Abstract
Catestatin (CST), which is emerging as a novel cardiac modulator, can protect the heart against excessive sympathetic drive in hypertensive cardiomyopathy. The aim of this study is to investigate whether exogenous CST decreases excessive cardiac sympathetic drive and improves autonomic function and exerts cardioprotective effects in myocardial infarction (MI) rats. Rats were divided into a sham group, MI group, and MI plus CST (MI + CST) group. Four weeks later, the autonomic function of the animals was assessed by analyzing heart rate variability (HRV) and measuring plasma catecholamine. Cardiac function was evaluated via echocardiography. Electrophysiological characteristics were assessed in Langendorff-perfused hearts. Compared to the MI group, the chronic administration of CST significantly increased the standard deviation of normal–normal intervals (P < .01) and low-frequency (LF) and high-frequency (HF) HRV and decreased the ratio of LF–HF HRV (P < .01 for all). Additionally, the level of plasma catecholamine was reduced in the MI + CST group compared to the MI group (P < .01). Treatment with CST significantly increased ejection fraction (EF) and fraction shorting (FS) and significantly decreased the left ventricular end-systolic diameter and left ventricular end-diastolic diameter at 28 days postmyocardial infraction (P < .05 for all). After MI, the ventricular repolarization duration, such as QTc intervals and action potential duration (APD) at 90% repolarization, was prolonged, and this prolongation could be decreased by CST (P < .05 for all). The CST also increased the threshold of ADP alternans (P < .01). Moreover, ventricular arrhythmias were induced in 83% of the MI group but only 33% of the MI + CST group (P < .05). These results suggested that the chronic administration of CST plays a role in cardioprotection in MI rats, which may function by decreasing the cardiac sympathetic drive and improving autonomic function.
Introduction
Chromogranin A (CHGA) is an acidic secretory proprotein costored and coreleased with catecholamine from the storage vesicles in adrenal chromaffin cells and adrenergic neurons. 1,2 Via posttranslational proteolytic processing, CHGA gives rise to several bioactive peptides including pancreastatin, 3 vasostatin, 4 and catestatin. 5 Although catestatin (CST) was preliminarily known as a potent nicotinic–cholinergic antagonist, subsequent studies indicated that CST exerts signaling effects in several systems, including the cardiovascular system. 6 The plasma level of CST is diminished in not only patients with essential hypertension but also in normotensive individuals with a family history of hypertension. 7 This indicated that an early deficiency of this catecholamine release inhibitory peptide may play a pathogenic role in the subsequent development of hypertension. Consistent with human findings, Chga knockout mice experienced severe hypertension and 8 dampened baroreflex sensitivity 9 and heart rate variability (HRV) 10 that could be rescued by the replacement of the CST peptide. Three naturally occurring amino acid substitution variants of CST have been discovered, G364S, P370L, and R374Q, which exhibited differential potencies toward the inhibition of catecholamine secretion. 11,12 Notably, human carriers of the Gly364Ser allele showed profound changes in human autonomic activity, both parasympathetic and sympathetic, which reduced the risk of developing hypertension, particularly in males. 13 Additionally, accumulating evidence showed that CST acts directly on the heart to inhibit the inotropic and lusitropic properties of the rodent heart both under basal conditions and after β-adrenergic stimulation. 14,15 This evidence suggested that CST has therapeutic potential for the treatment of hypertensive cardiomyopathy.
A recent study demonstrated that the occurrence rate of adverse cardiac events in patients with acute myocardial infarction (AMI) significantly increased with CST levels. 16 Pei et al also found that the incidence of malignant arrhythmia in patients with AMI significantly increased during hospitalization as the CST level increased. 17 This suggested that the increase in CST may be a compensatory response of sympathetic nerve activity. Additionally, plasma CST is positively correlated with norepinephrine (NE) and was elevated in parallel with that of NE in different myocardial ischemia states. 18 This finding indicates that CST most likely functions as an endogenous compensation to balance sympathetic activity and plays a role in cardioprotection in patients with MI. However, the precise role of CST in MI remains elusive. The purpose of this study is to investigate whether exogenous CST can decrease the overactivated sympathetic tone and balance the function of the autonomic system in rats with myocardial infarction (MI), thereby exerting cardioprotective effects.
Materials and Methods
Experimental Animals
All experimental procedures were performed according to the Guide for the Care and Use of Laboratory Animals Published by the US National Institutes of Health (NIH Publication #85-23, revised 1996), and the protocol was approved by the Animal Care and use Committee of Renmin Hospital of Wuhan University, China.
