Abstract
The role of lipid lowering in reducing the risk of mortality and morbidity from cardiovascular disease (CVD) is well established. Treatment particularly aimed at decreasing low-density lipoprotein cholesterol (LDL-C) is effective in reducing the risk of death from coronary heart disease and stroke. Statins form the cornerstone of treatment. However, in some individuals with a high risk of CVD who are unable to achieve their target LDL-C due to either intolerance or lack of efficacy, there is the need for alternative therapies. This review provides an overview of the different classes of currently available lipid-lowering medications including statins, fibrates, bile acid sequestrants (resins), and omega-3 fatty acids. Data are presented on their indications, pharmacology, and the relevant end point clinical trial data with these drugs. It also discusses the human trial data on some novel therapeutic agents that are being developed including those for homozygous familial hypercholesterolemia—the antisense oligonucleotide mipomersen and the microsomal transfer protein inhibitor lomitapide. Data are presented on phase II and III trials on agents with potentially wider applications, cholesterol ester transfer protein inhibitors and proprotein convertase subtilisin kexin 9 inhibitors. The data on a licensed gene therapy for lipoprotein lipase deficiency are also presented.
Introduction
Dyslipidemia is one of the risk factors for progression of atherosclerotic disease which may account for up to 55% of the cardiovascular disease (CVD) risk after correction for age and gender. 1 Epidemiological and prospective studies have established the benefit of reducing low-density lipoprotein cholesterol (LDL-C), with a 1% reduction associated with a 1% decrease in CVD events. 2 Alternatively, a 40 mg/dL (1 mmol/L) reduction in LDL-C is associated with a 21% reduction in major CVD events. 3 In the Helsinki heart study, a 1% increase in high-density lipoprotein cholesterol (HDL-C) is linked with a 3% reduction in cardiovascular events. 4 The effects of high triglycerides (TGs), TG-rich lipoprotein remnants, and very LDLs (VLDLs) are uncertain, 5,6 but they are also associated with increased CVD risk through the effects on HDL and LDL particle sizes increasing the atherogenicity of LDL. 7,8
There are different guidelines with practical recommendations on the management of dyslipidemia. Lifestyle modification that includes dietary changes, weight reduction, exercise, decreased alcohol consumption, and smoking cessation is important in the management of hyperlipidemia. 2 However, in certain patients who have a high risk of CVD, the addition of drug therapy will be necessary to achieve beneficial effects. Lipid-lowering agents in current use are generally effective, but some are limited by their side effects. Statins are the first choice lipid-lowering drugs given their almost universal efficacy for CVD. 3 However, only 50% of the high-risk patients attain LDL-C targets with statins, 9 and the role of additional lipid-lowering therapies remains unclear. 10 Hence, there is a need for development of new therapeutic agents aimed at those who cannot achieve current targets or who are intolerant to most or all of the existing therapies.
Currently Available Lipid-Lowering Therapies
Statins
Statins inhibit 2-hydroxyl-methyl-glutaryl coenzyme A (HMG-CoA) reductase, the rate-limiting step in cholesterol synthesis. 11,12 This leads to a reduced intracellular cholesterol concentration and subsequent upregulation of LDL receptors that encourages the removal of LDL-C from the circulation. “Mevastatin,” the first statin produced, was a fungal metabolite identified by Dr Endo and his team. 12 Statins are commonly prescribed drugs, and currently there are 7 available statins with 5 being off patent worldwide—lovastatin, simvastatin, pravastatin, fluvastatin, and atorvastatin (Table 1). Rosuvastatin and atorvastatin are the most potent drugs, 13,14 and pitavastatin is licensed in a few countries. 15 They display different pharmacokinetic properties, and some are metabolized by the cytochrome P450 3A4 (CYP3A4) pathway apart from pravastatin and rosuvastatin, which are metabolized by sulfation and conjugation or CYP2C9, respectively. Statins are effective in reducing LDL-C but with a wide range of efficacies by dose and drug. 13,16 Their benefits have been confirmed in CVD outcome studies (Table 2) and are indicated in their product licenses (Table 3). 3 The reduction in LDL-C is dose dependent, with a 5% to 7% decrease with every dose doubling; they can also reduce TGs, but they have little effect on HDL-C. 13,17 Statins are safe and generally tolerated by most patients. However, higher doses that achieve greater LDL-C reduction are associated with more side effects 3 such as gastrointestinal disturbances and myopathy. 18,19 Increase in liver enzymes particularly alanine and aspartate transaminases occurs in a small proportion of patients on statin therapy and may normalize on reducing the dose or stopping the statin. Hence, the baseline measurement of liver function test is important prior to commencing the statin therapy and stopping treatment if transaminases are raised more than 3 times the upper reference limit. 18 The use of statin has also been associated with an increased incidence of new onset diabetes mellitus. 20
The Effect of Treatment With Different Statins on Lipid Profiles.a

Abbreviations: TC, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
Summary of Major Monotherapy CVD Outcome Trials.

