Abstract
Pulmonary veno-occlusive disease (PVOD) is a fatal disease of pulmonary vascular lesions leading to right heart failure. Heritable PVOD (hPVOD) is related to biallelic mutation of EIF2AK4 (encoding GCN2), but its molecular mechanism remains unclear. In this study, we aimed to investigate the pathogenesis of PVOD and to find potential drug targets for PVOD. GCN2 dysfunction led to an enhanced transcription of collagen I gene (col1a1 and col1a2) through decreasing ATF3-dependent p38 phosphorylation inhibition in PVOD, which promotes the collagen I synthesis in pulmonary arterial smooth muscle cells (PASMCs) and eventually leads to increased collagen deposition in pulmonary artery. Four GCN2 knockout (KO) cell lines (exon 15 or 33 mutation) were successfully constructed by epiCRISPR system. Two induced pluripotent stem cells (iPSCs) were generated by reprogramming peripheral blood mononuclear cells (PBMCs) of PVOD patient. It was also comfirmed that GCN2 dysfunction could lead to increased expression of collagen I in lateral plate mesoderm lineage-smooth muscle cells (LM-SMCs) differentiated from both GCN2 KO cell lines and iPSCs. SB203580 (a specific inhibitor of p38) improved hemodynamics and pulmonary vascular remodeling in mitomycin C (MMC)-induced PVOD rats by right ventricle echocardiography. On the whole, we proposed that GCN2 deficiency decreased ATF3-dependent p38 phosphorylation inhibition in PVOD development and suggested a potential therapeutic reagent of SB203580 for the treatment of the disease.
Introduction
Pulmonary veno-occlusive disease (PVOD) is a fatal disease of pulmonary vascular lesions leading to right heart failure, which categorized as belonging to group 1 pulmonary hypertension. 1 The main pathological features of PVOD are pulmonary artery medial hypertrophy and intimal fibrosis and pulmonary vein intimal fibrosis. 2 The hemodynamic and clinical features of PVOD and pulmonary artery hypertension (PAH) are similar, but using of PAH therapeutic drugs in patients with PVOD may cause pulmonary edema. 2 To date, it has not been reported that crucial target or therapeutic drug to PVOD, so lung transplantation is still the preferred therapy. Loss of eukaryotic translation initiation factor 2 alpha kinase 4 (EIF2AK4), encoding GCN2, leads to heritable PVOD (hPVOD), which are identified by whole exon sequencing on 5 sporadic PVOD (sPVOD) and 13 PVOD families. 3 Recently, it has been shown that GCN2 deficiency reduced BMP9-induced SMAD1/5/8 phosphorylation in PAEC (pulmonary arterial endothelial cell) of heritable PVOD (hPVOD). 4 However, the molecular mechanism was mainly identified in PAEC, but not in PASMC (pulmonary arterial smooth muscle cell) of hPVOD. Therefore, study deeply on the pathogenesis of PVOD is great significance for the development of therapeutic drugs.
Activating transcription factor 3 (ATF3) is one of cAMP responsive element-binding families, and different stimuli or cytokines regulate ATF3 by triggering intracellular signaling pathways. It has recently been shown that ATF3 is involved in the pulmonary artery intimal layer proliferation of PAH. 5 Downregulation of ATF3 in vascular smooth muscle cells (VSMCs) can induce apoptosis, while overexpression of ATF3 can promote migration, indicating that ATF3 plays a critical regulatory role in the process of apoptosis and migration of VSMCs. 6 p38 mitogen-activated protein kinase (p38 MAPK) signaling pathway, as one of the MAPK signaling pathways, is involved in a variety of cellular activities such as cardiac hypertrophy, inflammatory response, apoptosis, differentiation, cell cycle and growth. 7 p38 MAPK signaling pathway can be triggered by various growth factors or stimuli in different cell types.
In this study, we examined the roles and effects of GCN2-ATF3-p38 MAPK signaling pathway in PVOD and evaluated the effects of SB203580 on MMC-exposed PVOD rat model. Our findings demonstrated that dysfunctional GCN2 enhanced transcription of collagen I gene (col1a1 and col1a2) through decreasing ATF3-dependent p38 phosphorylation inhibition, which promotes the collagen I synthesis in pulmonary arterial smooth muscle cells (PASMCs) and eventually leads to increased collagen deposition in pulmonary vascular wall. Moreover, SB203580 may be a potential therapeutic agent for the treatment PVOD.
Methods and Materials
Patient Samples
Human PVOD patient samples were obtained from Wuxi People’s Hospital affiliated to Nanjing Medical University (Jiangsu, China). All samples were from patients that gave informed consent and ongoing research protocols were approved by the ethical committee of Wuxi People’s Hospital No. (2015)36. All lung tissues of patients tested for genes associated with PAH by whole exome sequencing. The total protein of lung tissues was extracted by Minute Total Protein Extraction Kit for Animal Cultured Cells and Tissues (Invent Biotechnologies, USA) according to the manufacturer’s instructions. A detailed depiction on how the PASMCs were isolated from pulmonary artery in Morrell et al. 8 Then PASMCs were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (cell culture center, Institute of Basic Medicinal Sciences, CAMS) supplemented with 1% penicillin-streptomycin (ThermoFisher Scientific, USA) and 10% fetal bovine serum (FBS) (ThermoFisher Scientific, USA). PASMCs were used in the experiment after passage 3.
Experimental Animals
The experimental protocol was approved by the animal care and use committee of the Second Affiliated Hospital, Zhejiang University School of Medicine (AIRB-2021-967). Twenty-four male Wistar rats (female rats were more sensitive to the toxicity of MMC exposure 9,10 ) weighing 160 g to 180 g were purchased from Beijing Vitalriver company (Beijing, China). Rats were randomly divided into 4 groups (n = 6 for each group): (a) control group, an equal volume of saline, intraperitoneal (i.p.); (b) model group, MMC (Target Molecule, USA) 3 mg/kg/week/2 weeks, and an equal volume of vehicle (0.03 N HCl 0.9% saline) once a week for 3 weeks, i.p.; (c) low-dose group: MMC 3 mg/kg/week/2 weeks and SB203580 (Target Molecule, USA) 2 mg/kg/week/3 weeks, i.p.; (d) high-dose group: MMC 3 mg/kg/week/2 weeks and SB203580 5 mg/kg/week/3 weeks, i.p. One hour before MMC challenge, rats were injected with SB203580. All the rats were examined using echocardiography and right heart catheterization at 26 days after the first injection. Right ventricle and pulmonary artery function were assessed, and lung and heart tissues were collected.