Male Sprague-Dawley rats (Experimental Animal Center of the Medical College of Wuhan University), each weighing 220 to 250 g, were randomly divided into 3 groups: a sham-operated group (sham group, n = 15), an MI group (MI group, n = 25), and an MI plus CST group (MI + CST group, n=25). Myocardial infarction was created via ligation of the left anterior descending coronary artery. Coronary artery ligation was performed as described previously. 19 Briefly, rats were anesthetized (pentobarbital sodium, 40 mg/kg, intraperitoneally (IP); Sigma, St. Louis, USA), intubated, and ventilated using a volume–constant rodent ventilator, and a left thoracotomy was performed. The heart was exteriorized from the thorax, and the left anterior descending artery was ligated using an 8-0 Prolene suture between the right ventricular outflow tract and the left atrium. The heart was then returned to its normal position, and the thorax was closed. The sham group was operated using the same protocol, with the exception that the coronary artery was not ligated. The MI model was confirmed according to the ST-segment of the electrocardiogram (ECG) and regional cyanosis of the myocardial surface. The MI + CST group was given CST (synthetized by Wuhan Moon Biosciences Co Ltd, China,) 0.25 mg/kg/12 hours via IP injection 24 hours after AMI for 28 days. In the MI group, normal saline was administered.
Echocardiographic Measurements
The changes in the left ventricular (LV) function were evaluated via transthoracic echocardiography at the 28th day postsurgery. The LV end-diastolic diameter (LVEDD), LV end-systolic diameter (LVESD), LV ejection fraction (LVEF), and fractional shortening (FS) were calculated via M-mode tracing.
Telemetry Probe Implantation
Rats were anesthetized with pentobarbital sodium (40 mg/kg, IP). The body of the ETA-F10 ECG radio telemetry transmitter (Data Sciences International [DSI], St Paul, Minnesota] was positioned in the abdominal cavity, and the 2 biopotential leads were anchored subcutaneously on the lower left side of the chest wall and over the right pectoral muscle, respectively. 20 The telemetry transmitters were turned on following 1 week of recovery time and were housed individually with cages placed on separate receive pads (model RPC-1. DSI), thereby enabling simultaneous ECG recording.
Data Acquisition and Analysis
Data were converted into TXT data format files, which were imported into Chart 7.0 software (AD instruments, New South Wales, Australia) for analyses. The RR, PR, QRS, and QT duration were measured, and the mean values were calculated. The QT was corrected for heart rate using the modification of Bazett formula: QTc = QT/(RR/100)1/2. Heart rate variability was determined from 5-minute segments of ECG data. The choice of segments used for the analysis was based on the relative lack of movement artifacts and arrhythmias. Arrhythmic beats were manually removed. The standard deviation (SD) of normal–normal intervals (SDNN) was determined from 5-minute segments of data from each hour. Fast Fourier transformation of the same 5-minute segments of the ECG was performed after the removal of linear trends and the application of a Welch window with a fast Fourier transform setting of n = 1024 points with 50% overlap. Spectral power was quantified within the following frequency bands: a low frequency (LF) power of 0.06 to 0.6 Hz and a high frequency (HF) power of 0.6 to 3.0 Hz. 21
Catecholamine Assay
Testing of the concentration of plasma catecholamine was performed as described previously. 9 Briefly, the rats were anesthetized using pentobarbital sodium (40 mg/kg, IP), and blood was collected from the left ventricle in potassium EDTA tubes. Plasma catecholamine was measured via high-performance liquid chromatography connected to an electrochemical detector. Separation was performed on an Atlantis dC18 column from Waters (Massachusetts, USA). An internal standard, [3, 4-dihydroxybenzylamine (1 ng)], and antioxidant sodium metabisulfite were added to 0.25 mL of plasma. After a 30-minute incubation and centrifugation (5000 rpm for 5 minutes at room temperature), the supernatant was discarded, and the beads were washed with water. The catecholamine was eluted with 80 μL of 0.1 N HCl supplemented with 0.1 mmol/L sodium metabisulfite. The data were analyzed using Empower software (Multiskan MK3, Thermo). Catecholamine levels were normalized with recovery of the internal standard.
