Abstract
Background:
Allogeneic cardiosphere-derived cells (CDCs) exert cardioprotective effects when administered intracoronarily after reperfusion in animal models of acute myocardial infarction (AMI). The “no-reflow” phenomenon develops rapidly post-reperfusion and may undermine the efficacy of cell therapy, due to poor cell delivery in areas of microvascular obstruction (MVO). We hypothesized that CDC-induced cardioprotection would be enhanced by cell administration prior to reperfusion, when microvasculature is still relatively intact, to facilitate widespread cell delivery within the ischemic area.
Methods and Results:
We studied 81 farm pigs; 55 completed the specified protocols. A dose-optimization study in infarcted pigs demonstrated that the doses of 5 million and 10 million CDCs are the maximum safe doses that can be administered intracoronarily at 5 minutes prior to and at 5 minutes post-reperfusion, respectively, without aggravating MVO. Quantification of acute cell retention by polymerase chain reaction demonstrated that cell delivery prior to reperfusion resulted in higher cardiac cell retention compared to delivery post-reperfusion. We then performed a randomized, placebo-controlled study to assess the long-term efficacy of intracoronary infusion of 5 million allogeneic CDCs, delivered at 5 minutes prior to reperfusion, in a porcine model of AMI. The CDC therapy resulted in decreased scar size, improved regional systolic function, and attenuation of adverse cardiac remodeling (manifested as preserved global systolic function, preserved end-systolic volume, and decreased interstitial fibrosis) compared to placebo at 30 days post-MI.
Conclusions:
Dose-optimized intracoronary infusion of allogeneic CDCs prior to reperfusion in a porcine model of AMI is feasible, safe and confers long-term benefits.
Keywords
Introduction
Percutaneous coronary revascularization has led to significant improvement in the outcomes after acute myocardial infarction (AMI). 1 Still, despite timely reperfusion and state-of-the-art medical treatment, up to 30% of patients will develop heart failure within 5 years of the index event 2,3 due to left ventricular (LV) systolic dysfunction and adverse remodeling triggered by cardiomyocyte loss and replacement fibrosis. 4,5
Allogeneic cardiosphere-derived cells (CDCs) have been shown to exert cardioprotective effects (manifested as infarct size reduction and LV remodeling attenuation) when administered intracoronarily 30 minutes postreperfusion in animal models of AMI. 6,7 However, ischemia and reperfusion injury along with microvascular obstruction (MVO) can result in the “no-reflow” phenomenon, defined as a state of impaired myocardial perfusion in the microvascular level despite restoration of blood flow in the epicardial infarct-related artery. 8 No-reflow areas can be detected as early as 5 minutes after restoration of blood flow to the ischemic myocardium and the phenomenon is aggravated as reperfusion evolves 9 -11 ; specifically, the area of no-reflow rapidly expands during the first hour of reperfusion, with minimal further expansion thereafter. 12 The no-reflow phenomenon is of particular clinical relevance, given that its presence is associated with development of heart failure and increased mortality. 13 In addition, development of no-reflow prior to intracoronary administration of cellular products may undermine the efficacy of cell therapy, due to poor delivery of cells in areas of no-reflow. We hypothesized that the magnitude of CDC-induced cardioprotection would be enhanced if CDC administration was temporally shifted toward an earlier phase of the ischemia–reperfusion timeline, when microvasculature is still intact, to facilitate widespread and homogeneous delivery of cells within the ischemic area.
In this study, we aimed to assess the feasibility, safety, and efficacy of allogeneic CDCs therapy administered intracoronarily prior to development of significant no-reflow, in a porcine model of ischemia–reperfusion. We first performed a dose-optimization study, in order to define the maximum CDC dose that can be safely delivered intracoronarily—without aggravation of MVO—at 2 different time points: at 5 minutes before reperfusion (when microcirculation is still patent and no-reflow has not yet developed) and at 5 minutes postreperfusion (when no-reflow is still minimal). 11 We then assessed the acute cardiac cell retention after administration of the maximum safe CDC dose at each time point, in order to define the optimal timing for CDC administration (5 minutes pre-reperfusion vs 5 minutes post-reperfusion). Finally, after defining the maximum safe dose and the optimal timing for CDC administration, we performed a randomized pivotal study to investigate the long-term effects of intracoronary infusion of allogeneic CDCs in a porcine model of AMI.
Methods and Materials
Eighty-one, 14 to 16 weeks old, outbred (genetically non-identical) farm pigs, weighing 30 to 40 kg, were used in the studies. Study protocols were approved by the Scientific Committee of our institution and the Veterinary Committee of Attica. Detailed methodology is provided in Online Supplement.
Myocardial Tissue Isolation and Cell Culture
Myocardial tissue samples were obtained from 10 male farm pigs and cell culture was performed according to published protocols, 14,15 as described in detail in the Online Supplement.
