Abstract
Phytogenic cannabinoids from Cannabis sativa and synthetic cannabinoids are commonly used substances for their recreational and medicinal properties. There are increasing reports of cardiotoxicity in close temporal association with cannabinoid use in patients with structurally normal hearts and absence of coronary arterial disease. Associated adverse events include myocardial ischemia, conduction abnormalities, arrhythmias, and sudden death. This review details the effects of phytogenic and synthetic cannabinoids on diverse receptors based on evidence from in vitro, human, and animal studies to establish a molecular basis for these deleterious clinical effects. The synergism between endocannabinoid dysregulation, cannabinoid receptor, and noncannabinoid receptor binding, and impact on cellular ion flux and coronary microvascular circulation is delineated. Pharmacogenetic factors placing certain patients at higher risk for cardiotoxicity are also correlated with the diverse effects of cannabinoids.
Introduction
According to the 2019 World Drug Report, phytogenic cannabinoids from Cannabis sativa (cannabis) and synthetic cannabinoids are the most commonly used drugs, with 188 million people, or roughly 3.8% of the global population aged 15 to 64 years, using cannabinoids at least once in the past year. 1 The rate of worldwide cannabis use has risen by 19% over the current decade and is projected to rise as legalization for recreational and medical use advances. 1 In the United States, an estimated 44 million Americans aged 12 or older, roughly 16% of the population, used cannabis in the past year, with adolescents aged 12 to 17 years representing the group with highest rate of initiation. 2
An association between cannabis use, cardiac arrhythmia, and acute coronary syndromes (ACS) was first recognized in the early 1970s, when several small prospective studies highlighted the significant increase in heart rate immediately after smoking cannabis. 3 -7 This effect could be mitigated with β-blocker pretreatment, which suggested involvement of the sympathetic nervous system. 3,8 Since then, research has delineated the complex interaction between the endocannabinoid system and the autonomic nervous system, cardiac conduction, and circulation. Nearly all prospective studies to date involving cannabis in healthy volunteers have demonstrated elevation of heart rate immediately after cannabis administration. However, there exist numerous case reports of cannabis-induced bradycardia and other bradyarrhythmias, such as sinus arrest and atrioventricular block. This suggests that cannabis use results in more complex effects beyond sympathetic nervous system activation.
The association of cannabis use and increased risk of cardiac arrhythmia and ACS is supported by plausible physiological mechanisms (Table 1) and large number of published reviews, studies, case series, and reports. The majority of these articles involved younger individuals with no structural heart defects or coronary arterial disease. 9 This comprehensive review explores the definitive and theoretical polypharmacologic, electrophysiologic, and neuroendocrine evidence linking cannabis and synthetic cannabinoid use with cardiac arrhythmias and ACS. Articles for this review were chosen from an extensive query of PubMed, Google Scholar, and OpenGrey from inception to May 1, 2020, using a broad search string (“cannabis”[TW] OR “marijuana”[TW] OR “cannabinoid”[TW]) AND (“coronary”[TW] OR “ischemia”[TW] OR “infarction”[TW] OR “dysrhythmia”[TW] OR “arrhythmia” OR “fibrillation”[TW] OR “tachycardia”[TW] OR “bradycardia”[TW] OR “asystole”[TW] OR “electrocardiogram”[TW] OR “toxicity”[TW]). All pertinent human, animal, and in vitro studies as well as case series, case reports, and reviews in any language were considered in the literature search.
Summary of Known and Theoretical Mechanisms for Phytogenic and Synthetic Cannabinoid Agonist-Induced Cardiac Arrhythmias.

Abbreviations: ATP, adenosine triphosphate; AV, atrioventricular; CB1, cannabinoid 1 receptor; CB2, cannabinoid 2 receptor; CYP, cytochrome P450; EET-EAs, endocannabinoid epoxides; GIRK, G protein-activated inwardly rectifying K+ channel; GPR, G protein-coupled receptors; 5-HT, 5-hydroxytryptamine (serotonin); PPAR, peroxisome proliferator-activated receptors; ROS, reactive oxygen species; THC, Δ9-tetrahydrocannabinol; TRPA, transient receptor potential ankyrin; TRPM, transient receptor potential melastatin; TRPV, transient receptor potential vanilloid.
