Abstract
Background:
Cardiosphere-derived cells (CDCs) have yielded promising efficacy signals in early-phase clinical trials of ischemic and nonischemic cardiomyopathy. The potential efficacy of CDCs in acute myocarditis, an inflammatory cardiomyopathy without effective therapy, remains unexplored. Given that CDCs produce regenerative, cardioprotective, anti-inflammatory, and anti-fibrotic effects (all of which could be beneficial in acute myocarditis), we investigated the efficacy of intracoronary delivery of CDCs in a rat model of experimental autoimmune myocarditis.
Methods:
Lewis rats underwent induction of experimental autoimmune myocarditis by subcutaneous footpad injection of purified porcine cardiac myosin supplemented with Mycobacterium tuberculosis on days 1 and 7. On day 10, rats were randomly assigned to receive global intracoronary delivery of 500 000 CDCs or vehicle. Global intracoronary delivery was performed by injection of cells or vehicle into the left ventricular (LV) cavity during transient occlusion of the aortic root. Rats were euthanized 18 days after infusion. Cardiac volumes and systolic function were assessed by serial echocardiography, performed on days 1, 10, and 28. Myocardial inflammation, T-cell infiltration, and cardiac fibrosis were evaluated by histology.
Results:
Experimental autoimmune myocarditis was successfully induced in 14/14 rats that completed follow-up. Left ventricular ejection fraction (LVEF) and volumes were comparable on days 1 and 10 between groups. CDC infusion resulted in increased LVEF (81.5% ± 3% vs 65.4% ± 8%, P < .001) and decreased LV end-systolic volume (43 ± 15 vs 100 ± 24 μL, P < .001) compared to placebo administration at 18 days post-infusion. Cardiosphere-derived cell infusion decreased myocardial inflammation (7.4% ± 7% vs 20.7% ± 4% of myocardium, P = .007), cardiac fibrosis (16.6% ± 13% vs 38.1% ± 3% of myocardium, P = .008), and myocardial T-cell infiltration (30.4 ± 29 vs 125.8 ± 49 cells per field, P = .005) at 18 days post-infusion compared to placebo administration.
Conclusion:
Intracoronary delivery of CDCs attenuates myocardial inflammation, T-cell infiltration, and fibrosis while preventing myocarditis-induced systolic dysfunction and adverse remodeling in rats with experimental autoimmune myocarditis.
Keywords
Introduction
Myocarditis is a potentially lethal cardiomyopathy characterized by myocardial inflammation and cardiomyocyte injury. Common causes include myocardial infection by viruses or other pathogens, hypersensitivity reactions, and autoimmune diseases. 1 Myocarditis most commonly affects the young and has a highly variable clinical course, ranging from asymptomatic disease to fulminant heart failure. 1,2 Since current treatment options for acute myocarditis are generally limited to supportive care for heart failure and arrhythmias, the search for novel effective therapeutic strategies is certainly warranted.
Over the past decade, cardiosphere-derived cells (CDCs) 3 have emerged as a potential therapeutic option for a wide spectrum of cardiac diseases; early-phase clinical studies of CDCs in convalescent myocardial infarction, 4,5 dilated cardiomyopathy, 6 dystrophic cardiomyopathy, and hypoplastic left heart syndrome 7,8 have demonstrated safety and have yielded intriguing hints of efficacy. Mechanistic studies have shown that CDCs exert regenerative, 9 –13 cardioprotective, 14,15 antifibrotic, 16 and immunomodulatory/immunosuppressive 15,17,18 effects, all of which could—from a theoretical standpoint—potentially be beneficial in acute myocarditis. However, the effect of cell therapy with CDCs in acute myocarditis is currently unknown. Thus, we performed—for the first time to our knowledge—a preclinical study to evaluate the efficacy of intracoronary delivery of CDCs in experimental autoimmune myocarditis.
