Abstract
Introduction:
Dexamethasone (DEX)-induced hypertension and cardiac remodeling are still unclear, especially in spontaneously hypertensive rats (SHR). On the other side, exercise training is a good strategy to control hypertension. Therefore, this study investigated the effects of DEX treatment and physical training on arterial pressure and cardiac remodeling in SHR.
Material and Methods:
SHR underwent treadmill training (5 days/week, 1h/session, at 50-60% of maximal capacity, 0% degree, 75 days) and received low-dose of DEX (50µg/kg, s.c.) during the last 15 days. Sedentary Wistar rats (W) were used as control. Echocardiography and artery catheterization were performed for cardiac remodeling and function, arterial pressure and autonomic nervous system analyses. In addition, left ventricle (LV) capillary density, myocyte diameter and collagen deposition area were analyzed using specific histological staining.
Results:
Low-dose of DEX treatment did not exacerbate arterial pressure of SHR and trained groups had lower values, regardless of DEX. DEX and training decreased relative left ventricle wall thickness (RWT) and determined LV angiogenesis (+19%) and lower collagen deposition area (−22%). In addition, it determined increased left ventricular diastolic diameter. These changes were followed by improvements on systolic and diastolic function, since it was observed increased posterior wall shortening velocity (PWSV) and reduced isovolumetric relaxation time (IVRT).
Conclusion:
In conclusion, this study is unique to indicate that low-dose of DEX treatment does not exacerbate arterial pressure in SHR and, when associated with training, it improves LV systolic and diastolic function, which may be due to LV angiogenesis and reduction of wall collagen deposition area.
Introduction
Hypertension induces cardiac remodeling, such as pathological hypertrophy, decreased capillary density and increased fibrosis. 1,2 Although dexamethasone (DEX) has a widespread use for inflammation and allergies, 3,4 there are growing evidence that it induces several side effects, like muscle atrophy, hyperglycemia, baroreflex alterations, autonomic imbalance to the heart, as well as hypertension and cardiac remodeling in normotensive animals. 5 -11 In contrast, pre-treatment with DEX was able to decrease area of infarction and reduce apoptosis of cardiomyocytes in animals that had their anterior descending coronary occluded, due to an activation of anti-apoptotic protein Bcl-xL transcription. 12 In agreement, cardiomyocyte cells treated with corticosteroids showed reduced doxorubicin-induced apoptotic response due to an increase in Bcl-xL. 13 In addition, treatment with DEX has been shown to reduce the protein production of metalloproteinases and improve cardiac function by decreasing tumor necrosis factor alpha (TNF-α) in animals with chronic heart failure. 14 But nothing is known about the effects of DEX treatment on arterial pressure and heart remodeling in hypertensive rats (spontaneously hypertensive rat-SHR), condition in which there are established hypertension and cardiac remodeling. 2,15
On the other hand, physical training has been recommended as a non-pharmacological treatment for hypertension. 16,17 It is already known that training may mitigate DEX-induced hypertension through an attenuation of autonomic dysfunctions associated with an improvement of the skeletal muscle microcirculation. 7 Also, training improves baroreflex activity in DEX-induced hypertensive animals. 8 Some studies have demonstrated that training may attenuate cardiac remodeling in old SHR rats 18 and in patients with heart failure, 19 but nothing was known about the effects of training in DEX-treated SHR. Therefore, the objective of the present study was to investigate the effects of DEX treatment and aerobic training on arterial pressure and cardiac remodeling process in SHR. Our hypothesis was that DEX treatment, associated or not with training would have beneficial effects on myocardial function of spontaneously hypertensive rats.
Methods and Materials
Animal Care
Male spontaneously hypertensive rats (SHR, 12 weeks) were obtained from the Institute of Biomedical Sciences at the University of São Paulo, Brazil and Wistar rats (W, 12 weeks) were obtained from the animal breeding facility at São Paulo State University (Botucatu, SP). Rats were housed in collective cages (5 each) in the Animal Facility Maintenance at School of Sciences, São Paulo State University (Bauru, SP), at a controlled temperature room (20 ± 2ºC) and 12:12 hours light: dark cycle. Water and food (Biobase, Águas Frias, Santa Catarina, Brazil) were provided ad libitum. Experimental protocols were approved by the Committee for Ethical Use of Animals (CEUA) of School of Sciences, São Paulo State University, UNESP, campus of Bauru, SP (# 779/2017 Vol. 1).