Histological Stains
Heart and lung tissues of rats and lung tissues of human PVOD patients were fixed by 4% paraformaldehyde (PFA) for 48 hours. The tissues were processed through paraffin embedding and then were sectioned in 5 µm thickness. The hematoxylin-eosin (HE) stain and masson stain were performed using HE and masson stain kits (Beijing Solarbio Science & Technology Co., China) following the instructions provided with the kit. Images were analyzed by the NDP View 2 software (Hamamatsu photonics K.K., Japan).
Western Blotting
Total protein concentration was determined using the BCA assay kit (ThermoFisher Scientific, USA). 40 µg total protein was loaded in 12% SDS-PAGE gels and were transferred to PVDF membranes (Millipore, USA). Next, the membranes were blocked by 5% fat-free milk in TBS containing 0.1% Tween 20 (TBST) for 1 hour at room temperature (RT) and incubated with primary antibody overnight at 4 °C. Later, the membranes were washed and incubated with HRP-conjugated secondary antibody for 1 hour at RT. Targeted protein was detected with ECL reagents (Millipore, USA) in the end.
Primary antibodies used in the assay were purchased either from Cell Signaling Technology: GCN2 (3302), Phospho-eIF2α (Ser51) (3398), p38 MAPK (9212), Phospho-p38 MAPK (Thr180/Tyr182) (4511), Phospho-mTOR (Ser2448) (5536), AKT (4691), Phospho-AKT (Thr308) (13038), Phospho-AKT (Ser473) (4060), Phospho-p44/42 MAPK (Erk1/2) (Thr202/Tyr204) (4376), Phospho-SAPK/JNK (Thr183/Tyr185) (9251); from TransGen Biotech: GAPDH (HC301); from Sigma: α-Tubulin (T8203), or from Novus Biologicals: ATF3 (NBP1-85816).
Immunofluorescence
Cells were fixed with 4% PFA for 20 min and permeabilized with PBS containing 0.3% Triton X-100 for 15 min at RT. After blocking with PBS containing 3% donkey serum, primary antibody was incubated at 4 °C overnight. Next, anti-rabbit Alexa Fluor 594 and anti-mouse Alexa Fluor 488 (ThermoFisher Scientific, USA) were incubated for 1 hour at RT. Nuclei were stained with DAPI reagent for 8 min at RT. Images were captured by an Olympus confocal microscope. Primary antibodies used in the assay were purchased either from Cell Signaling Technology: Nanog (3580); from Abcam: Sox2 (ab97959), Oct4 (ab18976), SM22-α (ab14106); from Sigma: Calponin (C2687), or from Abclonal: MYL9 (A3039).
Quantitative Real-Time PCR (qRT-PCR)
Total RNA was extracted by TRIzol reagent (Invitrogen, USA). One microgram total RNAs were reverse transcribed to cDNA using PrimeScript RT reagent Kit with gDNA Eraser (Takara, Dalian, China) according to the manufacturer’s instructions. One microliter of cDNA was mixed with components supplied by TransStart Tip Green qPCR SuperMix (TransGen Biotech). The CFX ConnectTM Real-Time PCR Detection System (Bio-Rad, USA) and CFX Manager Software were used to analyze the mRNA expression levels of targeted genes. The mRNA expressions of target gene were normalized to β-actin mRNA level. All qPCR primers are listed in Table 1.
Primers for Quantitative Real-Time Polymerase Chain Reaction.

Abbreviations: fwd, forward; rev, reverse.
Reverse Transcription PCR (RT-PCR)
Total RNA was extracted from iPSCs and reversely transcribed as described above. The cDNA as a template, PCR analysis was performed using Golden Star T6 Super PCR Mix (TSINGKE) under standard conditions (95 °C, 5 min, 1 cycle; 95 °C 45 s, 60 °C 30 s, 72 °C 1 min, 29 cycles; 72 °C 5 min, 1 cycles; 4 °C hold). All PCR primers were listed in Table 2.
Primers for Reverse Transcription Polymerase Chain Reaction.

Abbreviations: fwd, forward; rev, reverse.
Genome Editing With epiCRISPR System
We selected 2 EIF2AK4 mutation sites, c.4414_4417del and c.2458C>T, as mutant targets. The gRNAs were designed using online “CRISPR Design” software (https://zlab.bio/guide-design-resources). The gRNA1, gRNA2 and gRNA3 were designed for mutation site c.4414_4417del. The gRNA4, gRNA5 and gRNA6 were designed for mutation site c.2458C>T. The gRNAs were constructed into epiCRISPR vector digested with BspQI (NEB, USA), obtaining epiCRISPR-gRNA. Plasmid epiCRISPR-gRNA3 and epiCRISPR-gRNA6 were verified the higher editing efficiency in 293 T using T7 endonuclease I (NEB, USA).
hiPSC-B1 was maintained in Essential 8TM basal medium (Gibco, Burlington, Canada) and subcultured using 0.5 mM EDTA digestion. 10 µg of the epiCRISPR-gRNA3 and epiCRISPR-gRNA6 were transfected into hiPSC-B1 using P3 Primary Cell 4D-Nucleofector™ X Kit (Lonza, Switzerland) following the manufacturer’s protocol, respectively. The gRNA targeting sites were PCR-amplified by Golden Star T6 Super PCR Mix (TSINGKE). The PCR products were sequenced by gRNA123 or gRNA456. All primers are listed in Table 3.
Primers for Construction epiCRISPR-gRNA Plasmids and Sequencing.