Preparation of Langendorff-Perfused Hearts
The rats were anesthetized with pentobarbital sodium (40 mg/kg, IP) and heparinized with heparin sodium (2000 U). After a thoracotomy, the hearts were quickly isolated, cannulated rapidly into the Langendorff perfusion system (AD Instruments), and secured for retrograde perfusion at 37.0°C ± 0.5°C with oxygenated Tyrode solution at a rate of 6 to 8 mL/min. The composition of Tyrode solution (in mmol/L) at a pH of 7.40 ± 0.05 (adjusted with NaOH) was as follows: NaCl, 130; KCl, 5.4; CaCl2 1.8; MgCl2, 2; Na2HPO4, 0.3; hydroxyethyl piperazine ethanesulfonic acid (HEPES), 10; and glucose, 10 in deionized water. Prior to further experimental procedures, the isolated hearts were perfused for 20 minutes. Hearts that had irreversible myocardial ischemia or did not recover a regular spontaneous rhythm were discarded.
Monophasic Action Potential Recording
The monophasic action potential (MAP) recording was performed in LV using a custom-made Ag–AgCl electrode, which consisted of two 0.25-mm, Teflon-coated silver wires (99.99% purity). 22 All MAP signals were recorded and analyzed using the PowerLab system and Chart 7.0 software (AD instruments). The amplifier bandwidth signal was between 0.3 and 1 kHz.
Action Potential Duration at 90% Repolarization and Action Potential Duration Alternans
The paired platinum-stimulating electrodes were positioned on the basal surface of the right ventricle, and the pacing stimuli were in 2-millisecond pulse widths with 3-fold the diastolic threshold current. The S1–S1 pacing protocol was performed with a series of pulse trains at a regular pacing cycle length (PCL). Starting at 300 milliseconds, the PCL was shortened to 160 milliseconds in 20-millisecond intervals and then to 40 milliseconds in 10-millisecond intervals. The regular pacing at each PCL lasted for 15 seconds to ensure a steady rhythm, and each pacing was separated by at least 30 seconds to minimize the pacing memory. The action potential duration (APD) at the PCL of 300 milliseconds in each site was determined at 90% repolarization (APD90) to calculate the average APD90 of the left ventricle. The APD alternans was assessed at each site by subtracting the APD90 for 2 consecutive beats when the alternate APD90 differed by 5% over 10 beats. The threshold was defined as the maximal PCL (PCLmax) that induced APD alternans.
Ventricular Arrhythmia Inducibility
Burst pacing protocols were performed to determine the ventricular arrhythmia (VA) susceptibility. Burst pacing (2-millisecond pulses at 50 Hz and 2 spacing durations) was performed at the left anterior free-wall (LAF), which was repeated 3 times after a 2-second rest period. Ventricular arrhythmia was defined as a run of 2 seconds or more consecutive premature ventricular contractions. Ventricular arrhythmia was considered nonsustained when the contractions lasted 2 to 30 seconds and sustained when they lasted >30 seconds according to the clinical criteria. The vulnerability of VA was evaluated based on the incidence of VA and the ratio of sustained VA.
Statistical Analysis
SPSS 19.0 for Windows (SPSS Inc, Chicago, Illinois) was used for the statistical analyses. Continuous data are expressed as the mean ± SD and compared via analysis of variance followed by the Newman-Keuls multicomparsion test. The median threshold values of APD alternans were compared using the Kruskal-Wallis H test followed by the Nemenyi test. The incidence of VA was compared using the χ2 test. A value of P < .05 was considered statistically significant.
Results
Mortality and Heart–Body Weight Ratio
A total of 65 rats were included (15 in the sham group, 25 in the MI group, and 25 in the MI + CST group). In the MI group, 8 rats died during the study period; of these, 3 died within the first 24 hours after coronary ligation. Seven rats died in the MI + CST group during the study period; of these, 2 died within the first 24 hours after coronary ligation. The sham-operated controls survived until the end of the study period. The heart–body weight ratio in MI rats was significantly increased compared to the sham group (P < .01). However, no significant differences were observed in the heart–body weight ratio in both MI groups (P > .05; Table 1).
Effect of CST on Cardiac Function.