We employed a rat model of experimental autoimmune myocarditis, induced by rat immunization with porcine myosin. 19,20 The natural history of this myocarditis model comprises 3 distinct phases: an initial antigen priming phase, a subsequent autoimmune phase characterized by myocardial lymphomononuclear infiltration and giant cell formation, and a final healing phase, characterized by reparative fibrosis. 20,21 Experimental autoimmune myocarditis in rats closely mimics human giant cell myocarditis, 19,20 a rare and frequently fulminant form of myocarditis, characterized by myocardial formation of multinucleated giant cells, in the absence of granulomas. 22 Human giant cell myocarditis generally results in rapidly progressive left ventricular (LV) failure and is characterized by a dismal prognosis; the rate of heart transplantation or death approaches 70% at 1 year. 22 While myocarditis in humans encompasses a diverse group of cardiac inflammatory conditions with a broad spectrum of clinical manifestations, 1,2 we chose to employ a model of giant cell myocarditis to test the efficacy of cell therapy with CDCs in a “worst-case scenario.”
Methods and Materials
Myocarditis Model
Lewis rats underwent induction of autoimmune myocarditis by a 2-stage footpad injection protocol. 23 Specifically, purified porcine cardiac myosin (1 mg administered in 105 μL; Sigma-Aldrich, USA) supplemented with Mycobacterium tuberculosis (2.2 mg of complete Freund adjuvant, administered in 200 μL; Sigma-Aldrich, USA) was subcutaneously injected into the rat footpads on days 1 and 7. The animal protocol was reviewed and approved by the Directorate of Agricultural and Veterinary Policy of the Region of Attica.
Cell Culture
Rat CDCs were grown from hearts explanted from healthy 5- to 6-week-old male Lewis rats. CDCs were cultured as described previously.
24
In brief, following explantation of the heart, the myocardium was dissected in ∼1-mm3 pieces, which underwent partial digestion with 0.05% trypsin. The partially digested myocardial fragments (termed cardiac explants) were subsequently cultured on fibronectin-coated dishes in cardiac explant media (Iscove Modified Dulbecco Medium supplemented with 20% fetal bovine serum 20%, 100 U/mL penicillin/ streptomycin, and 0.1 mmol/l 2-mercaptoethanol). After 3 to 5 days, stromal-like “cardiac outgrowth” cells started emerging from the cardiac explants. Once they became confluent, cardiac outgrowth cells were harvested and plated on poly-
Cardiosphere-Derived Cell/Vehicle Intracoronary Infusion
Ten days after induction of autoimmune myocarditis, animals were randomly assigned to receive global intracoronary delivery of CDCs or vehicle solution. Following induction of anesthesia with 4% isoflurane, animals were intubated and mechanically ventilated. Anesthesia was subsequently maintained with 2% isoflurane. A left lateral thoracotomy followed by pericardiotomy was performed to expose the heart. Global intracoronary infusion was performed as follows: The aortic root was transiently occluded for 10 seconds while cells (500 000 CDCs suspended in 200 μL of PBS)/vehicle solution were infused through a 29 g needle into the LV cavity. 25,26 We administered 500 000 CDCs based on previous dose-ranging studies demonstrating that this is the optimal cell dose for global intracoronary delivery of CDCs in rats (as higher doses result in significant microvascular obstruction and myocardial injury). 25
Echocardiography
Global LV function and LV volumes were evaluated by echocardiography. Serial echocardiographic imaging (using a Vivid Q machine [GE Healthcare, UK]) was performed at the following time points: at baseline (just prior to induction of autoimmune myocarditis), at 10 days (prior to cell/ placebo infusion), and at 28 days. Following induction of anesthesia with 4% isoflurane, animals were intubated, mechanically ventilated, and anesthesia was subsequently maintained with 2% isoflurane. Left ventricular ejection fraction (LVEF), LV end-systolic (LVESV), and end-diastolic (LVEDV) volumes were measured from the long-axis view using the EchoPAC* software (GE Healthcare, UK).
