Abstract
Chronic heart failure (CHF) is characterized by left ventricular (LV) dysfunction along with impaired autonomic control functions. Herbal drugs are increasingly being used in the treatment of cardiovascular disorders. The present study was designed to examine the protective effect of Terminalia arjuna (T arjuna) bark extract on LV and baroreflex function in CHF and to elucidate the possible mechanistic clues in its cardioprotective action. The baroreflex was evaluated by measuring the changes in heart rate (HR) with changes in arterial blood pressure induced by bolus injections of phenylephrine (vasoconstrictor) and sodium nitroprusside (vasodilator). T arjuna bark extract and fluvastatin were tested/administered therapeutically and prophylactically in isoproterenol-induced rat model of CHF. Fifteen days after isoproterenol administration, rats exhibited cardiac dysfunction, hypertrophy, and LV remodeling along with reduced baroreflex sensitivity. Prophylactic and therapeutic treatment with T arjuna improved cardiac functions and baroreflex sensitivity. It also attenuated hypertrophy and fibrosis of the LV. Fluvastatin treatment exerted a similar protective effect against myocardial remodeling and heart failure. Further, T arjuna and fluvastatin significantly reduced oxidative stress and inflammatory cytokine level in CHF rats. In conclusion, T arjuna exerts beneficial effect on LV functions, myocardial remodeling, and autonomic control in CHF possibly through maintaining endogenous antioxidant enzyme activities, inhibiting lipid peroxidation and cytokine levels.
Keywords
Introduction
Chronic heart failure (CHF) is a major health problem with limited and complex therapy options. Considerable progress has been made in the management of heart failure over the past few decades. However, it remains a major public-health problem, with high prevalence, poor clinical outcomes, and high health care costs. Neurohormonal activation, 1,2 sympathoexcitation, 3 disturbed sympathovagal balance, impaired arterial baroreflex function, 4 cardiac hypertrophy along with cardiac remodeling, and endothelial dysfunction 1 are characteristics which are invariably associated with CHF.
Arterial baroreceptor reflex normally contributes to cardiovascular homeostasis by maintaining blood pressure during changes in volume and posture. 5 The baroreceptor reflex is a negative feedback reflex that buffers autonomic alterations by sympathetic inhibition and parasympathetic activation in response to blood pressure elevations and by the reverse actions when the blood pressure falls. Blunted arterial baroreflex regulation has been directly implicated as an enhanced risk factor for sudden cardiac death and cardiovascular mortality. 6 Deterioration of cardiac function in CHF may be caused by ventricular remodeling, a dynamic process in which alteration in ventricular size, shape, and function occurs in response to hemodynamic overload, neurohormonal activation, activation of renin–angiotensin system, inflammatory cytokines, and oxidative stress. 7 Hence, there is a need to identify the drugs that could inhibit the progression of LV remodeling and improve autonomic function in patients with CHF, thus lowering mortality rate from sudden death.
Statins or hydroxymethylglutaryl CoA reductase inhibitors have also been shown to reduce the incidence of CHF and reduce mortality in patients with preexisting CHF. 8 Statins exert additional beneficial pleiotropic effects independent of their lipid-lowering action, such as free radical scavenging activity, anti-inflammatory, antithrombotic, antioxidant actions, and improvement in endothelial function. 9 Simvastatin treatment improved autonomic function in experimental heart failure. 10 However, adverse effects of long-term use of statins have been reported. 11,12 Thus, such therapies have a narrow margin of therapeutic safety and can be associated with severe side effects and toxicities. Alternative forms of therapy have attracted recent interest. One of these is herbal products which are becoming increasingly popular as these herbal preparations have no or minimal side effects and are also cost-effective.
Terminalia arjuna (Family Combretaceae), an important medicinal plant, has been widely used in Indian systems of medicine in cardiac ailments. Use of Arjuna bark in the management of hypercholesterolemia has been reported. 13 Pharmacological studies have shown its antimutagenic, 14 anticancer, 15 and hypotensive properties. 16 Both experimental and clinical studies have shown that the dried bark powder of T arjuna has significant protective effects in ischemic heart disease. 17 However, its effect on restoring normal baroreflex in CHF is not known.
Therefore, the present study was designed to evaluate the therapeutic and prophylactic efficacy of T arjuna bark extract on myocardial and autonomic neural control in isoproterenol-induced CHF rats. Further, the cardioprotective effect of T arjuna bark extract in CHF was compared with fluvastatin, a known lipid-lowering agent.