Groups and Experimental Protocol
After an adaptation period on a treadmill (10 days), all rats underwent maximal exercise capacity test (Tmax) as published. 20 After that, rats were randomly allocated into 5 groups, which underwent 75 days of the experimental protocol: 5 days/week of treadmill aerobic exercise training, during 1h/session, at 50-60% of Tmax, 0% degree, as previously described 7 or were kept sedentary. After 4 and 8 weeks, Tmax was repeated to adjust the speed in order to maintain training intensity and to evaluate training effects on maximal capacity. Five groups are follow: (1) WISTAR control (W = 20), sedentary for 75 days and received saline subcutaneous injection (s.c.) during the last 15 days; (2) SHR sedentary control (SC = 21), sedentary for 75 days and received saline (s.c.) during the last 15 days; (3) SHR sedentary DEX (SD = 21), sedentary for 75 days and received DEX (Decadron®; 50µg/kg per day, at 9 a.m., s.c.) during the last 15 days; (4) SHR trained control (TC = 11), trained for 75 days and received saline (s.c.) during the last 15 days and (5) SHR trained DEX (TD = 11), trained for 75 days and received DEX (Decadron®; 50µg/kg per day, at 9 a.m., s.c.) during the last 15 days. This dose and route of administration of DEX was chosen since previous studies from our group, carried out on W rats, showed increases in arterial pressure. 7 Body weight was weekly measured during training and daily during treatment periods, as demonstrated in Figure 1.

Experimental design. After an adaptation period on a treadmill (10 days), all rats underwent maximal exercise capacity test (Tmax). After that, rats were randomly allocated into 5 groups, which underwent 75 days of the experimental protocol: 5 days/week of treadmill aerobic exercise training, during 1 h/session, at 50-60% of Tmax, 0% degree, or were kept sedentary. After 4 and 8 weeks, Tmax was repeated. Control groups received saline during the last 15 days. Echocardiogram and hemodynamic analyses were performed at the end of experimental protocol.
Ecocardiographic Analysis
On the 74th day of the experimental protocol, all rats were subjected to a transthoracic echocardiography. In brief, echocardiography was performed with a 5 – 11.5 MHz multifrequency probe (General Electric Medical Systems, Vivid S6, Tirat Carmel, Israel), as previously described. 18
Arterial Pressure Measurement
On the 75th day, all rats were anesthetized with ketamine hydrochloride and xylasine hydrochloride (50 and 10 mg/kg, i.p. respectively; Ceva Santè Animale, São Paulo, Brazil). Then, a catheter was inserted into the carotid artery for arterial pressure measurement, as published. 7,21 After 24 h recovery, arterial pressure was recorded in awake rats, using a pressure transducer (DPT100; Utah Medical Products Inc., Midvale, Utah, USA) and an amplifier (Quad Bridge Amp, ADInstruments, NSW, Australia), connected to an acquisition board (Powerlab 4/35, ADInstruments, NSW, Australia).
Autonomic Nervous System Analysis
Cardiac interval pulse from 15-30 min recordings (2,000 Hz) was processed by LabChart pro v7.1 software (ADInstruments, NSW, Australia) that applies an algorithm to detect cycle-to-cycle inflection points in the pulsatile arterial pressure signal, as previously published. 21 Heart rate (HR) and systolic arterial pressure (SAP) variability analyses, within frequency domain, were managed using DIAS software (CardioSeries V2.7, http://www.danielpenteado.com). Low frequency band power and high frequency band power were determined, corresponding to sympathetic and parasympathetic (LF) and parasympathetic (HF) nervous activity, respectively. The sympathovagal balance was analyzed by the LF/HF ratio. 22 Spontaneous baroreflex sensitivity was assessed by the sequence method also using a CardioSeries v2.7 computer software, over the physiological range of fluctuations in the arterial pressure. 23
Withdrawal of the Heart
At the end of the experimental protocol, rats were euthanized under anesthesia overload (xylasine hydrochloride 20 mg/kg and ketamine hydrochloride 160 mg/kg). Heart was removed, washed, weighed and its chambers were separated. Tibia bone length was used for tissue normalization and adrenal glands were removed for confirmation of DEX treatment efficacy.
Histological Analysis
A transverse section was made in the left ventricle (LV) and it was fixed in 4% buffered paraformaldehyde solution for 24 h. Tissues were embedded in Paraplast (Sigma, USA). Histological sections were performed using a manual microtome (Microm HM 325 Microtome, Southeast Pathology Instrument Service, SC) with disposable blades and placed on glass slides. Sections of 5 µm thickness (hematoxylin-eosin staining, Easy Path, SP) were used for capillary density and myocyte diameter, 7 while 7 µm thickness (Picrosirius Red staining, Merck-Millipore, USA) were used for collagen deposition area analyses. 24 All images were captured using Leica MC170 HD camera, coupled to the Leica DM4 B microscope (20 or 40x magnification). Analyses (off-line) were performed using ImageJ software, on blinded slides.
Statistical Analysis
Results are presented as mean ± standard error of the mean. Student’s t-test was used to compare 2 groups. Two-way analysis of variance (ANOVA) was used to compare 4 groups. In the presence of interactions, Tukey Post hoc was used (P < 0.05).
