Abstract
Introduction:
Recent evidence suggests that transcriptional reprogramming is involved in the pathogenesis of cardiac remodeling (cardiomyocyte hypertrophy and fibrosis) and the development of heart failure. 5-Azacytidine (5aza), an inhibitor of DNA methylation approved for hematological malignancies, has previously demonstrated beneficial effects on cardiac remodeling in hypertension. The aim of our work was to investigate whether pressure overload is associated with alterations in DNA methylation and if intervention with low-dose 5aza can attenuate the associated pathological changes.
Methods and Results:
C57Bl6/J mice underwent surgical constriction of the aortic arch for 8 weeks. Mice began treatment 4 weeks post-surgery with either vehicle or 5aza (5 mg/kg). Cardiac structure and function was examined in vivo using echocardiography followed by post mortem histological assessment of hypertrophy and fibrosis. Global DNA methylation was examined by immunostaining for 5-methylcytosine (5MeC) and assessment of DNA methyltransferase expression. The results highlighted that pressure overload-induced pathological cardiac remodeling is associated with increased DNA methylation (elevated cardiac 5MeC positivity and Dnmt1 expression). Administration of 5aza attenuated pathological remodeling and diastolic dysfunction. These beneficial changes were mirrored by a treatment-related reduction in global 5MeC levels and expression of Dnmt1 and Dnmt3B in the heart.
Conclusion:
DNA methylation plays an important role in the pathogenesis of pressure overload-induced cardiac remodeling. Therapeutic intervention with 5aza, at a dose 5 times lower than clinically given for oncology treatment, attenuated myocardial hypertrophy and fibrosis. Our work supports the rationale for its potential use in cardiac pathologies associated with aberrant cardiac wound healing.
Introduction
Heart failure (HF) is becoming an ever more prevalent condition in the general aging population as risk factors that promote disease progression such as hypertension, obesity and diabetes continue to be under managed. 1,2 Persistent injury or exaggerated wound healing in response to these risk factors can lead to aberrant cardiac tissue remodeling that impacts cardiac function. Two prominent remodeling features that are now accepted as key determinants of the clinical course of HF 3,4 are enlargement of myocyte size (cardiac hypertrophy) and increased extracellular matrix deposition (cardiac fibrosis). Although anti-hypertensive treatments that antagonize the renin-angiotensin-aldosterone system (RAAS) (e.g. ACE inhibitors or ARBs) have demonstrated anti-remodeling action and benefit for HF patients, several clinical studies have demonstrated considerable mortality rates in patients receiving anti-RAAS medication. 5,6 As such, there remains a need for therapies that target aberrant remodeling processes in the heart.
Cardiac remodeling is associated with transcriptional reprogramming due to alterations in mechanisms that regulate gene expression. 7,8 Considering epigenetic modifications play a central role in controlling chromatin structure and gene expression, it is no surprise that alterations in these regulatory mechanisms contribute to HF pathogenesis. 9 In recent years, several studies have identified altered DNA methylation signatures in cardiac tissue from HF patients 7,10 -13 and in pre-clinical models of cardiac dysfunction. 14 -16 DNA methylation involves the addition of methyl groups to cytosine residues already incorporated into DNA sequences. The reaction is catalyzed by the DNA methyltransferase (Dnmt) enzymes and results in the formation of 5-methylcytosine (5MeC). This generally causes transcriptional repression as it can prevent binding of transcription factors to the DNA or lead to chromatin condensation. 17
DNA methylation is an attractive target for therapy as this epigenetic process can be pharmacologically influenced by inhibiting Dnmt activity using agents such as 5-azacytidine (5aza) and its structural analog 5-aza-2-deoxycytidine, that are clinically approved for the treatment of myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). 18 We have previously highlighted the potential involvement of DNA methylation in chronic hypertension-induced cardiac dysfunction as treatment with the Dnmt inhibitor, 5aza, was shown to attenuate cardiac hypertrophy and fibrosis in the spontaneous hypertensive rat (SHR) model. 19 In the oncology setting 5aza treatment generally involves daily infusions of 75 mg/m2 for 7 days. The FDA consider 5aza to be well tolerated but at these high doses there is clear evidence of side effects such as nausea and weight loss. Before a strong case for repurposing 5aza can be made it is important to establish whether low-doses are effective at reducing pathological cardiac remodeling.
In the present study, we investigated whether DNA methylation is altered in the heart in response to acute pressure overload injury and whether pharmacologically targeting this epigenetic process with low-dose 5aza could attenuate the associated cardiac hypertrophy and fibrosis.
Methods
Animals
Male C57BL6/J wild type (WT) mice were purchased from Envigo UK (formerly Harlan) and maintained in a specific pathogen free environment. They were fed laboratory rodent chow and water ad libitum. Mice were maintained on a 12-hour light/dark cycle. All animals used in the study received humane care, and the study protocol complied with the institution’s guidelines and was approved by the local Animal Research Ethics Committee (Protocol number: P-13-69) and Health Products Regulatory Authority (HPRA) (AE18982/P025).