Dose-Optimization Study
The objective of this non-randomized study was to define the maximum CDC dose that can be safely delivered intracoronarily without aggravating MVO (ie, without aggravating no-reflow) in infarcted pigs, at 2 different time points: at 5 minutes before reperfusion (5′ pre-rep group) and at 5 minutes post-reperfusion (5′ post-rep group). Escalating CDC doses were administered to each group and ischemic area at risk (AR) and MVO were measured 2 hours following cell delivery in both groups by histopathology, as described in detail in the Online Supplement. A schematic depiction of the study protocol is presented in Figure 1A (5′ pre-rep group) and 1B (5′ post-rep group). A total of 45 female farm pigs were used in this study (5′ pre-rep group n = 22, 5′ post-rep group n = 23).

Schematic depiction of study protocols. Dose-optimization study: 5′ pre-rep group (A) and 5′ post-rep group (B). Cell retention study: 5′ pre-rep group (C) and 5′ post-rep group (D). Pivotal study (E). CDCs indicates cardiosphere-derived cells; ic, intracoronary; LAD, left anterior descending artery; LVG, left ventriculography; MI, myocardial infarction; MVO, microvascular obstruction.
Cell Retention Study
The objective of this non-randomized study was to quantify acute cardiac cell retention after administration of the maximum safe CDC dose (as determined by the dose-optimization study) at each time point (5 minutes pre-reperfusion vs 5 minutes post-reperfusion), in order to define the optimal timing for CDC administration. Allogeneic CDCs isolated from male donors were administered to infarcted female recipients. We administered cells obtained from male donors to female recipients in order to quantify cell retention by real-time polymerase chain reaction (PCR), using the SRY gene located on the male-specific Y chromosome as target. Acute cell retention was quantified at 2 hours post-delivery. A schematic depiction of the study protocol is presented in Figure 1C (5′ pre-rep group) and 1D (5′ post-rep group). A total of 8 female farm pigs were used in this study (5′ pre-rep group n = 4, 5′ post-rep group n = 4).
Pivotal Study
The objective of the randomized pivotal study was to investigate the long-term effects of intracoronary infusion of allogeneic CDCs in pigs with AMI. The data obtained from the dose-optimization and the cell-retention studies were used to inform the dose selection and the timing of cell administration tested in the pivotal study. Infarct size (assessed by staining with triphenyltetrazolium chloride [TTC]), cardiomyocyte size, and fibrosis (assessed by histology), as well as indexes of LV remodeling and global and regional systolic function (assessed by echocardiography and left ventriculography [LVG]) were assessed at 30 days post-MI, as described in detail in the Online Supplement. A schematic depiction of the study protocol is presented in Figure 1E. A total of 18 farm pigs were used (CDC group n = 8, control group n = 10).
Statistical Analysis
Data are presented as mean ± standard deviation in the text/tables and as boxplots in the figures. The horizontal line within each box is the median, the length of each box is the interquartile range, and the whiskers extend to the highest and lowest values that are no greater than 1.5 times the interquartile range. The nonparametric Mann-Whitney U test was used for comparisons between 2 independent groups. The nonparametric Wilcoxon signed-rank test was used when comparing changes within groups. A P value <.05 was considered statistically significant.
Results
Mortality and Adverse Events
A total of 81 pigs were used in the studies; 55 completed the specified protocols. A detailed description of the mortality and the adverse events in each protocol is provided in the Online Supplement. Myocardial tissue was successfully isolated from all (10/10) male animals without any complications. Of the 45 pigs, 26 completed the dose-optimization study (5′ pre-rep group n = 13, 5′ post-rep group n = 13). Of the 8 pigs, 7 completed the cell retention study (5′ pre-rep group n = 3, 5′ post-rep group n = 4). Of the 18 animals, 12 successfully completed the pivotal study (CDC, n = 6, control n = 6). No adverse events associated with cell infusion (eg, arrhythmias, hypotension) were observed in any study.
Dose-Optimization Study
In the dose-optimization study, we assessed MVO after administration of different doses of allogeneic CDCs at either 5 minutes prior to reperfusion (5′ pre-rep group) or at 5 minutes post-reperfusion (5′ post-rep group). In both groups (5′ pre-rep group and 5′ post-rep group), we initially tested the intracoronary delivery of 10 million CDCs, based on previous work by Kanazawa et al, 6 demonstrating that this is the maximum safe dose that can be administered at 30 minutes post-reperfusion in a porcine ischemia–reperfusion model. The dose in each group was subsequently increased or decreased depending on the extent of MVO when compared to vehicle administration.
In the 5′ pre-rep group, administration of 10 million CDCs resulted in increased area of MVO (45.4% ± 11.5% of AR, n = 3) compared to controls (21.3% ± 5.2% of AR, n = 6; P = .02). The CDC dose was subsequently reduced to 5 million; this dose did not aggravate the extent of MVO (21.2% ± 4.8% of AR, n = 4) compared to vehicle-treated controls and thus was deemed to be the maximum safe dose that can be administered intracoronarily at 5 minutes prior to reperfusion. The AR was similar in all 3 groups (25.5% ± 5.5%, 23.1% ± 3.2%, and 26.2% ± 5.4% of LV in the control, 5 million, and 10 million groups, respectively; Figure 2).

