reportedly promote Cl− efflux. ClC-3 inhibition can potentially slow endosomal NOX activity, and NOX1 is unable to generate ROS in VSMCs. 13,67 ClC-3-mediated phosphorylation and activation of the p38 MAPK signaling pathway are reportedly involved in NOX activation induced by LPS, IL, AngII, AT1, TNF-α, and TGF-β1. 64 ClC-3 silencing markedly inhibited the phosphorylation and activation of the p38 MAPK signaling pathway, as well as reduced NOX-derived ROS production. However, these effects can be reversed by ClC-3 overexpression. 13,64,66,68 In addition, a previous study has demonstrated that VECs excrete ROS via ClC-3 channels, suggesting that ClC-3 may be involved in the extracellular transport of ROS. 13,69 Collectively, ClC-3 may play a significant role in dysregulated ROS production, leading to multiple pathological events of AS, including oxidative stress, LDL oxidative modification, and dysfunction of endothelial cells, macrophages, and VSMCs.
ClC-3 and Macrophage Pro-Inflammatory Activation
One critical factor contributing to the pathological development of AS is circulating monocytes recruited to the endothelial intima, which transmigrate to the superficial layer, and subsequently differentiate into macrophages and form foam cells. An event known to trigger this process is the accumulation of LDL particles in subendothelial spaces, which stimulate overlying VECs to produce numerous pro-inflammatory molecules, including adhesion, chemotactic, and activation molecules. All these pro-inflammatory molecules result in a hyper-inflammatory macrophage phenotype. 70,71
ClC-3 is involved in monocyte adhesion and migration by mediating the transcription of pro-inflammatory molecules. Overexpression of ClC-3 enhances the adhesion and trans-endothelial migration of monocytes; however, ClC-3 knockout significantly inhibited these effects. 42,65 Vascular cell adhesion molecule 1 (VCAM1), monocyte chemoattractant protein 1 (MCP1), and intercellular adhesion molecule 1 (IAM1) reportedly promote monocyte adhesion to the endothelium. ClC-3 overexpression and reduced intracellular Cl− levels promote the expression of VCAM1, MCP1 and IAM1; in contrast, ClC-3 deletion and elevated intracellular Cl− levels inhibit their expression. 42,72 Moreover, transfection with ClC-3 siRNA decreased the secretion of LPS, IL, AngII, AT1, TNF-α, and TGF-β1, which can induce the transcription of VCAM1, MCP1, and IAM1. 68,73 In addition, ClC-3-mediated RVD contributes to cellular morphological changes, which allow monocytes to squeeze through pores present on the VEC layer. 74 These findings suggest that ClC-3 may be involved in atherogenesis by promoting monocyte aggregation in the arterial intima.
ClC-3 is involved in the hyper-inflammatory phenotype of macrophages. In high-fat diet-induced obese mice, ClC-3−/− mice demonstrated significantly attenuated macrophage activity, as well as accumulation in adipose tissue. 45 NF-κB, which promotes macrophage differentiation, migration, accumulation, and proliferation, is one of the most important factors that increase macrophage pro-inflammatory activation, as well as the inflammatory response. 71,75 Phosphorylation and activation of NF-κB in macrophages reportedly parallel outward rectifying Cl− currents and ClC-3 upregulation, suggesting that ClC-3 may be involved in activation of NF-κB signaling. 42 ClC-3−/− or Cl− channel blockers inhibit LPS-, TGF-β1-, and TNF-α-induced NF-κB activation, and the expression and secretion of cytokines or chemokines, including macrophage colony-stimulating factor. However, ClC-3 overexpression or lower intracellular Cl− levels can reverse these effects and dramatically enhance macrophage activity. 42,44,45 These results indicate that the NF-κB-mediated hyper-inflammatory phenotype of macrophages is Cl− dependent. In addition, a serine on D0 (N-terminal) of ClC-3 has been identified as an active site of CamKII, and the activation of CamKII is required for the IL1β-induced hyper-inflammatory phenotype of macrophages. 21 Overall, ClC-3 displays pro-inflammatory activity and can maintain the hyper-inflammatory phenotype of macrophages.
