Abstract
Dyslipidemia, particularly increased low-density lipoprotein cholesterol (LDL-C) levels, are associated with an increased risk of atherosclerotic cardiovascular disease (ASCVD) events. There is an unmet need for ASCVD risk reduction even with the optimal use of statin therapy, which has led to an ongoing search for novel targets for cholesterol reduction to decrease ASCVD events.
Objectives:
The main aim of this review was to summarize current evidence on approved and emerging non-statin lipid-lowering therapies.
Methods and Materials:
Recent literature on U.S. FDA approved non-statin lipid-lowering therapies and evolving lipid-lowering drugs currently under development was reviewed.
Results and Discussion:
In the past 20 years, the emergence of non-statin cholesterol-lowering drugs has changed the landscape of dyslipidemia management. Food and Drug Administration approval of non-statin lipid-lowering therapies such as ezetimibe, proprotein convertase subtilisin/Kexin type 9 (PCSK9) inhibitors (evolocumab, alirocumab), bempedoic acid and combination of bempedoic acid and ezetimibe, evinacumab and other triglyceride-lowering agents (eg, icosapent ethyl) has emerged. The European Commission has also recently approved inclisiran for treatment of hypercholesterolemia and mixed hypercholesterolemia even though FDA has put the approval of this drug on hold. Recent guidelines have incorporated PCSK9 inhibitors to treat patients with primary hyperlipidemia and patients with very high-risk ASCVD, who could not achieve adequate lipid-lowering with combination therapy of maximally tolerated statin and ezetimibe. Icosapent ethyl use as an adjunct therapy to statins is also recommended to reduce the risk of ASCVD in patients with hypertriglyceridemia.
Conclusion:
Despite cost limitations, the uptake of PCSK9 inhibitors is increasing. Approval of bempedoic acid alone or in combination with ezetimibe has provided additional oral lipid-lowering drug alternatives to ezetimibe. Various lipid-lowering drug targets are under investigation.
Introduction
Lipids are vital biomolecules, but any biomolecule in excess is detrimental to human health. Elevated levels of lipids; hyperlipidemia poses a constant threat to health. In particular, coronary arteries are most vulnerable to hyperlipidemia. 1 Cardiovascular disease is a leading cause of death and a major public health problem. 2 Increased low-density lipoprotein cholesterol (LDL-C), triglycerides (TGs) or Apolipoproteins, and low levels of high-density lipoprotein cholesterol (HDL-C) are associated with the development and progression of atherosclerotic lesions. Both elevated fasting and non-fasting triglycerides are linked with CVD. However, the link between TG and CVD is confounded by its inverse metabolic relationship with HDL-C and the heterogeneity of TG lipoproteins. It is recommended that diagnosis of hypertriglyceridemia should be made post 9-12 hrs of fasting. Although non-fasting TG levels better reflect the risk of CVD, the lack of standardization of non-fasting TG measurements, lack of specific reference ranges, and the variability of TG levels after food limits their routine clinical use. 3 Lowering LDL-C is crucial to reduce the risk of atherosclerotic cardiovascular disease (ASCVD) in primary and secondary prevention. 4 Lowering Apolipoprotein B (apo B), increasing levels of HDL-C, and lowering non-HDL-C levels can lower ASCVD risk. Recently apo B is emerging as a more reliable and more accurate marker to estimate treatment effectiveness of lipid-lowering therapy and ASCVD risk reduction than LDL-C. 5 Various risk factors such as old age, family history, and diabetes, high blood pressure, being overweight, physical inactivity, poor diet, and smoking are associated with an increased level of LDL-C. 4 There is an increased interest in the use of new classes of hypoglycemic drugs with cardioprotective effects in patients with diabetes. Glucagon-like peptide 1 receptor agonists (GLP-1 RA) and sodium-glucose transport protein 2 inhibitors (SGLT2i) have shown a favorable impact on blood glucose, body weight, blood pressure (BP), and the lipid profile. Due to their pleiotropic benefit, these drugs have been shown to reduce cardiovascular events and mortality. Hence, the American diabetic association recommends the use of these drugs early on in the management of diabetes to reduce the risk of CVD. 6
Identifying patients at the highest risk of ASCVD is essential to initiate or intensify preventive therapy. 7 Recent guidelines recommend personalizing lipid-lowering therapy based on lipid profile, ASCVD risk estimate, and the presence of additional ASCVD risk enhancers. 7 Based on risk estimates, patients need to undergo a therapeutic lifestyle change, use a statin, or add on a non-statin lipid-lowering agent (eg, ezetimibe) if there is inadequate control of LDL-C. The FDA has approved additional non-statin lipid-lowering therapies (eg, PCSK9 inhibitors, bempedoic acid, a combination of ezetimibe and bempedoic acid, icosapent ethyl, evinacumab) in the last decade. According to European Medical Agency, bempedoic acid is indicated in combination with a statin or statin with other lipid-lowering therapies in patients who fail to reach LDL-C goals on maximum tolerated doses of a statin and alone or in combination with other lipid-lowering therapies in patients who are statin-intolerant or who have a contraindication to statin therapy. The fixed-dose combination of bempedoic acid and ezetimibe has similar indications as bempedoic acid. Besides, the combination is indicated in patients who fail to achieve LDL-C goals despite using bempedoic acid and ezetimibe as separate tablets with or without a statin. In this paper, we present a summary of recent literature on FDA approved and evolving non-statin lipid-lowering therapy.