5-Azacytidine Treatment of a Murine Model of Pressure Overload
Transaortic constriction was used to induce pressure overload in 8-10 week old male C57BL6/J mice. 20 Briefly, surgical constriction was carried out under anesthesia, induced with 4% isofluorane vapor in oxygen, which was reduced to 2% for maintenance. Once anaesthetized, animals were placed in a supine position and temperature was maintained at 37 °C using a heating pad. Upon confirmation of an adequate plane of anesthesia and loss of pedal reflex, surgery was commenced. A small incision was made at the level of the suprasternal notch, the thymus retracted, and a cut made in the proximal portion of the sternum allowing visualization of the aortic arch. The arch was cleared of any fat tissue and once clean, a silk suture was passed under the aorta between the origin of the right innominate and the left common carotid artery. A 27-gauge needle (0.4 mm outside diameter) was placed next to the aortic arch and the suture snugly tied around the needle and the aorta. The skin was sutured closed and mice were allowed to recover in a heating chamber at 37 °C until full consciousness was reached. Post-operative pain management was done with a single pre and post-operative dose of buprenorphine (0.05 mg/kg). The sham procedure was carried out in an identical manner except that the aorta was not constricted. After 4 weeks TAC animals were randomly allocated to receive treatment with 5aza at a dose of 5 mg/kg or vehicle control (DMSO-PBS) by intraperitoneal injection (n = 6 per group). This dose was determined to be equivalent to a human dose of 15 mg/m2 using the FDA conversion calculators (as described in 21 ). Treatment was re-administered every 4 days until the end of the study at 8 weeks post-surgery. For organ harvesting, animals were humanely killed by exsanguination while under 4% isoflurane anesthesia and the heart was removed immediately, rinsed in PBS, weighed and cut in half with the apex side fixed with 10% formalin (Sigma) and remaining cardiac tissue snap frozen in liquid nitrogen for molecular analysis.
Quantitative Real-Time Polymerase Chain Reaction
RNA was isolated from frozen mouse myocardial tissue stored in Tissue Reagent (Qiagen) and extracted using the RNeasy fibrous mini kit according to the manufacturer’s instructions (Qiagen). RNA quality and concentration were then determined by spectrophotometry (Nanodrop-Thermo Scientific). First strand complementary DNA (cDNA) synthesis was carried out using SuperScript II RT (Invitrogen). Quantitative real-time polymerase chain reaction (QPCR) primers were designed so that one of each primer pair was exon/exon boundary spanning to ensure only mature messenger RNA (mRNA) was amplified.
Primer sequences used are as follows:

QPCR was performed using Platinum SYBR Green qPCR SuperMix-UDG (Invitrogen). Amplification and detection were carried out in duplicate with Mx3000P System (Stratagene). The PCR cycling program consisted of 40 three-step cycles of 15 seconds/95 °C, 30 seconds/TA, and 30 seconds/72 °C. To confirm signal specificity, a melting program was carried out after the PCR cycles were completed. Relative fold change in gene expression was calculated with the ΔΔCT method with Gapdh used as a reference gene. GAPDH was validated as a suitable housekeeping gene by assessing expression across all groups (treatment with 5aza had no impact on GAPDH expression—data not shown).
Echocardiography
Cardiac structure and function was assessed using the Vevo 770 In-vivo Micro-Imaging System (Visualsonics) equipped with Real-Time Micro Visualization 707B transducer (30 MHz). During the procedure, the animals were under inhaled anesthesia (Isoflurane 2%) and body temperature was maintained using a heat lamp. M-mode and 2-dimensional (2D) images were obtained in the parasternal short-axis view. The interventricular septal (IVS) thickness, posterior wall (LVPW) thickness, and left ventricular diameter were measured in systole and diastole at the tips of the papillary muscle. Measurements were taken over 3 consecutive cardiac cycles and averaged. Relative wall thickness was calculated by the following formula (IVSd + LVPWd/LVIDd). 22 LV mass was calculated according to Devereux’s formula and indexed to tibia length (LVMi). Ejection fraction was calculated by the Teicholz formula. 23 For assessment of diastolic function, the mouse was positioned backward in the Trendelenburg position to obtain an apical 4 chamber view to provide an adequate angle for Pulsed Wave (PW) Doppler of the mitral valve. All echocardiographic data is represented by the mean of 3 measurements on sequential cardiac cycles per animal assessed. All image analysis was conducted in a blinded fashion.
Cardiomyocytes Size Measurement
Relative cardiomyocyte size was quantified in 5 µm thick hematoxylin and eosin (H&E)-stained left ventricular sections. Cell area was measured in transverse section by planimetry in the presence of a centrally located nucleus. Two independent observers were blinded, and 5 fields of 10 myocytes per slide (50 cells in total per section) were measured using the Aperio digital image analysis system (Aperio ScanScope XT Slide Scanner; Aperio Technologies).