Cannabinoid Receptors
Cannabinoid receptors are found in vertebrates (mammals, birds, reptiles, fish) and some invertebrates, with the exception of the Phyla Protozoa and Insecta. 129 These receptors participate in myriad essential biological processes such as cardiovascular regulation, central nervous system (CNS) regulation and plasticity, immune function, inflammation, metabolism, pain, bone growth, and allostasis. Cannabinoid agonists interact with the endocannabinoid system primarily via cannabinoid 1 receptor (CB1) and cannabinoid 2 receptor (CB2), which are coupled to G proteins. These G proteins are heterotrimeric, with α, β, and γ subunits. Cannabinoid stimulation of CB1 and CB2 leads to G protein activation and phosphorylation of guanosine diphosphate on the α subunit to guanosine triphosphate (GTP). This induces an allosteric change of the α subunit and disassociation from the rest of G protein. 10 G proteins are involved in many different signal transduction pathways, such as cyclic adenosine monophosphate (cAMP)/protein kinase A, ion channels, Rho GTPase, mitogen-activated protein kinase, phosphoinositide 3-kinase, and inositol triphosphate/diacylglycerol (DAG). Cannabinoid agonists result in Gi protein-mediated inhibition of adenylyl cyclase (Figure 1), leading to decreased cAMP, inhibition of voltage sensitive N-, P/Q-, and L-type calcium channels, D-type outward potassium channels; and activation of G protein-activated inwardly rectifying potassium channels. 10,11 Alternatively, CB1 receptor activation may in rare cases be coupled to Gs proteins, which stimulate adenylyl cyclase. 131
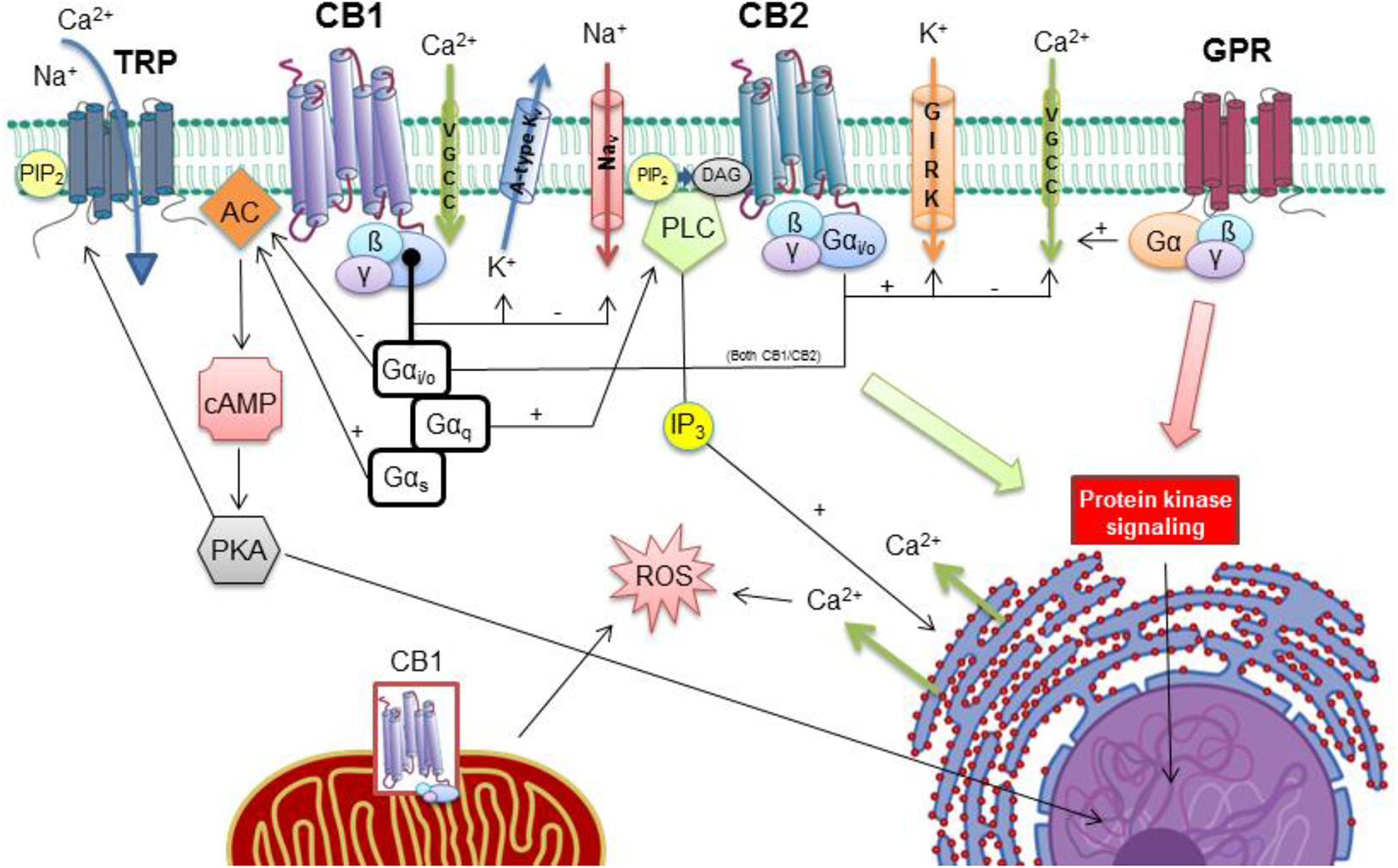
Cannabinoid (CB1 and CB2) and noncannabinoid receptors, voltage- and ligand-gated ion channels, and downstream cellular processes. Cannabinoid agonists interact with noncannabinoid receptors transient receptor potential (TRP), for example, TRP vanilloid-1 (TRPV1), and orphan G protein-coupled receptors (GPR), for example, GPR55/18. CB1 and CB2 activation result in modulation of voltage-gated calcium (VGCC), sodium (Nav), potassium (K v) channels, and the G protein-coupled inwardly rectifying potassium channel (GIRK). This may result in conduction disturbances within the heart and increased risk of arrhythmia. In CB1, the G protein α subunit has either inhibitory (Gαi/o) or stimulatory (Gαs) actions on adenylyl cyclase (AC), or it stimulates phospholipase C (PLC). Adenylyl cyclase catalyzes cyclic adenosine monophosphate production (cAMP) and protein kinase A (PKA) activation, which results in changes in gene expression in the nucleus. Phospholipase C catalyzes production of inositol triphosphate (IP3) and diacylglycerol (DAG) from phosphatidylinositol bisphosphate (PIP2), and IP3 results in calcium release from the endoplasmic reticulum and further changes in nuclear gene expression. Cannabinoid-induced protein kinase signaling and intracellular calcium release are responsible for vasoregulation in coronary arteries and arterioles and balance between myocardial inflammation, fibrosis, and apoptosis. Cannabinoids are lipophilic and may enter the cell by passive diffusion to interact with CB1 located in mitochondria leading to altered production of energy and reactive oxygen species (ROS). Reprinted and modified with permission from: Amin MR and Ali DW. 130 Copyright, Springer Nature (2019).