Materials and Methods
Isoproterenol, urethane, phenylephrine, and sodium nitroprusside were purchased from Sigma Chemical Co (St Louis, Missouri, USA). Fluvastatin was procured from Ranbaxy Pharmaceuticals, India. All other chemicals and reagents used in the study were of analytical grade.
Terminalia arjuna Bark Extract
Bark extract was procured from Jamia Hamdard, Delhi, India. Terminalia arjuna bark was used in the form of 50% aqueous ethanol extract. The shade-dried T arjuna bark was coarsely powdered and extracted with ethanol (50%) using Soxhlet apparatus (H L Scientific Industries, Haryana, India). The extract was filtered and concentrated to obtain solid residue. The yield of the total aqueous ethanolic extract was 28.76%. Primary phytochemical screening of the ethanolic extract of T arjuna bark revealed the presence of glycosides, triterpenoids, phenols, flavonoids, tannins, and saponins. Dose–response studies starting with the smaller doses were carried out. T arjuna bark extract in 500 mg/kg dose produced significant effect. In total, 500 mg/kg of T arjuna per day was found more efficacious and without any apparent side effects. 18
Study Design
Wistar rats (weight 200-240 g; either sex) were obtained from the animal house of Maulana Azad Medical College ([MAMC] Delhi) and housed at 25°C ± 2°C with a 12-hour light/dark cycle. All animal experiments were carried out in accordance with the recommendations of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India. Ethical clearance for the experimental protocol was obtained from the Institutional Animal Ethical Committee (IAEC), MAMC, University of Delhi, Delhi, India. Rats were fed commercial pellet diet and water ad libitum. They were divided randomly into 8 groups having 10 animals in each group. Group I (control) rats were fed with normal pellet diet. Group II (CHF) rats were administered isoproterenol (85 mg/kg, subcutaneously [sc]) for 2 consecutive days and were kept for 15 days for induction of CHF. 19,20 Some of the animals were kept for 30 days after administration of isoproterenol. Groups III and IV (fluvastatin/T arjuna alone) rats were treated with fluvastatin (10 mg/kg, oral gavage) or T arjuna (500 mg/kg, oral gavage) for 15 days. Groups V and VI (fluvastatin/T arjuna therapeutic) CHF rats were treated with fluvastatin (10 mg/kg, oral gavage) or T arjuna (500 mg/kg, oral gavage) for 15 days. Groups VII and VIII (fluvastatin/T arjuna prophylactic) rats were pretreated with fluvastatin (10 mg/kg, oral gavage) or T arjuna (500 mg/kg, oral gavage) for 15 days. On the 15th day, isoproterenol (85 mg/kg, sc) was administered for 2 consecutive days. Treatment with fluvastatin/T arjuna was continued further for 15 days.
Hemodynamic Measurements
After completion of treatment, rats were anesthetized with urethane (1 g/kg, intraperitoneally). Tracheal intubation was done to allow free air breathing. Body temperature of the rat was maintained at 37 to 38°C. Femoral artery of one side was exposed and a polyethylene catheter filled with heparin solution (500 IU/mL, volume/volume [v/v]) was inserted in the artery through a small incision for recording arterial blood pressure (ABP). The catheter was attached to a 23-gauge needle connected to a pressure transducer (Statham P23Db, Oxnard, California, USA). The femoral vein was cannulated for injecting drugs. Mean arterial pressure (MAP) and heart rate (HR) were derived using ABP. A Miller mikro-tip catheter (Millar Instruments, Houston, Texas, USA) was placed in the left ventricle (LV) via the right carotid artery to record left ventricular pressure (LVP). To assess LV function, left ventricular end-diastolic pressure (LVEDP) and maximal rate of rise of LVP divided by LV systolic pressure (LV(dP/dt)max/P) were measured offline from the LVP data. A period of approximately 20 minutes was allowed for stabilization of cardiovascular parameters before the hemodynamic data were recorded on a Power Lab data-acquisition system (4SP, AD Instruments, Australia) with a computerized analysis program (Chart 5.4.2, AD Instruments, Australia). For measuring cardiac output, animals were mechanically ventilated by a small animal ventilator (Columbus, Ohio) through a tracheal cannula with room air at a rate of 60 to 70 breaths/min and tidal volume of 8 to 10 mL/kg. A median sternotomy was performed. After the pericardium was opened, the ascending aorta was isolated by blunt dissection. A 2-mm Transonic flowmeter perivascular flow probe was positioned around the ascending aorta, perpendicular to the curvature of the arch. Cardiac output (mL/min) was measured from continuous aortic blood flow measurements using blood flowmeter (model T101, Transonic Systems, Ithaca, New York). After recording of hemodynamic parameters, blood was withdrawn by cardiac puncture, serum separated, and stored at −20°C for biochemical estimations.