Picrosirius Red Staining and Automated Digital Quantification
Cardiac tissue sections (5 µm) were deparaffinized and rehydrated prior to incubating with 0.2% phosphomolybdic acid for 2 minutes. After rinsing in distilled water, the slides were stained with picrosirius red (Direct Red 80 dissolved in picric acid, Sigma) for 90 minutes. Finally, the slides were placed in 0.4% HCl for 2 minutes, 70% ethanol for 45 seconds, dehydrated, and cover-slipped for analysis. The degree of collagen deposition was quantified by automated digital image analysis (Aperio ScanScope XT Slide Scanner; Aperio Technologies) at 20× magnification.
Automated image analysis was performed using Imagescope (Aperio). A positive pixel count algorithm was used to automatically quantify the area occupied by the dark pink stain colors representing collagen within each scanned slide image. To detect the dark pink color of collagen with picrosirius red, a hue value of 0.8 was specified. The hue width value of 0.5 was used to allow inclusion of a moderate range of color shades. Quantification of perivascular collagen deposition was carried out on 6-8 vessels in cross section per cardiac section. Collagen positivity values for each vessel were normalized to the corresponding vascular luminal area.
5-Methylcytosine Staining
Cardiac sections (5 µm thick) were deparaffinized, rehydrated and underwent antigen retrieval using 1X Trilogy solution (Cell Marque) deparaffinized and rehydrated with xylene and an alcohol gradient. Endogenous peroxidases were blocked by incubation in 3% H2O2 for 10 minutes. Sections were denatured by incubation in 1 M HCL for 2 hours at 37 °C. Tissue sections were blocked for 30 minutes in serum-free protein block (DAKO) followed by incubation with primary anti-5-methylcytosine (5MeC) (Eurogentec, 1:500) for 1 hour at room temperature. Following washes in 0.1% TBS Tween, The EnVision Detection System Kit (DAKO) was used to complete the staining process which contains a pre-diluted HRP-conjugated secondary antibody and 3,3′-diaminobenzidine (DAB) peroxidase chromogen and substrate solution. Quantitative analysis of 5MeC staining was carried out the Aperio Imagescope Nuclear Algorithm V9 which grades the levels of nuclear 5MeC staining across the same area of LV tissue section. Strong positive staining was set at a threshold value of 160. Cytoplasmic threshold was set at 230. Hematoxylin was used as the nuclear counter-stain.
Statistical Analysis
All statistical analyses were performed using Graphpad Prism software (Version 6). Normally distributed data are expressed as mean and standard deviation of the mean (SD), while non-normally distributed data are presented as medians and interquartile range (IQR). For normally distributed data, statistical significance was determined by ANOVA with correction for multiple post hoc comparisons by Holms-Sidak step-down. For non-normally distributed data, statistical significance was determined by Kruskal-Wallis test with Dunn’s post-hoc test for multiple comparisons. A repeated measure 2-way ANOVA was used to assess differences in weight gain between treatment with vehicle and 5aza over the 4-week treatment period. Values of P < .05 were accepted as statistically significant.
Results
Sustained Pressure Overload-Induced Cardiac Remodeling Is Associated With Increased DNA Methylation
Mice subjected to transaortic constriction (TAC) displayed a significant induction of pathological cardiac remodeling which was persistent until the end of the study (8 weeks post-surgical constriction). Cardiac hypertrophy was apparent in mice that were subjected to 8 weeks TAC with significant increases in heart weight (Figure 1A), indexed left ventricular mass (LVMi) and relative wall thickness (Figure 1B and C), cross sectional area of myocytes (Figure 1D) and genetic markers (Figure 1E-G), all upregulated relative to the Sham surgical control. Cardiac fibrosis was present after 8 weeks TAC, with significant increases in collagen 1a1 mRNA expression and perivascular collagen deposition (Figure 1 H-J). Evidence of diastolic dysfunction was present after 8 weeks of TAC with a significant decrease in the E/A ratio (Figure 1K) and increase in isovolumetric relaxation time (IVRT) (Figure 1L). In response to 8 weeks of TAC, we found that there was a significant increase in the percentage of strongly positive nuclei staining for 5-methylcytosine (5MeC, Figure 1N), suggesting a global increase in cardiac DNA methylation. Interestingly, this increased global DNA methylation was mirrored by an upregulation of Dnmt1 (Figure 1O, P < .01) but not Dnmt3A or Dnmt3B (Figure 1P, P = .192 and 1Q, P = .08).