Abbreviations: fwd, forward; rev, reverse.
a The underline indicates the BspQI cleavage site.
iPSCs Cell Generation
Peripheral blood mononuclear cells (PBMCs) of PVOD patient were reprogrammed by the integration-free CytoTuneTM-iPS 2.0 Sendai Reprogramming kit (ThermoFisher Scientific, USA). The calculated volumes of each of the 3 CytoTune 2.0 Sendai virus (SeV) vectors were added to the cells with Multiplicity of Infection (MOI) of 10-10-8 (KOS MOI = 10, hc-Myc MOI = 10, hKlf4 MOI = 8). The cells were cultured in incubator for 24 hours before being replaced with fresh medium to remove the SeV vectors. On day 3 post-transduction, the cells were plated in 6-well plate coated with mice embryonic fibroblasts CF-1. At least 20 days after transduction, colonies with embryonic stem cell-like were picked and seeded into 96-well plate.
iPSCs colonies were cultured with feeder free in Essential 8TM basal medium.
Echocardiography
Rats were anesthetized by inhaled isoflurane and subjected to transthoracic echo by VisualSonics Vevo 2100 (VisualSonics Inc., Toronto, ON, Canada). Indicators of right ventricular and pulmonary arteries function, including tricuspid annular plane systolic excursion (TAPSE), cardio output (CO), right ventricle ejection fraction (RVEF), pulmonary vessel peak velocity, were measured in 2-dimensional M-mode echocardiograms.
Right Heart Catheterization and Hemodynamic Measurement
Rats were anesthetized by intraperitoneal injection of 12% urethane (100 g weight/ml) before catheterization. A Millar pressure transducer catheter was inserted in the right jugular vein. Right ventricular systolic pressure (RVSP) was monitored by a BL-420 S physiological experiment system. The heart was divided into 2 part, right ventricle (RV), left ventricle (LV) plus interventricular septum (LV+S). The index of right ventricle hypertrophy, the weight ratio of RV divided by (LV+S), was measured.
Statistical Analysis
The western blotting was quantified by Image J (National Institutes of Health, USA). Differences were analyzed using t-test, 1-way or 2-way ANOVA. Statistical calculations and graphs were completed by GraphPad Prism Version 8.0 (GraphPad Software Inc., San Diego, CA, USA). All the experiments were repeated for more than 3 times. Data were presented as means ± SEM. P < 0.05 was considered statistically significant.
Results
Deficient GCN2 Caused Pulmonary Vascular Remodeling in PVOD Patient
Using whole exome sequencing, we detected 3 types of biallelic mutation of EIF2AK4, which were not reported yet, in the 3 PVOD patients (Table 4). The premature of a termination codon led to the GCN2 encoded by EIF2AK4 unable to synthesize normally. Compared with the donors, the GCN2 expression was significantly reduced in the PVOD patients, which was consistent with the results of whole exome sequencing (Figure 1A). In addition, we also found that the medial layer of pulmonary artery and vein in PVOD patient were severely thickened, resulting lumen area reduction via HE staining (Figure 1B). HE staining showed that both pulmonary artery and vein had pathological features of vascular remodeling in PVOD patient with EIF2AK4 mutation, which was consistent with Esther et al. 11 Blue-dye collagen was significantly increased in pulmonary artery and vein wall of PVOD patient, as well as collagen deposition increased (Figure 1C).
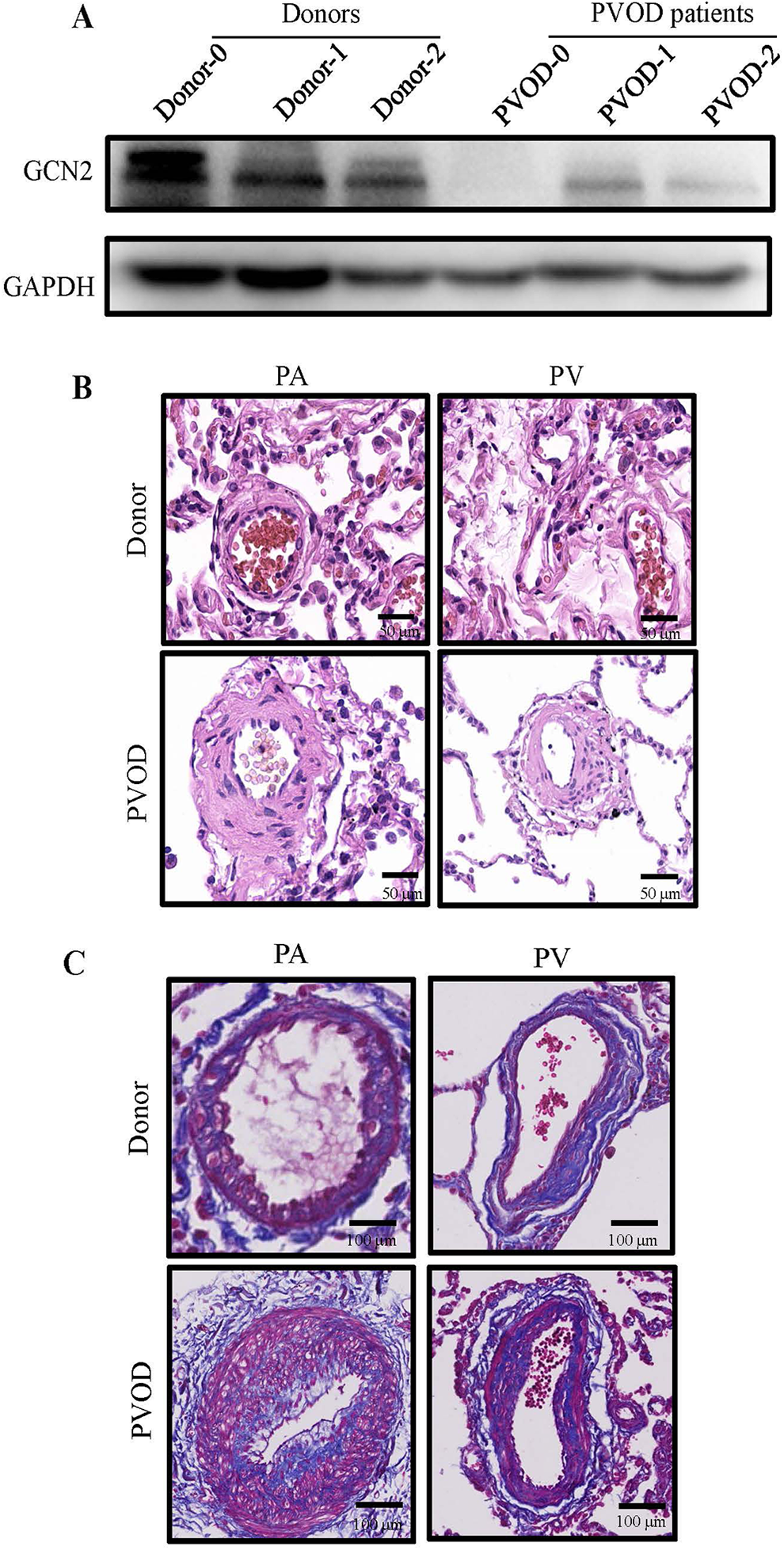
Deficient GCN2 was associated with pulmonary vascular remodeling in PVOD patients. A, Expression of GCN2 in lung tissues from donors and PVOD patients. B, HE staining of donor and PVOD patient. C, Masson’s trichrome staining of donor and PVOD patient. PA, pulmonary artery; PV, pulmonary vein.