Measurement of Baroreflex Sensitivity
Baroreflex sensitivity was assessed as previously described (Tauseef et al) 21 by administering increasing doses of vasoconstrictor phenylephrine (20-40 μg/mL per kg) and vasodilator sodium nitroprusside (20-40 μg/mL per kg) as bolus injections through a venous catheter. An appropriate interval (15 minutes) between doses was allowed for ABP and HR to return to basal levels. The resultant changes in HR at corresponding rise or fall in systolic blood pressure were measured at different time intervals. The relationship between increase in systolic blood pressure evoked by phenylephrine and associated bradycardia or decrease in systolic blood pressure evoked by sodium nitroprusside and associated tachycardia was assessed by regression analysis for individual animals. The regression coefficient (slope of regression line), expressed as beats per minute per millimeter of mercury (beats/min/mm Hg), was taken as an index of baroreflex sensitivity measurement.
Cardiac Hypertrophy
At the end of the experiments, the animals were euthanized with an overdose of anesthesia. Hearts were excised and weighed. Cardiac hypertrophy was evaluated by measuring the ratio of heart weight (mg) to body weight (g).
Histology
After weighing, hearts were fixed in 10% formalin solution for histopathological analysis. Tissues were embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (H&E). The sections were examined under light microscope, and photomicrographs were taken.
Biochemical Estimations
Lipid peroxidation was estimated by the method of Wright et al. 22 Reduced glutathione (GSH) was determined by the method of Jollow et al. 23 Superoxide dismutase ([SOD] Cayman Chemicals, Ann Arbor, Michigan) and inflammatory marker tumor necrosis factor-α ([TNF-α] Alpco immunoassays Salem, New Hampshire) were measured in serum using the enzyme-linked immunosorbent assay (ELISA) kits.
Statistical Analysis
The results were presented as mean ± standard error of the mean (SEM). All data were analyzed by analysis of variance (ANOVA) followed by Tukey multiple comparison tests, for the analysis between the groups. P value of < .05 was considered statistically significant.
Results
Mean Arterial Pressure and HR
In CHF group, no significant differences in MAP and HR were observed as compared to control rats (Table 1). Also, no change was observed in MAP and HR among all the different treatment groups.
Effect of Terminalia arjuna and Fluvastatin on MAP and HR in Control and Chronic Heart Failure Groups a

aValues are means ± standard error of the mean ([SEM] n = 10 animals in each group). Mean arterial pressure (MAP) and heart rate (HR) in control group (group I), chronic heart failure group (group II), fluvastatin per se group (group III), Terminalia arjuna per se group (group IV), fluvastatin therapeutic group (group V), T arjuna therapeutic group (group VI), fluvastatin prophylactic group (group VII), and T arjuna prophylactic group (group VIII).
Left Ventricular Function and Cardiac Output
To evaluate LV function, various hemodynamic parameters were measured in anesthetized rats of all the groups. Myocardial function was significantly altered in the CHF group. Left ventricular end-diastolic pressure was markedly (P < .05) increased (11.45 ± 0.76 mm Hg vs 2.44 ± 0.21 mm Hg in control), whereas LV(dP/dt)max/P (58.81 ± 4.40 s−1 vs 114.1 ± 3.06 s−1 in control), and CO (93.19 ± 5.05 vs 169.6 ± 6.23 mL/min per kg in control) were significantly (P < .05) decreased in CHF rats compared to control rats (Figure 1A-C). Treatment of CHF animals prophylactically as well as therapeutically with fluvastatin or T arjuna significantly improved the isoproterenol-induced depression in LV(dP/dt)max/P and cardiac output, whereas the isoproterenol-induced increase in LVEDP was significantly attenuated (Figure 1A-C). No statistically significant difference was found when fluvastatin or T arjuna therapeutic groups were compared with each other, and similar results were obtained in the prophylactic groups. Oral administration of fluvastatin and T arjuna in normal rats did not produce any significant change in LV function as compared to control rats.

Effect of Terminalia arjuna and fluvastatin on (A), LVEDP; (B), LV(dP/dt)max/P; (C) cardiac output in control group (group I), chronic heart failure group (group II), fluvastatin per se group (group III), T arjuna per se group (group IV), fluvastatin therapeutic group (group V), T arjuna therapeutic group (group VI), fluvastatin prophylactic group (group VII), and T arjuna prophylactic group (group VIII). All values are expressed as mean ± standard error of the mean ([SEM] n = 10). *P < .05 vs group I. #P < .05 vs Group II.