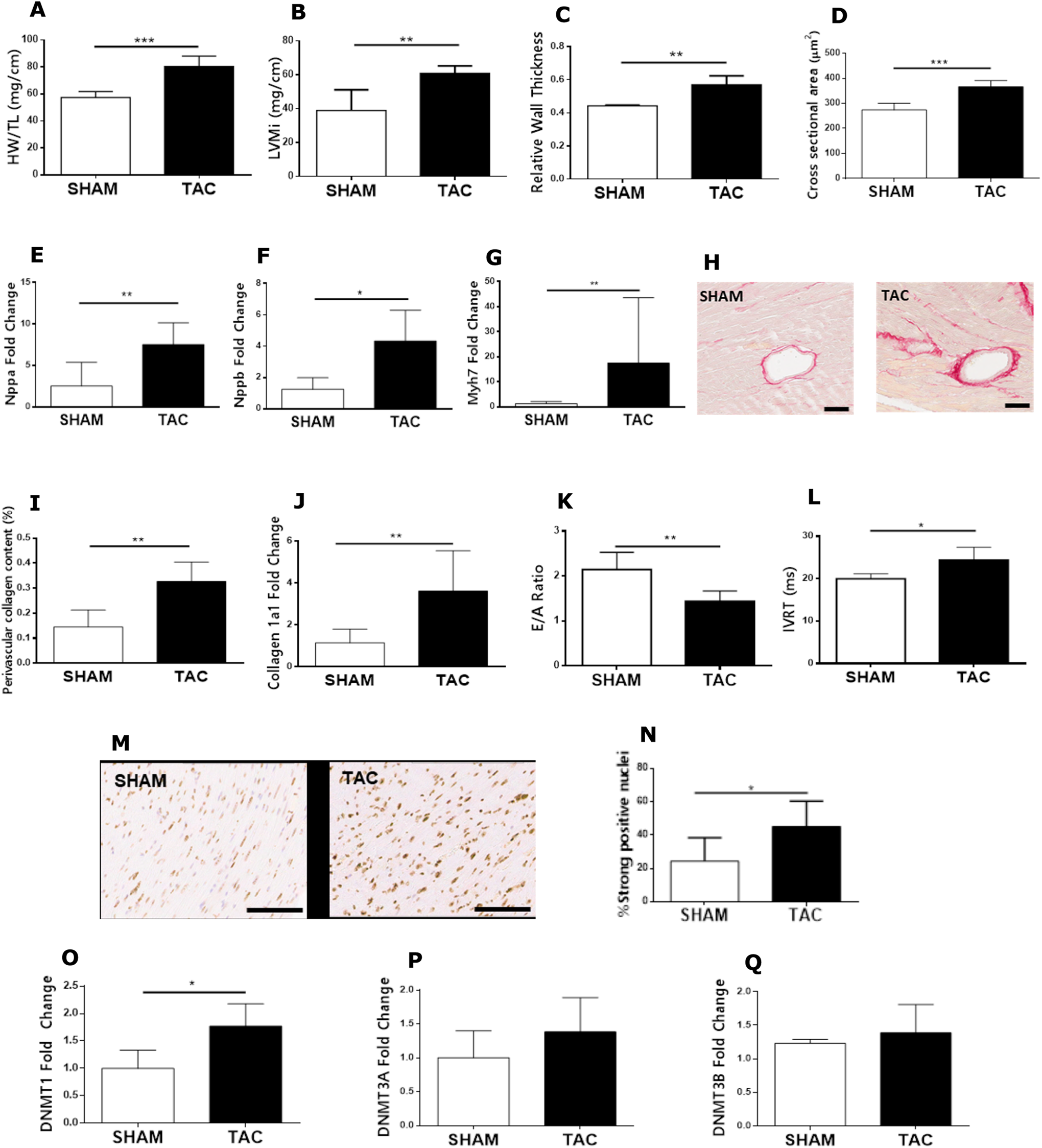
Established cardiac remodeling induced by pressure overload is associated with increased DNA methylation. (A) Heart weight (indexed to tibia length) measured at post mortem. (B) Left ventricular mass (indexed to tibia length; LVMi) and (C) relative wall thickness at 8 weeks after transaortic constriction (TAC) or sham surgery measured by echocardiography. (D) Myocyte cross sectional area quantified in hematoxylin and eosin stained cardiac sections. Myocardial mRNA levels of (E) Nppa, (F) Nppb and (G) Myh7 were examined by qPCR analysis. (H) Representative images of picrosirius red-stained perivascular collagen deposition (pink staining, scale bar = 100 μm) which was subsequently quantified (I) by application of the positive pixel count algorithm (indexed to luminal area). (J) Myocardial mRNA levels of collagen 1a1 from murine hearts. Parameters of diastolic function, (K) E/A ratio and (L) isovolumetric relaxation time (IVRT), were examined by pulse-wave Doppler imaging. Global DNA methylation was examined by (M) Representative immunohistochemical staining for nuclear 5-methylcytosine (brown staining; scale bar 100μm) where (N) strong positive staining was quantified in cardiac sections by application of the Aperio nuclear algorithm V9 and myocardial mRNA expression of (O) Dnmt1, (P) Dnmt3a and (Q) Dnmt3b. Data are expressed as mean ± standard deviation and median ± interquartile range (C, G, Q) with n = 6 for each group. *P < .05, ** P < .01, ***P < .001.