Cannabinoid receptors are found throughout the body, but CB1 predominates in the central and peripheral nervous system. Other anatomical locations for CB1 include the myocardium, vasculature, gastrointestinal tract, pulmonary parenchyma, integument, adipose tissue, and reproductive organs. Endocannabinoid activation of CB1 in the heart and blood vessels primarily leads to vasorelaxation, but exogenous cannabinoids, such as Δ9-tetrahydrocannabinol (THC), have more heterogeneous effects on different arterial types and may also result in vasoconstriction. 16 Cannabinoid 1 receptor is also present within mitochondria and regulate energy metabolism of the cell by reducing the activity in the electron transport chain. 24
Cannabinoid 2 receptor is found mainly in cells and organs of the immune system and regulates inflammatory responses. In the blood vessels, CB2 modulates endothelial adhesion and permeability for certain ligands and leukocytes but not vasoregulation. 10,16,25 More recently, an endothelial cannabinoid receptor (CBe) distinct from CB1 and CB2 has been identified and also has a role in vasorelaxation. 16 The pleiotropic effects of exogenous phytogenic and synthetic cannabinoids on the vascular system (Figure 2) may cause sudden, unpredictable vasoconstriction or vasodilation of coronary and/or peripheral arteries and arterioles, which increases theoretical risk for ACS and cardiac arrhythmia. 17

Balance between myocardial injury and repair mediated by cannabinoid receptor (CB) activation. Cannabinoids, which include endocannabinoids, phytocannabinoids, and synthetic cannabinoids, activate CB1 and CB2 with potential for myocardial injury or healing based on specific compound and dosage. Cannabinoids stored in adipose tissue from chronic use also represent an important source which may be released acutely during periods of stress, exercise, and fasting. Certain cannabinoids may inhibit metabolic pathways, including cyclooxygenases (COXs), cytochromes P450 (CYPs), fatty acid amide hydrolase (FAAH), and monoacylglycerol lipase (MAGL) and lead to supranormal levels of reactive oxygen species (ROS).
The Endocannabinoid System
The endogenous activators of cannabinoid receptors are arachidonic acid derivatives, the most well-defined being arachidonoylethanolamine (anandamide) and 2-arachidonyl glycerol. cannabinoid receptor 1 is localized to the presynaptic membrane, but endocannabinoids are produced postsynaptically in a calcium-dependent manner after neuronal depolarization. Endocannabinoids are not stored in vesicles like other neurotransmitters but are rather produced “on demand” by phospholipase C and DAG lipases following depolarization of the postsynaptic neuron. 132 Newly synthesized endocannabinoids then cross the synapse in a paracrine manner to activate presynaptic cannabinoid receptors. This negative feedback loop inhibits further neurotransmitter release by blocking calcium influx into the presynaptic neuron. In this manner, endogenous cannabinoids play an important role in the physiology and behavioral expression of hypothalamic-pituitary-adrenal (HPA) axis responses to stress. 39,40 Endocannabinoids have a short in vivo half-life and are rapidly metabolized into arachidonic acid and other metabolites through the activity of fatty acid amide hydrolase (FAAH), monoacylglycerol lipase (MAGL), cyclooxygenase 2 (COX-2), and lipoxygenases. 10
Physical and psychological stressors result in paraventricular nucleus activation within the hypothalamus, with release of corticotropin-releasing hormone, adrenocorticotropic hormone, and downstream adrenal stimulation. Catecholamines and glucocorticoids are thus released, which have short-term benefit to an organism responding to stress. However, protracted catecholamine and glucocorticoid secretion results in damaging side effects, such as immunosuppression and catabolism. The endocannabinoid system provides critical negative feedback mechanisms to regulate HPA axis activity and achieve allostasis or return to a normal state. 41 The balance provided by the endocannabinoid system in response to stress affects conditioned fear, memory, emotion, and response to pain. 39
Phytogenic and Synthetic Cannabinoids
Cannabis contains over 500 different chemical compounds, of which over 100 are classified as cannabinoids. 109 The psychoactive properties of the partial cannabinoid receptor agonist THC are responsible for the ongoing popularity of cannabis as a recreational drug since 5000 BC. 12 Modern-day selective breeding techniques of phytogenic cannabis and cultivation practices have tripled the THC content of Cannabis sativa over the past 2 decades, whereas the concentration of other phytocannabinoids remains essentially unchanged. 109 This increasing ratio of THC relative to other cannabinoids and terpenes present in cannabis which may have medical benefit, such as cannabidiol (CBD), may negate this “entourage effect” and have potential for increased cardiac and CNS toxicity. 4,110 In addition to CBD, other phytocannabinoids obtained from cannabis include cannabigerol and cannabichromene, but these are not intoxicants and have a wide spectrum of medicinal use (Figure 3), including treatment of pain, epilepsy, emesis, inflammation, and appetite. 111,112 To date, hundreds of THC analogues have been synthesized with much higher potency and affinity for CB1, such as nabilone, which is prescribed as an antiemetic for the treatment of chemotherapy-induced nausea and vomiting. Examples of nonprescribed or illicit synthetic cannabinoids include JWH-018 and CP-47,497 found in “Spice” and “K2” and AMB-FUBINACA. 114,115 Both phytogenic and synthetic cannabinoids with higher potency and CB1 affinity have the potential to overwhelm the endocannabinoid system and disrupt the balance of the autonomic nervous system. 41,113