Microvascular obstruction (MVO) after administration of different doses of allogeneic cardiosphere-derived cells at 5 minutes prior to reperfusion. A, Representative cardiac slices stained with gentian violet and thioflavin S under ultraviolet light 2 hours following cell delivery. B, Quantitative assessment of MVO. C, Quantitative assessment of area at risk. LV indicates left ventricle.
In the 5′ post-rep group, administration of 10 million CDCs resulted in a comparable area of MVO (15.5% ± 3.2% of AR, n = 4) compared to controls (15.6% ± 4.5% of AR, n = 5; P = .81). Increasing the cell dose to 15 million CDCs (n = 4) resulted in a significant increase in MVO area (39.6% ± 12.1% of AR, n = 4) compared to controls. Thus, the dose of 10 million CDCs was deemed to be the maximum safe dose that can be administered intracoronarily at 5 minutes postreperfusion. The AR was similar in all groups (19.5% ± 3.9%, 19.6% ± 2.5%, and 20.1% ± 6.3% of LV in the control, 10 million, and 15 million groups, respectively, Figure 3).
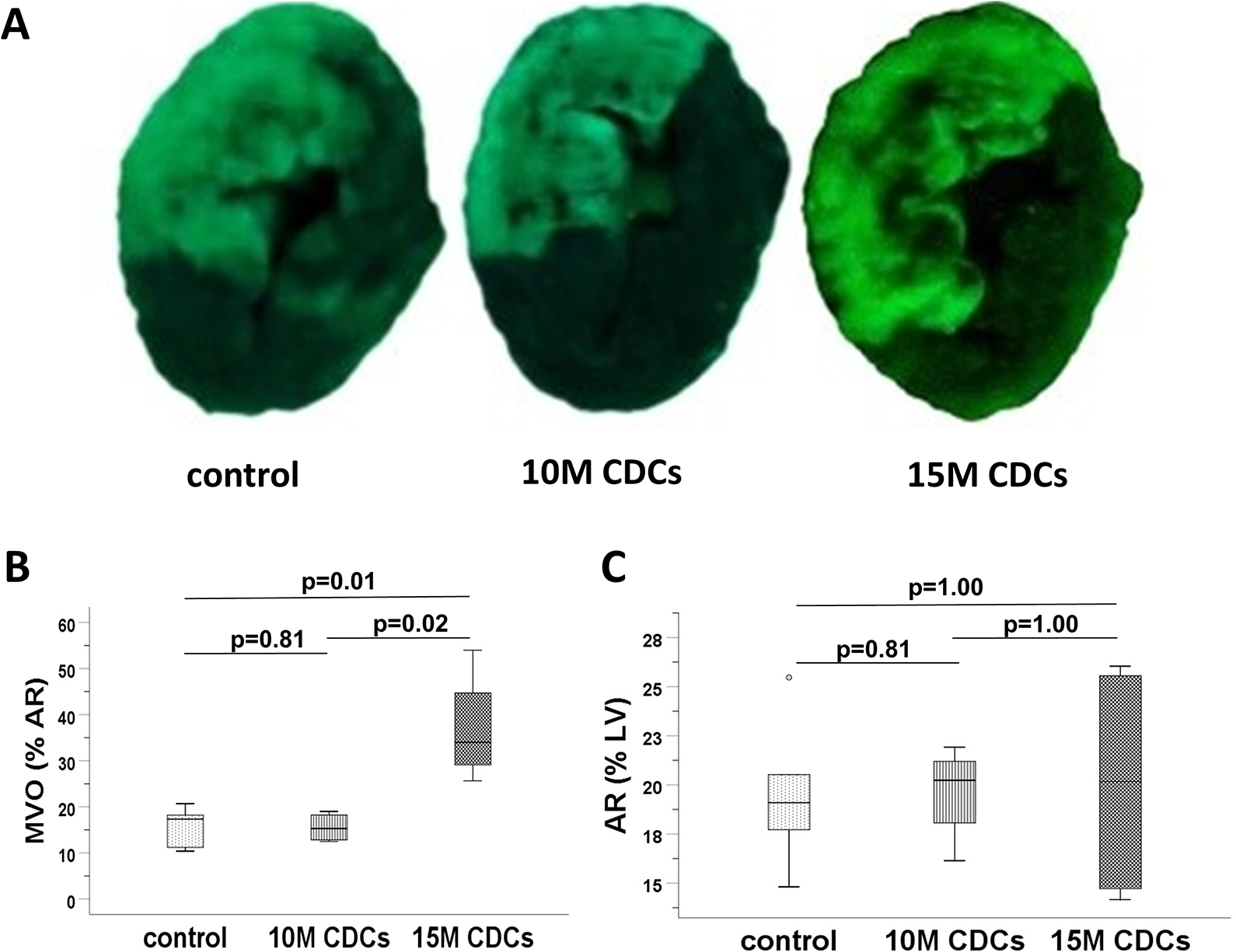
Microvascular obstruction (MVO) after administration of different doses of allogeneic cardiosphere-derived cells at 5 minutes postreperfusion. A, Representative cardiac slices stained with gentian violet and thioflavin S under ultraviolet light 2 hours following cell delivery. B, Quantitative assessment of MVO. C, Quantitative assessment of area at risk. LV indicates left ventricle.