Targeting DNA Methylation With Low Dose 5-Azacytidine Attenuates Pressure Overload Induced Cardiac Hypertrophy
Due to the increase in global cardiac DNA methylation in response to pressure overload induced by TAC, we sought to investigate whether inhibiting DNA methylation could attenuate the established pathological cardiac hypertrophy and fibrosis. To investigate this concept, we treated mice with TAC induced pressure overload with a low dose of the nucleosidal DNA methylation inhibitor, 5aza, every 4 days for 28 days. Compared to the vehicle treated controls, 5aza-treated mice displayed a significant reduction in the parameters associated with pressure overload-induced cardiac hypertrophy such as lower heart weight (Figure 2A), indexed LV mass (Figure 2C), myocyte cross sectional area (Figure 2E) and myocardial gene expression of Nppa (Figure 2F). 5aza treated animals also displayed a reduction in relative wall thickness, but this failed to reach a level of statistical significance (P = .09). Throughout the course of the study, body weight was carefully measured to examine any potential adverse drug effects. Over the 4 week drug treatment period, there was no significant effect on body weight observed with treatment of 5aza (Figure 2G).

Effects of low dose 5-azacytidine (5aza) treatment on parameters of cardiac hypertrophy and body weight. (A) Overall heart weight (indexed to tibia length) was measured at study endpoint. Echocardiography was used to examine (B) relative wall thickness and (C) left ventricular mass (indexed to tibia length; LVMi). (D) Representative images of hematoxylin and eosin stained cardiomyocytes (scale bar = 50 µm) with (E) cross sectional area quantified. (F) Quantification of myocardial mRNA levels of Nppa in vehicle and 5aza treated murine hearts. (G) Body weight profile of animals treated with vehicle or 5 mg/kg 5-azacytidine over the 8 week study period. Data are presented as mean ± standard deviation and median ± interquartile range (B), with n = 6 for each group. *P < .05, ** P < .01, ***P < .001.
5-Azacytidine Treatment Reduces Cardiac Fibrosis and Improves Diastolic Function in Mice With Pressure Overload
The impact of 5aza treatment on cardiac fibrosis was examined. Histological analysis of perivascular collagen deposition was assessed by picrosirius red staining of cardiac sections followed by automated digital image analysis. Compared to the vehicle control, perivascular collagen content was significantly reduced in the hearts of 5aza-treated mice (Figure 3A). This decrease in collagen deposition was also mirrored by a reduction in levels of myocardial collagen 1a1 and collagen 3a mRNA (Figure 3B and C). With a reduction in pathological remodeling with 5aza treatment, we examined whether treatment with 5aza had any impact on improving cardiac function. Although within the normal range, there was no difference in ejection fraction measured for either treatment group (Figure 3D). Interestingly, mice that received 5aza demonstrated significant improvements in parameters of diastolic function, with elevated E/A ratio and reduced IVRT values (Figure 3E and F) compared to that of the vehicle treated group.

Effect of low dose 5-azacytidine (5aza) treatment on myocardial collagen deposition and cardiac function. (A) Perivascular collagen deposition was quantified by application of the positive pixel count algorithm (indexed to luminal area). Gene expression of (B) collagen 1a1 and (C) collagen 3a quantified by qPCR analysis in myocardial tissue from vehicle and 5aza treated mice. Echocardiography was used to measure cardiac function at study endpoint examining (D) ejection fraction by M-mode imaging and both (E) E/A ratio and (F) Isovolumetric relaxation time (IVRT) by pulse-wave Doppler imaging in vehicle and 5aza treated mice. Data are presented as mean ± standard deviation and median ± interquartile range (C) with n = 6 for each group. *P < .05, ** P < .01.
5-Azacytidine Treatment Reduces Increased Cardiac DNA Methylation Associated With Pressure Overload
Since 5aza is a known inhibitor of DNA methylation, we examined whether 5aza treatment could influence the increased cardiac DNA methylation seen in response to pressure overload. Compared to the vehicle-treated group, there was a significant reduction in cardiac cell nuclei staining strongly positive for 5MeC (Figure 4A and B). Interestingly, this reduction in nuclear 5MeC staining was mirrored by a significant decrease in gene expression of both Dnmt1 and Dnmt3b in mice treated with 5aza (Figure 4C and E). There was no difference in mRNA levels of Dnmt3a between the 2 treatment groups (Figure 4D).