Results
Spontaneously Hypertensive Rats Have a Cardiac Remodeling as a Result of High and Sustained Arterial Pressure Levels
It has been well described in the literature that SHR have high arterial pressure levels that are responsible for promoting cardiac remodeling. 2,15,18 This first set of results refers to a comparison of W versus SHR (Table 1) and confirms that SHR had higher SAP (+72%), diastolic arterial pressure (DAP, +59%) and mean arterial pressure (MAP, +76%) without changes in HR. Investigating the mechanisms that may be involved in SHR hypertension, Table 1 also shows that sympathetic nervous activity to the vessels was higher in SHR compared with W, without a significant alteration on autonomic balance to the heart. Also, significant changes in cardiac structure were found in SHR, like higher heart weight (+25%), LV mass index (LVMI) and relative thickness of the ventricle (RWT) (+27% and +28%, respectively), driven by higher myocyte diameter. Consequently, SHR had smaller LV diastolic diameter (LVDD, −11%) and LV systolic diameter (LVSD, −10%). These myocardial changes probably contributed to compromise LV systolic and diastolic function, since SHR presented lower values of posterior wall shortening velocity (PWSV, −15%) and higher values of isovolumetric relaxation time (IVRT, +16%), respectively, while Endocardial Fractional Shortening (EFS %) and Left Ventricle Ejection Fraction (LVEF) were preserved. (Table 1).
Hypertension Promotes Cardiac Remodeling in SHR.
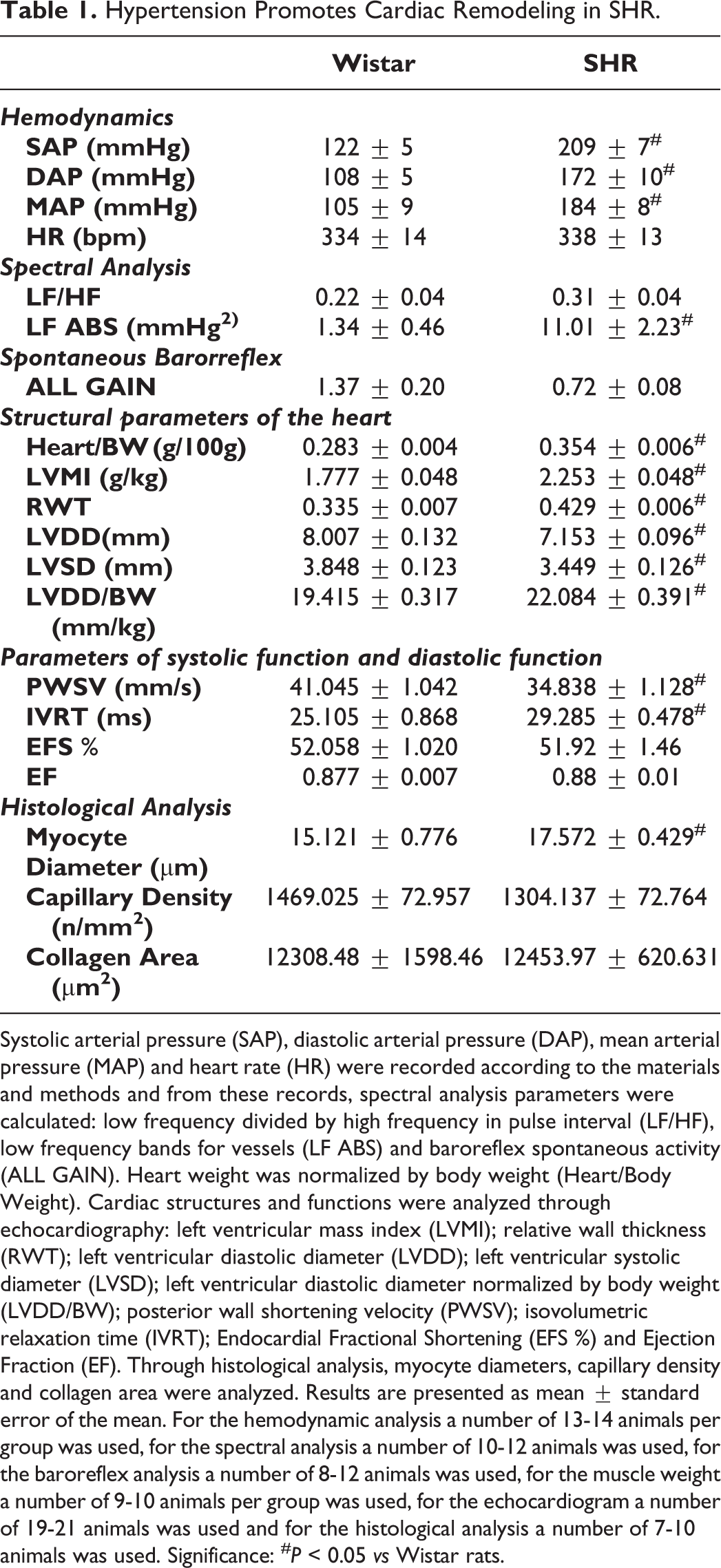
Systolic arterial pressure (SAP), diastolic arterial pressure (DAP), mean arterial pressure (MAP) and heart rate (HR) were recorded according to the materials and methods and from these records, spectral analysis parameters were calculated: low frequency divided by high frequency in pulse interval (LF/HF), low frequency bands for vessels (LF ABS) and baroreflex spontaneous activity (ALL GAIN). Heart weight was normalized by body weight (Heart/Body Weight). Cardiac structures and functions were analyzed through echocardiography: left ventricular mass index (LVMI); relative wall thickness (RWT); left ventricular diastolic diameter (LVDD); left ventricular systolic diameter (LVSD); left ventricular diastolic diameter normalized by body weight (LVDD/BW); posterior wall shortening velocity (PWSV); isovolumetric relaxation time (IVRT); Endocardial Fractional Shortening (EFS %) and Ejection Fraction (EF). Through histological analysis, myocyte diameters, capillary density and collagen area were analyzed. Results are presented as mean ± standard error of the mean. For the hemodynamic analysis a number of 13-14 animals per group was used, for the spectral analysis a number of 10-12 animals was used, for the baroreflex analysis a number of 8-12 animals was used, for the muscle weight a number of 9-10 animals per group was used, for the echocardiogram a number of 19-21 animals was used and for the histological analysis a number of 7-10 animals was used. Significance: # P < 0.05 vs Wistar rats.