Review Questions
What are the available U.S. FDA approved non-statin lipid-lowering medications?
What are the non-statin lipid-lowering therapies currently under investigation?
Methods and Materials
Recent literature on U.S. FDA approved non-statin lipid-lowering therapies and evolving lipid-lowering drugs currently under development was reviewed. Google Scholar, PubMed, UpToDate, clinicaltrials.gov databases were searched to identify relevant literature and ongoing studies. Randomized controlled trials that described the advantage and disadvantages of non-statin lipid-lowering therapy and current guidelines that guide lipid-lowering therapies are included. Articles not written in English are excluded. To ensure up-to-date literature is summarized the articles published between 2011 and 2020 are included in this study. Subjects taking non-statin hypolipidemic agents for the management of primary hyperlipidemia or primary and secondary ASCVD prevention are included. Non-statin lipid-lowering therapy taken as a monotherapy compared with placebo or combination of statin and non-statin lipid-lowering drugs compared with statin therapy alone are summarized. The relevant outcomes are lipid-lowering potential, cardiovascular disease prevention, or effective management of hyperlipidemia using these agents and potential side effects associated with the non-statin agents. To identify relevant literature a combination of medical subject headings (MeSH) terms and text words were used. MeSH terms and text words used include “ezetimibe” OR “Omega-3 fatty acid,” OR “Fibrate” OR “Pemafibrate” OR “niacin” OR “Bile acid sequestrant,” OR “Proprotein convertase subtilisin/kexin,” OR “Cholesteryl ester transfer protein,” OR Lomitapide OR Mipomersen OR Bempedoic Acid OR Inclisiran OR Antisense apolipoprotein A oligonucleotide OR Gemcabene OR Evinacumab OR Non-statin AND lipid-lowering OR Major cardiovascular events OR adverse drug events. No statistical tests were used to summarize the data.
Results
Lipid-Lowering Strategies
Before initiation of any lipid-lowering strategy, there is a need to identify to classify the cause of dyslipidemia as primary or secondary. Secondary causes such as clinical or drug-related factors can be linked with elevated LDL-C, non-HDL-C, or triglycerides levels. Risk factors for increased cholesterol are multifactorial, so are lipid-lowering strategies. Non-pharmacological and pharmacological lipid-lowering strategies are equally important to lower cholesterol levels. Lifestyle strategies such as the cessation of smoking, dietary factors, weight loss, lowering of blood pressure or blood glucose levels, and regular exercise are critical aspects of lipid-lowering strategy for all patients. These can be used as first-line therapy for patients with low ASCVD risk. The current 2019 ACC/AHA guidelines emphasize the incorporation of lifestyle changes for the primary prevention of ASCVD. 3,5 Pharmacological strategies are needed in conjunction with lifestyle changes for patients with high ASCVD risk and patients with high-risk equivalent conditions such as diabetes and primary hypercholesterolemia. Lipid-lowering agents can be selected based on the underlying abnormalities in lipid profile, targets for LDL-C reduction, and patient acceptance. The classes of established lipid-lowering agents include statins, fibrates, bile acid sequestrants (resins), nicotinic acid derivatives, ezetimibe, and PCSK9 inhibitors. In patients where combination therapies of statin and non-statin lipid-lowering medications failed to control LDL-C, LDL apheresis, gene therapy, liver transplantation, and surgery may be considered as adjunct strategies.