Abbreviations: HW, heart weight; BW, body weight; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; FS, fractional shorting; LVEF, left ventricular ejection fraction; CST, catestatin; MI, myocardial infarction.
a P < .01 versus sham group.
b P < .05 versus MI group.S
Effect of CST on Cardiac Function
After MI, the LV dilatation and systolic dysfunction were observed as shown by the increased LVEDD and LVESD and the decreased LVEF and FS (P < .01 for all). The CST exerted cardioprotective effects by improving LV systolic performance (LVEF: MI, 50 ± 5.36 and MI ± CST, 58 ± 4.41; FS: MI, 36.4 ± 4.74 and MI ± CST, 42.6 ± 4.55; P < .05 for all) and attenuating LV dilation (LVEDD: MI, 6.62 ± 0.55 and MI ± CST 5.96 ± 0.48; LVESD: MI, 3.77 ± 0.33 and MI ± CST, 3.09 ± 0.34; P < .05 for all; Table 1).
Changes in the Time Domain and Frequency Domain of HRV
After 4 weeks of operation, the time domain and frequency domain of HRV were performed to assess autonomic function. The MI rats showed a significant decrease in SDNN (sham: 12.1 ± 1.4; MI: 6.7 ± 1.3; P < .01) compared with the sham rats. However, this effect was relieved by the chronic administration of CST (MI: 6.7 ± 1.3; MI ± CST: 9.3±1.2; P < .01; Figure 1). Additionally, the LF (sham: 7.0 ± 1.1; MI: 4.5 ± 0.8; P < .01) and HF (sham: 7.2 ± 1.3; MI: 1.87 ± 0.48; P < .01) were reduced, whereas the LF–HF (sham: 0.96 ± 0.09; MI: 2.4 ± 0.28; P < .01) was increased in the MI group compared with the sham group. However, after the administration of CST for 4 weeks, the MI + CST rats demonstrated an increase in LF (MI: 4.5 ± 0.8; MI ± CST: 5.6 ± 0.7; P < .01) and HF (MI: 1.87 ± 0.48; MI ± CST: 3.66 ± 0.58; P < .01) and a decrease in the LF–HF ratio (MI: 2.4 ± 0.28; MI ± CST: 1.5 ± 0.12; P < .01). This finding indicated that the chronic administration of CST could improve cardiac autonomic function in MI rats (Figure 2).

Time domain parameters of HRV among 3 groups. (A), (B), and (C) represents the typical Lorenz return maps acquired from the sham, MI, and MI + CST groups. Plotted on abscissa is (R-Rn) against the next (R-Rn + 1) interval time on the ordinate. (D) SDNN was decreased after MI compared with the sham group. In contrast, the chronic administration of CST increased the SDNN in the MI + CST group. *P < .01 versus sham group; # P < .01 versus MI group. HRV indicates heart rate variability; SDNN, SD of N-N; MI, myocardial infarction; CST, catestatin.

The frequency domain parameters of HRV in 3 groups. A, FFT spectrum for HRV obtained from the sham, MI, and MI + CST groups. The vertical boundaries are LF = 0.06-0.6 Hz and HF = 0.6-3 Hz. B-D, The LF and HF was reduced, whereas the LF/HF was increased in the MI group compared to the sham group; LF and HF were relieved through the chronic administration of CST. *P < .01 versus sham group; # P < .01 versus MI group. FFT indicates fast Fourier transform; LF, low frequency; HF, high frequency; HRV, heart rate variability; MI, myocardial infarction; CST, catestatin.
Level of Plasma Catecholamine
The level of plasma epinephrine (EPI; sham: 239 ± 11.9; MI: 425 ± 44; P < .01) and NE (sham: 79 ± 10; MI: 180 ± 11; P < .01) were higher in the MI group than in the sham group, which indicated that sympathetic activity was hyperactive after MI. However, the levels of EPI (MI: 425 ± 44; MI ± CST: 330 ± 18; P < .01) and NE (MI: 180 ± 11; MI + CST: 127 ± 5.1, P < .01) in the MI + CST group were lower than those in the MI group, indicating that sympathetic hyperactivity after MI was inhibited by the chronic administration of CST (Figure 3).

Quantification of plasma epinephrine (EPI) and norepinephrine (NE). Exogenous catestatin (CST) significantly reduced elevated plasma EPI and NE after myocardial infarction. *P < .01 versus sham group; # P < .01 versus myocardial infarction (MI) group.
Changes in Surface ECG and Ventricular Repolarization
As shown in Figure 4A, frequent premature ventricular contractions were observed in MI rats; however, fewer were observed in MI + CST rats. Additionally, MI rats exhibited widened QRS complexes and prolonged QTc (P < .01 for all) intervals. However, the chronic administration of CST markedly attenuated these alterations after myocardial infarction (QRS: MI 26 ± 1.5, MI ± CST 18 ± 2.0, P < .01; QT: MI 76 ± 4.5, MI ± CST 64±5.0, P < .05; QTc: MI 60 ± 3.5, MI + CST 50 ± 3.9, P < .05; Figure 4).