DEX Treatment and Treadmill Training Effects on Arterial Pressure, Autonomic Nervous System and Spontaneous Baroreflex of SHR
In order to evaluate the effects of DEX and training on hemodynamic values and autonomic nervous system, hypertensive rats were submitted to the training protocol or remained sedentary for 75 days and, during the last 15 days, they received DEX treatment or saline. SHR groups had similar body weight increase during the training period, but analysis of the delta body weight during DEX treatment indicated effect of treatment (P < 0.001), and post hoc comparisons revealed that DEX treatment significantly reduced body weight in both SD (-15%) and TD (−16%) groups, as well as reduced adrenal gland weight, independent of training (treatment effect, P < 0.001) as shown in Table 2. As expected, trained SHR, regardless of DEX treatment, had higher physical capacity (+96% and +110%, for TC and TD, respectively), compared with sedentary rats (Table 2). All these results confirm the effectiveness of the treatment with DEX and treadmill training.
DEX Treatment and Treadmill Training Effectiveness.

Body weight was measured before and after training period (delta body weight training period); and before and after 15 days of DEX treatment (delta body weight treatment period). Maximal physical capacity was measured before and after training period (delta Tmax). After euthanasia adrenal gland weight was measured and normalized by tibia bone length. Results are presented as mean ± standard error of the mean. For the body weight a number of 11-21 animals per group was used, for the maximal physical capacity a number of 11-21 animals was used and for the adrenal gland weight a number of 10-11 animals was used. Significance: *P < 0.05, for effects of treatment (vs control), + P < 0.05, for effects of training (vs sedentary).
As shown in Figure 2, DEX treatment did not exacerbate SAP, DAP and MAP in SHR, on the other hand, analysis of the arterial pressure indicated effect of training (P = 0.002; P = 0.003 and P = 0.002, for SAP, DAP and MAP respectively). Post hoc comparisons revealed that trained SHR group (TC) showed lower SAP (−20%), DAP (−22%) and MAP (−21%) compared with sedentary SHR. Similarly, trained and treated SHR group (TD) showed lower arterial pressure compared with SD group (−16%, −19% and −18%, for SAP, DAP and MAP, respectively). HR was similar between SHR groups. Values of LF/HF ratio were not different between SHR groups. However, analysis of the sympathetic activity to the vessels indicated effect of training (P = 0.041), without influence of DEX treatment (P = 0.097). Post hoc comparisons revealed that sympathetic activity to the vessels was reduced in SD and TC (−51% and −51%, when compared with SC, respectively). Trained and treated group (TD) presented similar results to the TC group. In addition, spontaneous baroreflex (gain) was similar between groups, as shown in Figure 2.

Effect of DEX treatment and treadmill training on hemodynamic values of SHR animals. Values of systolic arterial pressure (SAP), diastolic arterial pressure (DAP), mean arterial pressure (MAP) and heart rate (HR) were evaluated according to materials and methods (Panel A). From the records of pulsatile arterial pressure, ratio of low frequency band and high frequency band (LF/HF) and low frequency bands for vessels (LF ABS, mmHg2) were analyzed (Panel B), as well as baroreflex activity (ALL GAIN) (Panel C). Results are presented as mean ± standard error of the mean. For the hemodynamic analysis, a group of 6-15 animals was used; for the spectral analysis, a group of 6-12 animals was used and for the baroreflex analysis, a group of 8-13 animals was used. Significance: *P < 0.05, for effects of treatment (vs control), + P < 0.05, for effects of training (vs sedentary).