Pharmacological actions and physiologic effects of nonpsychotropic, medicinal cannabinoids. Although Δ9-tetrahydrocannabinol (THC) is predominant, myriad other phytogenic and synthetic cannabinoids have potentially beneficial effects on a wide range of pathophysiological processes. The “Entourage Effect” of phytocannabinoids may even be cardioprotective, but this denouement has been compromised by high THC concentrations in modern-day cannabis and synthetics. 5-HT1A, 5-hydroxytryptamine receptor subtype 1A; Δ9-THCA, Δ9-tetrahydrocannabinolic acid; Δ9-THCV, Δ9-tetrahydrocannabivarin; CB1, cannabinoid 1 receptor; CB2, cannabinoid 2 receptor; CBC, cannabichromene; CBD, cannabidiol; CBDA, cannabidiolic acid; CBDV, cannabidivarin; CBG, cannabigerol; FAAH, fatty acid amide hydrolase; GABA, γ-aminobutyric acid; Id-1, inhibitor of differentiation/DNA binding; PPARγ, peroxisome proliferator-activated receptor γ; ROS, reactive oxygen species; TRPV1, transient receptor potential vanilloid type 1;. (+), direct or indirect activation; (−), antagonism; ↑, increase; ↓, decrease. Reprinted with permission from Izzo et al. 112 Copyright, Elsevier (2009).
Cannabinoid Receptors and the Autonomic Nervous System
In humans, the primary cardiovascular effects of exogenous phytogenic and synthetic cannabinoid agonists are a CB1-dependent increase in heart rate accompanied by a small increase in blood pressure and cardiac output, which is attributed to an increase in cardiac sympathetic nerve activity and a decrease in parasympathetic nerve activity. 26 This is the opposite effect to animal models, in which exogenous cannabinoids evoke a vasodepressor and bradycardic response. 27 Involvement and distribution of cannabinoid receptors in the peripheral nervous system and CNS, heart, and blood vessels are responsible for these responses and differences between species (Figure 4). Animal studies of CNS administration of cannabinoids into the medulla, cisterna magna, and periaqueductal gray evoke sympathoexcitatory and vagal inhibitory responses which can be prevented by CB1 antagonism. 27 -30 These areas within the CNS are highly integrative and involved in cardiovascular regulation and emotional responses. 27 For example, cannabinoid receptors are also found in the nucleus tractus solitarii of the medulla, where cardiac baroreceptor and chemoreceptor afferent nerves terminate. Cannabinoids inhibit the effects of certain central neurotransmitters such as acetylcholine, glutamate, γ-aminobutyric acid, and monoamines, leading to an increase in sympathetic nerve activity. 27 Cannabinoids also have significant effects on peripheral autonomic neurons, such as presynaptic inhibition of monoamines from postganglionic sympathetic neurons. 13,31 The balance of human and animal sympathovagal response to exogenous cannabinoid administration is highly dependent on type, dose, route, duration, and pharmacogenetic sensitivity to certain cannabinoids.

Tachyarrhythmia and acute coronary syndrome: Cannabinoid agonists such as Δ9-tetrahydrocannabinol (THC) cause sympathetic nervous system activation and parasympathetic nervous system (Vagus nerve) inhibition at low to moderate doses via cannabinoid 1 (CB1) and transient receptor potential (TRP) receptors in the hypothalamus, brainstem, circulation, and heart. Stimulation of sympathetic fibers to the heart and release of monoamines norepinephrine (NE), epinephrine (EPI), dopamine (DA), and serotonin (5-hydroxytryptamine, or 5-HT) from the paravertebral ganglia and adrenal glands results in increased cardiac chronotropy, inotropy, and sinoatrial (SA) and atrioventricular (AV) nodal dromotropy, which increases the risk of tachyarrhythmias. These effects and subsequent activation of the renin–angiotensin–aldosterone system (RAAS) result in cardiac and peripheral vasoconstriction, with increased afterload. These effects increase myocardial oxygen demand. Other vasoregulatory receptors modulated by cannabinoids include orphan G protein-coupled receptors (GPR), peroxisome proliferator-activated receptors (PPAR), 5-HT, and adenosine. Additional cannabinoid effects include alteration of nitric oxide (NO) and reactive oxygen species (ROS) within the coronary arterial endothelium, myocardium, and prothrombotic effects from platelet activation, further increasing the danger of ACS. Bradyarrhythmia and syncope: Acute exposure to higher cannabinoid agonist doses and/or chronic use with large adipose tissue stores may result in parasympathetic nervous system activation, which may result in bradyarrhythmias, syncope, and asystole. Evocation of the Bezold-Jarisch reflex via cannabinoid-activated cardiac TRP and 5-HT receptors may also contribute to this potentially fatal phenomenon.