Thus, the dose-optimization study demonstrated that the doses of 5 million and 10 million CDCs are the maximum safe doses that can be administered intracoronarily at 5 minutes prior to and at 5 minutes post-reperfusion, respectively, without aggravating MVO. A head-to-head comparison revealed that both the MVO area (P = .10) and the AR (P = .14) were comparable between these 2 groups.
Cell-Retention Study
In this study, we assessed acute cardiac cell retention after administration of the maximum safe CDC dose (as determined by the dose-optimization study) at 5 minutes pre-reperfusion and 5 minutes post-reperfusion. Acute cell retention of the maximum safe doses (5 million CDCs in the 5′ pre-rep group, n = 3; and 10 million CDCs in the 5′ post-rep group, n = 4) was quantified at 2 hours post-delivery by real-time PCR. No donor cells could be detected in one recipient animal in the 5′ post-rep group (the animal was included in the analysis). No significant statistical difference was detected in the absolute number of retained cells (383 753 ± 414 152 in the 5′ pre-rep group vs 141 551 ± 154 326 in the 5′ post-rep group; P = .48) and in the percentage of retained cells (7.7% ± 8.3% vs 1.4% ± 1.5%, P = .29; Figure 4). Given that acute cell retention was numerically higher in the 5′ pre-rep group, infusion of allogeneic CDCs at 5 minutes prior to reperfusion was deemed to be the optimal timing (in terms of acute retention of administered cells) for CDC administration.
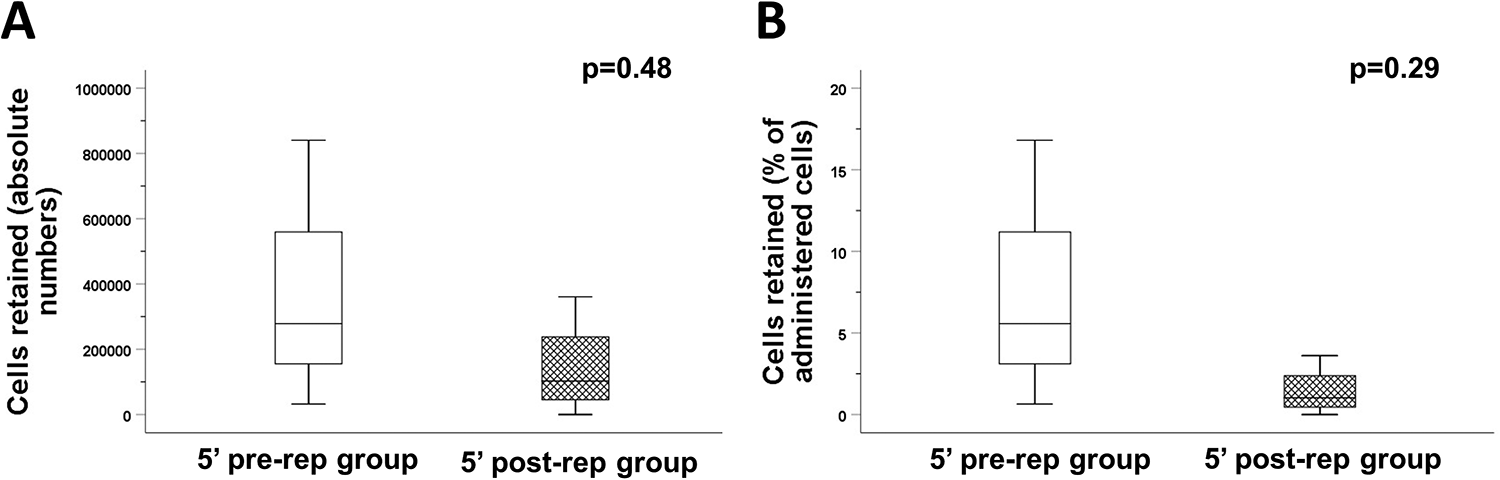
Acute cardiac retention of cardiosphere-derived cells. Quantitative assessment of the absolute cell number (A) and the percentage of cells (B) retained in the infarcted heart at 2 hours postdelivery.
Pivotal Study
In the pivotal study, we assessed the long-term efficacy of intracoronary infusion of allogeneic CDCs in a porcine model of AMI in a randomized, placebo-controlled manner. Based on the results of the dose-optimization and cell-retention studies, we chose to intracoronarily infuse 5 million allogeneic CDCs 5 minutes prior to reperfusion, as this combination of dose and timing of administration does not aggravate MVO and is associated with increased acute cardiac cell retention.
Assessment of infarct size, cardiomyocyte size, and interstitial fibrosis by histopathology
Morphometric analysis of TTC-stained myocardium at 30 days post-MI demonstrated a significant reduction in infarct size in CDC-treated animals (37.4% ± 9.9% of AR, n = 6) compared to controls (49.9% ± 7.4%, n = 6, P = .04). The AR was similar in both groups (Figure 5).