Whole Exome Sequencing of EIF2AK4 in 3 PVOD Patients.

Abbreviations: H-PVOD, heritable pulmonary veno-occlusive disease; S-PVOD, sporadic pulmonary veno-occlusive disease.
a GenBank reference sequence number of EIF2AK4 gene is NM_001013703.3.
Detection of GCN2-ATF3-p38 MAPK Signaling Pathways Associated With PVOD
It has been reported that GCN2 could phosphorylates the α-subunit of eukaryotic translation initiation factor 2 (eIF2α), which is involved in inhibiting global protein synthesis and regulating activating transcription factor (ATF) (Supplementary Figure 1A). 2,12 Smooth muscle cell markers, calponin, smooth muscle 22-α and myosin light chain 9 (MYL9), were highly expressed in PASMCs from both donor and PVOD patient (Supplementary Figure 2). We found that the relative expression of ATF3 increased with the increasing salubrinal concentration in PASMCs of donor (Supplementary Figure 1B). The expression levels of GCN2 and ATF3 strongly decreased, but only the expression levels of p-p38 MAPK was significantly increased, indicating that p38 MAPK may be downstream effectors of ATF3 in PASMCs of PVOD patient (Figure 2A). Meanwhile, we noticed that the expression level of p-eIF2α had no significant difference between the PASMCs of donor and PVOD patient. It may be that GCN2 deficiency has pathological effects independently of eIF2α as it has already been shown recently by Manaud et al. 4

Detection of GCN2-ATF3-p38 MAPK signaling pathways in PVOD. A, Proteins leves of GCN2, p-elF2α, ATF3 and p-p38 in PASMC’s from both donor and PVOD patient. B, Expression of proteins associated with mTOR, AKT, ERK or JNK signaling pathways. C, The mRNA expression levels of GCN2, ATF3, collal and colla2 of PASMCS from both donor and PVOD patient. D, PASMCs of donor and PVOD patient were treated with 10 µM salubrinal (an inhibitor of dephosphorylation of phospho-elF2a) and 30 µM SB203580 (a specific inhibitor of p38) for 48 or 72 h to detect the mRNA level of collal and colla2. E, GCN2-ATF3-p38 MAPK signaling pathway. EIF2AK4 mutation induces decreased GCN2 expression and leads to an enhanced transcription of collagen I gene (collal and colla2) in PASMC’s through decreased ATF3-dependent p38 phosphorylation inhibition. Data is relative to mRNA expression level of ß-actin. n-3, P < 0.05, **P < 0.01, *** P < 0.001 and **** P < 0.0001. CN, control; sal, salubrinal; SB, SB203580.
Additionally, it was no significant difference in the expression levels of p-mTOR, p-AKT(Thr308), p-AKT(Ser473), p-ERK1/2 and p-JNK in PASMCs of PVOD patient compared with donor (Figure 2B). In further study, we also found that the mRNA levels of collagen I (col1a1 and col1a2) were notably enhanced in PASMCs of PVOD patient (Figure 2C). The mRNA levels of col1a1 and col1a2 in PASMCs of both donor and PVOD patient were significantly reduced after 48 or 72 h stimulation by 10 µm salubrinal and 30 µm SB203580, and it’s always higher in PVOD patient (Figure 2D). The mRNA level of ATF3 was notably increased after 48 or 72 h stimulation by 10 µm salubrinal in PASMCs of both donor and PVOD patient (Supplementary Figure 1C). The expression levels of p-eIF2α and ATF3 were enhanced in PASMCs of both donors and PVOD patients by western blot analysis, but p-p38 was decreased (Supplementary Figure 3).
Therefore, we hypothesized that reduced GCN2 by EIF2AK4 mutation increased the transcription of collagen I gene (col1a1 and col1a2) in PASMCs of PVOD patient by decreasing ATF3-dependent p38 phosphorylation inhibition, which promoted the collagen I synthesis in PASMCs (Figure 2).