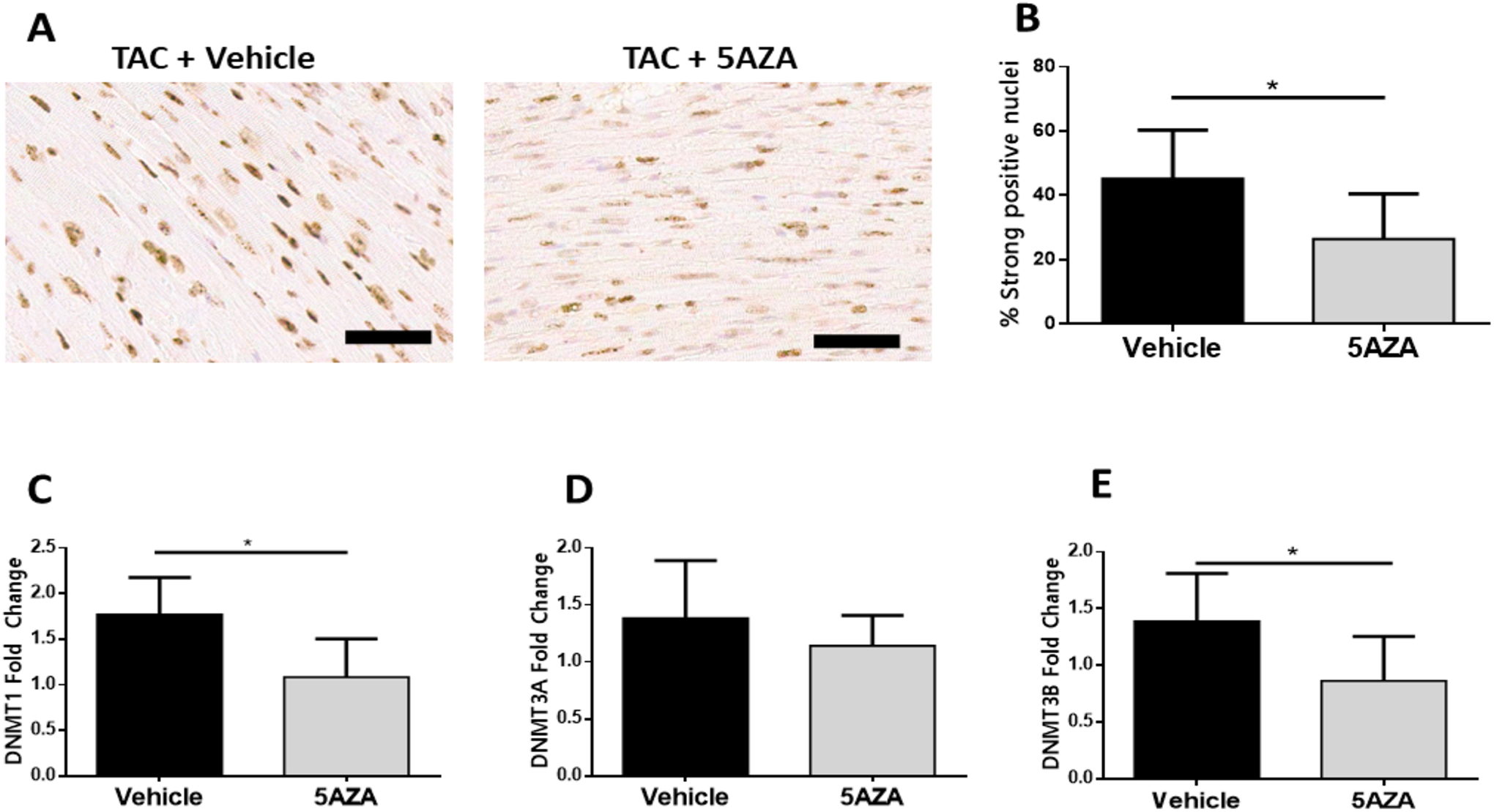
The impact of 5azacytidine treatment on DNA methylation in the pressure overloaded heart. (A) Representative images of myocardial tissue immunohistochemically stained for 5-methylcytosine (nuclear counterstain hematoxylin) from vehicle and 5aza treated mice with pressure overload (scale bar = 50 μm). (B) The percentage of cells staining strongly positive for 5MeC was quantified in cardiac sections by application of the Aperio nuclear algorithm V9. Gene expression of (C) Dnmt1, (D) Dnmt3a and (E) Dnmt3b in mice which had pressure overload and treated with either vehicle or 5 mg/kg 5aza. Data are presented as mean ± standard deviation and median ± interquartile range (E) with n = 6 for each group. *P < .05.
Discussion
Pathological left ventricular (LV) remodeling is one of the hallmarks of pressure overload strain on the heart. These include structural, metabolic and functional changes in the myocardium associated with hypertrophy of cardiomyocytes, increased extracellular matrix (ECM) deposition and inflammation. 4 Sustained activation of these cellular processes ultimately results in cardiac dysfunction and the development of HF. Most HF patients have underlying insidious comorbidities, such as hypertension, with aberrant remodeling and dysfunction preceding HF diagnosis by up to a decade(s). While therapeutic advances in HF have been encouraging, the outlook for patients with HF remains a concern as roughly half of all patients die within 5 years of diagnosis. 24 Therefore, there is a great need to develop novel therapeutic strategies to specifically target aberrant cardiac remodeling for the prevention of HF.