Cannabinoid-Receptor Associated Arrhythmias
Initial human studies of the 1970s established that smoking cannabis results in an immediate increase in heart rate lasting approximately 1 hour with a substantial rise in serum norepinephrine concentration at 30 minutes. 3,8,32 Pretreatment with propranolol and rimonabant, an inverse agonist of CB1, blocks this response and thereby confirms sympathetic nervous system involvement. 3,8,32 -35 Involvement of the parasympathetic nervous system was first suggested after a human study reported atropine pretreatment resulted in an exaggerated heart rate following cannabis smoking and decreased P-wave amplitude. 3 A later electrophysiological study demonstrated THC administration enhanced sinus automaticity and facilitated sinoatrial and atrioventricular dromotropy, thus creating favorable conditions for arrhythmia formation. 133,36
Differences in regional myocardial neuronal distribution account for the variety of tachy- and bradyarrhythmias associated with acute and chronic cannabinoid use. Parasympathetic neurons predominate at or near the sinoatrial node whereas sympathetic neurons are concentrated in the ventricles. 37,42 Low to moderate doses of exogenous cannabinoids cause sympathetic stimulation with subsequent decrease in action potential duration, atrial refractory period, and alteration of myocardial electrophysiological properties, which favors automaticity and micro-reentry. 3,25,36,37,38 Higher doses drive parasympathetic stimulation, with increase in action potential duration and atrial refractory period favoring macro-reentry, which may predispose to bradyarrhythmias especially in younger individuals with higher baseline vagal tone. 43,44 Both autonomic mechanisms, however, increase the risk for tachyarrhythmias such as atrial fibrillation and paroxysmal supraventricular tachycardia. 44,45,70
The association of sympathetic overactivity and atrial fibrillation is well-established, but vagal-induced atrial fibrillation is a lesser-known phenomenon. 42,45 Vagal stimulation increases the heterogeneity of the action potential and atrial effective refractory period and regional variations within the atrium, which promotes re-entry tachyarrhythmias. 44,45,46 P-wave dispersion, or the time difference between the maximum and minimum of the P-wave, is a noninvasive measure of disorganized atrial repolarization and has been associated with increased risk of paroxysmal atrial fibrillation, panic disorders, and cardiovascular disease. 134 Higher P-wave dispersion has also been linked to synthetic cannabinoid use. 56,116 High doses of cannabinoids leading to vagal stimulation have the potential to provoke bradyarrhythmias including sinus arrest, syncope, atrioventricular block, and asystole as highlighted in several cases. 47 -53 Postural hypotension and associated vasovagal reactions have also resulted in dizziness and syncope. 50,51,53 -55
Cannabinoid use is associated with modest increases in blood pressure and enhanced left ventricular systolic function, but this response is blunted in chronic users. 18 -20,135 -137 Cannabinoid dysregulation of coronary circulation may be an important factor in the genesis of ACS and acute arrhythmias. Case reports describe a temporal association between cannabinoid use and ventricular tachyarrhythmias and implicate myocardial ischemia as a potential etiology. The ventricular arrhythmias described after cannabinoid use may result from diminished coronary microcirculatory flow with subsequent triggered activity in Purkinje fibers or micro-reentry in a small region of the posteroinferior left ventricle. 21 -23 During angiography, these patients often have slow reflow or no reflow from vasospasm of smaller coronary arterioles, 21,23 which resolves with administration of verapamil.
The QT interval, a marker of the ventricular action potential, represents the duration of ventricular depolarization and repolarization. Dispersion (QTd) is the maximum interlead difference in QT and is a marker for heterogeneity in ventricular excitability and recovery. 138 Prolongation of both the QT interval and QTd may be caused by myriad medications and electrolyte imbalance, such as magnesium deficiency. 42 Increased QT interval associated with synthetic cannabinoid use has been described in case reports and an animal study. 56 -59 Impaired myocardial repolarization is an important predictor of malignant ventricular arrhythmias and ACS. 139 Cannabinoids inhibit cardiac sodium, potassium, and calcium channels, resulting in QT prolongation and increased total dispersion of repolarization. 15,58,60,61 In particular, inhibition of the rapid component of the delayed rectifier potassium current, IKr is a major cause of ventricular arrhythmias such as Torsades de pointes. 140 These unique cation channel interactions are further detailed in the following section. Several cases of ventricular arrhythmias, including atrioventricular block, ventricular tachycardia, and fibrillation, induced by exogenous cannabinoid use have been published. 21,22,47,48,50,53,71,141 -145
Noncannabinoid Receptors
Endocannabinoids and exogenous cannabinoids, such as THC and CBD, are promiscuous, interacting with sundry noncannabinoid receptors with pleiotropic effects. These include orphan G protein-coupled receptors (GPRs), voltage- and ligand-gated ion channels, peroxisome proliferator-activated receptors (PPARs), transient receptor potential (TRPs) ion channels (TRPs), 5-hydroxytryptamine (5-HT) receptors, α2-adrenoceptor, glycine α1 and α1b receptors, adenosine membrane transporters, as well as metabolic enzymes phospholipase A2, LOX, COX-2, and various cytochrome P450 s (CYPs). 10,112 The diverse functional effects of these non-cannabinoid receptors likely play important roles in cannabinoid-associated ACS and cardiac arrhythmia.