Infarct size and area at risk (AR) at 30 days following myocardial infarction. A, Representative cardiac slices stained with gentian violet and 2,3,5-triphenyltetrazolium chloride obtained from a placebo- and CDC-treated pigs. Quantitative assessment of infarct size (B) and AR (C). CDCs indicates cardiosphere-derived cells; LV, left ventricle.
Myocardial tissue from the infarct border zone and the remote myocardium was stained with hematoxylin and eosin to assess cardiomyocyte size, and with Masson trichrome to evaluate fibrosis. No difference in cardiomyocyte size was noted between CDC-treated animals (n = 6) and controls (n = 6), both in the border zone and in the remote myocardium (Figure 6A). Animals treated with CDCs had significantly less interstitial fibrosis in remote myocardium (1.4% ± 0.4%) compared to controls (3.4% ± 1.5%, P = .004, Figure 6B), indicating a CDC-induced attenuation of adverse remodeling. 16 No sign of increased lymphoplasmacytic infiltration (a hallmark of immune rejection) was observed in hematoxylin and eosin–stained myocardial tissue obtained from animals treated with allogeneic CDCs compared to controls.

Cardiomyocyte size and interstitial fibrosis at 30 days following myocardial infarction. A, Representative images of cardiac sections stained with hematoxylin and eosin and quantitative assessment of cardiomyocyte size in infarct border zone and remote myocardium in CDC- and placebo-treated animals. B, Representative images of cardiac sections stained with Masson trichrome and quantitative assessment of myocardial fibrosis in CDC- and placebo-treated animals; fibrotic tissue appears blue. CDCs indicates cardiosphere-derived cells.
Assessment of global LV systolic function by LVG
Left ventriculography was performed to quantify LV systolic function prior to ischemia and at 30 days post-MI. There was no statistically significant difference in left ventricular ejection fraction (LVEF) between controls (n = 6) and CDC-treated animals (n = 5), both at baseline and follow-up. Nevertheless, control animals exhibited a significant reduction in LVEF at 30 days post-MI (35.7% ± 5.5%) compared to baseline (41.5% ± 7.2%, P = .046), while CDC-treated animals had preserved LVEF at 30 days post-MI (41% ± 4.5%) compared to baseline (43% ± 8.2%, P = .89; Figure 7).

Global left ventricular systolic function assessed by left ventriculography. Quantitative assessment of left ventricular ejection fraction at baseline and at 30 days post-myocardial infarction in placebo- and CDC-treated pigs. CDCs indicates cardiosphere-derived cells.
Assessment of LV dimensions, global and regional systolic function by echocardiography
Left ventricular dimensions and volumes, LVEF, and systolic thickening of the infarcted anteroseptal LV wall were measured by echocardiography to evaluate LV adverse remodeling, and global and regional systolic function, respectively (control group n = 6, CDC group n = 6, Figure 8A). Both groups exhibited a significant increase in LV end-diastolic diameter and LV end-diastolic volume at 30 days post-MI compared to baseline (Figure 8B and C). Control animals exhibited a significant increase in LV end-systolic diameter (LVESD; 32.6 ± 3.8 mm) and LV end-systolic volume (LVESV; 43.3 ± 12.4 mL) at 30 days follow-up compared to baseline (22.3 ± 2 mm, P = .03; and 17.8 ± 4.4 mL, P = .03, respectively), whereas LVESD and LVESV were preserved in CDC-treated animals at 30 days post-MI compared to baseline (29.8 ± 6.6 mm from 22.3 ± 2 mm, P = .07, and 36.8 ± 19.2 mL from 17.3 ± 3.8 mL, P = .12, respectively; Figure 8D-E). A significant decrease in LVEF was observed in control animals at 30 days post-MI compared to baseline (50.8% ± 7.9% vs 62.5% ± 6.4%, P = .046); in contrast, LVEF was preserved in the CDC group (58% ± 17% at 30 days compared to 67.8% ± 7% at baseline, P = .46, Figure 8F). Between-group comparisons of cardiac dimensions and LVEF at 30 days post-MI did not reveal any statistically significant differences (Online Table 1). However, end-systolic dimensions (LVESV and LVESD) were numerically lower and LVEF was numerically higher in the CDC group. These trends are in accordance with the statistically significant differences observed in the within-group comparisons of the aforementioned parameters. Regarding regional LV systolic function at 30 days post-MI, animals treated with allogeneic CDCs showed increased systolic thickening of the anteroseptal LV wall (24.2% ± 4.3%) compared to controls (13.2% ± 4.6%, P = .004, Figure 8G).