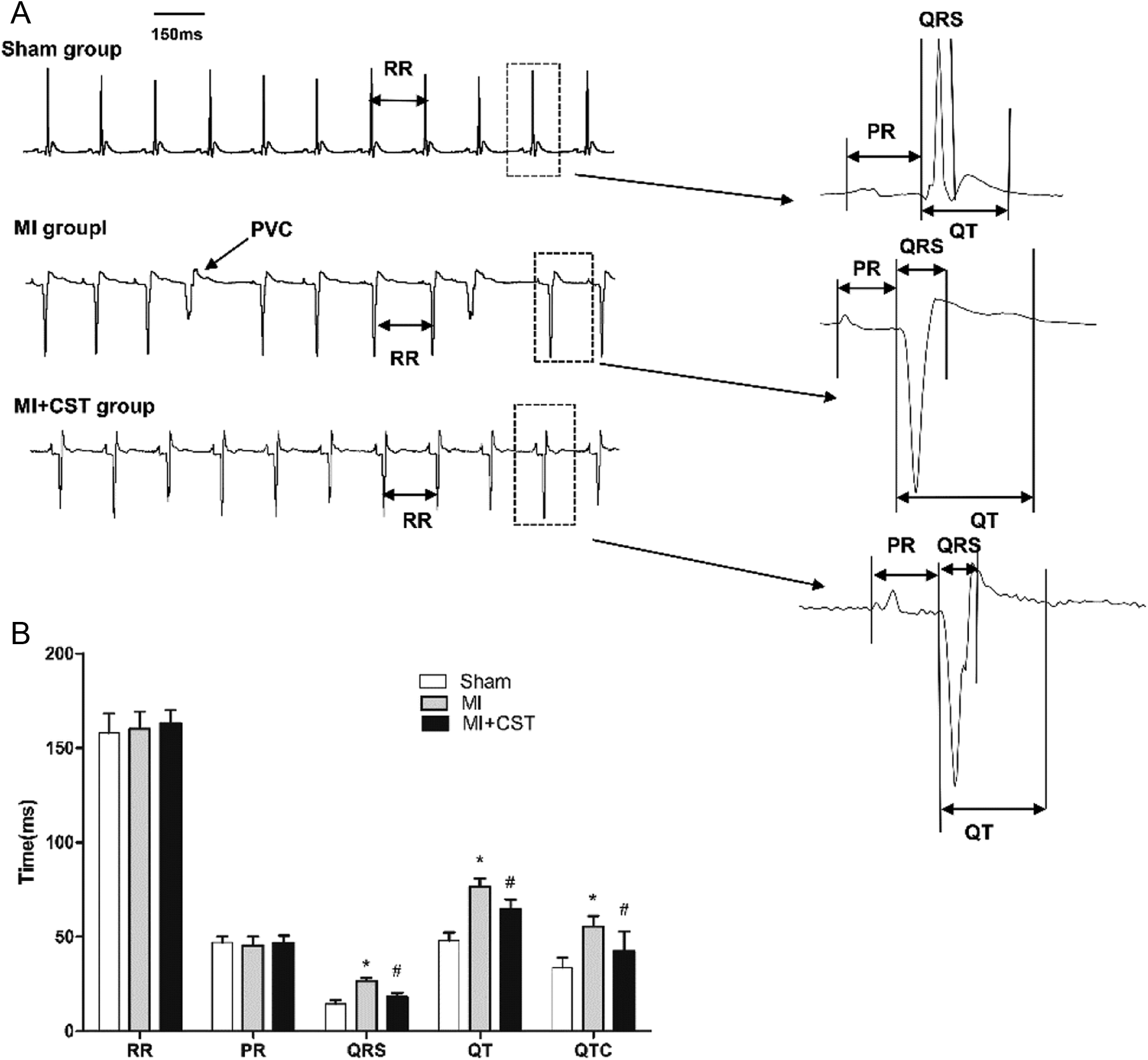
Representative electrocardiogram (ECG) and the comparison of ECG parameters. A, Frequent premature ventricular contraction were observed in the MI rat; these were markedly reduced in myocardial infarction (MI) + catestatin (CST) rats. B, There were longer QRS, QT, and QTc intervals in the MI group compared with the sham group. However, these parameters were normalized after the administration of CST. The RR and PR intervals were similar between the 3 groups. *P < .01 versus sham group; # P < .05 versus MI group.