Abbreviations: LRC, lipid research clinics primary prevention trial; CDP, Coronary Drug Project; HHS, Helsinki heart study; 4S, Scandinavian simvastatin survival trial; WOSCOPS, West of Scotland coronary prevention study; CARE, Cholesterol and recurrent events; LIPID, long-term intervention with pravastatin in ischemic disease; AF/TexCAPS, Air Force/Texas coronary atherosclerosis prevention study; VA-HIT, Veterans Administration HDL-intervention trial; HPS, heart protection study; GREACE, The Greek atorvastatin and coronary heart disease evaluation; ASCOT, AngloScandinavian coronary outcomes study; CARDS, Collaborative atorvastatin diabetes study; JUPITER, Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin; MI, myocardial infraction; PCI/CABG, percutaneous coronary intervention/coronary artery bypass graft; CVD, cardiovascular disease; N/A, not available.
Information on the Doses and Licensed Indications (UK and/or USA) of Different Classes of Currently Existing Lipid-Lowering Therapies.

Fibrates
Fibrates are agonists of peroxisomal proliferator activated receptor alpha (PPAR-α), a nuclear transcription factor that regulates the expression of many enzymes and genes. 21 Their primary action is to decrease TGs and increase HDL-C in patients with hypertriglyceridemia and mixed hyperlipidemia. The TG synthesis is decreased by stimulating fatty acid uptake and beta-oxidation in the mitochondria, noncompetitive inhibition of diacylglycerol acyltransferase-2 (DGAT2), increased lipoprotein lipase (LPL) levels via induction of LPL synthesis and reduction in apolipoprotein C3, and directly through decreased VLDL secretion and increased LDL receptor-mediated clearance of TG-rich lipoproteins. The HDL-C is increased by stimulating the expression of apolipoprotein A1 and A2. Treatment with a fibrate may be associated with increase in LDL-C due to increased LPL-dependent conversion of VLDL to LDL-C. Clofibrate was the first drug of this class developed in the 1960s; however, it has now been discontinued due to adverse effects associated with it. 22 Fenofibrate, gemfibrozil, and bezafibrate are the commonly used therapies. A dose of 267 mg of fenofibrate is slightly more potent than the 200 mg dose that is equivalent in potency to other fibrates. Fibrates (Table 2) have been shown in meta-analyses to reduce CVD events by 10% to 13% 23,24 in monotherapy and may also improve microvascular disease in patients with diabetes. Fenofibrate is the only fibrate shown to lower the plasma urate levels. 25 They do not add to risk reduction with statins in patients with diabetes and good control of LDL-C. 26 They may offer greater benefit if added to a statin in patients with low HDL-C and high TGs, although this remains to be confirmed in a large clinical trial. 7 However, a combination of gemfibrozil with a statin is discouraged due to the increased risk of myopathy and hepatotoxicity, as gemfibrozil decreases glucuronidation of statins thereby reducing its excretion. 27
Bile Acid Sequestrants
Cholesterol is secreted into the gut through the biliary system and directly through the action of perilipin in enterocytes in the ileum. 28 Bile acid sequestrants decrease LDL-C by binding to bile acids in the gut and preventing their reabsorption leading to decreased enterohepatic concentration of bile acids and cholesterol. The decreased level of bile acids stimulates upregulation of hepatic LDL receptors leading to increased LDL-C clearance from the circulation. They increase HDL-C by 3% to 5% and increase TG levels by stimulating hepatic VLDL production through its action on the liver-X receptor. 29 Bile acid sequestrants also reduce blood glucose through their effect on the farnesoid-X receptor. Cholestyramine, colestipol, and colesevelam are currently available therapies, and colesevelam has been shown to decrease hemoglobin A1c by 0.4% to 0.6% and is thus also licensed for glycemic control. 30 Colesevelam decreases LDL-C by about 16% to 19%, and combination with statins produces a greater than 40% decrease. 29 Some end point evidence exists for the use of bile acid sequestrants. In the Lipid Research Clinics trial, cholestyramine decreased CVD events by 25% (Table 2). 31 The use of these agents is limited by their frequent gastrointestinal side effects. They interfere with the absorption of micronutrients such as vitamin A, D, E, K, iron, folic acid, and magnesium. They also reduce the absorption of medications such as warfarin and to a lesser extent thiazide diuretics and digoxin. 30
Niacin/Nicotinic Acid