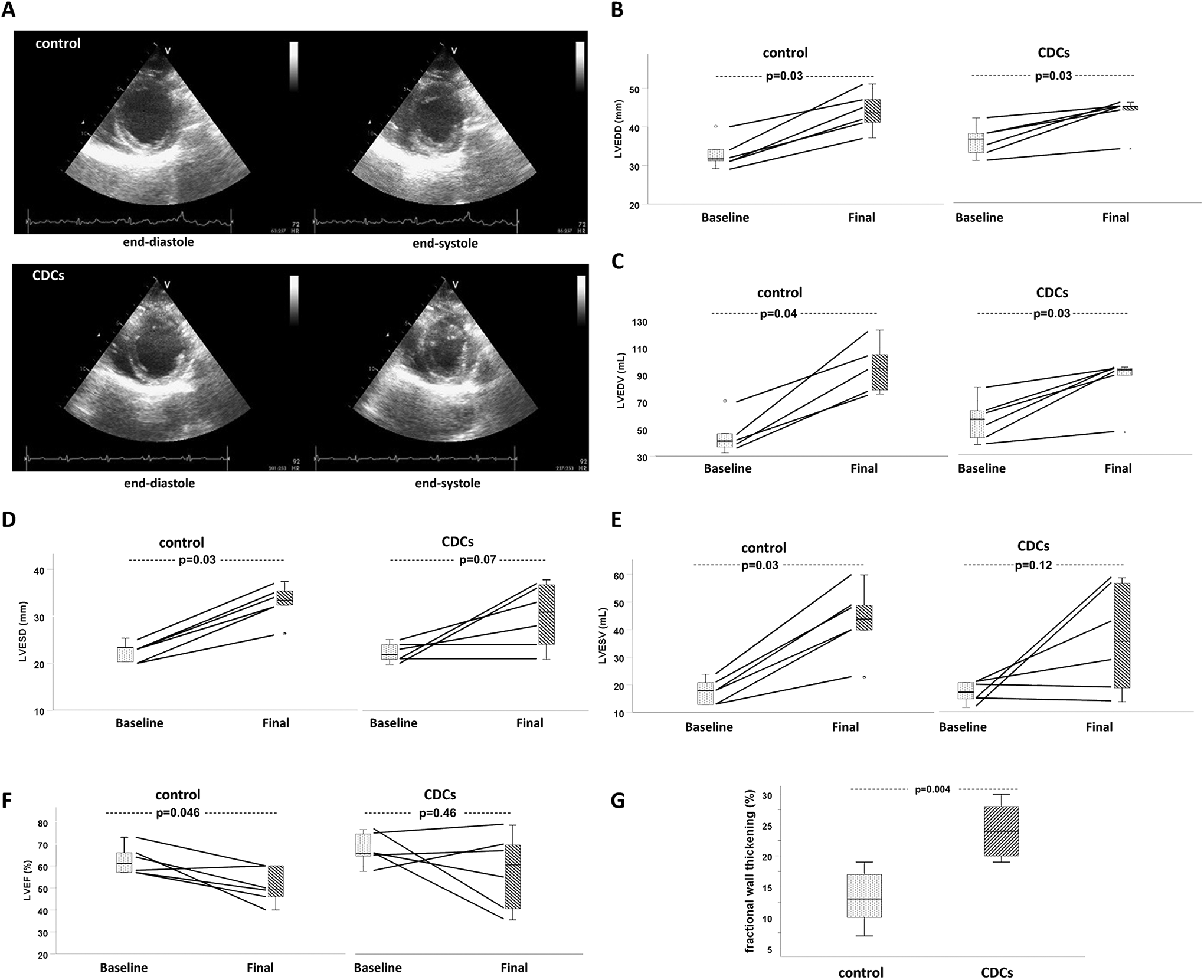
Assessment of left ventricular (LV) dimensions, and global and regional systolic function by echocardiography. (A) Representative short axis, end-diastolic, and end-systolic echocardiographic LV images at 30 days post-MI, obtained from a CDC- and placebo-treated pig. Quantitative assessment of LV end-diastolic diameter (B), LV end-diastolic volume (C), LV end-systolic diameter (D), LV end-systolic volume (E), and LV ejection fraction (F) at baseline and at 30 days post-myocardial infarction in placebo- and CDC-treated pigs. (G) Quantitative assessment of systolic thickening of the infarcted anteroseptal LV wall at 30 days post-MI. * P < .05. CDCs indicates cardiosphere-derived cells.
Discussion
In the present study, we tested—in a systematic manner—the feasibility, safety, and efficacy of intracoronary delivery of allogeneic CDCs in a porcine model of ischemia–reperfusion. We demonstrate that, following careful dose optimization, intracoronary infusion of 5 million allogeneic CDCs prior to reperfusion in AMI is safe, results in robust acute cardiac cell retention and confers long-term structural and functional benefits, manifested as reduced infarct size, improved regional systolic function, preservation of global systolic function, and attenuation of LV remodeling.