Statin remains the first-line therapy for primary and secondary prevention of cardiovascular disease (CVD) due to its efficacy and long-term safety data. 4 Several clinical studies using statins have shown a significant reduction of mortality and cardiovascular events which led to recommendations that all patients having known vascular disease, or those who are at high risk for vascular disease, should be on statins. These drugs lower cholesterol by interfering with the cholesterol biosynthetic pathway by inhibiting 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase and thus suppressing cholesterol biosynthesis. Moderate-intensity statin therapy can reduce LDL-C by 30% to 50% while high-intensity therapy (eg. atorvastatin 40-80 mg OD and rosuvastatin 20-40 mg OD) reduces LDL-C by ≥ 50%. 2
Challenges of Lipid-Lowering With Statin
Despite the wide availability and use of statins, there is a significant gap between the expected and actual benefits of using these agents. One of the reasons is poor adherence to statin therapy. Non-adherence to statins is a significant issue for the prevention and treatment of cardiovascular disease. 8 Poor adherence is linked to an increase in cardiovascular events. Interventions such as improving awareness among patients, safer prescribing and a focus on the alliance between health care providers and the patients have improved adherence rates. However, even in the cases of optimal statin use, there is a residual risk of ASCVD. Statins reduce cardiovascular events by 20%-40%. 2 Besides, statin therapy may not adequately lower LDL-C in all patients to achieve target LDL-C levels. Furthermore, there are challenges associated with statin therapy such as an increased risk of drug interaction, statin intolerance, and altered levels of liver enzymes. 9 Statin intolerance encompasses a wide range of statin-related side effects including muscle symptoms, headache, sleep disorders, dyspepsia, nausea, rash, alopecia, erectile dysfunction, gynecomastia, and/or arthritis. Statin intolerance seems to occur in 10%-15% of patients. 10 Statin-associated muscle symptoms (SAMS) are one of the main reasons that promote statin non-adherence. 11 This suggests that non-statin therapies to reduce LDL-C by mechanisms that do not involve inhibition of HMG-CoA reductase are likely to be useful for patients requiring LDL-C reduction; particularly those who either cannot take statins or respond only partially or not at all to statins alone.
Non-Statin Lipid-Lowering Therapy
Although statins are currently the major hypolipidemic drugs, an increasing number of patients on this type of therapy have side effects or do not respond well to the therapy. Non-statin alternatives to address the epidemic of hyperlipidemia remain of vital importance. The addition of non-statin therapy allows further reduction of LDL-C, thereby reducing the chance of future cardiac issues or stroke. 1 The characteristics of randomized controlled trials of non-statin lipid-lowering therapies are indicated in Table 1, while the pros and cons of FDA-approved lipid-lowering medications are shown in Table 2.
Characteristics of Completed Randomized Clinical Trials and Ongoing Studies of Antihyperlipidemic Agents.
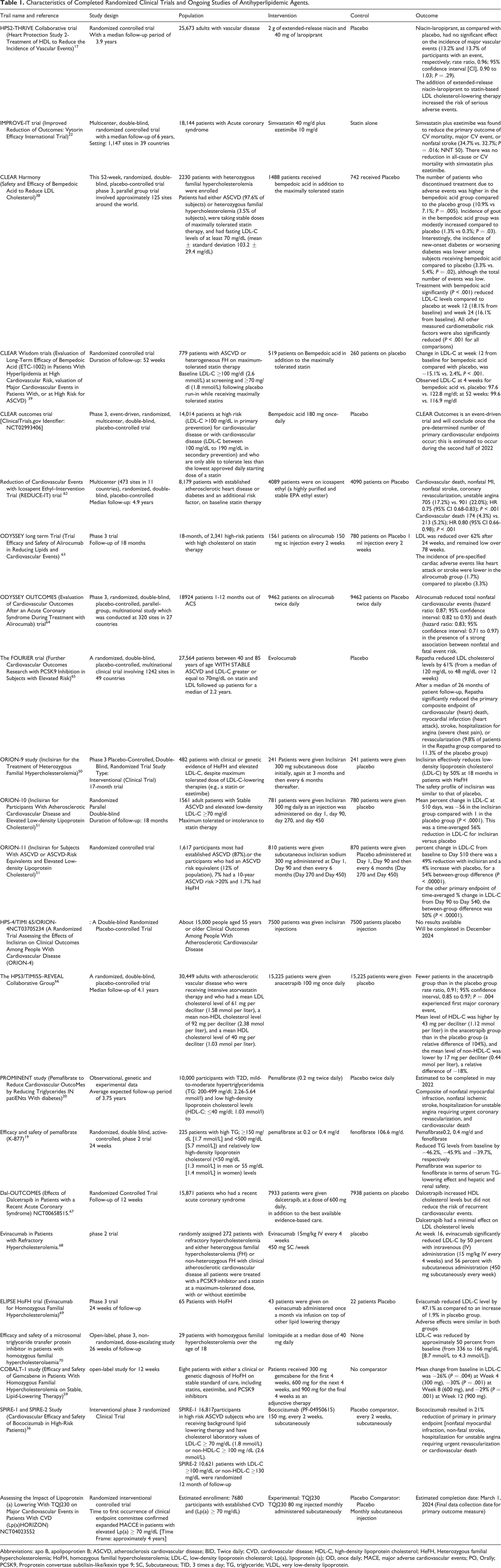
Abbreviations: apo B, apolipoprotien B; ASCVD, atherosclerosis cardiovascular disease; BID, Twice daily; CVD, cardiovascular disease; HDL-C, high-density lipoprotein cholesterol; HeFH, Heterozygous familial hypercholesterolemia; HoFH, homozygous familial hypercholesterolemia; LDL-C, low-density lipoprotein cholesterol; Lp(a), lipoprotein (a); OD, once daily; MACE, major adverse cardiovascular events; PO, Orally; PCSK9, Proprotein convertase subtilisin-like/kexin type 9; SC, Subcutaneous; TID, 3 times a day; TG, triglyceride; VLDL, very low-density lipoprotein.