Niacin or vitamin B3 is the oldest lipid-lowering therapy. Its mode of action is still not entirely clear, but it has favorable effects on all aspects of an abnormal lipid profile. 32,33 It decreases VLDL and TG synthesis in the liver by inhibiting diacylglycerol O-acyltransferase 2 (DGAT-2) and hormone-sensitive lipase activity. 33 The LDL-C is decreased by increased catabolism of apolipoprotein B (apo B) and TG levels are decreased by niacin, leading to increased LDL particle size and decreased LDL particle number. Niacin increases HDL-C by promoting production of apolipoprotein A-1 and reducing its clearance through the ATP synthase β-chain holoparticle receptor. Niacin monotherapy reduced the CVD risk by 22% in the Coronary Drug Project (Table 2). 34 It, unlike ezetimibe, also leads to significant regression of carotid intima–media thickness when combined with statin therapy as demonstrated by the Arterial Biology for the Investigation of the Treatment Effects of Reducing Cholesterol 6-HDL and LDL Treatment Strategies in Atherosclerosis (ARBITER 6-HALTS) trial. 35 The use of niacin is limited by flushing due to increased prostaglandin D2 synthesis. Niacin treatment is also associated with increased rates of new diabetes and deterioration in glycemic control. 36,37 End point studies of niacin combined with statins have been disappointing. In the Atherothrombosis Intervention in Metabolic Syndrome with Low HDL/High Triglycerides: Impact on Global Health Outcomes (AIM-HIGH) study, patients with established high CVD risk and dyslipidemia where secondary optimization of LDL-C was allowed, extended release (ER) of niacin did not reduce CVD events. 38 The Heart Protection Study-2/Treatment of HDL to Reduce the Incidence of Vascular Events (HPS-2/THRIVE) study investigated the effect of the combination of niacin and laropiprant (Tredaptive) with a statin on CVD events in patients at high risk of CVD after initial optimization of LDL-C levels. 10,37 Niacin–laropiprant failed to show any significant reduction in CVD events but showed an increased incidence of nonfatal side effects, and it has since been withdrawn.
Ezetimibe
Ezetimibe inhibits cholesterol absorption through inhibition of the duodenal Niemann-Pick C1-like protein. 39 In combination with statin therapy, it decreases LDL-C although its effect in reducing cardiovascular events is controversial. Studies of adding ezetimibe to statins in familial hypercholesterolemia (FH) such as ezetimibe and Simvastatin in Hypercholesterolaemia Enhances Artherosclerosis Regression (ENHANCE) of carotid intima-media thickness 40 and of combination therapy on CVD events in the underpowered Simvastatin and Ezetimibe in Aortic Stenosis (SEAS) 41 have not shown any beneficial effect. Recently, in the Study of Heart And Renal Protection (SHARP) combination therapy with statin reduced CVD events by 27% in patients with advanced renal failure. 42 Some small studies have suggested ezetimibe may improve hepatic steatosis. 39 Ezetimibe is generally well tolerated but can cause nausea or bloating. The outcome data on the effect of ezetimibe and simvastatin combination therapy in reducing the risk of CVD in patients with acute coronary syndrome in the IMProved Reduction of Outcomes: Vytorin Efficacy International Trial (IMPROVE-IT) study are still awaited. 10
Omega-3 Fatty Acids
Omega-3 fatty acids that include docosahexaenoic acid and eicosapentaenoic acid (EPA) have been shown to decrease CVD events in monotherapy in patients with CVD or at high risk of developing CVD. 43,44 However, individual studies examining the CVD effect of omega-3 fatty acids have shown conflicting results with early studies being positive and later studies negative. Meta-analyses of omega-3 fatty acids added to optimal statin therapy suggest they give no added benefit. 45 Their mechanism of action is complex and not fully understood, but it may involve the regulation of genes involved in lipid metabolism. They are known to reduce TGs in a dose-dependent manner. In the Multi-center plAcebo-controlled Randomised double-blINd 12-week study with an open label extension (MARINE; AMR-101; Amarin Pharmaceuticals, Dublin, Eire), an EPA preparation has been shown in patients with high TGs (>750 mg/dL; 8.5 mmol/L) to reduce TGs by 33% to 45%. 46 The suggested mechanism for TG reduction includes suppression of lipogenesis, decreased TG synthesis through inhibition of DGAT-2 and increased beta-oxidation in the mitochondria. 44 They are well tolerated with mild gastrointestinal disturbance being the most commonly reported adverse symptom. 46
New Therapies for Dyslipidemia
Statins remain the cornerstone of drug-based lipid management allied with lifestyle changes such as dietary modification, weight reduction, and exercise. However, if the desired level of LDL-C is not achieved with statins even after titration to the highest tolerable dose, then other treatment options may need to be considered. A considerable number of new therapies for dyslipidemia are in development (Table 4). 47,48 New lipid-lowering therapies can be classified based on their role, either as LDL-C reducing agents or as drugs that target other lipids such as HDL-C or TGs.