Histology
Rats were killed 28 days after myocarditis induction (18 days after CDC/vehicle infusion). Hearts were arrested with KCl solution, explanted, fixed with 10% formalin, and sectioned into 5-μm sections. Myocardial fibrosis was visualized by Masson trichrome staining and performed in 4 to 6 myocardial sections per heart (sections collected at 400-μm intervals). Myocardial inflammation (ie lymphomononuclear infiltration) was evaluated by a blinded cardiac pathologist, following hematoxylin and eosin staining of 4 to 6 myocardial sections per heart (sections collected at 400-μm intervals). Both myocardial inflammation and fibrosis were evaluated by whole-field microscopy and are expressed as %/cardiac section. In addition, immunostaining against CD3 was performed in 4 to 6 sections per heart (sections collected at 400-μm intervals) to evaluate myocardial infiltration by T cells. Myocardial T-cell infiltration was assessed in 5 random high-power (×40) fields in each section. Manual cell counting was performed in each field, and T-cell infiltration is expressed as absolute number of T cells/field.
Statistical Analysis
Results are reported in the text as means ± standard deviation (SD). Comparisons between groups were performed using the independent samples t test. Differences were considered statistically significant when P < .05.
Results
Induction of Autoimmune Myocarditis, Adverse Events, and Mortality
Twenty-six rats were used in the study. One rat died after induction of autoimmune myocarditis but prior to surgery. Three rats died during lateral thoracotomy but prior to randomization. Nine rats were randomized to receive CDCs, whereas 13 rats were randomized to undergo vehicle infusion. Three rats infused with CDCs and 5 rats infused with vehicle died during the surgical procedure (due to pneumothorax or hemorrhagic shock following traumatic aortic injury). All rats that survived surgery (6 CDC-treated rats and 8 control rats) successfully completed the 18-day follow-up and were included in the analysis. Experimental autoimmune myocarditis was successfully induced in all 14 rats that completed follow-up (as deemed by histological evaluation of the explanted hearts). A schematic depiction of the experimental protocol is presented in Figure 1.
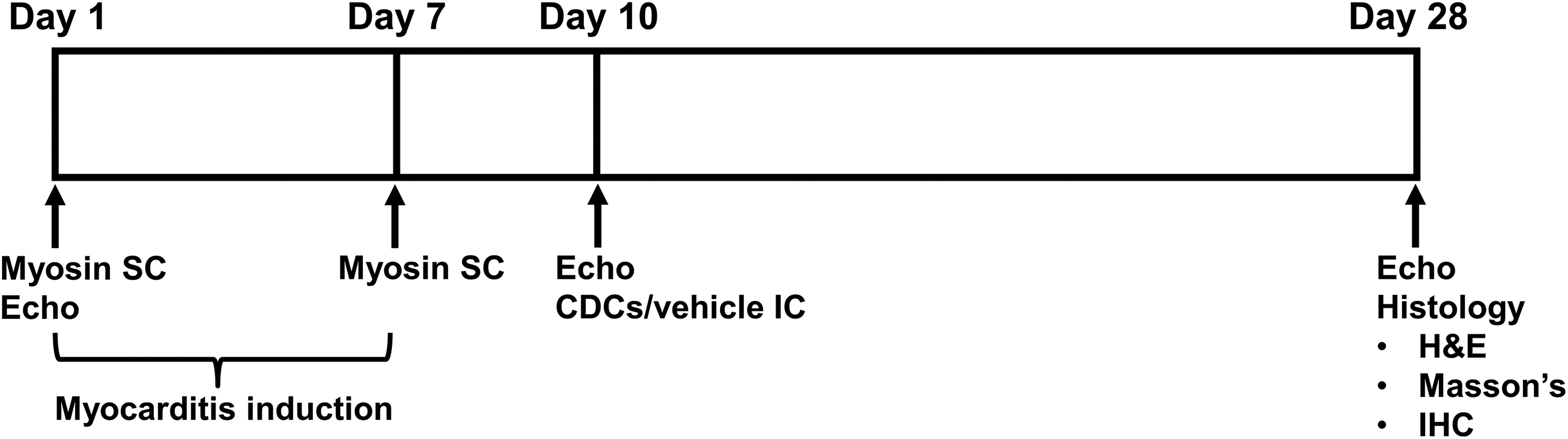
Schematic depiction of study outline. H&E indicates hematoxylin and eosin; IC, intracoronarily; IHC, immunohistochemistry; SC, subcutaneously.