FDA Approved Non-Statin Lipid-Lowering Medications.

Abbreviations: apo B, apolipoprotien B; ASCVD, atherosclerosis cardiovascular disease; BID, Twice daily; CV, cardiovascular; CVD, cardiovascular disease; EMA, European Medical Agency; HDL-C, high-density lipoprotein cholesterol; HeFH, Heterozygous familial hypercholesterolemia; HoFH, homozygous familial hypercholesterolemia; LDL-C, low-density lipoprotein cholesterol; Lp(a), lipoprotein (a); OD, once daily; PO, Orally; PCSK9, Proprotein convertase subtilisin-like/kexin type 9; SC, Subcutaneous; TID, 3 times a day; TG, triglyceride; VLDL, very low-density lipoprotein.
FDA Approved Non-Statin Lipid-Lowering Medications
Niacin, bile acid sequestrants, and fibrates
Niacin, bile acid sequestrants, or fibrates may be used as add-on therapy if the goals of LDL-C reduction are not met on the maximum tolerated doses of statins. However, these drugs are not routinely considered for primary or secondary prevention of CVD, because their use is linked with a high risk of adverse drug reactions. 1 Bile acid sequestrants (eg, cholestyramine, colestipol, colesevelam) are one of the oldest and generally considered safest lipid-lowering agents and have been used in patients with statin intolerance or statin contraindication. 12 The bile acid sequestrants are highly positively charged molecules that bind to the negatively charged bile acids in the intestine and block cholesterol absorption. 1 Fibrate can lower LDL-C, total cholesterol, triglycerides (by 20%-70%), apolipoprotein B (Apo-B), and increase high-density lipoprotein cholesterol (HDL). 13 Fibrates act via activation of a transcription factor, peroxisome proliferator-activated receptor (PPAR). 14 When used in combination with statins fibrates show no additional benefit in secondary prevention of CVD, 15 and in high-risk patients fibrates (e.g. gemfibrozil) increase the risk of muscle injury. 14 The combination of fibrates and statin is associated with muscle pain, myositis, and rhabdomyolysis. The risk of these adverse effects is significantly higher with gemfibrozil than bezafibrate and fenofibrate. Niacin raises HDL-C significantly and lowers lipoprotein (a) levels by an average of 25% and TGs by 15%-25%. 16 However, the use of niacin as add on therapy to a statin to prevent CVD is limited by the absence of conclusive evidence on improved clinical outcomes and poor tolerability. 16 The European Medical Agency in 2013 has recommended the suspension of niacin/laropiprant products worldwide after the failure of the HPS-2THRIVE study. 17 Drug-induced hyperuricemia and gout has been reported with nicotinic acid use. 18
Pemafibrate is a selective PPARα modulator (SPPARMα) modulator that reduces TG which is a risk factor for the development of CVD. Pemafibrate has a better benefit-risk profile than other fibrates. 19 Pemafibrate has an equivalent lipid-lowering capacity as fenofibrate but has superiority over fenofibrate because it can be safely taken in patients with renal dysfunction. However, more clinical data on the long-term efficacy and safety of pemafibrate is needed. 20
Ezetimibe
Ezetimibe reduces blood cholesterol by inhibiting the absorption of cholesterol by the small intestine by blocking Niemann-Pick C1-Like 1 (NPC1L1) protein on the small intestinal epithelial cells and hepatocytes. It also blocks aminopeptidase N and interrupts a caveolin 1-annexin A2 complex that is part of the cholesterol metabolic pathways. When added to statin ezetimibe reduces 21 LDL-C by 15%-20%. 22 The IMPROVE-IT trial based on data of 18,000 patients showed the clinical benefit of ezetimibe as an add-on therapy to a statin. In this trial patients who took ezetimibe plus simvastatin had a lower rate of cardiovascular-related death, myocardial infarction, or stroke (32.7%) than those who took statin alone (34.7%). 