The novelty of our study lies in the timing of cell administration, as this is the first cell therapy study to our knowledge in which cells were administered prior to reperfusion. We hypothesized that development of no-reflow (which rapidly expands during the first hour of reperfusion) would undermine the efficacy of cell therapy, as it would prevent cells from reaching a large portion of the infarcted myocardium (due to impairment of the microvasculature). We attempted to circumvent this issue by administering cells prior to development of significant no-reflow. We thus investigated cell administration at 2 discrete time points: at 5 minutes prior to reperfusion (when microvasculature is still relatively intact, since the vast majority of no-reflow occurs due to reperfusion injury 10,12 and at 5 minutes after reperfusion (prior to the development of significant no-reflow 12 ). Since CDCs are larger (mean diameter ≈21 μm) 17 than the diameter of the coronary capillaries, 18 we carefully assessed, in a dose-ranging study, the number of cells that can be safely infused in the setting of an AMI without resulting in significant microembolization and aggravation of the no-reflow phenomenon. In the 5′ pre-rep group, CDCs were intracoronarily infused over 30 seconds. We deliberately chose to deliver CDCs in such a rapid manner in order to avoid significant delays in prompt myocardial reperfusion, which has a central role in the clinical management of AMI as it has been proven to salvage ischemic myocardium. Cell clumping due to rapid cell infusion in this setting could account for the smaller dose (5 million CDCs) that we were able to safely deliver compared to the 5′ post-rep group and previous studies. 6,7 In the 5′ post-rep group, CDCs were administered using a stop flow technique over 10 minutes, beginning at 5 minutes after reperfusion, when peak reactive hyperemia has subsided, to minimize washing of cells. 11,19,20 Ten million CDCs could be safely delivered in this setting, which is similar to the findings by Kanazawa et al 6 who administered allogeneic CDCs at 30 minutes postreperfusion.
Cell retention is known to be poor following cardiac delivery of exogenous cells. 21,22 Given that higher rates of acute cell retention have been associated with improved long-term benefits after cell therapy, 23,24 development of more effective cell delivery strategies is of great importance. Here, we report a 5-fold increase in retention rate when cells are administered prior to reperfusion; this resulted in a 2.7-fold increase in the absolute number of retained cells in the 5′ pre-rep group compared to the 5′ post-rep group, despite the lower cell dose that was administered in the 5′ post-rep group. The reasons for the increased acute cardiac retention observed when cells are administered prior to reperfusion, compared to post-reperfusion, are unclear; it could be postulated that delivery of cells to the ischemic myocardium prior to development of reperfusion injury may facilitate cell survival and acute cardiac retention. In addition, prolonged absence of coronary blood flow at the time of cell delivery may have also contributed to the increased acute retention observed when cells were administered prior to reperfusion (coronary flow resumed after ∼4.5 minutes following cell administration in the 5′ pre-rep group compared to 2 minutes in the 5′ post-rep group). Lack of coronary flow during cell delivery has been associated with increased acute cell retention in rodents, 22 as it presumably prevents cell washout and promotes adherence to the recipient myocardium.
Based on the findings of the dose-ranging and cell-retention studies, 5 million CDCs were administered intracoronarily 5 minutes prior to reperfusion in the pivotal study. In the pivotal study, allogeneic CDCs exhibited a favorable safety profile; infusion was well- tolerated, without cell-related arrhythmias, and no signs of significant immune rejection were detected by histopathology, confirming previous reports. 6,7,21 Of note, administration of allogeneic CDCs conferred long-term structural and functional benefits; at 30 days post-MI, CDC-treated animals exhibited smaller infarcts, improved regional systolic function, preserved global systolic function, and attenuation of LV remodeling compared to vehicle-treated controls. The CDC-induced benefits presented here are comparable to those reported by Kanazawa et al, 7 who assessed intracoronary infusion of allogeneic CDCs at 30 minutes post-reperfusion. While we show that administration of cells prior to reperfusion may result in greater acute cardiac retention, a head-to-head comparison assessing the long-term effects of the 2 therapeutic approaches (ie, administration of cells at 5 minutes pre-reperfusion vs 30 minutes post-reperfusion) is merited.
It should be acknowledged that administration of cell therapy prior to reperfusion would inevitably delay reperfusion of the ischemic myocardium; using the approach tested here, this cell therapy-associated delay in reperfusion would be 5 minutes. Although it is established that prompt reperfusion in AMI improves prognosis, time delays of ∼5 minutes in reperfusion do not appear particularly relevant from a therapeutic standpoint; in stable patients with ST-elevation AMI, a 5-minute treatment delay is estimated to result in ∼0.17 additional deaths per 100 patients treated with percutaneous coronary intervention. 25 The long-term structural and functional benefits observed in this study (if reproduced at the clinical level) would certainly justify this cell therapy-associated delay in reperfusion. For example, a ∼25% relative decrease in scar size alone (as observed in our study) would translate into a ∼1% absolute decrease in 6-month mortality. 26,27
Our study does not provide insight into the potential mechanisms accounting for the observed functional and benefits. However, previous mechanistic studies have demonstrated that CDCs are potent secretors of paracrine factors and exosomes 28,29 that modulate the inflammatory response, decrease apoptosis and promote angiogenesis, myocyte proliferation, and recruitment of endogenous progenitor cells. 30 -35 Thus, the observed benefits should be attributed to indirect mechanisms of action of the administered CDCs.