CDCs Improve Myocardial Function and Structure in Rats With Autoimmune Myocarditis
To investigate whether CDC infusion leads to functional and structural improvement in rats with autoimmune myocarditis, global LV systolic function and LV volumes were measured by serial echocardiography. LVEF and LV volumes were comparable on day 1 (LVEF: 81.8% ± 3% vs 82.9% ± 4%, P = .55; LVEDV: 200 ± 66 vs 216 ± 69 μL, P = .67; LVESV: 40 ± 18 vs 39 ± 17 μL, P = .90 in the CDC and control groups, respectively) and on day 10 (LVEF: 78.3% ± 3% vs 78.4% ± 4%, P = .96; LVEDV: 213 ± 79 vs 220 ± 49 μL, P = .85; LVESV: 47 ± 21 vs 45 ± 14 μL, P = .86 in the CDC and control groups, respectively) between the 2 groups. CDC therapy prevented myocarditis-induced LV systolic dysfunction and LV dilation; LVEF was significantly increased (81.5 ± 3 vs 65.4 ± 8, P < .001) and LVESV was significantly decreased (43 ± 15 vs 100 ± 24 μL, P < .001) in CDC-treated compared to placebo-treated animals at 18 days post-infusion (Figure 2). Furthermore, a trend toward decreased LVEDV (225 ± 63 vs 288 ± 61 μL, P = .085) in CDC-treated compared to placebo-treated animals at 18 days post-treatment was observed (Figure 2).
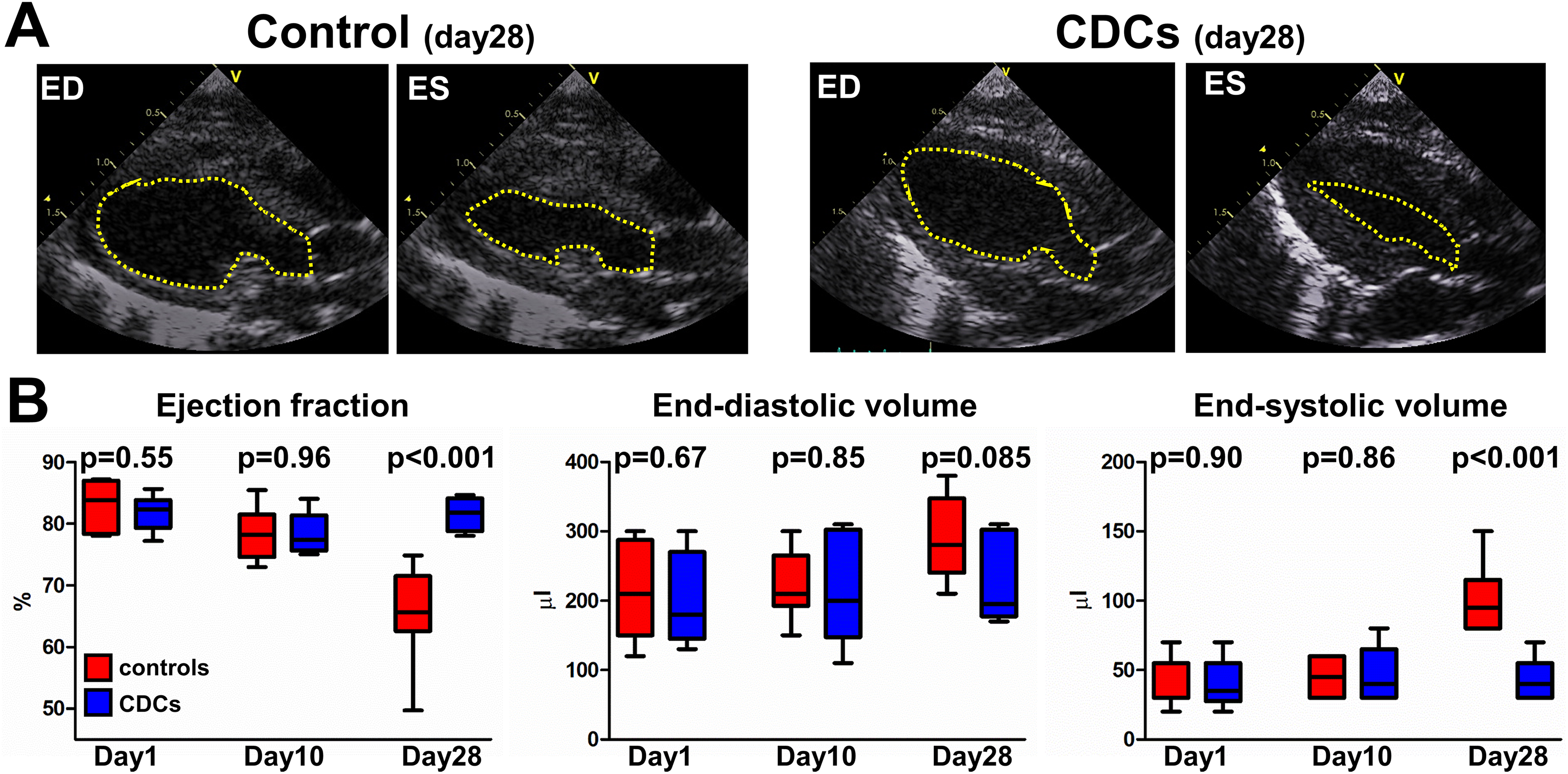
Cardiosphere-derived cells (CDCs) preserve systolic function and attenuate adverse remodeling in myocarditis. A, Representative echocardiographic images at end-diastole (ED) and end-systole (ES) from a control and a CDC-treated rat with myocarditis, 18 days post cell/vehicle infusion. Note the significantly increased end-systolic volume of the control rat. A scale (in cm) is provided at the side of each sector. B, Quantitative analysis of ejection fraction (left), end-diastolic volume (middle), and end-systolic volume (right). Horizontal line within each box is the median, length of each box is the interquartile range, ends of the whiskers are the 10th and 90th percentile. n = 6 to 8/group. Comparisons between groups were performed using the independent samples t test.