22 Caution should be exercised during the administration of ezetimibe and the dose of simvastatin exceeding 20 mg, in patients with moderate to severe renal impairment due to increased risk of statin-associated myopathy. 23 In a different study the combination therapy of simvastatin 20 mg plus ezetimibe 10 mg daily has been shown to effectively reduce LDL cholesterol and the incidence of major atherosclerotic events in patients with advanced chronic kidney disease. 24 However, favorable results were not observed for statin plus ezetimibe therapy in patients with stable angina after successful coronary stenting despite evident lipid-lowering. 25 On the contrary, the combination of ezetimibe and statin therapy has been shown to effectively reduce the risk of cardiovascular events after vascular surgery within the first year period. However, recommendations on routinely using this combination therapy to reduce cardiovascular events risk require further evidence. 26
Evolocumab and alirocumab
The gene encoding for proprotein convertase subtilisin/Kexin type 9 protein (PCSK9) was previously implicated in familial hypercholesterolemia. The PCSK9 protein increases LDL-C levels by facilitating the uptake and degradation of LDL receptors and limiting LDL-receptor recycling. 27 The loss of function variations in the gene resulted in low levels of LDL-C respectively and this fueled the development of PCSK9 inhibitors. PCSK9 inhibitors are monoclonal antibodies that act by inhibiting the PCSK9 protein. These agents have superior LDL-C lowering capacity than other non-statin cholesterol-lowering drugs. PCSK9 inhibitors result in a 60% reduction in LDL-C when taken as add-on therapy with a high-intensity statin. 28 Some studies have even suggested an 80% to 90% reduction in cardiovascular disease. 29 The FDA approved evolocumab and alirocumab in 2015 following positive results of FOURIER 30 and ODYSSEY trial. 31 Both trials of Evolocumab and Alirocumab have shown that these drugs significantly lower LDL-C and the risk of ASCVD. However, mortality benefits are only evident from the Alirocumab trials. These trials show that PCSK9 inhibitors reduce LDL-C significantly to very low levels. However, very low LDL-C levels had not been linked with a significant detrimental effect on neurological and neurocognitive function. 30,31 In addition to its impact on LDL level evolocumab significantly reduced Lp(a) levels, and patients with higher baseline Lp(a) levels experienced greater absolute reductions in Lp(a) and tended to derive greater coronary benefit from PCSK9 inhibition. 32 Recent evidence also suggests that PCSK9 inhibition could represent a therapeutic strategy in patients with calcific aortic valve stenosis (CAVS). 33 This was suggested after finding that CVAS is less prevalent in carriers of the PCSK9 R46 L variant compared with non-carriers. 34
The need for subcutaneous injection and minor side effects such as influenza-like symptoms, nasopharyngitis, upper respiratory tract infection, rash, nausea, backache, arthralgia, and injection-site reactions, the high acquisition cost, and lack of long-term safety data are some of the drawbacks of using these drugs. 30,31 Regardless of these drawbacks a study has shown that there is an increasing trend of PCSK9 prescription fills in the United States between the years 2015 and 2019. 35 The manufacturers have also reduced the cost of these drugs to improve affordability. However, the drugs are not yet considered cost-effective. Due to this PCSK9 inhibitors are recommended only as a third-line approach if statins plus ezetimibe fail to produce the desired results.