Summary of Major Trials on Novel Cholesterol-Lowering Therapies.
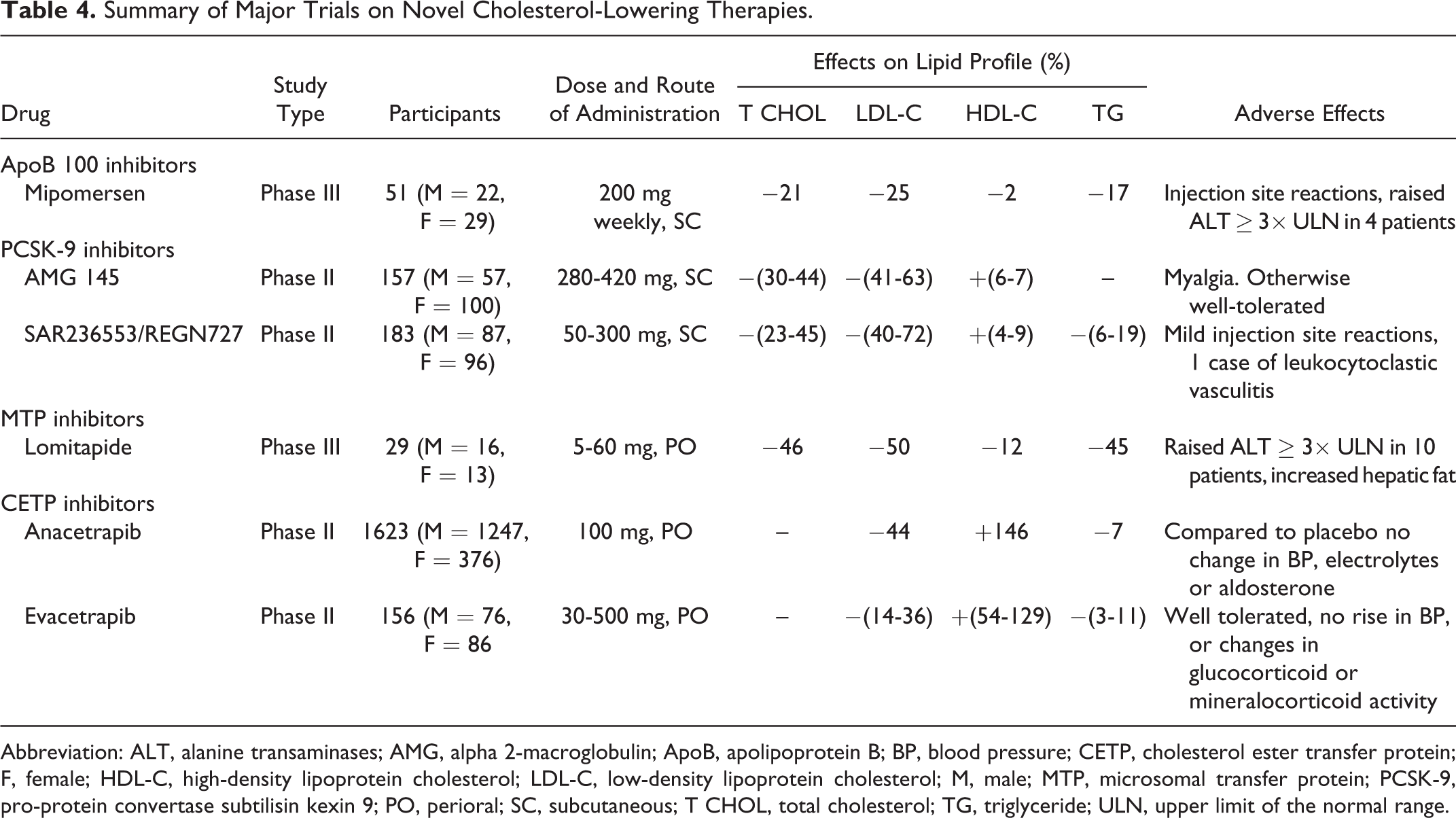
Abbreviation: ALT, alanine transaminases; AMG, alpha 2-macroglobulin; ApoB, apolipoprotein B; BP, blood pressure; CETP, cholesterol ester transfer protein; F, female; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; M, male; MTP, microsomal transfer protein; PCSK-9, pro-protein convertase subtilisin kexin 9; PO, perioral; SC, subcutaneous; T CHOL, total cholesterol; TG, triglyceride; ULN, upper limit of the normal range.