CDCs Attenuate Myocardial Inflammation in Rats With Autoimmune Myocarditis
Whole-field microscopy of cardiac sections stained with hematoxylin and eosin revealed that CDC administration resulted in significant attenuation of myocardial inflammation compared to control animals at 18 days postinfusion (7.4% ± 7% vs 20.7% ± 4% of the myocardium, P = .007; Figure 3). Given that T-cells orchestrate the development of experimental autoimmune myocarditis, 27 we performed immunohistochemistry for CD3 to specifically quantify myocardial T cell infiltration. We found that cell therapy with CDCs resulted in significant attenuation of myocardial T-cell infiltration compared to control animals at 18 days post-infusion (30.4 ± 29 vs 125.8 ± 49 T cells per high-power field, P = .005; Figure 4).

Cardiosphere-derived cells (CDCs) attenuate cardiac lymphomononuclear infiltration in myocarditis. Representative Images of hematoxylin and eosin-stained cardiac sections from a control (A) and a CDC-treated (B) rat with myocarditis, 18 days post cell/vehicle Infusion. (C) Quantitative analysis of lymphomononuclear infiltration. Horizontal line within each box is the median, length of each box is the interquartile range, ends of the whiskers are the 10th and 90th percentile. n = 4 to 6/group. Comparisons between groups were performed using the independent samples t test. Scale bars 2 mm.

Cardiosphere-derived cells (CDCs) attenuate cardiac T-cell infiltration in myocarditis. Representative high-power field images of cardiac sections from a control (A) and a CDC-treated (B) rat with myocarditis, 18 days post cell/vehicle infusion. Sections underwent immunohistochemistry against CD3 (a marker of T cells). (C) Quantitative analysis of T-cell infiltration. Horizontal line within each box is the median, length of each box is the interquartile range, ends of the whiskers are the 10th and 90th percentile. n = 4 to 6/group. Comparisons between groups were performed using the independent samples t test. Scale bars: 20 μm.