Furthermore, ClC-3 leads to foam cell formation and accelerates AS. Under ox-LDL stimulation, macrophages exhibit increased endocytic activity, accompanied by augmented transmembrane Cl− movement and ClC-3 expression, thus suggesting that ClC-3 may play a role in stimulus-induced phagocytosis in macrophages. 14,76 Overexpression of ClC-3 significantly increased the binding and phagocytosis of ox-LDL in both RAW264.7 macrophages and primary isolated mouse peritoneal macrophages; ClC-3 ablation significantly reversed these effects. Accordingly, ClC-3 overexpression increased intracellular cholesterol accumulation and accelerated foam cell formation. 14,15 In terms of molecular mechanisms, the rapid phagocytosis of ox-LDL particles by macrophages is mediated by a group of scavenger receptors (SRs), such as SRa. SRa expression can be regulated by the JNK/p38 MAPK signaling pathway. ClC-3 ablation or increased intracellular Cl− levels reduce JNK/p38 MAPK phosphorylation levels, along with SRa expression in RAW264.7 macrophages. 14,15 Accordingly, targeting ClC-3 could be employed as a potential therapeutic strategy for AS, at least in part, by regulating JNK/p38 MAPK-dependent SRa expression and foam cell formation (Figure 3). 77
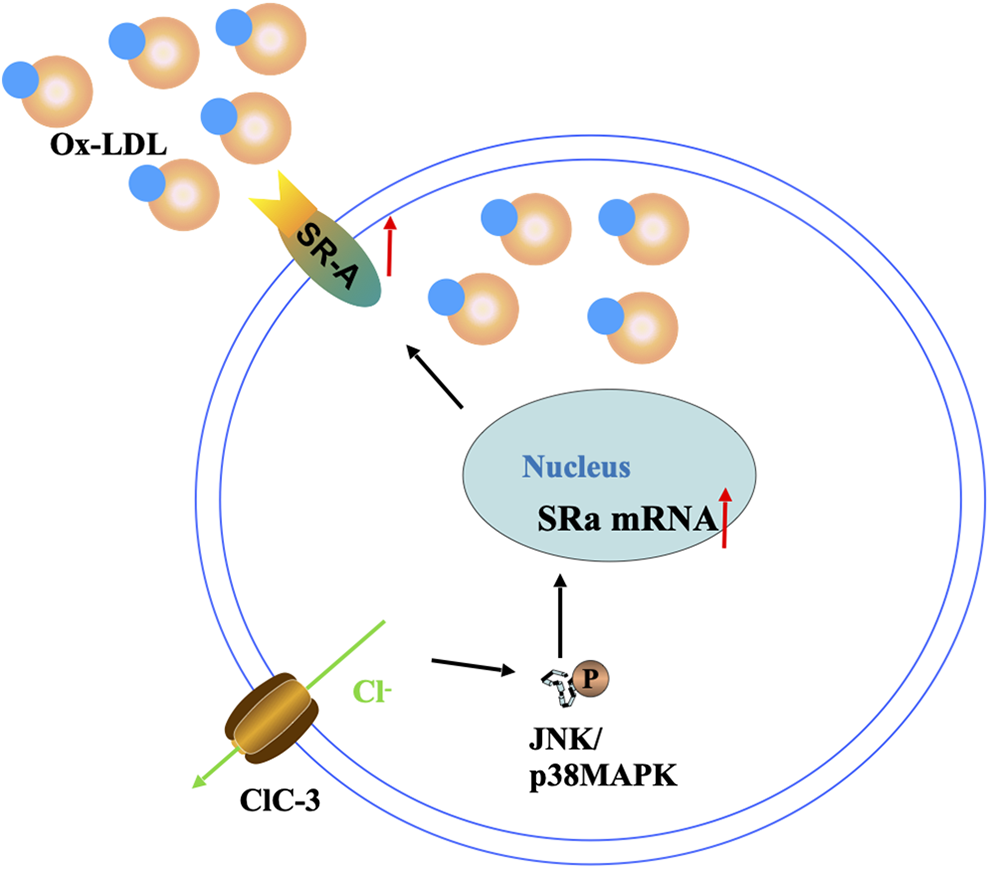
ClC-3 may induce macrophages to phagocytose ox-LDL. ClC-3 mediated decrease in cytoplasmic Cl− levels may contribute to the activation of JNK/p38 MAPK pathway, thus promoting the expression of SRa mRNA in macrophages. SRa mediates phagocytosis of ox-LDL by macrophages. Therefore, ClC-3 may be involved in the formation of foam cells. ClC-3 indicates chloride channel 3; ox-LDL, oxidatively modified low-density lipoprotein; JNK, c-Jun N-terminal kinases; MAPK, mitogen-activated protein kinase; SRa, scavenger receptor a.