G Protein-Coupled Receptors
Cannabinoids have been shown to interact with several distinct GPRs unrelated to the CB1 and CB2 GPRs, including GPR55 and GPR18. These orphan GPRs are mainly localized to vascular endothelium, and their activation results in vasodilation, increased membrane permeability, angiogenesis, and endothelial wound healing. 74 -76 Other locations include lymphoid tissues, lungs, brain, and reproductive organs. The most studied of these receptors is GPR55, which modulates calcium, potassium, and sodium currents resulting in membrane hyperpolarization within cardiac endothelial cells. 74 GPR55 is also involved in the control of adrenergic signaling within the heart, and its dysfunction is characterized by adverse ventricular remodeling and the development of cardiomyopathy. 77 The theoretical contribution of these noncannabinoid GPRs to the genesis of cannabinoid-induced cardiac arrhythmias and ACS may be from alteration of endothelial vasoregulation and membrane cation currents of the cardiac conduction system. 78
Ion Channels
Voltage- and ligand-gated ion channels are essential to each phase of the cardiac action potential and are implicated in many types of arrhythmia from autonomic nervous system overstimulation, electrolyte imbalance, certain drugs, and myocardial ischemia. 146 These channels are direct targets for cannabinoids as demonstrated by several animal studies. 62 Endocannabinoids have a protective effect against myocardial ischemia, reperfusion injury, arrhythmia, and cardiomyopathy. 147,148 Anandamide at various concentrations shortened action potential duration and amplitude in a study of rabbit sinoatrial nodes. 69 This research group also found anandamide suppresses calcium overload during myocardial ischemia in a murine model by inhibiting the sodium/calcium exchanger (I NCX). 66 An in vitro murine study demonstrated THC blocked voltage-gated sodium channels (Nav) and altered 3 parameters associated with the inward sodium current: Peak amplitude was decreased, whereas time to both peak and recovery were increased. 63 Experiments with anandamide and CBD have shown similar inhibition of Nav. 64,65 In addition to contributing to cardiac arrhythmias, Nav dysfunction is implicated in the pathophysiology of seizures and chronic pain. As a result, the inhibitory effect of cannabinoids on Nav highlights their potential for epilepsy treatment and analgesia. 62
Cannabinoids also directly interact with voltage-gated potassium channels (K v). The endocannabinoid anandamide and exogenous THC both decreased or completely inhibited human Kv1.5 and murine Kv1.2 channels in a dose-dependent fashion. 61,14 The Kv1.5 channels underlie the ultrarapid delayed rectifier current (I Kur), which is critical for determining the height and duration of the atrial action potential. Anadamide also inhibits adenosine triphosphate (ATP)-gated potassium channels (K ATP) in another in vitro study. 15 Various cannabinoid agonists block inwardly rectifying potassium channels (K ir) with downstream inhibition of calcium influx in murine oligodendrocytes. 67 Calcium channels are also unmediated targets for cannabinoids. Cannabinoid agonists, including THC, inhibit current through voltage-gated (Cav) and T-type calcium channels by altering deactivation and calcium entry during repolarization. 68 Anadamide directly suppresses myocardial L-type calcium channels in murine ventricles and calcium-mediated excitation–contraction, resulting in decreased myocardial contractility. 64 The net effect of these alterations is hyperpolarization of the cardiac membrane, with decreased amplitude and rate of rise of the action potential upstroke. 60 This alteration of cation membrane transport by cannabinoids represents a potentially important etiology for the inception of cardiac arrhythmias (Figure 5), in particular the Brugada phenocopy described in several published case reports. 55,70 -73

Overview of potential cannabinoid agonist electrophysiological effects on cardiac action potentials. A, Voltage- and ligand-gated ion channels at risk from cannabinoid agonist modulation and their temporal relation to action potential phase. Phase 0: depolarization from rapid inflow of sodium current (I Na) and slow inflow of calcium current, I Ca(L); phase 1: inactivation of sodium inflow and brief rapid potassium outflow (I to; I Kur in the atrium); phase 2: plateau and initiation of atrial repolarization from potassium outflow (I Kr and I Ks), continued calcium inflow, I Ca(L), and sodium–calcium exchange, INCX. This plateau is not seen with nodal action potentials. Phase 3: rapid repolarization from ongoing potassium outflow (I Kr and I Ks), closure of I Ca(L), and opening of I K1; phase 4: rest period (diastole) with restoration of ion balance from ATP-dependent sodium–potassium pump, sodium–calcium exchanger, and initiation of I KATP and I KACh in the atrium. B, Correlation of progressing action potentials with electrocardiogram. C, Dose-dependent Δ9-tetrahydrocannabinol (THC) effects on nodal action potentials.