Cardiosphere-Derived Cells Attenuate Myocardial Fibrosis in Rats With Autoimmune Myocarditis
Experimental autoimmune myocarditis results in extensive cardiomyocyte injury and replacement fibrosis. 19,20,27 Importantly, from a clinical standpoint, detection of cardiac fibrosis by magnetic resonance imaging in patients with histologically-proven myocarditis is associated with a dramatic increase in cardiac mortality. 28 We, therefore, quantified myocardial fibrosis using whole-field microscopy of cardiac sections stained with Masson Trichrome. We found that CDC therapy resulted in significant reduction in myocardial fibrosis compared to control animals at 18 days postinfusion (16.6% ± 13% vs 38.1% ± 3% of the myocardium, P = .008; Figure 5).

Cardiosphere-derived cells (CDCs) decrease cardiac fibrosis in myocarditis. Representative images of cardiac sections stained with Masson Trichrome in a control (A) and a CDC-treated (B) rat with myocarditis, 18 days post cell/vehicle infusion. Viable myocardium appears red, while fibrotic tissue appears blue. (C) Quantitative analysis of myocardial fibrosis. Horizontal line within each box is the median, length of each box is the interquartile range, ends of the whiskers are the 10th and 90th percentile. n = 4 to 6/group. Comparisons between groups were performed using the independent samples t test. Scale bars 2 mm.
Discussion
We report that global intracoronary delivery of CDCs attenuates myocardial inflammation, T-cell infiltration, and fibrosis while preserving cardiac function and structure in rats with experimental autoimmune myocarditis. Two aspects of our study are notable. First, this is the first study to our knowledge to investigate a potential role of CDCs in the treatment of acute myocarditis. CDCs are attractive for cardiac repair and regeneration, as they have yielded promising efficacy signals in early-phase clinical trials of convalescent myocardial infarction, 4,5 dilated cardiomyopathy, 6 dystrophic cardiomyopathy, and hypoplastic left heart syndrome. 7,8 Importantly, preclinical studies performing head-to-head comparisons between different cell types demonstrate that CDCs appear to be more 29,30 (or at least equally) 11 potent compared to other clinically-applicable cell types. Second, this is the first study to our knowledge employing intracoronary delivery of progenitor cells in myocarditis; in previous preclinical studies of cell therapy in myocarditis, cells were administered via the intravenous 21,31,32 or the intramyocardial 33,34 route. We chose to administer CDCs via global intracoronary infusion because this delivery method achieves broad and homogeneous myocardial coverage 26,35 (which appears particularly important for the treatment of diffuse myocardial diseases such as myocarditis) and is relatively simple in execution, thus potentially enabling widespread clinical application. However, given that no data are available comparing different delivery methods in the setting of myocarditis, it remains unknown whether intracoronary delivery confers increased benefits compared to intravenous or intramyocardial delivery.
Myocarditis in humans can produce a wide range of clinical outcomes, ranging from asymptomatic disease to fulminant heart failure and death. 1,2 In this study, we employed a model of giant cell myocarditis to test the efficacy of CDCs in a fulminant and devastating form of myocarditis, which typically results in rapidly progressive heart failure. Although in clinical practice viral myocarditis is more common than giant cell myocarditis, it has been shown that viral myocardial infections can trigger an autoimmune reaction to cardiac auto-antigens, resulting in myocardial inflammation without viral persistence. 2,36 Thus, given that cardiac autoimmunity appears to play a pivotal role in the pathophysiology and progression of both viral and giant cell myocarditis, it is logical to assume that the CDC-induced salutary effects observed in the present study could perhaps be extrapolated to the setting of viral myocarditis.