Furthermore, as shown in Figure 5A, the APD90 of MI rats was significantly prolonged compared with the sham rats (sham: 58 ± 5.9; MI: 79 ± 6.0; P < .01) and was decreased by the chronic administration of CST (MI: 79 ± 6.0; MI ± CST: 72 ± 5.8; P < .05). These data indicated that the chronic administration of CST could attenuate the abnormal ventricular repolarization induced by MI, which may contribute to reducing the vulnerability to VA (Figure 5).

Comparisons of ventricular repolarization between the 3 groups. A, Representative APD recorded from the LV of each group. B, Chronic administration of CST significantly reduced the prolonged APD90 after MI. *P < .01 versus sham group; # P < .05 versus MI group. APD90 indicates action potential duration at 90% repolarization; LV, left ventricular; CST, catestatin; MI, myocardial infarction.
Action Potential Duration Alternans and Burst Pacing-Induced VA
Figure 6 shows that the APD alternans occurred in 3 groups. The threshold of APD alternans was significantly decreased in MI rats but was increased by the chronic administration of CST (P < .01). Additionally, nonsustained ventricular tachycardia or ventricular fibrillation was induced in 83% of the MI rats but in only 33.3% of the MI + CST rats. Furthermore, sustained ventricular tachycardia (VT) or ventricular fibrillation (VF) was induced in 66% of the MI rats and 16% in the MI + CST rats (P < .05 for all; Figure 7).

The APD alternans was induced in the 3 groups. A, MAP tracingsduring the APD alternans in 1 rat of each group. B, The threshold of APD alternans was lower in the MI group compared with the sham group. The CST prevented the reduction in the APD alternans threshold induced by MI. *P < .01 versus sham group; # P < .01 versus MI group. MAP indicates monophasic action potential; CST, catestatin; MI, myocardial infarction; APD, action potential duration.
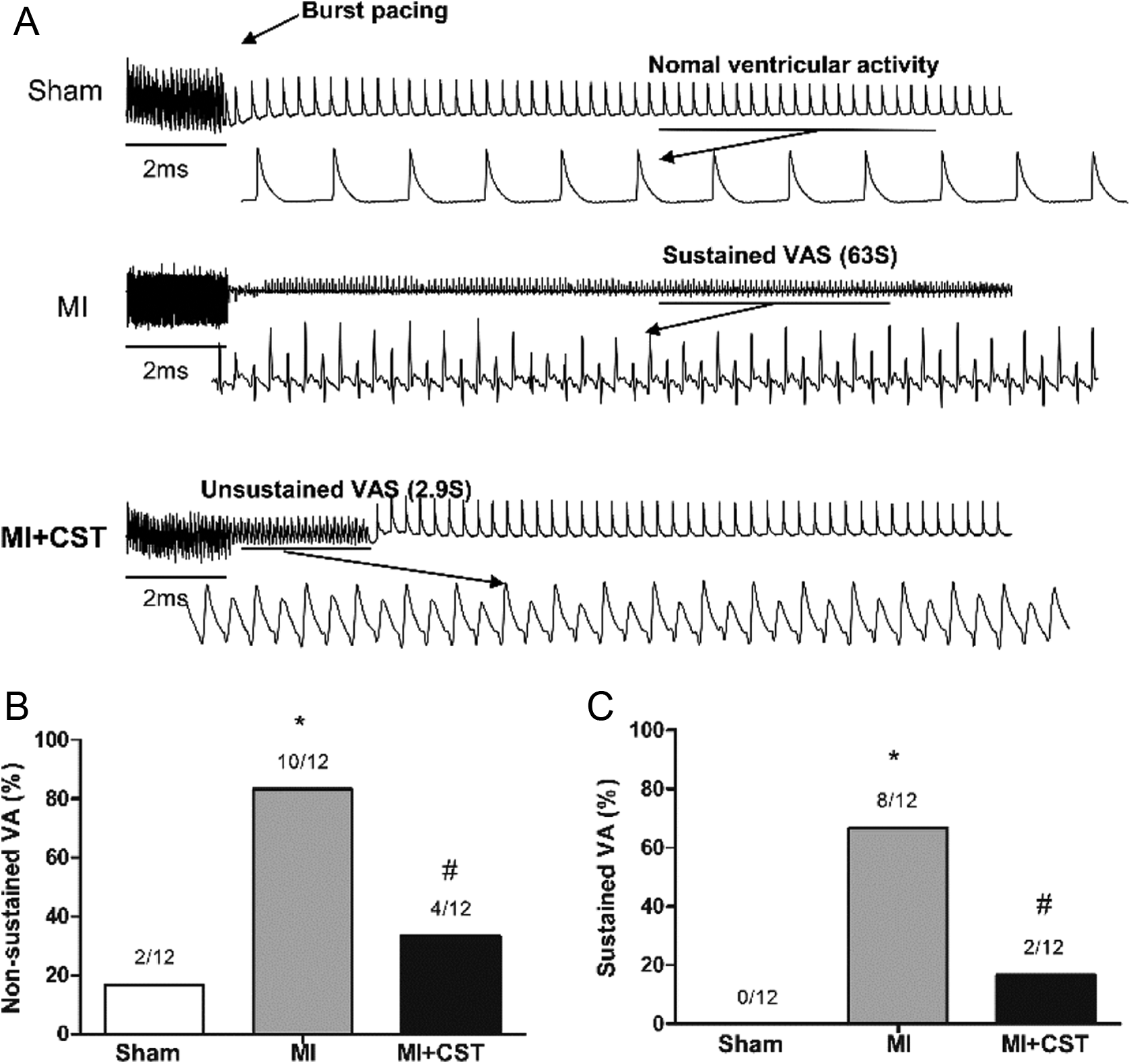
VA was elicited by burst pacing in each group. A, Examples of VA in the 3 groups. B, The nonsustained VA was induced in 10 of 12 MI rats, and 4 of 12 CST + MI rats. C, Sustained VA was induced in 8 of 12 MI rats and 2 of 12 MI + CST rats. *P < .01 versus sham group; # P < .05 versus MI group.VA indicates ventricular arrhythmia; CST, catestatin; MI, myocardial infarction.
Discussion
Overview