Effects of DEX and Treadmill Training on Hypertension-Induced LV Remodeling
To confirm if DEX, associated or not with training, would ameliorate cardiac remodeling and function of SHR, analyses of the structures of the LV and cardiac function through echocardiogram and histology were performed. The analysis of cardiac weight indicated effect of training (P < 0.001), without influence of DEX treatment (P = 0.057). Post hoc comparisons revealed that training increased cardiac weight in both controls (+10%, TC vs SC) and treated with DEX (+12%, TD vs SD) groups, with no change in myocyte diameter (Figure 3). From echocardiographic analysis, associated with histology of LV, analysis of LVSD, LVDD/BW and LVDD (Figure 3 and Table 3) indicated effect of treatment (P = 0.006; P < 0.001 and P = 0.009, respectively). Post hoc comparisons revealed that DEX treatment alone or associated with training increased LVSD (+10% and +14%, for SD and TD, vs respective control groups) and LVDD/BW (+14% and +18%, for SD and TD, vs respective control groups). Even without normalization by body weight, LVDD was higher in SD group, suggesting an increase in the heart cavity diameter (+5%, SD vs SC). On the other side, analysis of the relative wall thickness indicated effect of treatment (P < 0.001) and training x treatment interaction (P = 0.020). Post hoc comparisons revealed that relative wall thickness decreased after DEX treatment or training (−12% and −20%, for SD and TD vs respective control groups). The analysis of LVMI indicated training x treatment interaction (P < 0.045) and post hoc comparisons revealed that DEX treatment increased LVMI (+7%) and training prevented this increase, as shown in Figure 3.
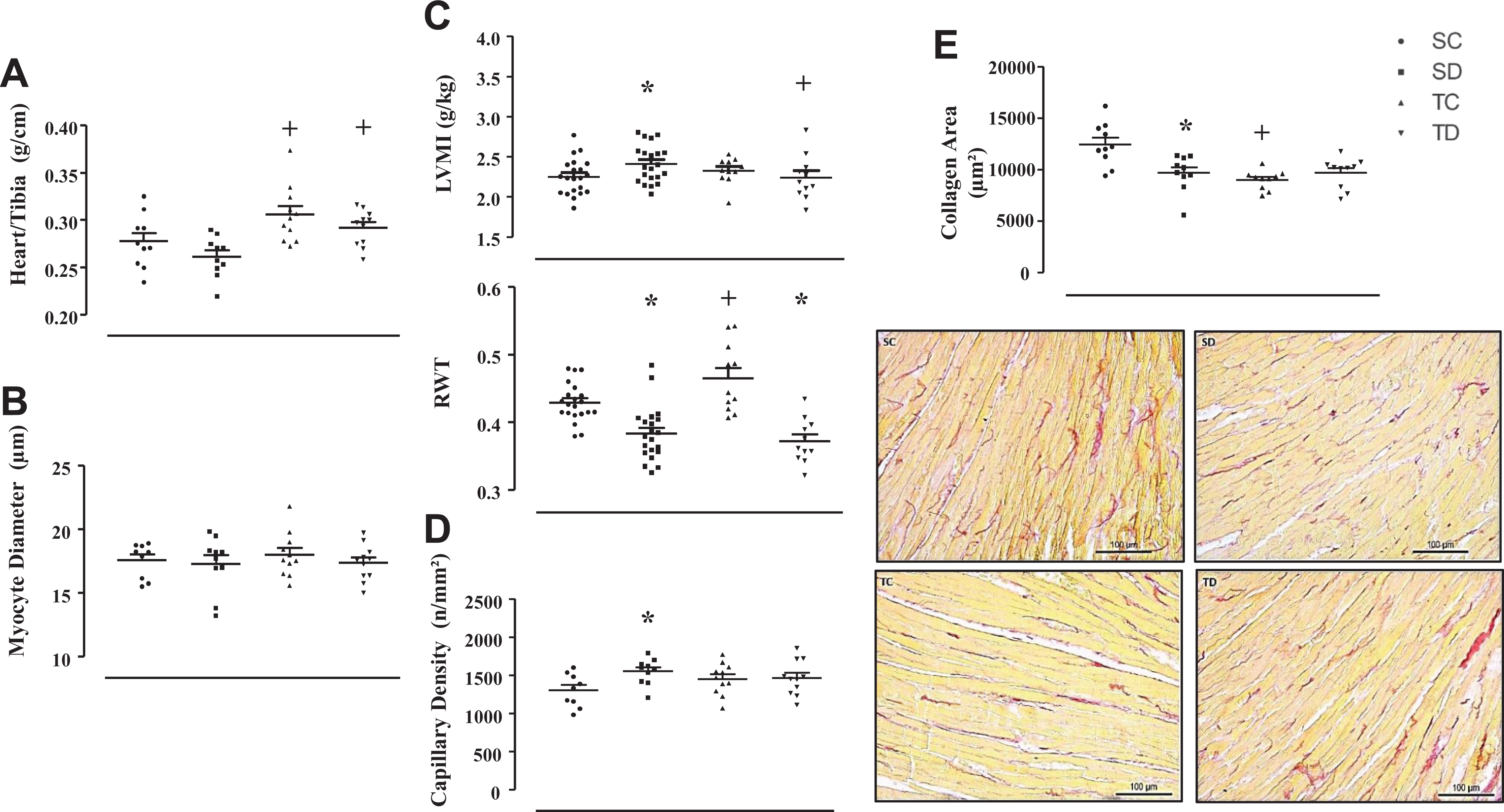
Effect of DEX treatment and treadmill training on LV remodeling of SHR animals. Heart weight (g) was normalized by tibia bone length (cm) in Panel A. Through hematoxylin-eosin technique, analyses of myocyte diameter (µm) on the left ventricle and capillary density (n/mm2) were performed (Panels B and D, respectively). Echocardiogram was used to analyze left ventricular mass index (LVMI) and relative wall thickness (RWT) (Panel C). Panel E (top): densitometric results of the left ventricle (LV) collagen deposition area (μm2) from picrosirius red staining images. Panel E (bottom). Histological sections from cardiac muscle of one animal from each group stained with Picrosirius Red, in which collagen fibers can be seen in red and myocardium in yellow. Results are presented as mean ± standard error of the mean. For the muscle weight, a group of 10-11 animals was used; for the histological analysis, a group of 9-11 animals was used and for the echocardiogram analysis, a group of 11-21 animals was used. Significance: *P < 0.05, for effects of treatment (vs control), + P < 0.05, for effects of training (vs sedentary).