Global DNA methylation analysis and gene-targeted studies have demonstrated that significant alternations in DNA methylation contribute to the onset and development of disease pathologies including cancer, neurological diseases and more recently HF. 10,11,13 It is perceived that injury to the myocardium, whether it be an acute ischemic injury as a result of myocardial infarction, pressure overload due to acute aortic stenosis or chronic hypertension, or metabolic dysfunction due to diabetes and obesity, drives dysfunctional changes to the DNA methylation machinery in cardiac cells leading to aberrant remodeling in the heart. Indeed, our study has highlighted increased Dnmt1 expression and a global increase in myocardial DNA methylation associated with pathological cardiac remodeling (robust myocyte hypertrophy and increased perivascular fibrosis) after 8 weeks of pressure overload. Although considered to be involved predominately in maintenance methylation, it is becoming apparent that Dnmt1 may itself possesses de novo methylation activity. 25 Indeed, studies are starting to highlight alterations in Dnmt1 levels in response to cardiac injury, especially in states of metabolic stress. Dnmt1 expression was found to be elevated in cardiomyocytes exposed to high levels of glucose, 26 homocysteine, 27 and most recently an environmental pollutant, phenanthrene. 28 This increased Dnmt1 expression was accompanied by increased oxidative stress and myocyte hypertrophy. 27,28 Similarly, increased global DNA methylation and expression of Dnmt1 has been reported in cardiac fibroblasts exposed to low oxygen for 8 days, mirrored with acquisition of a pro-fibrotic cellular phenotype. 29 It seems likely, in the context of our study, that pressure overload-induced cardiac injury promotes a change in the methylation status in murine cardiac cells that contributes to driving pathological remodeling in the heart. This provided the rationale to examine the therapeutic potential of 5aza in pressure overload cardiac injury.
Although 5aza has received regulatory approval for the treatment of hematological malignancies, its use, at the doses that are used in clinical oncology (daily infusions of 75 mg/m2 for 7 days) is associated with a variety of side effects. These include gastrointestinal upset, weight loss and myelosuppression. Considering nucleosidal DNA methylation inhibitors have been shown to maintain their demethylating ability at lower doses, 30 we set out to investigate whether the beneficial effects of 5aza on cardiac remodeling could be demonstrated at significantly lower doses than those used in oncology, and at a lower dose than used in our previous work in the hypertensive rat model. 19 Our results demonstrated that administration of 5aza every 4 days at a dose of 5 mg/kg, which according to FDA calculators is equivalent to a dose of 15 mg/m2 in humans and thus 5 times lower than what is clinically given to MDS and AML patients, was well tolerated (no impact on body weight profile) and significantly attenuated pathological cardiac hypertrophy and fibrosis induced by TAC. The anti-hypertrophic and anti-fibrotic actions of 5aza were associated with reduced LV isovolumetric relaxation times and increased E/A ratios suggesting a potential improvement in diastolic function, but there was no impact on ejection fraction, which remained normal. Taken together with our previous study, that demonstrated that treatment of SHRs with 10 mg/kg 5aza actually lead to improved ejection fraction, these data suggest that treatment with low-dose 5aza may lead to improved cardiac function in the setting of pressure overload. This is important because a recent study identified 4 oncology cases (out of a database of over 9000 adverse events) where high-dose 5aza treatment was associated with alterations in ejection fraction and temporary symptoms of heart failure. 31 Unfortunately it is impossible to know whether the cardiac events reported were caused by 5aza treatment or related to the ongoing disease process. For example, myelodysplastic syndromes are associated with a complex array of systemic complications such as chronic anaemia 32 and iron overload. 33 Iron overload can impair cardiac function through increased oxidative stress and induction of iron-related endothelial cell dysfunction. 34,35 It has also been shown that patients with myeloid malignancies experience systemic inflammatory and autoimmune manifestations which were found to be strongly associated with an increased frequency of cardiovascular complications. 36 On balance we acknowledge a slight risk of cardiac complications in complex MDS patients that are being treated with high-dose 5aza but believe that our data support the use of low-dose 5aza in the context of pressure overload.
The anti-remodeling effects seen with 5aza treatment in the heart were associated with a reduction in global DNA methylation in myocardial cells mirrored by lower Dnmt1 and Dnmt3b expression. Demethylation by 5aza is associated with its ability to form tight covalent complexes with Dnmt enzymes, preventing their activity and promoting their degradation. 18 While we cannot conclusively determine the mechanism by which 5aza treatment led to a reduction in mRNA expression of both Dnmt1 and 3b, we speculate that this finding could be a result of increased methylation of the Dnmt genes 37 or indirectly related to effects on other signaling pathways or non-coding elements such as reactivation of miRNAs 28 that modulate Dnmt expression.