The main finding of the present study are as follows: (1) The chronic administration of CST decreased the cardiac sympathetic and improved autonomic function after MI, as indicated by the HRV indices and the level of plasma NE and EPI. (2) Administration of CST could improve the impaired cardiac function observed in MI rats. (3) The chronic administration of CST could relieve the electrical remodeling and reduce the vulnerability to VA after MI.
In vitro, CST is a potent inhibitor of chromaffin cells and sympathetic neurite catecholamine release. 5 This peptide also functions as a nicotinic–cholinergic antagonist by blocking agonist-induced increases in both the secretion and the transcription of catecholamine in sympathochromaffin cells in vivo. 23 However, the studies that discovered CST’s ability to inhibit catecholamine release were performed under normal conditions. In the present study, we found that exogenous CST could reduce the level of plasma NE and EPI after MI.
Previous studies found that patients with MI had significantly higher plasma CST levels than patients with angina pectoris. Additionally, the level of CST was positively correlated with the levels of NE. 17,18 This finding suggested that CST most likely functions as an endogenous compensation to balance sympathetic activity and that it plays a cardioprotective role in MI. However, this compensation effect may not be sufficient, which would explain why a balance between CST and catecholamine could not be achieved at a higher level. Exogenous CST may help in balancing sympathetic activity in MI rats. Another important antiadrenergic mechanism of the CST is that it directly affects the heart. 15,24,25 Angelone et al reported that the CST abolished isoproterenol-induced positive inotropism and lusitropism in Langendorff-perfused rat hearts. 15
The SDNN, which is the time domain of HRV, reflects the amount of HRV available. Generally, if this measure is extremely low, true autonomic dysfunction can be assumed. 26 Heart rate variability (such as SDNN) was previously demonstrated to be reduced in Chga knockout mice; CST replacement reversed these differences. 10 This finding indicates that CST has the potential to minimize adverse autonomic traits, thereby favorably influencing cardiovascular outcomes. In this study, we found that the SDNN was significantly depressed in MI rats, which was reflected by nonlinear (Lorenz) beat-to-beat measurements of R-R intervals (Figure 1A). This finding suggested that autonomic function was impaired after myocardial infarction. Excitingly, this effect was alleviated by the chronic administration of CST, suggesting that exogenous CST could improve depressed autonomic function in MI rats.