Careful consideration was given to the selection of the dose of intracoronarily-infused CDCs and the timing of administration employed in our study. The dose of 500 000 CDCs was selected based on previous preclinical studies, demonstrating that this is the maximum dose that can be intracoronarily delivered without causing microvascular obstruction and cardiac injury in rats. 25 This dose falls within the lower end of the spectrum of cell dosages administered in other studies, investigating the effects of cell therapy in rats with experimental autoimmune myocarditis (ranging from 500 000 to 5 million cells delivered either intravenously or intramyocardially). 21,31 -34 Furthermore, CDCs were administered 10 days after rat immunization with porcine myosin, a time point shown to be associated with the greatest cell therapy-induced benefits in rats with experimental autoimmune myocarditis. 32
In our study, global intracoronary infusion of CDCs attenuated myocardial inflammation and fibrosis while preventing the myocarditis-induced decline in systolic function and increase in cardiac volumes. The observed functional (increased LVEF) and structural (decreased LV volumes and decreased myocardial fibrosis) benefits appear particularly important from a clinical standpoint; decreased LVEF, increased LV volumes, and increased scar mass have all been associated with increased all-cause mortality, increased cardiac mortality, and increased incidence of sudden cardiac death in patients with biopsy-proven myocarditis. 28
A limitation of our study is that it does not provide insight into the mechanisms through which CDCs exert their salutary effects in myocarditis. However, previous mechanistic studies have demonstrated that CDCs possess significant immunomodulatory properties and can regulate the immune response of both T cells 17 and macrophages, 15 which represent 2 most prevalent cell types in the inflammatory myocardial infiltrates in experimental autoimmune myocarditis. 37 With regard to T cells (which orchestrate the development of experimental autoimmune myocarditis 27 ), Lauden et al demonstrated that CDCs can robustly suppress an ongoing inflammatory T-cell response in vitro. 17 This immunosuppressive effect of CDCs is achieved through CDC-induced stimulation of suppressive regulatory-T cells. This occurs via contact-dependent interaction of PD-1 (located on the surface of activated-T cells) with its ligand, PD-L1 (located on the surface of CDCs). 17 Interestingly, this interaction between PD-1 and PD-L1 has been shown to govern T-cell function in several inflammatory diseases, 38,39 including experimental autoimmune myocarditis. 40 Specifically, Tarrio et al demonstrated that PD-1-deficient mice with experimental autoimmune myocarditis display increased lymphomononuclear infiltration, elevated cardiac expression of pro-inflammatory cytokines and increased cardiomyocyte injury compared to wild-type controls. 40 With regard to the immunomodulatory effects of CDCs on macrophages, De Couto et al demonstrated that CDCs (administered in the setting of ischemia–reperfusion) polarize macrophages toward a distinctive phenotype (different from M1 or M2), characterized by reduced expression of cytotoxic, proinflammatory cytokines, and increased expression of the anti-inflammatory cytokine IL 10. Importantly, this unique macrophage phenotype conferred cardioprotective effects through activation of an antiapoptotic program in myocytes. 15 Thus, the aforementioned immunomodulatory effects of CDCs on T cells and macrophages could explain the decreased lymphomononuclear infiltration observed in CDC-treated hearts with myocarditis. Furthermore, on top of their immunomodulatory/immunosuppressive effects, CDCs also exert robust regenerative 9 -13 and antifibrotic effects, 16 both of which could (at least partially) underlie the salutary effects observed in our study.
Conclusion
In conclusion, in the first preclinical study to date to evaluate the effect of CDCs in myocarditis, we found that global intracoronary infusion of CDCs dramatically attenuates myocardial inflammation, T-cell infiltration and fibrosis while preserving systolic function and preventing adverse remodeling. The observed intriguing efficacy signals warrant further exploration of intracoronary delivery of CDCs as a potential therapeutic strategy in acute myocarditis.
Footnotes
Author Contributions
Nana-Leventaki, E contributed to acquisition and analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy; Nana, M, Poulianitis, N, Sampaziotis, D contributed to acquisition, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy; Perrea, D, Sanoudou, D, Rontogianni, D contributed to analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Malliaras, K contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K. Malliaras has received consulting fees from Capricor Therapeutics, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