DEX Treatment and Treadmill Training on Cardiac Function of SHR.

From the analysis of the echocardiogram, that was described in the materials and methods, were analyzed: left ventricular diastolic diameter (LVDD) and left ventricular systolic diameter (LVSD). Left ventricular diastolic diameter was normalized by body weight (LVDD/BW). In addition, systolic and diastolic functions were analyzed: posterior wall shortening velocity (PWSV) and isovolumetric relaxation time (IVRT), respectively. Also Endocardial Fractional Shortening (EFS %) and Ejection Fraction (EF) were analyzed. Results are presented as mean ± standard error of the mean. For the echocardiogram analysis, a number of 19-21 animals per group was used. Significance: *P < 0.05, for effects of treatment (vs control), + P < 0.05, for effects of training (vs sedentary).
In addition, Figure 3 shows that the lower relative wall thickness induced by DEX treatment alone, or associated with training, was accompanied by a decrease in collagen deposition area (−22%), and this analysis indicated effect of training (P = 0.002), effect of treatment (P = 0.048) and training x treatment interaction (P = 0.002). Analysis of the capillary density indicated effect of treatment (P = 0.048), and post hoc comparisons revealed that DEX treatment alone, or associated with training increased the capillary density (+19%). Lower collagen deposition and higher capillary density probably contributed to improve the cardiac function, since PWSV was higher (+10% and +17%, for SD and TD vs respective control groups) and IVRT was lower (−13% and −16%, for SD and TD vs respective control groups). For both analyses, effect of treatment was indicated (P = 0.002 and P < 0.001, for PWSV and IVRT, respectively).
Training per se did not significantly change LVMI, but increased RWT, when compared with SC. Even though RWT was higher, collagen deposition area was lower (−28%) in TC vs SC, but it was not enough to change cardiac function, as shown in Table 3.
Discussion
The main results of this study were that low-dose of DEX treatment in SHR did not exacerbate arterial pressure, mainly because it did not change autonomic balance to the heart or spontaneous baroreflex activity, which was a benign response. On the other hand, association of low-dose of DEX treatment with training improved systolic and diastolic function of the heart, which may be associated with lower LV collagen deposition and higher vessel density.
It is well described that SHR is an experimental model that resembles essential hypertension, 25 just as it has been quite described the effects of hypertension on cardiac remodeling. 1,26,27 Although it has been shown that SHR may present an autonomic unbalance to the heart and higher sympathetic activity to the vessels 28 results of the present study revealed that hypertension was maintained mainly by the higher sympathetic nervous activity for the vessels, which contributes to increase arterial pressure, 7 with only few non-significant fluctuations of LF/HF ratio and spontaneous baroreflex activity.
Cardiac remodeling is another important factor to be considered during hypertension progress. Results of the present study showed that SHR had higher cardiac mass, which agrees with some other studies. 2,15,18,29,30 In addition, echocardiographic analysis revealed that structural parameters of the heart were significantly altered in SHR. For instance, SHR presented higher LVMI, accompanied by increased RWT, which may contribute to the reduced internal diameters (LVDD and LVSD) observed. These changes in wall thickness were further confirmed by histological procedures, which have shown an increased myocyte diameter, which could be induced by pressure overload 1,27 and fibrotic infiltration. 30 All these structural changes were followed by an impairment in LV systolic (PWSV) and diastolic (IVRT) function, while LV ejection fraction (LVEF) and LV fractional shortening (LVFS) were still preserved, which may suggest a compensatory phase of cardiac remodeling. 18,27 This cardiac remodeling could also be partially associated with increases in myocardial inflammatory cytokines, 27,29,30 oxidative stress and renin angiotensin system (RAS) hyperactivity, 27,29 as well as abnormal protein expression of sarcoplasmic reticulum Ca2+-regulating proteins. 30
Although DEX is widely used to treat inflammation, 3 allergies, 4 vomiting and nausea 31 and cancer, 32 some side-effects are well described in normotensive rats, even after different doses, for instance, body weight reduction, muscle atrophy, peripheral insulin resistance, skeletal muscle microcirculation rarefaction and hypertension. 5 -9,33 -35
In agreement with previous results, in the present study, DEX treatment induced a significant reduction of SHR body weight. Also, adrenal gland weight was reduced, which confirms the effectiveness of the DEX treatment. In addition, training was not able to attenuate these responses, as already shown in normotensive rats. 6 -8,20,33 Reduction of body weight after DEX treatment could be a consequence of several factors, such as muscle atrophy and/or food intake reduction, 5 but the exact mechanism of body weight reduction was not the focus of this work.