Baroreflex Sensitivity
Baroreflex sensitivity was measured as ratio of bradycardia response to rise in arterial pressure by phenylephrine and ratio of tachycardia response to fall in arterial pressure by sodium nitroprusside. The baroreflex sensitivity was markedly reduced in CHF rats as compared to normal rats of the control group, as indicated by a significant (P < .05) fall in both reflex bradycardia (0.75 ± 0.12 vs 2.43 ± 0.1 beats/min per mm Hg in control; Figure 2A) and tachycardia (0.6 ± 0.07 vs 1.7 ± 0.1 beats/min per mm Hg in control; Figure 2B) response elicited by phenylephrine and sodium nitroprusside, respectively. Prior treatment of CHF rats with fluvastatin or T arjuna significantly (P < .05) restored baroreflex responses. Baroreflex sensitivity to both phenyephrine and sodium nitroprusside was significantly (P < .05) improved in rats, given therapeutic fluvastatin or T arjuna treatment compared to CHF rats (Figure 2A and B). No significant change was observed in baroreflex sensitivity by fluvastatin or T arjuna alone treatment.

Baroreflex sensitivity to (A) phenylephrine and (B) sodium nitroprusside in control group (group I), chronic heart failure group (group II), fluvastatin per se group (group III), Terminalia arjuna per se group (group IV), fluvastatin therapeutic group (group V), T arjuna therapeutic group (group VI), fluvastatin prophylactic group (group VII), and T arjuna prophylactic group (group VIII). All values are expressed as mean ± standard error of the mean ([SEM] n = 10). *P < .05 vs group I. #P < .05 v/s group II.
Serum Malondialdehyde, SOD, and GSH Levels
Effects of fluvastatin and T arjuna on serum malondialdehyde (MDA), SOD, and GSH levels in control and experimental groups are shown in Table 2. The level of MDA increased, while SOD activity and GSH content decreased significantly (P < .05) in CHF rats when compared with the control group. Fluvastatin and T arjuna treatment both prophylactically and therapeutically significantly (P < .05) decreased the level of MDA in CHF rats. SOD activity and GSH content increased significantly (P < .05) in rats pretreated with fluvastatin or T arjuna when compared to CHF group.
Effect of Terminalia arjuna and Fluvastatin on Serum MDA, GSH, SOD, and TNF-α Levels in Control and Chronic Heart Failure Groups a

Abbreviations: MDA, malondialdehyde; GSH, reduced glutathione; SOD, superoxide dismutase; TNF-α, tumor necrosis factor-α in control group (group I), chronic heart failure group (group II), fluvastatin per se group (group III), Terminalia arjuna per se group (group IV), fluvastatin therapeutic group (group V), T arjuna therapeutic group (group VI), fluvastatin prophylactic group (group VII), and T arjuna prophylactic group (group VIII); SEM, standard error of the mean.
aValues are means ± SEM (n = 10 animals in each group).
b P < .05 vs group I.
c P < .05 vs group II.
Therapeutic treatment with fluvastatin or T arjuna also increased the SOD activity and GSH content significantly (P < .01) in CHF rats. The results showed that there were no significant alterations in the MDA levels, SOD enzyme activities, and GSH content in fluvastatin and T arjuna alone treatment groups when compared with that of the normal control group (Table 2).
Serum Cytokine TNF-α Level
The effects of fluvastatin and T arjuna on serum inflammatory cytokine TNF-α levels in control and experimental groups are shown in Table 2. Serum TNF-α levels increased significantly (P < .05) in CHF rats when compared with control group. Fluvastatin treatment both prophylactically and therapeutically significantly (P < .05) lowered the serum levels of TNF-α when compared with that of CHF group. Prophylactic and therapeutic treatment of CHF rats with T arjuna significantly prevented the rise in serum TNF-α levels. Fluvastatin and T arjuna alone did not have any effect on TNF-α level (Table 2).
Cardiac Hypertrophy
Figure 3 shows the effect of fluvastatin and T arjuna on cardiac hypertrophy expressed as ratio of heart weight to body weight in control and experimental groups. Chronic heart failure rats showed marked increase in heart weight to body weight ratio compared to control rats. Heart weight to body weight ratio of CHF rats treated prophylactically and therapeutically with fluvastatin or T arjuna decreased significantly (P < .05) when compared with that of CHF group.