ClC-3 and Arterial Intima Thickening and Remodeling
VSMCs, which are the most abundant cell type in the vasculature, are essential for maintaining vascular tone, tissue elasticity, wall stress homeostasis, and vessel stiffness. Atherosclerotic lesions are primarily located in the vascular walls. Thickening of the arterial intima is a key feature of AS progression, mainly due to the dispersion and migration of VSMCs into the superficial layer of the intima. 78 The migration and proliferation of VSMCs are crucial events in neointimal formation, a hallmark of AS. 79
In an inflammatory environment, ClC-3 could be implicated in VSMC migration by employing a mechanism similar to that mediating macrophage migration. Coordinated ClC-3 expression and plasmalemmal Cl− fluxes were detected during electrophysiological recordings of VSMC migration. 12 ClC-3 gene disruption reduced the Cl− current by 50%, and VSMC migration was also halved when compared with wild-type VSMCs. 12 Moreover, it has been reported that ClC-3 phosphorylation at threonine 532 is required for AngII-induced Cl− currents and VSMC migration. 23 Overall, the function of ClC-3 in arterial intima structural remodeling via VSMC migration warrants further in-depth study and exploration.
ClC-3 plays an important role in regulating cell proliferation. VSMC proliferation is accompanied by cell volume swelling or shrinkage. Moreover, the magnitude of Cl− currents in actively growing VSMCs is higher than that observed in growth-arrested or differentiated VSMCs, indicating that Cl− currents and volume changes may be necessary for VSMC proliferation. 80 -82 Undoubtedly, ClC-3 expression is upregulated during VSMC proliferation, and ClC-3 is required for the proliferation of VSMCs. 36,83 ClC-3−/− mice exhibit a reduction in the neointimal area of the carotid artery following injury. 14,15,45,46 Static pressure, insulin growth factor (IGF), ET1, TNF-α, and SGK1, which are concomitant and proliferation factors for VSMCs, can enhance ClC-3 protein expression and induce cell proliferation in cultured human or rat aortic VSMCs. In contrast, ClC-3 antisense oligonucleotide or 4-(2-Butyl-6,7-dichloro-2-cyclopentylindan-1-on-5-yl)oxybutyric acid (DCPIB) significantly inhibit VSMC proliferation. 84,85 Moreover, pharmacological inhibitors or transient ClC-3 silencing prolonged G0-G1 transition, arresting rat VSMC proliferation. 80,86 Mechanistically, ClC-3 can mediate ET1- and IGF-induced phosphorylation and activation of Akt/GSK-3β, thereby downregulating cyclin D1 and cyclin E and upregulating p27kip and p21cip, which promotes the entry of VSMCs into the cell cycle. 87,88 In addition, lower cytoplasmic Cl− induced by ClC-3 is required for ET-1 and TNF-α induced ERK phosphorylation and NF-κB activation, which accelerates the progression from G1 to S phase. 33,36 Furthermore, ClC-3 decreases the rate of premitotic condensation (PMC), which is associated with cytoplasmic and DNA condensation in the mitotic M phase to accelerate mitotic progression. 89 In conclusion, ClC-3 is involved in VSMC proliferation, known to be associated with arterial intima remodeling and AS progression.
Perspectives
The unique bioactive properties and molecular mechanisms of ClC-3 in several AS pathological processes remain poorly understood and need to be systematically explained. Studies indicate that disorders of adipocyte proliferation and apoptosis, hyperinsulinemia, insulin resistance, and EPC-mediated vascular endothelial repair are associated with AS occurrence and development. To date, few studies have investigated the role of ClC-3 in these processes. Moreover, VSMCs are another critical source of foam cells, and the role of ClC-3 in the formation of VSMCs derived foam cells is worth exploring. Furthermore, the RVD function of ClC-3 is one of the important mechanisms of cellular atrophy or morphological changes, providing a novel explanation for the expansion of arterial intimal permeability in facilitating trans-endothelial LDL transport and the transmigration of macrophages and VSMCs into the subendothelial layer of arteries.