DEX-induced hypertension has been described in normotensive animals, using different experimental models, dosages, routes of administration and treatment periods. 7 -9,11,34,36 -38 Recently, our group has demonstrated in normotensive rats that DEX treatment increases arterial pressure associated with autonomic imbalance to the heart, higher sympathetic activity to the vessels and reduction of baroreflex activity, 7,8 independent of RAS hyperactivity. 8 However, nothing was known about the effects of DEX in rats with already stablished hypertension, like SHR. In this study, we showed for the first time that chronic treatment with DEX did not induce any additional effect on arterial pressure. Although DEX decreased sympathetic activity to the vessels, it did not change autonomic balance to the heart or spontaneous baroreflex activity in SHR. However, regardless of DEX treatment, all trained SHR presented lower values of arterial pressure when compared with their respective sedentary groups. These results agree with previous results found in SHR 2,39 and suggest, for the first time, that low dose of DEX does not interfere with the beneficial response of training in reducing arterial pressure of SHR. In fact, training reduces arterial pressure due to reductions in peripheral vascular resistance, since it is known that training decreases skeletal muscle arterioles wall-to-lumen ratio in SHR 39,40 and, in agreement, the results of the present study showed that trained rats presented lower sympathetic nervous activity to the vessels, which, in turn, may help to decrease local vascular resistance.
Data in the literature are still controversial about the effects of DEX on myocardial remodeling, since some authors showed that DEX treatment causes cardiomyocyte hypertrophy, cardiac dysfunction, accompanied by hypertension, 9 -11,41 -43 while other authors showed a cardioprotective effect. 12 -14,41,43 -49 De Salvi Guimarães et al. 9 revealed that DEX-treated W rats showed increased heart weight, cardiac fibrosis, cardiomyocyte apoptosis and genetic damage in cardiomyocytes, suggesting a pathologic cardiac remodeling, followed by diastolic dysfunction. Cardiac remodeling observed after the treatment with DEX can be associated with activation of the cardiac calcineurin signaling pathway 9 or increased expression of heavy chain myosin alpha isoform (MHC) and decreased MHC-beta expression. 10 In agreement, cardiac hypertrophy is mediated through glucocorticoid receptor 41 and may be accompanied by left ventricle fibrosis and reduction of sarcoendoplasmic reticulum calcium transport ATPase 2a (SERCA2a) expression. 42 Moreover, it has been shown that treatment of pregnant ewes with DEX throughout gestation induces left ventricular hypertrophy in adult offspring and reduces cardiac functional reserve. 11
On the other hand, several cardioprotective effects induced by DEX have been shown, 12 -14,41,43,45 -49 especially under pathological situations, like infarction, heart failure, adrenalectomy or even in aging. For instance, DEX treatment was able to reduce the infarction area 47 or even to attenuate infarction area and apoptosis of the cardiomyocytes, when DEX treatment preceded the infarction surgery, and this response was associated with activation of Bcl-xL. 12 In agreement, Bcl-xL had a critical role in preventing doxorubicin-induced apoptosis of cardiomyocytes when these cardiomyocytes were treated with corticosteroids 13 and this response can be mediated by increases in Leucine Zipper (GILZ). 45 This glucocorticoid induced Leucine Zipper inhibits doxorubicin-induced mitochondrial cytochrome c release and caspase signaling activation. Furthermore, DEX treatment may protect cardiomyocyte apoptosis induced by TNF-α through phosphorylation of the adenosine monophosphate-activated protein kinase (AMPK) and Calcium Calmodulin Dependent Protein Kinase (CaMKI) phosphorylation. 48 This anti-apoptotic response induced by DEX was confirmed after treatment with a glucocorticoid receptor antagonist. 41 In another study, DEX improved cardiac function through reduction of the myocardial TNF-α, which, in turn, reduced metalloproteinases MMP-2 and MMP-9, oxygen reactive species and fibrosis in left ventricle of rats with heart failure. 14 Likewise, cardiac function improvements, induced by DEX treatment may be related to the maintenance of normal function of sarcoplasmic reticulum and, consequently, cellular Ca+2-homeostasis in the myocardium, especially in situations where Ca+2 dynamic is compromised, like adrenalectomy 45,49 and aging. 46
The present study demonstrated that low dose of DEX treatment decreased RWT and increased internal diameters (LVDD and LVSD). In addition, DEX increased LV vessel density and reduced collagen deposition, which most likely contributed to improve systolic and diastolic function, as illustrated in the schematic Figure 4.

Schematic view of the effects of DEX treatment and/or exercise in hypertension. This schematic Figure represents the results of DEX treatment, associated or not with exercise training, on cardiac remodeling and hypertension in SHR. While DEX treatment induced cardiac protection, DEX associated with exercise training on the treadmill induced a decrease on arterial pressure and both results improved cardiac function.