GCN2 Knockout Cell Lines Were Successfully Constructed by epiCRISPR System in hiPSC-B1
We chose 2 mutation site, an unreported mutation site c.4414_4417del (exon 33 mutation) and another reported mutation site c.2458C>T (exon 15 mutation), were as the mutation targets of EIF2AK4. GCN2 knockout (KO) cell lines, GCN2E1479X/E1479X and GCN2L1478X/L1478X (exon 33 mutation), GCN2D834X/G835X and GCN2R830X/R830X (exon 15 mutation), were successfully gained by epiCRISPR system in hiPSC-B1. Compared to wild allele, the 1479th glutamate mutated into a stop codon, which was caused by GCGTG deletion in EIF2AK4 of GCN2E1479X/E1479X mutant (Figure 3A, Middle panel). The loss of GAAGCGTG led to the mutation of the 1478th leucine into a termination codon in EIF2AK4 of GCN2L1478X/L1478X mutant (Figure 3A, Lower panel). Both of these 2 mutants were homozygous biallelic mutation of EIF2AK4. Two strands of DNA had different mutation in the GCN2D834X/G835X mutant. The absence of CTCTGGA in one strand of double-stranded DNA caused 834th aspartate mutation, which lead to early termination of GCN2 translation. Meanwhile, another strand had the loss of TCTG, resulting that 835th glycine mutated into a termination codon (Figure 3B, Middle panel). The lack of 19 bases led to nonsense mutation in the GCN2R830X/R830X mutant (Figure 3B, Lower panel). Moreover, the GCN2D834X/G835X mutant was heterozygous biallelic mutation of EIF2AK4, but the GCN2R830X/R830X mutant was homozygote.
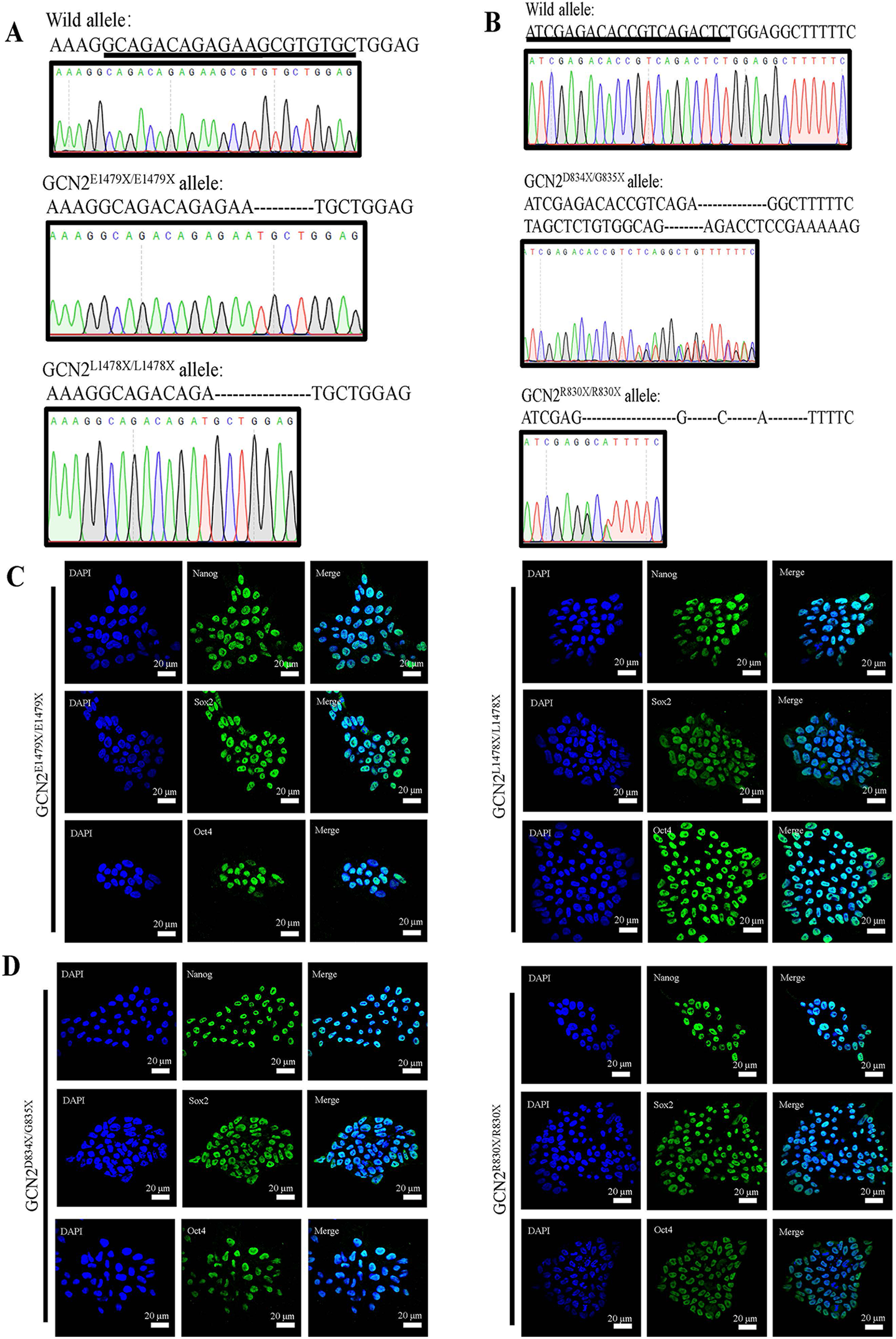
Construction of GCN2 KO cell lines using epiCRISPR system in hiPSC-B1. A and B, The sequencing results of EIF2AK4 exon 33 (GCN2E1479X/E1479X and GCN2L1478X/11478X) and exon 15 (GCN2D834X/G835X and GCN2R830X/R830X) of hiPSC-B1 via epiCRISPR system. PCR amplification and sequencing were performed on the sequence around the gRNA target area. The underline is gRNA and the short black lines represent deleted base. C and D, Immunofluorescence analysis of pluripotency markers (Oct4, Nanog and Sox2, green) in GCN2 KO cell lines. DAPI, blue.
We found that the pluripotent markers, Nanog, Sox2 and Oct4, were markedly expression in all GCN2 KO cell lines, showing that these cell lines have unaffected pluripotency (Figure 3C and D). Expression of GCN2 was lost in all GCN2 KO cell lines by western blot analysis (Figure 4D).