Recent studies have revealed that Cl− may act as a secondary intracellular messenger. Several signaling cascades are amplified upon low intracellular Cl− levels and are inhibited by high intracellular Cl− levels. 90 -92 Therefore, insights gained from these empirical studies may aid in elucidating underlying molecular mechanisms of ClC-3 in AS-related signaling pathways. Furthermore, ClC-3 is thought to interact with proteins or enzymes, at least as a key subunit or cooperative protein for the assembly of functional proteins or membrane receptors. For example, ClC-3 is a receptor-associated protein that interacts with MMP-2. 38 ClC-3 may function as a partner in the internalization and externalization of MMP-2, indicating that ClC-3 may be involved in the MMP-2-dependent migration of VSMCs and macrophages. Moreover, co-immunoprecipitation demonstrated that the N-terminal of ClC-3 shares an active site with CaMKII, a pivotal kinase for the activation of VSMCs and macrophages. Accordingly, inhibition of ClC-3 can reduce CamKII activation. 21,93 In addition, ClC-3 protein interactions with postsynaptic glutamate receptors and postsynaptic density protein 95 have been reported. 94 These findings suggest that ClC-3 may have complex physiological and pathological functions.
Finally, in VECs and VSMCs, non-phosphorylatable mutations of tyrosine 284 and threonine 532 in ClC-3 almost completely abolished ClC-3-mediated Cl− current and Cl− efflux; in contrast, phosphomimetic mutation significantly promoted ClC-3-mediated Cl− current. 22,23 In tumor cells, serine 109 in the N-terminal has been identified as an active site of ClC-3. 21 These findings suggest that some unique phosphorylation sites of the ClC-3 protein are associated with ClC-3 channel activity. Moreover, membrane translocation of ClC-3 is dependent on the interaction between the N-terminal dileucine cluster and clathrin, indicating that the N-terminal may provide a mechanism for ClC-3 channel inactivation, as well as for mediating its biological effects. 95 Accordingly, the N-terminal or amino acid site of ClC-3 may be a key target for AS therapy, presenting significant implications for further target validation and drug screening.
Conclusion
Over the last 15 years, accumulated evidence has revealed an intricate connection between ion channels and AS. Molecular identification of ClC-3 represents a significant breakthrough in the ion channel field. ClC-3 can be associated with AS occurrence and development. Recent studies offer insights into key roles and underlying mechanisms of ClC-3 in arterial homeostasis. (Figure 4). More broadly, several therapeutic agents or siRNAs can inhibit the expression of ClC-3, thus mitigating the pathophysiological processes of AS (Table 1).

The role of ClC-3 in atherosclerosis. ClC-3 mediated RVD or Cl− efflux may play a significant role in LDL infiltration, as well as in monocyte adhesion and trans-endothelial migration. Monocytes are stimulated by ClC-3-mediated production of inflammatory cytokines and molecules, thereby differentiating macrophages, demonstrating a hyper-inflammatory phenotype. ClC-3 might regulate ROS production, which oxidizes LDL to form ox-LDL and mediates the formation of foam cells by regulating phagocytosis of ox-LDL. Additionally, ClC-3 mediated hyper-migration and hyper-proliferation of VSMCs can form AP to induce arterial intimal thickening. ClC-3 indicates chloride channel 3; RVD, regulatory volume decrease; LDL, low-density lipoprotein; ROS, reactive oxygen species; ox-LDL, oxidatively modified low-density lipoprotein; VSMCs, vascular smooth muscle cells; AP, atherosclerotic plaques.
Therapeutic Agents That Inhibit ClC-3 Expression.

Abbreviations: VSMCs, vascular smooth muscle cells; LDL, low-density lipoprotein; ClC-3, Chloride channel 3; ROS, reactive oxygen species; VECs, vascular endothelial cells; DIDS, 4,4’-diisothiocyanato-2,2’-stilbenedisulfonic acid; MMP-2, matrix metalloproteinase-2; DCPIB, dichloro-2-cyclopentylindan-1-on-5-yl)oxybutyric acid.
In summary, ClC-3 downregulation or inhibition alleviates the occurrence and development of AS. However, the role of ClC-3 in AS has only begun to be unveiled. Further research is needed to elucidate detailed molecular mechanisms of ClC-3 in modulating plasma LDL, inflammation, oxidative stress, arterial intima remodeling, and endothelial dysfunction. Moreover, ClC-3 may be a potential biomarker for diagnosis and as a therapeutic target in AS. Additional information would help establish the association between ClC-3 and AS more precisely.
Footnotes
Author Contributions
Deng Niu contributed to the drafting of the manuscript. Dr. Li Lanfang contributed to the editing and revision of the manuscript critically for the important scientific content. Prof. Xie Zhizhong contributed to the design, conception, and edition of the manuscript and finally approved the upcoming version. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 81970431), Natural Science Foundation of Hunan Province (No. 2019JJ40251), and Open Fund of Hunan Provincial Key Laboratory of Novel Antibody-Based Drug and its Intelligent Transport System (No. 2020TP1044).