Testing of bococizumab, another monoclonal antibody drug, was discontinued due to attenuation of the drug response over time. This may have occurred because the antibody was humanized rather than fully human-derived. 36
Bempedoic acid
Bempedoic acid, a prodrug, activated by a hepatic enzyme, lowers LDL-C by inhibiting ATP citrate lyase, an enzyme involved in fatty acid and cholesterol synthesis. 37 In 2020 the FDA approved using bempedoic acid in addition to the maximally tolerated statin for patients with heterozygous familial hypercholesterolemia or those with established ASCVD based on significant LDL-C lowering noted in the CLEAR Harmony, 38 and CLEAR Wisdom trials respectively. 39 In addition to LDL-C lowering, bempedoic acid can lower non-HDL-C, apolipoprotein B, and high-sensitivity C-reactive protein (CRP). 39 Bempedoic acid can be used as an alternative for patients with statin-related muscle symptoms due to fewer muscle-related side effects. Bempedoic acid has also been used as add-on therapy with the maximum dose of statin and this combination has led to lower LDL cholesterol levels without a higher incidence of overall adverse events. 38 In the CLEAR outcomes trial, the impact of bempedoic acid on cardiovascular outcomes in statin-intolerant patients with, or at high risk for CVD is being evaluated [ClinicalTrials.gov Identifier: NCT02993406]. Further data to allow better risk-benefit decisions are expected. 40 The FDA has also approved bempedoic acid plus ezetimibe combination as a lipid-lowering agent for patients with ASCVD and Heterozygous FH on February 26, 2020. The CLEAR tranquility study stated that this drug reduces LDL-C by 28% more than placebo. 41
Omega-3 fatty acids
Fish oil products containing omega-3 fatty acids such as eicosapentaenoic acid [EPA] and docosahexaenoic acid [DHA] are effective in reducing triglycerides in patients with severe hypertriglyceridemia (TG levels >500 mg/dL). EPA monotherapy (dose of 4 g/d) or a combination of EPA and DHA (dose of >3 g/d) is an effective and safe option for reducing triglycerides as monotherapy or as an adjunct to other lipid-lowering agent. 42 High-dose icosapent ethyl (IPE), a highly purified form of EPA, reduced major adverse cardiovascular events by 25% over 5 years among patients who were on a statin in the REDUCE-IT trial. 43 Icosapent ethyl is approved by the FDA recently as TG lowering therapy on top of statin to reduce the risk of cardiovascular events in adult patients with hypertriglyceridemia. 43 This drug acts by reducing the synthesis and enhancing the clearance of triglycerides. The CV outcome trial [C linicalTrials.gov Identifier, NCT02104817] which evaluated the impact of EPA plus DHA combination therapy in patients with high TG and low HDL cholesterol on a statin was halted due to lack of benefit to patients. 44
Lomitapide and mipomersen
Lomitapide and mipomersen can be used to lower LDL-C levels in addition to standard hypolipidemic agents or LDL-apheresis in patients with HoFH but the cost and drug-related adverse effects limit their use. Lomitapide inhibits microsomal TG transfer protein (MTP) thereby decreasing the synthesis of TG-rich chylomicrons in the intestine and very-low-density lipoprotein (VLDL) in the liver. 45 This drug can reduce LDL-C (by 50%), total cholesterol, Apo B, and non-HDL Cholesterol. 46 Mipomersen as an add-on therapy reduces apo B-containing lipoproteins in patients at high risk of ASCVD who are contraindicated or intolerant of statins. Also, this agent reduces LDL-C (by 25%), total cholesterol, and non-HDL cholesterol. 47 Mipomersen significantly reduced LDL cholesterol, apolipoprotein B, and lipoprotein(a) in patients with hypercholesterolemia with, or at risk for, coronary heart disease not controlled by existing therapies. 48 Despite the positive results of LDL-C reduction, the use of mipomersen as an adjunct therapy in patients without a confirmed diagnosis of HoFH is not warranted due to increased risk of hepatotoxicity and undetermined effect on cardiovascular morbidity and mortality.
Non-Statin Lipid-Lowering Drugs Under Investigation
Inclisiran
It has been demonstrated that gain-of-function PCSK9 mutations cause familial hypercholesterolemia (FH), while loss-of-function PCSK9 mutations cause low circulating LDL-C levels. Inclisiran is the first small interfering RNA (siRNA) molecule designed to decrease the activity of PCSK9. It is a chemically modified PCSK9-specific molecule that was shown to inhibit hepatic synthesis of PCSK9 in adults with heterozygous FH (HeFH). 49 Inclisiran, is given biannually as 300 mg via a prefilled injection. ORION-9, 50 ORION-10, and ORION-11 51 trials showed a robust reduction in LDL-C levels when inclisiran is used in addition to statin (LDL-C reduction 39.7% to 52.3%). To check if the reduction of LDL-C translates into clinical outcomes, a long-term follow-up study (ORION-4) of patients with cardiovascular disease is ongoing. 52 So far, no serious inclisiran related-safety issues are noted except for injection-site side reactions. Inclisiran is recently approved by the European Commission for the treatment of adults with primary hypercholesterolemia (heterozygous familial and non-familial) or mixed dyslipidemia, as an adjunct to diet. Inclisiran is the first and only approved small-interfering RNA lipoprotein cholesterol (LDL-C) lowering treatment in Europe. The FDA has not yet approved this drug due to some concerns with the manufacturing site.