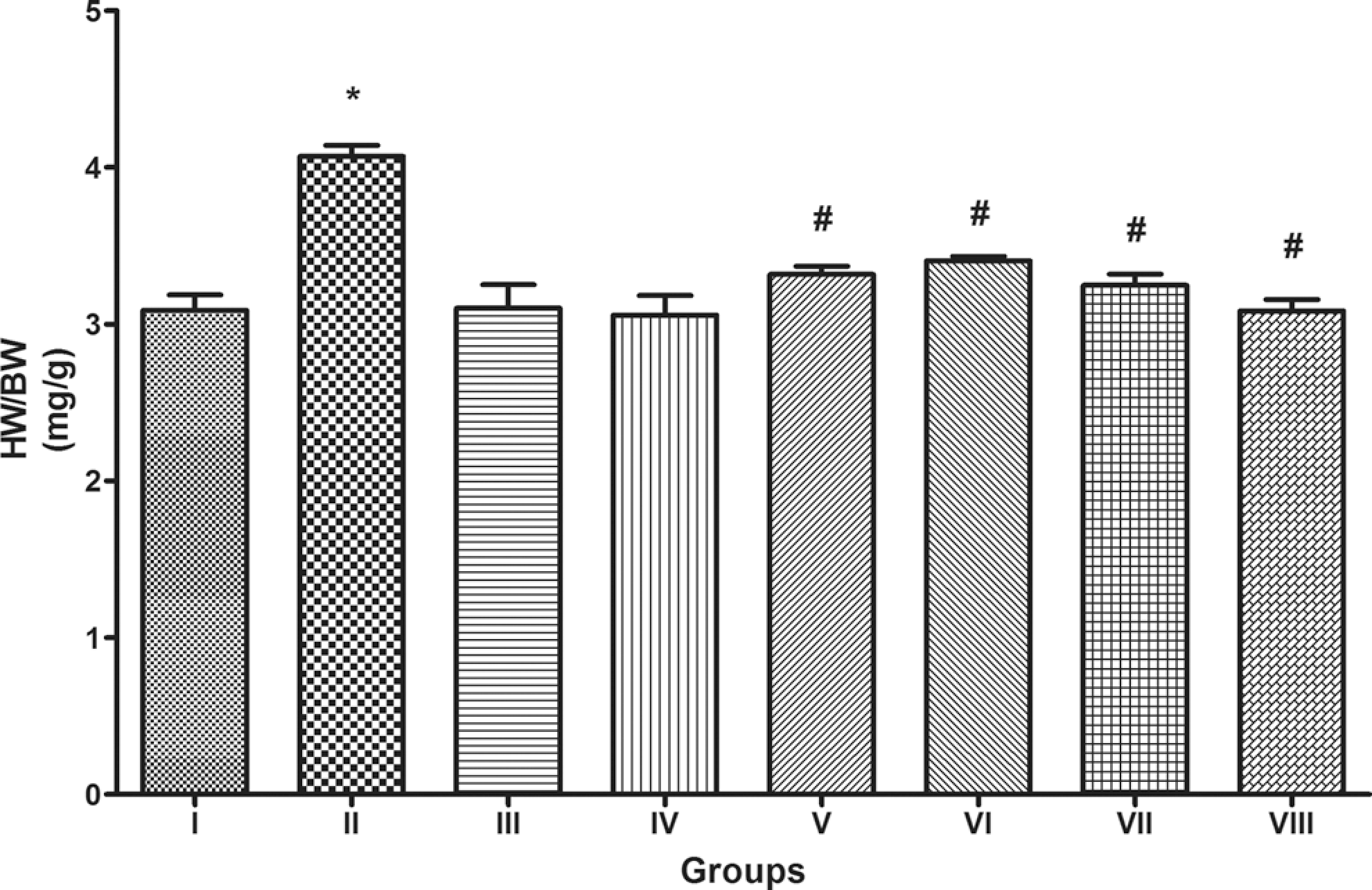
Heart weight to body weight ratio (mg/g) in control group (Group I), chronic heart failure group (Group II), fluvastatin per se group (Group III), Terminalia arjuna per se group (Group IV), fluvastatin therapeutic group (Group V), T arjuna therapeutic group (Group VI), fluvastatin prophylactic group (Group VII), and T arjuna prophylactic group (Group VIII). All values are expressed as mean ± S.E.M (n = 10). *P < .05 vs Group I. #P < .05 vs Group II. 209 × 134 mm (300 × 300 DPI).