The decrease in LF power and HF power in rats after MI compared with sham was likely a result of a reduction in parasympathetic activity. However, these effects could be rescued via exogenous CST. This finding suggested that CST could increase the parasympathetic activity in rats after MI. Furthermore, the increase in the LF–HF ratio after MI could be relieved by CST. This finding indicated that CST could decrease the excessively activated sympathetic and increase parasympathetic activities in MI rats.
The mechanism by which CST improves the HRV has not been explored in this study. As mentioned previously, the most thoroughly documented effect of CST is to inhibit catecholamine release 5,23 and cause vasodilation. 27,28 Catecholamine exerts important central effects that are central to efferent parasympathetic and sympathetic outflow. For example, antihypertensive clonidine acts as a central α2 adrenergic agonist to enhance efferent parasympathetic activation. 29 Alternatively, CST has been reported to exert a vital role in blocking the sympathoexcitatory effects of nicotinic receptors and adrenoceptors. 30,31 Thus, we speculated that CST may act on the central nervous system and thereby decrease the sympathetic efferent and increase the parasympathetic efferent in MI rats.
Abnormal repolarization or intracellular calcium regulation could act individually or in concert to decrease cardiac electrical stability and increase the propensity for VA. 32 Myocardial infarction induces repolarization abnormalities that can provide an electrophysiological substrate for VAs. 33,34 In our study, the heart weight–body weight (HW–BW) ratio significantly increased in both MI groups, which could significantly influence cardiac electrical activity. A previous study showed that exercise training can decrease APD in susceptible animals to such an extent that it no longer differed from animals without infarction. 32 This finding may be due to the improved autonomic function after exercise training. Thus, the prolongation of QTc intervals and APD90 in MI were abolished by the chronic administration of CST, which was most likely attributed to improved autonomic function. Notably, the HW–BW ratio did not significantly differ between the MI groups; however, the ventricular repolarization was significantly decreased in the MI + CST rats compared with the MI rats. We speculate that CST may more substantially influence cardiac electrical activity than cardiac hypertrophy.
Although the mechanism of APD alternans has not been completely elucidated, there is evidence that impaired Ca handling plays a critical role in the occurrence of APD alternans. 35,36 Because changes in intracellular calcium play a critical role in the signal transduction process (parasympathetic activation reduces, whereas sympathetic activation increases myocyte calcium), 32 the chronic administration of CST can be inferred to decrease cardiac sympathetic activity and increase cardiac parasympathetic activity, which can improve Ca handling and eventually increase the threshold of APD alternans. The decreased ventricular repolarization and increased threshold of APD alternans may contribute to reducing the vulnerability to VA in the MI + CST group.
Clinical Implications
Previous studies found that patients with MI had significantly higher plasma CST levels than patients with angina pectoris. However, the specific role of plasma-elevated CST in patients with MI is not known. In the present study, the chronic administration of CST decreased cardiac sympathetic activity and increased cardiac parasympathetic activity and improved cardiac function to reduce the vulnerability to VA in myocardial rats. Thus, the plasma-elevated CST in patients with MI most likely functions as an endogenous compensation to balance sympathetic activity and plays a key role in cardioprotection in MI.
Study Limitations
There are some limitations to the present study. First, HRV provides an indirect assessment of cardiac autonomic activity and does not provide a direct measurement of either cardiac parasympathetic or sympathetic nerve activity. Second, we used Langendorff-perfused rat hearts to assess the vulnerability to VA; however, this setting was not affected by autonomic function. Autonomic function plays an important role in electrical remodeling for 4 weeks following MI. Third, we only assessed the HRV at 4 weeks after MI. This may not be sufficient to describe the course of dynamic changes in HRV after MI. Finally, this study provides evidence indicating that CST exerts cardioprotective effects in MI rats; however, this effect was not causally linked to the improvement in autonomic function. The improvement in autonomic function may be one mechanism contributing to or associated with this finding.
Conclusion
The chronic administration of CST improved cardiac function and reduced vulnerability to ventricular tachyarrhythmia in MI rats. This effect may act through a mechanism of decreasing the cardiac sympathetic drive and increasing the parasympathetic outflow.
Footnotes
Author Contribution
Dandan Wang, Tao Liu, and Shaobo Shi contributed equally to this work. Huang, C contributed to conception and design, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Wang, D contributed to design, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Liu, T contributed to acquisition, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Shi, S contributed to acquisition, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Li, R contributed to analysis, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Huang, Y and Shan, Y contributed to acquisition, drafted manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Hu, D critically revised manuscript and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Basic Research Development of China (The “973” Program; no. 2012CB518604).