LDL-cholesterol-Lowering Therapies
Patients with heterozygous FH respond to statin and ezetimibe therapy, but statins show reduced or absent efficacy in patients with homozygous FH who do not have functioning LDL receptors. Novel treatments to reduce LDL-C include drugs with a primary license indication for orphan disorders such as homozygous FH and drugs that interfere with the production of VLDL and hence LDL either through disruption of apoB synthesis or through interference with transfer factors involved in loading lipids onto the nascent particle. Mipomersen is an antisense oligonucleotide to apoB which reduces LDL-C by 25% in homozygous FH but is limited by injection site reactions and hepatic steatosis. 49 Lomitapide is a microsomal transfer protein inhibitor 50 which reduces LDL-C by 50% but causes hepatic steatosis. 51 The side effects of these drugs restrict them at the moment to orphan indications.
Other novel agents reduce LDL-C by inhibiting pro-protein convertase subtilisin kexin 9 (PCSK-9), a plasma and hepatocellular protein that controls LDL receptor expression. 52 Antibodies to PCSK-9 deliver up to 70% reduction in LDL-C when added to other therapies or in statin-intolerant patients and may have a large potential application in patients with CVD. Large-scale CVD end point trials of adding these agents to optimal LDL-C-lowering strategies are just beginning.
HDL-C Raising Drugs
Statins do not address all lipid-related CVD risk. Epidemiological studies show that higher HDL-C is associated with a better prognosis even after statin treatment. Some of this residual risk is ascribed to HDL. 7,53 The newest agents that raise HDL-C are cholesterol ester transfer protein (CETP) inhibitors. 48 These can raise HDL-C by 30% to 100% and reduce LDL-C by 0% to 40%. The original compound in the class torcetrapib was highly effective in improving lipid profiles but increased CVD events by 30%, possibly by raising blood pressure. 54 Dalcetrapib raised HDL-C by 30% but had no effect on LDL-C and in reducing CVD events. 55 Two other CETP inhibitors—anacetrapib and evacetrapib—that raise HDL-C and also reduce LDL-C by 40% remain in development.
Gene Therapy for Lipid Disorders
Initial attempts at gene therapy for homozygous FH were unsuccessful. More recently, the development of adeno-associated viral vectors has led to the development of a gene therapy for lipoprotein lipase deficiency (LPLD). 56 The LPLD is associated with severe hypertriglyceridemia and pancreatitis, and there is no treatment for homozygotes apart from a highly fat-restricted diet. Treatment with alipogene tiparvovec transiently reduces TGs in patients with LPLD but does reduce the frequency of pancreatitis by 80%.
Conclusion
Many therapies that reduce LDL-C have been proven to reduce CVD risk in monotherapy. Statins remain as first-line treatment, and they are safe. Combination therapy with other currently used lipid-lowering drugs such as niacin and ezetimibe has not shown beneficial effects in CVD risk reduction. Despite the efficacy of statins in LDL-C lowering there still remains a residual risk of CVD, and it is important that this is addressed. Newer therapies are being developed to further reduce LDL-C. Some have been disappointing with respect to their safety and tolerability profiles. The PCSK-9 inhibitors and CETP inhibitors anacetrapib and evacetrapib appear promising. These newer agents can be used as alternatives to statins in patients who are statin intolerant, or they can be used in combination therapy to help achieve LDL-C targets, especially in high-risk individuals thereby reducing the risk of CVD. As noted with torcetrapib, LDL-C lowering is not always associated with CVD risk reduction. Therefore, these new lipid-lowering therapies should provide evidence of improved CVD outcome. Safety and efficacy data in large clinical trials will ascertain the benefits of these agents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