iPSCs were generated by reprogramming PBMCs of PVOD-0 patient. A, The change of cell morphology during reprogramming. (a) Mice mbryonic fibroblasts CF-1. (b) Small colony emerged on day 20 after Sendai virus vectors infection. (c) iPSCs grown on feeder free. B, The Sendai virus genome and transgenes of wildtype hiPSC-BI and iPSCs of PVOD patient (PVOD-0-iPS-C1 and PVOD-0-iPS-C2) were detected by RT-PCR. (virus)-SeV, 181 bp; (virus)-KOS, 528 bp; (virus)-Klf4, 410 bp; (virus)-c-Myc, 532 bp; Oct4, 144 bp; Sox2, 151 bp; Klf4, 220 bp; c-Myc, 68 bp. Mr indicates 2000 DNA Marker, from top to bottom were 2000 bp, 1000 bp, 750 bp, 500 bp, 250 bp, 100 bp. C, The expression of the pluripotent markers Nanog (green), Sox2 (green) and Oct4 (green) in iPSCs of PVOD patient (PVOD-0-iPS-C1 and PVOD-0-iPS-C2) were detected by immunofluorescence. DAPI, blue. D, Analysis of expression of GCN2 among 0202-iPS, hiPSC-B1, GCN2 cell lines and iPSCs of PVOD patient by western blotting. 0202-iPS and hiPSC-B1 were normal expressed GCN2 controls. n = 3, **** P < 0.0001 v.s. hiPSC-BI.
iPSCs From PBMCs of PVOD Patient Were Obtained by Reprogramming
In this study, we well generated 2 iPSCs (PVOD-0-iPS-C1 and PVOD-0-iPS-C2) from PVOD-0 patient with EIF2AK4 mutation, using non-integrating Sendai viral vectors expressing KOS, Klf4 and c-Myc (Figure 4A).
The iPSCs cultured in feeder free with typical morphology of pluripotent stem cells. Compared to wildtype hiPSC-B1, we discovered that PVOD-0-iPS-C1 and PVOD-0-iPS-C2 were completely free of SeV, KOS, Klf4 and c-Myc carried by Sendai virus after passage 6 and 10, respectively (Figure 4B). Furthermore, the endogenous pluripotent gene Oct4, Sox2, Klf4 and c-Myc were still existed in PVOD-0-iPS-C1 and PVOD-0-iPS-C2 after passage 6 and 10, respectively.
Immunofluorescence analysis showed that the pluripotent markers, Nanog, Sox2 and Oct4, were highly expressed in the PVOD-0-iPS-C1 and PVOD-0-iPS-C2 (Figure 4C). It was shown that the reprogrammed iPSCs have the pluripotency of stem cells. PVOD-0-iPS-C1 and PVOD-0-iPS-C2 lost expression of GCN2 in comparison with normal expressed GCN2 controls, 0202-iPS and hiPSC-B1 (Figure 4D).
Enhancement of the Expression of Collagen I in LM-SMCs Differentiated From GCN2 KO Cell Lines and iPSCs
hPSCs, namely GCN2 KO cell lines and iPSCs were differentiated into LM-SMCs according to the protocol published by Christine et al. 13 hPSCs had the ability to differentiate into LM-SMCs in vitro (Figure 5A). The brachyury (encoded by T) expression level was obviously increased, while the Nanog expression level was decreased significantly on day 1.5 (Figure 5B). It was shown that after 1.5 days the cells were in the early mesoderm. The expression of lateral plate mesoderm markers containing KDR, NKX2-5 and ISL1 were raised clearly on day 5 compared with on day 0 (Figure 5B). It was revealed that the cells from early mesoderm entered lateral plate mesoderm. The expression of calponin, SM22-α and myosin heavy chain 11 (Myh11), which represent smooth muscle cells markers, were greatly enhanced on day 17 (Figure 5B). hPSCs were successfully differentiated into LM-SMCs after 17 days of differentiation.

The expression of collagen I was regulated by GCN2 in LM-SMCs lines. A, The change of cell morphology during hPSC differentiated into LM-SMCs in vitro. (a) Small colonies of hPSC emerged before differentiation; (b) colonies of early mesoderm were observed after 1.5 days of treatment with FGF2, LY294002 and BMP4; (c) cell populations were observed during early mesoderm to lateral plate mesoderm differentiation; (d) 1 day after splitting of lateral plate mesoderm into medium including PDGF-BB and TGF-B1; (e) LM-SMCs were obtained after 12 days of treatment with PDGF-BB and TGF-B1. B, Analysis of relative gene expression of positive cell markers during differentiation. Nanog is one of pluripotent markers. T is a early mesoderm marker. KDR, NKX2-5 and ISL1 are lateral plate mesoderm markers. Calponin, SM22-a and Myh11 represent smooth smucle cells markers. C, The expression of LM-SMCs specific markers, calponin (red) and SM22-a (green), were detected by immunofluorescence. The negative control hiPSC-B1 was used. DAPI, blue. D, The mRNA expression levels of GCN2 and collal of LM-SMCs lines differentiated from 0202-iPS, GCN2E1479X/E1479X and PVOD-0-iPS-C2. Data is relative to mRNA expression level of ß-actin. n-3, P < 0.05 and **P < 0.01.
To further confirm the successful differentiation, we identified the expression of calponin and SM22-α by immunofluorescence staining. The result displayed that calponin and SM22-α were expressed strongly after 17 days of differentiation (Figure 5C). It indicated that hPSCs had the ability to differentiate into mesoderm. We further discovered that the mRNA level of GCN2 had decreased, but the mRNA level of col1a1 had increased in GCN2E1479X/E1479X and PVOD-0-iPS-C2 compared with 0202-iPS (Figure 5D).
SB203580 Improved the Right Ventricular and Pulmonary Vascular Function in MMC-Induced PVOD Rats
We found that GCN2 upregulated collagen I expression via ATF3-dependent p38 phosphorylation inhibition, leading to increased collagen deposition in pulmonary vascular wall of PVOD patient. To examine whether SB203580 (a specific inhibitor of p38) improved the pathological features of PVOD, we conducted a study in PVOD rat model. It has been reported that MMC could induce PVOD in mice, rats and rabbits, but the rats are more sensitive to the same dose of MMC. Moreover, the histological and hemodynamic characteristics of MMC-induced rats were similar to human PVOD. 9,10
There were significant medial hypertrophy and luminal stenosis in pulmonary artery and vein from model group compared with control group, but not in both low-dose group and high-dose group (Figure 6A). Tricuspid annular plane systolic excursion (TAPSE) significantly diminished in model group but not in both low-dose group and high-dose group (Figure 6B). Pulmonary vessel peak velocity (PV peak velocity) is one of the indexes to evaluate pulmonary vessels function. PV peak velocity was elevated obviously in model group, however, there was no difference in either low-dose group or high-dose group compared to control group (Supplementary Figure 4).