Peroxisome Proliferator-Activated Receptors
Peroxisome proliferator-activated receptors are a family of ligand-activated transcription factors, and several in vitro experiments have provided evidence that cannabinoids are agonists for certain PPAR subtypes. 13 Peroxisome proliferator-activated receptor α, PPARγ, and PPARδ are expressed in arterial smooth muscle and endothelial cells, and cannabinoid activation leads to vasorelaxation that is dependent on production of nitric oxide (NO), hydrogen peroxide, and superoxide dismutase (SOD) activity. 79 Other PPAR effects include decreased endothelial expression of prothrombotic mediators and monocyte adhesion in response to inflammation. 80 Alteration of PPAR-associated cardiac and peripheral vascular regulation by cannabinoids may be a factor in the development of reflex tachycardia in response to sudden hypotension, other cardiac arrhythmias, and precipitation of ACS. 81
Transient Receptor Potentials
Transient receptor potential channels are a group of membrane proteins involved in the transduction of a variety of physical and chemical stimuli, including the sensation of temperature, pressure, pain, acidity, smell, taste, and vision. Cannabinoids act on certain TRP vanilloid (TRPV), TRP ankyrin (TRPA), and TRP melastatin channels. 12,82 The TRP channels are widespread throughout the CNS and peripheral nervous systems, heart, and blood vessels and are involved in autonomic nervous system regulation and control of vascular responses. Various cannabinoid agonists such as anandamide and THC activate TRPA1, TRPV1, and TRPV2 channels localized to perivascular sensory neurons and vascular endothelium. 12,83 The direct effects of cannabinoids on TRP-mediated regional and organ-level vasoregulation are inconsistent and may depend on external factors such as exercise and temperature as well as the type of cannabinoid, dosage, and duration of use. 19,84
Cannabinoids predominantly cause an acute vasodilatory response possibly through activation of TRPA1 and TRPV1 channels with release of calcitonin gene-related peptide, a potent vasodilator. 85,86 However, this response is not universal, as TRP-mediated vasoconstriction also may occur in coronary, cerebral, and peripheral arterial systems. 87,88 It is possible exogenous cannabinoids at supratherapeutic doses may cause acute coronary or cerebral vasoconstriction, leading to ACS and stroke. 9,89 Cardiac afferent TRPV1 receptors may serve as a molecular detector of myocardial infarction to activate cardiac nociceptors, resulting in chest pain. 90 The activation of TRPV1 and 5-HT3 receptors results in a teleological response, the Bezold-Jarisch reflex, to counter the sympathoexcitatory effects induced by myocardial ischemia. 91 This cardioprotective reflex includes bradycardia, vasodilation, and hypotension. Exogenous cannabinoids may induce the Bezold-Jarisch reflex in the absence of myocardial ischemia, resulting in syncope, bradyarrhythmia, ACS, and sudden death. 9,54
Monoamine Receptors and Adenosine
Several 5-HT receptor subtypes are expressed in the cardiovascular system and are involved in regulation of vascular tone. 92 Activation of the 5-HT1A receptor subtype is involved in neuromodulation of blood pressure and heart rate through peripheral vasodilation and vagal nerve stimulation. These effects of cannabinoids are antipodal, as certain cannabinoids have been shown to activate or antagonize the 5-HT1A receptor. 92,93 In in vivo animal models of stroke, CBD, and not THC, activation of 5-HT1A increased cerebral artery blood flow, with reduction in infarct damage. 94 -96 Other studies demonstrated stress-induced hypertension can be inhibited by CBD. 97,98 Certain cannabinoids are also 5-HT3 receptor antagonists, which may explain their antiemetic properties. 12,99,100
Adenosine is an endogenous purine nucleoside involved in myriad physiological processes. There are 4 adenosine GPR subtypes (A1, A2A, A2B, and A3). Adenosine is used therapeutically to terminate certain tachyarrhythmias and for arterial vasodilation during angiography. Adenosine activation of A1 leads to membrane hyperpolarization from increased potassium efflux via inward rectifier potassium channels (K ir) and decreased inward calcium current. 149 This results in a transient heart block in the atrioventricular node. Certain cannabinoids, such as CBD, also activate A1 receptors and have an antiarrhythmic effect. 101 Cannabinoid–adenosine receptor interactions are further complicated by the existence of heteromers, such as A2A-CB1, which inhibit neurotransmitter release and modulate cannabinoid signaling. 102,103 Although experimental evidence is nascent, it is feasible that exogenous cannabinoids at high doses and/or prolonged exposure may result in cardiac arrhythmia from deleterious effects on 5-HT and adenosine receptors.
Metabolic Enzymes
As mentioned earlier, endocannabinoids are synthesized “on demand” and rapidly metabolized. In contrast, exogenous cannabinoids such as THC are preferentially stored in adipose tissue, thus resulting in a long elimination half-life lasting weeks. Chronic use leads to accumulation of THC and its metabolites in adipose tissue. Lipolysis from stress, fasting, or exercise can acutely elevate THC blood levels even during several days of abstinence, which may increase risk of cardiac arrhythmia and ACS. 104 Exogenous cannabinoids undergo hepatic metabolism, and more than 80 metabolites have been identified. In humans, oxidation of THC occurs primarily via CYP2C9, CYP2C19, and CYP3A4 resulting in the active metabolite 11-hydroxy-Δ9-THC, which is also a CB1 agonist. 104
Several studies have demonstrated the vascular effects of some cannabinoids are mediated by their metabolic products. 104,105 Further complicating this system, phytogenic and synthetic cannabinoids inhibit several different CYPs. 106 The vasorelaxant effects of endocannabinoids can be reversed by FAAH, MAGL, COX-2, and CYP inhibition. 105 One example of this potential adverse side-effect is cannabinoid inhibition of the primary CYP of cardiomyocytes CYP2J2, which is responsible for the metabolism of the endocannabinoids into endocannabinoid epoxides (EET-EAs). These EET-EAs are cardioprotective vasodilators with additional anti-inflammatory and anti-thrombotic properties. 107,108 Acute exogenous cannabinoid intake could theoretically lead to an acute, unanticipated EET-EA shortage within the heart and increase risk for arrhythmia and ACS. Pharmacogenetics could also be a factor in aberrant cannabinoid metabolism, whereupon polymorphisms of certain metabolic enzymes lead to higher concentrations of inhibitory metabolites. 105