Antisense apolipoprotein A oligonucleotide
Gene-silencing therapies in the future may help reduce the residual ASCVD risk. 53,54 A hepatocyte-directed antisense apolipoprotein A (apo A) oligonucleotide, reduced lipoprotein(a) levels in a dose-dependent manner in patients who had established CVD. 37,38,55
Cholesteryl ester transfer protein (CETP) inhibitors
Variants in the CETP gene were associated with higher blood HDL cholesterol, lower low-density lipoprotein cholesterol, and lower risk of coronary heart disease suggested that pharmacological inhibition of CETP may be beneficial. An increased level of HDL-C is not always linked with a reduction of cardiovascular risk or death. Though CETP inhibitors such as torcetrapib, dalcetrapib, and evacetrapib increase the levels of HDL-C adequately, their development has been hampered by an increase in mortality rates or an insignificant impact on CV events. Anacetrapib, a CETP inhibitor, resulted in a modest reduction in CV events as an add-on therapy to intensive statin. 56 However, FDA approval was not sought for this drug by the developer due to poor market potential and because the drug accumulates in adipose tissue.
A randomized, phase III multicenter study evaluated the effects of dalcetrapib on cardiovascular risk in a genetically defined population with a recent acute coronary syndrome. 57 The results indicate that a polymorphism in the adenylate cyclase type 9 (ADCY9) gene influenced the composite outcome of cardiovascular death, myocardial infarction, or stroke. A favorable benefit-risk ratio was depicted for patients with AA genotype at position rs1967309. A recent study that evaluated exposure to variants in the genes that encode the targets of CETP inhibitors and statins revealed a discordant reduction in LDL-C and apoB levels. This result may explain the failure of CTEP inhibitors to reduce cardiovascular events, which is mainly driven by apoB reduction.
Gemcabene
Gemcabene is an investigational drug that lowers LDL-C, reduces CRP, decreases TGs, and increases HDL-C with a mechanism of action independent of the LDL receptor. 58 It acts by increasing the clearance of VLDL via the reduction of hepatic apolipoprotein C-III (Apo C-III) messenger RNA. As an adjunct to other lipid-lowering therapy for HoFH, this drug can significantly reduce LDL-C levels. 59
Evinacumab
Evinacumab, an intravenously administered drug, is a monoclonal antibody against Angiopoietin-like protein 3 (ANGPTL3) a protein secreted by the liver that regulates the hydrolysis of VLDL triglycerides (TGs) and HDL phospholipids by lipoprotein lipase and endothelial lipase, respectively. 60 In 2017, the FDA granted breakthrough therapy designation for evinacumab for the treatment of hypercholesterolemia in patients with HoFH. Evinacumab nearly halves LDL cholesterol in HoFH atop even a PCSK9 inhibitor, according to ELIPSE HoFH trial[ClinicalTrials.gov Identifier, NCT03399786]. Evinacumab is well-tolerated and may improve clinical outcomes in patients with HoFH. 60 Relationship between loss of function mutations in ANGPTL3 and favorable reductions in TG and non-HDL-C has been noted in recent studies. This finding has made ANGPTL3 a promising molecular target for gene editing to reduce CHD risk. Unlike other antihyperlipidemic agents, ANGPTL3 inhibition may also improve insulin sensitivity. The US Food and Drug Administration has approved evinacumab for use on top of other cholesterol-modifying medications in patients aged 12 and older with HoFH. The drug is currently under regulatory review for the same indication in Europe.
Discussion
This review presents the summary of FDA approved non-statin lipid-lowering drugs and other emerging non-statin lipid-lowering agents. The review aims to serve as an update for clinicians, pharmacists, and other healthcare teams who are involved in the care of patients who require lipid-lowering therapy to manage primary hyperlipidemia or to prevent cardiovascular events. In the result section, the indication, advantages, and disadvantages of currently approved medication are discussed. This could facilitate the optimal selection and monitoring of approved non-statin lipid-lowering drugs by healthcare providers.