One possible explanation for the improvement of the cardiac function found in this present study could be related with the capacity of DEX in modulating ATP-dependent Ca+2 uptake rate of cardiac sarcoplasmic reticulum membrane vesicles, which is the target for DEX action in the heart. 49 SHR is a model of systemic hypertension that progresses to heart failure at older ages 50,51 and, under this circumstance, quantification of myocardial Ca+2-ATPase (SERCA2a) is highly reduced in SHR compared with W rats. In this present study, young SHR´s hearts (5-6 months) are not in failure yet (EF% is preserved), but it has been shown that even at this age, cardiac hypertrophy may be associated with changes in the mechanisms involved in the excitation–contraction coupling of the myocardium, like lower sarcoplasmic reticulum Ca+2 release, 52,53 reduced Ca+2-ATPase (SERCA2a) levels and activity, as well as, reduced phospholamban phosphorylation. 30 Since DEX may increase CaMKinaseII in cardiac sarcoplasmic reticulum membrane of adrenalectomized rats, 49 which, in turn, is responsible for the phosphorylation of SERCA2a and phospholamban, and also enhance the rate of Ca2+ uptake by sarcoplasmic reticulum in aged rats, 46 we may assume that a possible normalization of SERCA2a and phospholamban expression and phosphorylation could be an important factor for the improvement of cardiac function induced by DEX treatment in this present study using young SHR.
Another possibility could be related to the anti-inflammatory role of DEX. In this sense, since that young SHR present higher levels of interleukin-6 (IL-6) and TNF-α in the myocardium, followed by greater fibrotic infiltration and collagen deposition, 27,29,30 we can assume that treatment with DEX could help to decrease these inflammatory cytokines and improve cardiac fibrosis, in addition to reducing the death of cardiomyocytes induced by TNF-α. 13,14,41,48 Also, our results showed increased myocardial angiogenesis after DEX treatment, which may help to guarantee a good nutrition to the myocardium. Taken together these studies, we may speculate some insights into the mechanisms underlying DEX modulation of SHR cardiac contractile function found in this present work: 1/maintenance of Ca2+ homeostasis; 2/ cardiomyocytes apoptosis protection; 3/ angiogenesis and 4/ integrity of the myocardium.
When DEX treatment was associated with training, we observed about the same responses as we saw in DEX-treated SHR, for instance, LVMI and RWT were lower and both systolic and diastolic internal diameters were higher (LVDD and LVSD), compared with sedentary SHR, which may contribute to improve cardiac function, as already shown in trained SHR. 18 The mechanisms involved are probable the same as discussed earlier for DEX-treated sedentary rats, which helped to induce angiogenesis and decrease myocardial collagen deposition/fibrosis and cardiomyocytes apoptosis, since TD group presented similar values of vessel density and collagen deposition area as SD group. In addition, training has been shown to be effective in improving cardiac function by protecting against cardiomyocyte apoptosis 54,55 and modulating myocardial inflammatory cytokines and RAS hyperactivity, which also help to decrease cardiac fibrosis. 29
Moreover, regardless of DEX treatment, treadmill exercise training was effective in attenuating the increase in arterial pressure, since TC and TD groups presented lower arterial pressure compared with SC and SD, respectively. Training-induced arterial pressure lowering effects have been shown by several studies 29,39,40,56,57 and, recently, Silva et al. (2017) 29 have demonstrated a time course relationship between the effects of exercise training on plasma and cardiac RAS activation, redox status and inflammatory responses in a rat model of spontaneous hypertension, which contributed to decrease arterial pressure and improve cardiac remodeling. Since DEX has been shown to decrease inflammatory cytokines, oxidative stress and RAS activity, 14,41,43,48 we may believe that trained and treated SHR had an additional effect on arterial pressure. Also, training reduces arterial pressure by improving skeletal muscle arterioles resistance and microcirculation. 39,40,56,57
In summary, the results of the present study are unique and show that low-dose of DEX treatment does not exacerbate arterial pressure in SHR and, when associated with training, it decreases arterial pressure, improves LV systolic and diastolic function, which may be associated with cardiac angiogenesis and reduction of the LV collagen deposition. These results suggest that low-dose of DEX treatment, associated or not with exercise training may protect SHR heart and confirm that training is an important and efficient tool in controlling hypertension.
Limitations of this study: The main limitation of this study was the uneven numbers of rats in all groups. This problem occurred because of the mortality rate after several procedures involving anesthesia, but this difference did not negatively impact on statistics.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by São Paulo Research Foundation (FAPESP #2017/00509 -1, 2015/03965-2, 2018/00567-4). FD was recipient of scholarship from FAPESP/CAPES (#2017/14405-3) and from the Coordination for the Improvement of Higher Education Personnel (CAPES #88882.426908/2019-01). NAH was recipient of scholarship from FAPESP (#2016–12532-5). LPT was recipient of scholarship from CAPES (#88882.426901/2019-01).