Reactive Oxygen Species, Smoking, and Prothrombotic Effects
Yet another essential endocannabinoid system function is regulation of reactive oxygen species (ROS) creation and scavenging mechanisms to maintain redox homeostasis via CB1, CB2, TRPV1, and GPR55 interaction. 107 Reactive oxygen species result from the mitochondrial electron-transport chain and oxygen-metabolizing enzymatic reactions, such as CYPs, NO synthase, and lipoxygenase, and mediate signal transduction from membrane receptors to activate various proteins and enzymes critical to cell function. Excess intracellular ROS damages membranes, proteins, nucleic acid, and mitochondria resulting in cell injury or death. In the cardiovascular system, excessive ROS results in vasospasm, ischemia, infarction, arrhythmias, atherosclerosis, cardiomyopathy, and apoptosis. 108 Reactive oxygen species levels are maintained by various antioxidant enzymes such as SOD, catalase, and glutathione peroxidase, and small molecular weight antioxidants, such as cysteine, vitamin C (ascorbic acid), and vitamin E (α-tocopherol). Exogenous cannabinoids, such as THC, have been shown to result in oxidative stress and apoptosis in several in vitro studies. 107,117 -119
The fine particulate matter (<2.5 µm in diameter) from smoking cannabis and synthetic cannabinoids stimulates the formation of ROS and the sympathetic nervous system similar to smoking tobacco. 119 A human study demonstrated that 1 minute of exposure to cannabis secondhand smoke impaired femoral artery flow-mediated dilation to a similar extent as tobacco, but recovery was considerably slower for cannabis. 120 Smoking cannabis also increases carboxyhemoglobin levels, resulting in decreased oxygen-carrying capacity. 121 This cannabis smoking-impaired myocardial oxygen demand to supply ratio reduces the time to onset of chest pain during exercise in patients with stable angina. 122,123 The combination of sympathetic stimulation, ROS generation, impaired vasodilation, and carboxyhemoglobin production promotes a situation favoring the genesis of ACS and cardiac arrhythmia.
Certain endocannabinoids at physiological concentrations contribute to platelet activation. 124 Exogenous cannabinoids such as THC may also have prothrombotic effects that may also impact risk for ACS and cardiac arrhythmia, even in young patients without cardiac risk factors. 9,125 Both CB1 and CB2 are present on the cell membrane of human platelets and in vivo exposure to THC results in alteration of membrane phospholipids, which led to increased rate of platelet lysis and adhesion in one study. 126 In another human study, THC increased the in vivo surface expression of glycoprotein IIb to IIIa and P selectin, which are both involved in platelet activation. 127 Δ9-Tetrahydrocannabinol enhanced in vitro human tissue factor expression in activated monocytes, contributing to a prothrombotic effect. 128 In addition to increased risk of ACS from potential procoagulant effects of THC, idiopathic thrombocytopenic purpura from synthetic cannabinoid use has been described in case reports. 150
Conclusion
The use of phytogenic and synthetic cannabinoids has resulted in ACS, tachy- or bradyarrhythmias, and cardiac arrest in otherwise healthy patients with no underlying cardiac risk factors. A complex, synergistic interplay between autonomic dysregulation, altered cellular ion flux, impaired coronary microcirculation, and pharmacogenetic risk factors are the likely causes for these deleterious clinical events rather than any sole mechanism.
Cannabinoid 1 receptor, CBe, TRP, PPAR, GPR, monoamine, and adenosine receptors all regulate vascular tone. Endocannabinoids typically induce vasodilation, but exogenous cannabinoids cause unpredictable vasoconstriction or vasodilation in coronary arteries and arterioles. Simultaneously, myocardial oxygen demand increases due to CB1-mediated surge in sympathetic tone, while the associated tachycardia reduces diastolic coronary perfusion time. Coronary microvascular hypoperfusion predisposes to the development of arrhythmias and is compounded by cannabinoid-induced increase in sinoatrial node automaticity and dromotropy. Inhibition of cardiac sodium, potassium, and calcium conductance induces repolarization abnormalities leading to the development of early afterdepolarizations and subsequent ventricular tachyarrhythmia. Syncope and bradyarrhythmia from cannabinoid intoxication result from increased atrial parasympathetic tone as well as TRPV1-mediated activation of the Bezold-Jarisch reflex.
The optimal treatment of patients who use cannabinoids and present with cardiac arrhythmia remains to be determined. Electrical synchronized cardioversion is the first-line treatment for patients with hemodynamic instability, as is asynchronous cardioversion for patients with pulseless ventricular tachycardia or ventricular fibrillation. The pharmacological action of commonly used antiarrhythmic agents, such as amiodarone, parallels cannabinoid inhibition of sodium, potassium, and calcium conductance and theoretically may worsen a patient’s status. The short-acting β1 adrenoreceptor-specific antagonist esmolol may be advantageous for treatment of tachyarrhythmias. Patients who exhibit bradyarrhythmias or third-degree atrioventricular-nodal blockade should receive the parasympatholytic drug atropine as initial therapy. If pacing is required due to hemodynamic instability, external or transvenous pacing is the preferred approach. Based on published clinical cases, these conduction abnormalities usually resolve within 24 hours, making permanent pacemaker placement unnecessary. 151
Cardiotoxic effects remain an overall rare event despite widespread use of cannabis. Cannabis potency, cannabinoid ratios, cannabis use patterns, and individual patient pharmacogenetic factors, such as metabolic enzyme expression, likely render a small proportion of cannabis users susceptible. Cessation of cannabis use reduces patients’ risk to population baseline, and clinicians should counsel their at-risk patients regarding abstinence.
Footnotes
Author Contribution
J.R.R. conducted the relevant literature research, created and/or modified the figures and tables, wrote the manuscript, and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