The current guideline recommendations on the use of non-statin lipid-lowering therapy according to recent American College of Cardiology/American heart association and European cardiac society guidelines are as follows. The 2018 ACC/AHA guideline on the management of blood cholesterol recommends a personalized approach to identify and provide primary and secondary prevention interventions. Using the race- and sex-specific pooled cohort equations (PCE) 10-year ASCVD risk can be estimated for patients who are 40 to 75 years of age. 61 PCE is widely criticized for overestimating or underestimating cardiovascular risk. Due to this limitation of the PCE score, there is a need to refine the risk estimate by looking into the presence of risk enhancers (eg, family history of ASCVD, primary hypercholesterolemia, chronic kidney disease, chronic inflammatory conditions, abnormal biomarkers such as lipoprotein a, ethnicity). In patients who do not have established ASCVD, initiating therapy for primary prevention can be challenging. The 2018 ACC/AHA guideline recommends testing of coronary calcium score (CCS) to help refine the risk of ASCVD in patients with moderate ASCVD risk. 7 According to this guideline, moderate to high-intensity statin should be prescribed to patients with primary hyperlipidemia, with diabetes, or a 10-year risk of ASCVD ≥ 7.5%. PCSK9 inhibitors should be considered for patients with severe primary hypercholesterolemia if the LDL-C level on a statin in combination with ezetimibe remains ≥100 mg/dL and the patient has multiple ASCVD risks enhancing factors. Similarly, in patients with very high-risk ASCVD, if LDL-C level remains ≥70 mg/dL on a maximally tolerated statin and ezetimibe therapy, a PCSK9 inhibitor is recommended. 7 Other non-statin hypolipidemic agents such as niacin, bile acid sequestrants, or fibrates as add-on therapy to statins to achieve target LDL-C reduction is not routinely recommended. Recommendations on bempedoic acid and its combination with ezetimibe are not available in this guideline as these drugs are approved in 2019 and cardiovascular outcome trials are ongoing.
According to the 2019 ESC/EAS guidelines for the management of dyslipidemias, the treatment of patients is based on scores of systematic coronary risk evaluation (SCORE). 5 The cardiovascular imaging for assessment of ASCVD risk should also be considered. According to this guideline, the main target for lipid-lowering therapy is the reduction of LDL-C levels. In the secondary prevention of patients who are at very high risk (SCORE >10%), the target for cholesterol reduction is LDL-C <55 mg/dL or reduction of at least 50% from baseline is recommended. For those who have developed a second cardiovascular event, the target for LDL-C reduction is >40 mg/dL. To achieve this target, the addition of a PCSK-9 inhibitor would be essential for most patients with coronary artery disease. 5 PCSK-9 inhibitors can be prescribed for patients who have refractory hyperlipidemia after the initial treatment with a maximally tolerated statin and ezetimibe or for those with statin intolerance, and those with very high cardiovascular risk. 5 On the other hand, for high-risk patients whose triglyceride levels are between 135 to 499 mg/dL despite statin use, the addition of high-dose icosapent ethyl (IPE) can be considered.
In the future
Approval of inclisiran by the European Commission is anticipated to pave the way for FDA approval of this drug shortly. Incliciran is anticipated to improve adherence due to twice a year dosing and lesser cost. In the near future, trials of recently approved bempedoic acid and a combination of bempedoic/ezetimibe are likely to be completed. The availability of orally taken hypolipidemic agents will provide a good alternative for patients who are statin-intolerant or those who would like to avoid injectable lipid-lowering drugs. More drugs acting on different targets to reduce cholesterol are likely to emerge.
Conclusion
Non-statin hypolipidemic drugs serve as an alternative therapy for patients unable to take statins and for those with residual cardiovascular risk while taking a high-intensity statin. Current guidelines recommend targeting lower LDL-C for patients with higher CVD risk. Ezetimibe and PCSK9 inhibitors can further lower LDL-C levels and ASCVD risk when used in addition to statins. The availability of PCSK9 inhibitors has changed the landscape of lipid-lowering therapy, but the cost is a limiting PCSK9 uptake. Bempedoic acid and its combination with ezetimibe have proved to offer the benefit of lipid-lowering as an add-on therapy to statin or as monotherapy for patients with hyperlipidemia intolerant to statins. However, further long-term safety data for most non-statin lipid-lowering drugs and cardiovascular outcome data for recently approved drugs such as bempodoic acid and combination of bempedoic acid, ezetimibe, inclisiran [approved by in Europe], and evinacumab [approved in Europe] is pending. For cases where a combination of statin and non-statin therapy is inadequate to control LDL-C to desirable levels, referral to clinicians who specialize in treating complex lipid disorders is necessary.
Footnotes
Author Contributions
S.A.B. and N.H. conceived of the primary idea for the review. S.A.B, N.H, A.A.E, and A.S.S contributed to identifying and selection of relevant studies to be included in the review. All authors discussed the results and contributed to the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