Even though several porcine studies, from independent laboratories, have shown allogeneic heart-derived cells to be effective in AMI/convalescent MI, 6,7,36 -40 clinical translation has been problematic. Although allogeneic heart-derived cells were well tolerated in both the ALLSTAR trial (which enrolled patients with 1- to 12-month-old infarcts) 41 and the CAREMI trial (which enrolled patients with 1-week-old infarcts), 42 efficacy of allogeneic cell therapy was not demonstrated. Specifically, even though signs of CDC-induced improvements in LV volumes and brain natriuretic peptide (BNP) were observed in the ALLSTAR trial, in both the ALLSTAR and the CAREMI trials, allogeneic heart-derived cells failed to produce significant reductions in scar size compared to placebo treatment. The reasons for the inconsistent results between preclinical and clinical studies remain unclear. However, administration of allogeneic cells early in the MI process could maximize the potential for cell therapy-induced benefits; given that most ischemic cardiomyocytes die in the first hours post-MI, 43 delivery of allogeneic cells as an adjunctive therapy to reperfusion (as reported here) would potentially allow exogenous cells to exert a cardioprotective role.
Our study has an important limitation: The choice of the optimal timing of cell administration was based on a nonsignificant result in the “cell-retention” study. Specifically, although administration of 5 million CDCs at 5 minutes pre-reperfusion resulted in a 2.7-fold absolute number of retained cells compared to delivery of 10 million cells at 5 minutes post-reperfusion (383 753 ± 414 152 cells in the 5′ pre-rep group vs 141 551 ± 154 326 cells in the 5′ post-rep group), the between-groups comparison did not yield statistically significant results, most likely due to the large measured standard deviations. This high variance can be rationalized as follows: although real-time PCR after sex-mismatch cell transplantation has many advantages for quantification of cell retention (it provides absolute quantification, no genetic modification of cells is required, and limitations of cell labeling techniques are avoided), it is prone to sampling error. As described in Online Supplement, in our study, real-time PCR was performed in ∼0.5% of the AR in each pig. Therefore, sampling error is most likely responsible for the significant variance in the measured cell retention. Considering the observed variance, detecting a statistically significant difference in cell retention between the 2 groups would require a substantially larger sample size. Neither testing more animals nor testing additional samples from each animal could be performed due to financial and logistical constraints. Thus, we followed a qualitative approach and chose to intracoronarily infuse 5 million CDCs 5 minutes prior to reperfusion in the pivotal study, as this cell delivery method was associated with an almost 3-fold (albeit statistically non-significant) increase in cardiac cell retention.
Conclusion
We demonstrate that dose-optimized intracoronary infusion of allogeneic CDCs prior to reperfusion is feasible and safe, results in robust acute cell retention and produces a reduction in infarct size, improvements in global and regional LV function, and attenuation of LV remodeling. The therapeutic approach employed here is conceptually attractive, as (1) cells are infused into a patent microcirculation (prior to development of significant no-reflow), allowing for homogeneous delivery throughout the AR; and (2) allogeneic cells as an adjunctive therapy to reperfusion allow cells to exert a cardioprotective role (as ischemic cardiomyocytes are still salvageable).
Supplemental Material
Supplemental Material, CDC_no_reflow_paper_Online_Supplement_revision_clean - Intracoronary Administration of Allogeneic Cardiosphere-Derived Cells Immediately Prior to Reperfusion in Pigs With Acute Myocardial Infarction Reduces Infarct Size and Attenuates Adverse Cardiac Remodeling
Supplemental Material, CDC_no_reflow_paper_Online_Supplement_revision_clean for Intracoronary Administration of Allogeneic Cardiosphere-Derived Cells Immediately Prior to Reperfusion in Pigs With Acute Myocardial Infarction Reduces Infarct Size and Attenuates Adverse Cardiac Remodeling by Vasileios Sousonis, Titika Sfakianaki, Argirios Ntalianis, Ioannis Nanas, Christos Kontogiannis, Dionysios Aravantinos, Chris Kapelios, Lampros Katsaros, Maria Nana, Dimitrios Sampaziotis, Despina Sanoudou, Apostolos Papalois and Konstantinos Malliaras in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Vasileios Sousonis and Titika Sfakianaki contributed equally to this work. Vasileios Sousonis, Titika Sfakianaki, Argirios Ntalianis, Ioannis Nanas, Christos Kontogiannis, Dionysios Aravantinos, Chris Kapelios, Lampros Katsaros, Maria Nana, Dimitrios Sampaziotis, Despina Sanoudou, Apostolos Papalois and Konstantinos Malliaras performed the study and collected and/or interpreted data. Vasileios Sousonis, Titika Sfakianaki and Konstantinos Malliaras drafted the manuscript with input from all authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Scholarship—Grant by the Experimental Research Center ELPEN Pharmaceuticals (E.R.C.E.). Despina Sanoudou was supported by CURE-PLaN, a grant from the Leducq Foundation for Cardiovascular Research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