Histopathological Findings
Figure 4 shows photomicrographs of heart tissue sections from control and experimental groups. Histological sections from the control group show the architecture of the normal cardiac tissue with typical myocardial structure (Figure 4A). Heart tissues from CHF rats showed widespread myocardial structural alterations and subendocardial necrosis with interstitial edema and leukocyte infiltration (Figure 4B). Therapeutic treatment with T arjuna or fluvastatin showed partial improvement in myocardial disordered structure (Figure 4 C and D). Prophylactic treatment with T arjuna or fluvastatin showed marked improvement in subendocardial necrosis, capillary dilatation, and leukocyte infiltration (Figure 4 E and F). Histological sections of heart from T arjuna or fluvastatin alone group showed the normal architecture of the rat myocardium.
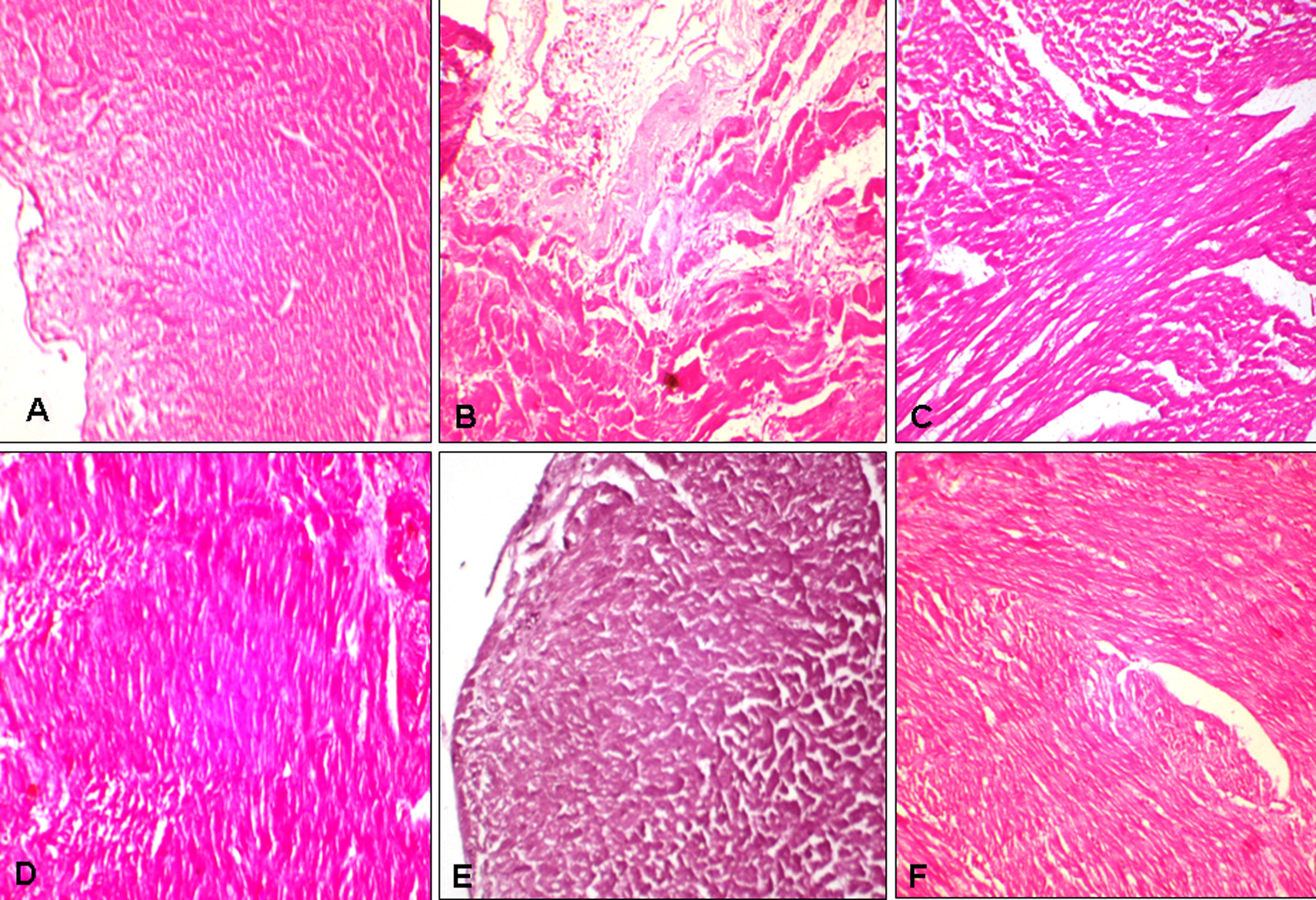
Photomicrographs of rat heart from control group (A), chronic heart failure group (B), therapeutic fluvastatin group (C), therapeutic Terminalia arjuna group (D), prophylactic fluvastatin group (E), and prophylactic T arjuna group (F) (hematoxylin and eosin [H&E]; ×200).