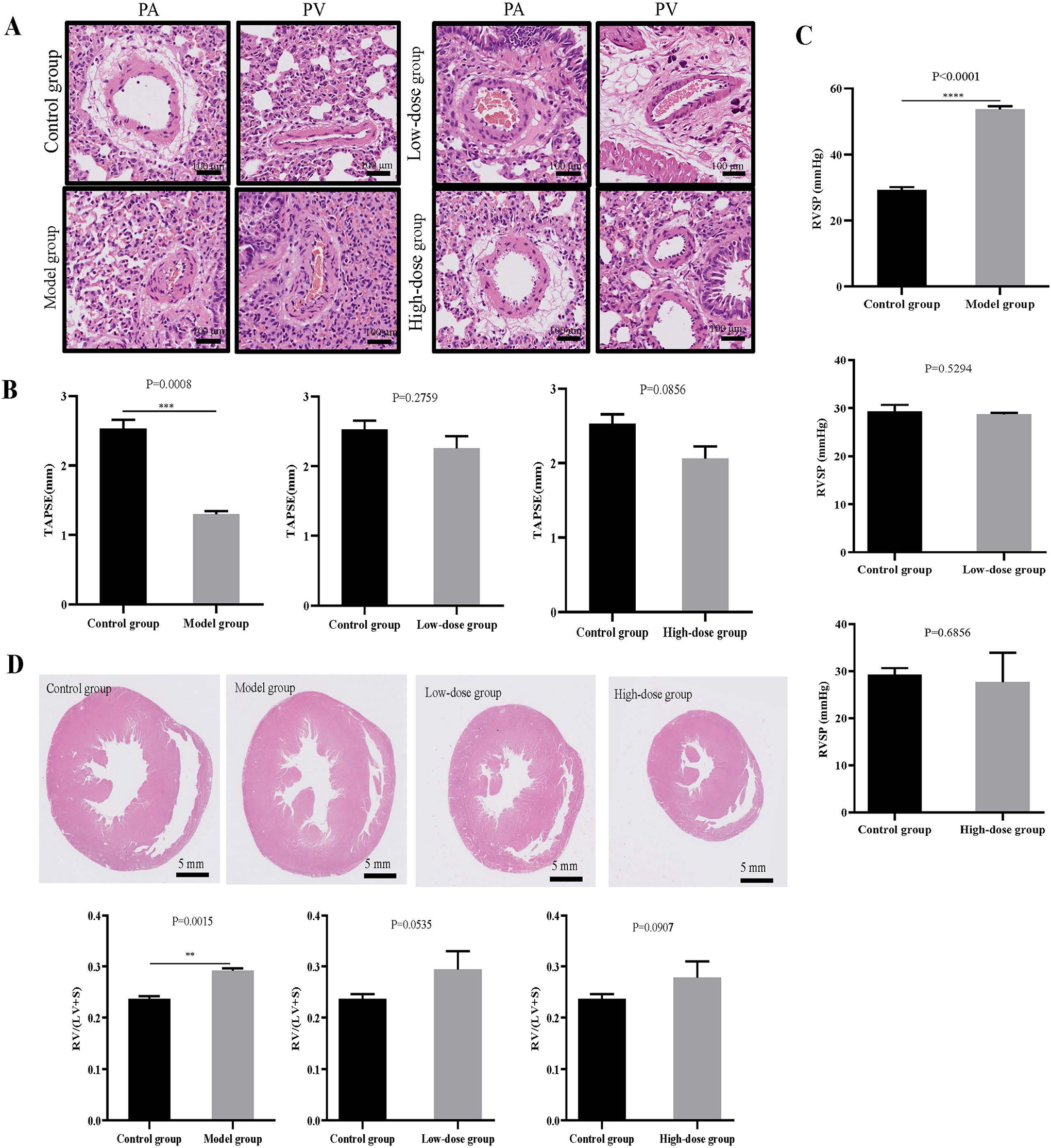
SB203580 improved the right ventricular and pulmonary vascular function in MMC-induced PVOD rats. A, HE staining of lung tissues in rats. B, Measurement of TAPSE in rats. C, Measurement of RVSP in rats. D, HE staining of heart tissues and determination the index of right ventricular hypertrophy (RV/(LV+S) in rats. n-6 in each group. **P < 0.01, ***P < 0.001, and ****P < 0.0001. PA, pulmonary artery; PV, pulmonary vein; TAPSE, tricuspid annular plane systolic excursion; RVSP, right ventricular systolic pressure.
Cardio output (CO) and right ventricle ejection fraction (RVEF) are the main indicators to assess whether the contraction of the heart is normal. In model group, CO and RVEF were reduced evidently (Table 5). Furthermore, there was no significant difference in CO and RVEF of low-dose group and high-dose group in comparison with control. A remarkable raised right ventricular systolic pressure (RVSP) was noticed in model group, more importantly, RVSP values of both low-dose group and high-dose group were close to control group (Figure 6C). Significant right ventricular hypertrophy was observed in model group, but did not exist in low-dose group and high-dose group (Figure 6D).
Measurement of CO and RVEF in Rats.

Abbreviations: CO, cardio output; RVEF, right ventricle ejection fraction.
*P < 0.05 and **P < 0.01, relative to control. n = 6 in each group.