Injury to the heart as a result of increased afterload evokes a diverse and complex array of cellular responses involving cardiomyocytes and non-muscle cells, including fibroblasts, endothelial cells and immune cells in order to maintain cardiac function. While we have shown that targeting DNA methylation with 5aza has demonstrated beneficial effects on pressure overload-induced cardiac remodeling, the precise mechanism of action for this therapeutic outcome is currently unknown. There have been efforts in recent years to investigate the role of DNA methylation and other epigenetic modifications in individual cardiac cell populations to understand the role they play in cardiac disease and whether therapeutic intervention can influence these changes. A recent study by Stenzig and colleagues has demonstrated similar anti-remodeling effects by pharmacologically inhibiting DNA methylation using the non-nucleosidal Dnmt inhibitor, RG108, in rats subjected to TAC for 4 weeks. 38 While our study primarily looked at global DNA methylation status in cardiac tissue, their work focused on examining methylation changes in cardiomyocytes from the treated pressure overload environment. In response to pressure overload injury, they saw a trend toward a higher degree of methylation in cardiomyocytes with a significant association of differential CpG methylation occurring at annotated loci related to cardiac hypertrophy. 38 Interestingly, while they were able to demonstrate that RG108 could reduce the number of methylated cytosine residues in terminally differentiated cardiomyocytes, their pathway analysis revealed no significant association with cardiac hypertrophy with RG108 treatment in TAC, suggesting that the therapeutic influence of targeting DNA methylation may not be directly mediated by methylation changes in the cardiomyocyte. 38 The influence of 5aza on attenuation of myocyte size in our study may also be mediated indirectly via effects on paracrine regulators of myocyte function such as fibroblasts, endothelial cells and inflammatory cells. Indeed, in vitro treatment with 5aza has been shown to reduce the pro-fibrotic effects of TGFβ on human ventricular cardiac fibroblasts 19 and also on activated fibroblasts from different organ systems such as the kidney 39 and lung. 40 DNA methylation has been shown to influence various aspects of immune cell differentiation, cell phenotype and cell function. 9 Of particular relevance, 5aza and its structural analog 5azadC have been shown to modulate macrophage phenotype and behavior in the context of myocardial infarction, 41 atherosclerosis 42 and obesity. 43 Further understanding the role of specific alterations in DNA methylation that occur in the individual cell types of the heart coupled with its influence on gene regulation, especially in the context of cardiac injury, will improve our understanding and ultimately enable better therapeutic targeting with epigenetic-modifying therapies. Indeed, advancements in epigenetic therapy are currently underway. Most of the developments in this area have occurred for histone-modifying therapies, that are now designed to target specific cell types including monocytes (Tefinostat) 44 and hepatic myofibroblasts (3-Deazaneplanocin A in a C1-3-liposomal vector). 45 For the clinically available Dnmt inhibitors, advancements include the development of novel oral formulations of 5aza 46 along with the use of enzyme inhibitors to prevent its breakdown that are making Dnmt inhibitor therapy more convenient, increasing drug bioavailability and improving therapeutic outcome for MDS and AML patients. 47 These features suggest that there is still great opportunities for enhancing DNA methylation inhibitor therapy and provide hope that advancements in this area may yield targeted approaches for therapeutic application in other disease contexts.
To conclude, this study further supports the hypothesis that aberrant DNA methylation plays an important role in pathological cardiac remodeling and presents novel evidence indicating that low-dose 5aza treatment has the potential to reduce hypertrophy and fibrosis in the setting of pressure overload. While we did not elucidate a precise mechanism of action in this study, our work supports the rationale for repurposing low dose 5aza for the prevention of heart failure in the context of hypertrophic and fibrotic pathologies.
Study Limitations
There are some limitations in the present study that should be acknowledged. Firstly, the TAC model was used for our investigation and although this model is well established in the literature to induce pressure overload cardiac hypertrophy and fibrosis, the nature of the injury is acute and mimics the clinical scenario of aortic stenosis rather than the gradual progression of systemic hypertension experienced by HF patients. Secondly, only young, healthy male mice were used during this investigation and we acknowledge that this is not the representative nature of HF and therefore cannot conclude the degree to which these findings apply to female mice. Thirdly, assessment of diastolic function was only conducted in this study by pulse wave Doppler echocardiography. We acknowledge that the use of PV catheter system/ cardiac MRI may provide a more in depth and conclusive assessment (e.g. LVEDP, EDPVR, LV dP/dtmin, or tau) into the therapeutic impact of 5aza on diastolic function.
Footnotes
Acknowledgments
The authors would like to acknowledge Ms. Janet McCormack and the UCD Research Pathology Core Facility for their assistance with automated image analysis.
Author Contributions
Adam Russell-Hallinan and Roisin Neary contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CJW and JAB are co-founders of AzaCor, a university spin out that is trying to develop 5aza for the treatment of hypertrophic cardiomyopathy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding support was provided by Enterprise Ireland.