Discussion
The present study demonstrated that prophylactic and therapeutic treatment with T arjuna and fluvastatin in CHF rats exerts beneficial effects on cardiac hemodynamics, LV remodeling, baroreflex function, and reduces serum oxidative stress and serum TNF-α levels.
Isoproterenol, a β-adrenergic receptor agonist, administration induces myocardial remodeling with LV hypertrophy and fibrosis in rats. 20,24 Several mechanisms have been proposed to explain these effects such as intracellular cyclic adenosine monophosphate (cAMP) increase, intracellular calcium overload, and modification of electrophysiological properties of cardiomyocytes, ischemia, and oxidative stress. 25 In this study, CHF was well characterized after 15 days following isoproterenol administration, by significant elevation of LVEDP, an alteration of contractility, and significant cardiac remodeling with the development of fibrosis in cardiac tissue. No significant change in MAP and HR was observed among all the groups.
LV(dP/dt)max/P a close index of myocardial contractility 26 was decreased in CHF rats as compared to control rats, indicating derangement in contractility. Left ventricular end-diastolic pressure increases both in systolic and in diastolic LV dysfunctions as in myocardial infarction and heart failure 27 and is a critical determinant of LV function. Increased LVEDP in CHF indicates an increased preload or incomplete emptying of the LV and impaired LV performance. Fluvastatin and T arjuna both markedly attenuated the reduction in LV(dP/dt)max/P and rise in LVEDP, thus showing a protective effect of these 2 drugs on myocardial function in the present CHF model. Cardiac output was also restored by treatment with these drugs in CHF rats, indicating improvement in cardiac performance. Isoproterenol produced cardiac hypertrophy as reflected by increased heart weight to body weight ratio. Hypertrophy is a process of remodeling of ventricle myocardial structure which deteriorates cardiac functioning and progresses into heart failure at the later stage. 28 Reduced cardiac output observed in CHF rats aggravated the work load on the heart due to reflex activation of the sympathetic nervous system. 29 Fluvastatin treatment improved LV function, cardiac performance, and hypertrophy owing to its cardioprotective property as reported in the earlier studies. 30,31 Treatment with T arjuna bark extract prophylactically as well as therapeutically improved cardiac performance and reduced the LV weight to body weight ratio. This is in contrast to a study by Kumar et al 32 in which T arjuna bark aqueous extract in doses lower than used in the present study did not prevent hypertrophy in isoprenaline-induced model of cardiac hypertrophy although treatment duration was longer than in our study. Terminalia arjuna bark alcoholic extract in 500 mg/kg dose was beneficial in reducing hypertrophy in isoproterenol-induced CHF rats when given for 2 weeks both prophylactically and therapeutically. This indicates that alcoholic extract may possibly contain additional constituents (components) in comparison to aqueous extract. It has been shown earlier that the major chemical compounds identified in the alcoholic extract of T arjuna were tannins, flavonoids, and glycosides, and that they have significant antioxidant properties. 33 Further studies are needed to elucidate the exact mechanisms of action of the various constituents and their potential in the treatment and/or prevention of heart failure. Our results suggest strong cardioprotective effect of T arjuna against isoproterenol-induced LV dysfunction and hypertrophy. Histological studies further support our observation as fluvastatin and T arjuna treatment alleviated the isoproterenol-induced alterations in myocardial structure, fibrosis, and cardiac myocyte hypertrophy.
Chronic heart failure is accompanied by depression in the sensitivity of the arterial baroreflex as described in several clinical 34,35 and experimental studies. 10,36 In the present study, decrease in baroreflex sensitivity was observed in CHF rats as reported in the earlier studies. Changes in central sympathetic and vagal outflow in the CHF state may be responsible for diminished baroreflex sensitivity. Therefore, restoration of autonomic function should also enhance baroreflex sensitivity. The present study revealed an increase in baroreflex sensitivity with both fluvastatin and T arjuna treatment in CHF rats. Statin therapy has already been shown to have a modulatory effect on autonomic function and sympathetic tone in an animal model of heart failure. 10 Several potential mechanisms for the beneficial effects of statins on autonomic nervous system function in heart failure have been described. Angiotensin II, Nicotinamide Adenine Dinucleotide Phosphate (NADPH)-oxidase-derived reactive oxygen species (ROS), and nitric oxides have been implicated in the modulation of sympathetic tone by statins in heart failure. 10,36,37 Thus, T arjuna improved sympathovagal balance and neurohormonal activation in CHF animals.