Discussion
For the past few years, the intracellular signaling pathway and the expression of downstream target genes have been considered as the key molecular mechanisms for the development of PAH. 14,15 The expression of transcription factors plays an increasingly important role in the development of PAH. 16 -18 ATF3 is an important transcription factor in cardiovascular diseases, it play roles in cardiac hypertrophy, heart failure, atherosclerosis, ischemic heart disease, diabetes mellitus and hypertension. 19
In the cardiac fibroblasts of ATF3 KO mice, p38 was activated instead of JNK or ERK1/2, and ATF3 improved hypertensive ventricular remodeling, heart failure and cardiac fibrosis by suppressing MAP2K3-p38 signaling. 20 Further more, when angiotensin II was infused into wild-type and ATF3 KO mice, the collagen deposition in ATF3 KO mice was significantly increased compared with wild-type mice, and the mRNA levels of collagen I and TGF-β in ATF3 KO mice were also increased. In human retinal pigment epithelial cells ARPE-19, TGF-β2 upregulates the expression of collagen I gene (col1a1 and col1a2) by activating the p38 signaling pathway, leading to the synthesis of extracellular matrix protein (ECM), and suggesting that MAPK signaling pathway may be a new therapeutic target for many ocular diseases. 21
In the present study, we have focused on roles and effects of GCN2-ATF3-p38 MAPK signaling pathway in PVOD. We discovered that the decreased ATF3 only affected the expression of p38 MAPK, not mTOR or AKT or ERK1/2 or JNK, indicating that the p38 MAPK signaling pathway may play an important role in PASMCs of PVOD patients. In addition, we also found that the p-p38 protein level decreased when the increase of the ATF3 protein level in PASMCs of PVOD patients. It implied that a negative correlation between ATF3 and p38 MAPK. The mRNA level of collagen I were decreased after stimulation by salubrinal and SB203580 in PASMCs of PVOD patients. It demonstrated that ATF3 and p-p38 had the capacity to regulate expression of collagen I. Taken together, these results suggest decreased GCN2 by EIF2AK4 mutation enhances the mRNA levels of collagen I by decreasing ATF3-dependent p38 phosphorylation inhibition, which promotes the collagen I synthesis in PASMCs and eventually leads to increased collagen deposition in pulmonary vascular wall.
Because PVOD is a rare form of pulmonary hypertension, few PVOD patient samples are available. hPSCs containing EIF2AK4 mutation solve the difficulty of obtaining PVOD patient samples to some extent. hPSCs obtained in this study could still maintain the pluripotency after several passages, and the expression of GCN2 was significantly decreased. The cardiovascular system is developed from the mesoderm, therefore, we determined to hPSCs differentiate into LM-SMCs in vitro. Both qRT-PCR and immunofluorescence showed that hPSCs had the ability to differentiate into LM-SMCs successfully. It verified that GCN2 dysfunction led to greatly increased expression of collagen I in LM-SMCs lines differentiated from GCN2 KO cell lines and iPSCs, which the same as PASMCs of PVOD patients.
SB203580, a pyridinyl-imidazole compound, specifically inhibits the activity of p38 MAPK by binding to the ATP pocket. However, SB203580 does not block the phosphorylation of p38 MAPK itself. 22,23 P2X7 receptor is involved in the development of PAH through the p38 MAPK signaling pathway, and SB203580 could reverse P2X7 receptor mediated lung injuries in PAH rats. 24 In non-obese type 2 diabetic Goto-Kakizaki rats, metformin or SB203580 can significantly reduce the size of myocardial infarction, and the combination of metformin and SB203580 can enhance the cardioprotection. 25
In this study, we examined right ventricle and pulmonary artery hemodynamics and morphological parameters of rats from control and experiment groups. We discovered that low- or high-dose SB203580 ameliorated pulmonary vascular remodeling and reduced pulmonary vascular lesion of PVOD. What’s more, low- or high-dose SB203580 significantly improved right ventricle and pulmonary vascular function of MMC-induced PVOD in rats. These results support the idea that SB203580 shows positive preventive effect on right ventricle and pulmonary artery in PVOD.
In summary, we report the finding that deficient GCN2 by EIF2AK4 mutation enhances the mRNA levels of collagen I by decreasing ATF3-dependent p38 phosphorylation, which promotes the collagen I synthesis in PASMCs and eventually leads to increased collagen deposition in pulmonary artery wall. It was also confirmed that GCN2 dysfunction led to enhanced expression of collagen I in LM-SMCs lines differentiated from GCN2 KO cell lines and iPSCs. In addition, SB203580 may be a promising therapeutic agent that can be used against PVOD.
Supplemental Material
Supplemental Material, sj-jpg-1-cpt-10.1177_10742484211015535 - GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease
Supplemental Material, sj-jpg-1-cpt-10.1177_10742484211015535 for GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease by Zhongqiu Chen, Jingyuan Zhang, Dong Wei, Jingyu Chen and Jun Yang in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-2-cpt-10.1177_10742484211015535 - GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease
Supplemental Material, sj-jpg-2-cpt-10.1177_10742484211015535 for GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease by Zhongqiu Chen, Jingyuan Zhang, Dong Wei, Jingyu Chen and Jun Yang in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-3-cpt-10.1177_10742484211015535 - GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease
Supplemental Material, sj-jpg-3-cpt-10.1177_10742484211015535 for GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease by Zhongqiu Chen, Jingyuan Zhang, Dong Wei, Jingyu Chen and Jun Yang in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-4-cpt-10.1177_10742484211015535 - GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease
Supplemental Material, sj-jpg-4-cpt-10.1177_10742484211015535 for GCN2 Regulates ATF3-p38 MAPK Signaling Transduction in Pulmonary Veno-Occlusive Disease by Zhongqiu Chen, Jingyuan Zhang, Dong Wei, Jingyu Chen and Jun Yang in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
We thank for the epiCRISPR plasmid donated by Professor YM. Wang (Fudan University, Shanghai, China). We would like to thank Lihong Sun of the animal center, Institute of Basic Medical Sciences, Chinese Academy of Medical Science and Peking Union Medical College for her help.
Author Contributions
ZC, JC, and JY designed the research study; DW and JC provided human samples; ZC participated in data collection and analysis; JZ and JY revised and finalized the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Program of China-stem cell and translational research [grant number 2016YFA0102300]; and the Nature Science Foundation of China [grant number 81870051].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