Increased oxidative stress resulting from an increased cardiac generation of ROS is implicated in the progression of cardiac hypertrophy and heart failure. 38 The most abundant ROS generated in cells are superoxide anion and its derivatives, particularly the highly reactive and damaging hydroxyl radical, which induces peroxidation of cell membrane lipids. Various studies have demonstrated that isoproterenol administration provokes cardiac oxidative stress. 25,39 Increase in lipid peroxidation and depletion in endogenous antioxidants such as SOD and GSH observed in the present study are in confirmation with the earlier studies. The results of the present study showed protective effect of fluvastatin against cardiac oxidative stress in CHF rats as reported in the earlier studies. 30 T arjuna treatment also prevented the cardiac oxidative stress in CHF rats. Its treatment also showed a significant reduction in MDA level and significant improvement in GSH and SOD activity, suggesting that T arjuna bark extract reduces oxidative stress thereby preventing the generation of free radicals in accordance with the earlier studies. 13,32 The inverse relationships observed between SOD, GSH activity, and LVEDP reinforce the possibility of increased oxidative stress in CHF rats. Indeed, the reflex response to phenylephrine and sodium nitroprusside administration was positively correlated with SOD and GSH activity and negatively correlated with lipid peroxidation, suggesting that an increase in oxidative stress of the heart is associated with a reduced reflex response to blood pressure alterations. It is possible that changes in the heart produced by changes in oxidative profile may be related to the changes in baroreflex sensitivity, probably by modulating cardiopulmonary afferents. The observed increase in LVEDP in CHF clearly suggests involvement of cardiac reflexes. Reactive oxygen species have been shown to directly inhibit the firing of arterial baroreceptors 40 and increase the firing of cardiac sympathetic afferent fibres. 41 The observed increase in oxidative stress seems to be associated with reduced reflex control of the circulation, reinforcing the role of oxidative profile not only in the metabolic and biochemical changes but also in hemodynamic features of isoproterenol-induced CHF.
Inflammatory cytokine activation has been implicated in the progression of cardiac remodeling and LV dysfunction in both clinical and experimental studies. 42,43 Cytokines exert unfavorable effect on LV function, induce abnormalities in cardiac metabolism and energetics, and promote hypertrophy, necrosis, and apoptosis. 44 Cytokines activate ROS generation in cardiac myocytes and peripheral muscles and pretreatment with antioxidants abolishes the cytokine-induced cellular effects. 45 Increased levels of TNF-α contributes significantly to cardiac dysfunction in heart failure rats. 42 Our results also corroborate with the findings of the earlier studies showing elevation in serum TNF-α level in CHF rats. This rise was inhibited by fluvastatin and T arjuna bark extract treatment, prophylactically as well as therapeutically, suggesting anti-inflammatory activity of fluvastatin and T arjuna.
T arjuna bark contains various active constituents such as tannins, cardenolide, triterpenoid saponins (arjunic acid, arjunolic acid, arjungenin, and arjunglycosides), flavonoids (arjunone, arjunolone, and luteolin), oligomeric proanthocyanidins (OPCs), phytosterols, calcium, magnesium, zinc, and copper. 46,47 Flavonoid possesses antioxidant, anti-inflammatory, antiproliferative, and antiplatelet activities. 33,48 Thus, combined or independent action of several constituents such as flavonoids, tannins, and glycosides might be responsible for the beneficial effects of T arjuna in various cardiovascular disorders. It may be correlated with statins as a potential agent having similar pharmacological benefits. Therefore, T arjuna could have improved autonomic nervous system control and cardiac remodeling through several different mechanisms, either by improving hemodynamics or by reducing endothelial dysfunction through its antioxidant, free radical scavenging, and anti-inflammatory property.
Conclusion
In conclusion, our study demonstrated prophylactic and therapeutic beneficial effect of T arjuna in regulating autonomic function and preventing LV remodeling in CHF rats. T arjuna shows a similar effect in improving left ventricular functions, hypertrophy, and oxidative stress as seen with fluvastatin, a synthetic clinically used drug. Thus, T arjuna can be used as a potent alternative herbal drug in protection against cardiovascular disorders.
Footnotes
Acknowledgments
The authors would like to acknowledge Professor M S Y Khan, Jamia Hamdard (India) for providing Terminalia arjuna bark extract.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
