Abstract
Background:
The “no reflow” phenomenon (microvascular obstruction despite restoration of epicardial blood flow) develops postreperfusion in acute myocardial infarction and is associated with poor prognosis. We hypothesized that increased reperfusion pressure may attenuate the no reflow phenomenon, as it could provide adequate flow to overcome the high resistance of the microvasculature within the no reflow zone. Thus, we investigated the effect of modestly elevated blood pressure during reperfusion on the extent of no reflow area and infarct size in a porcine model of ischemia–reperfusion.
Methods:
Eighteen farm pigs underwent acute myocardial infarction by occlusion of the anterior descending coronary artery for 1 hour, followed by 2 hours of reperfusion. Just prior to reperfusion, animals were randomized into 2 groups: in group 1 (control group, n = 9), no intervention was performed. In group 2 (n = 9), aortic pressure was increased by ∼20% (compared to ischemia) by partial clamping of the ascending aorta during reperfusion. Following 2 hours of reperfusion, animals were euthanized to measure area at risk, infarct size, and area of no reflow.
Results:
Partial clamping of the ascending aorta resulted in modest elevation of blood pressure during reperfusion. The area at risk did not differ between the 2 groups. The no reflow area was significantly increased in group 2 compared to control animals (50% ± 13% vs 37% ± 9% of the area at risk;
Conclusions:
Modestly elevated blood pressure during reperfusion is associated with an increase in no reflow area and in infarct size in a clinically relevant porcine model of ischemia–reperfusion.
Keywords
Introduction
Prompt myocardial reperfusion is the most effective therapeutic strategy for reducing infarct size and improving clinical outcome following acute myocardial infarction (AMI). 1 However, in approximately 30% of patients with AMI, complete restoration of myocardial tissue perfusion does not occur, despite restoration of blood flow in the epicardial coronary vessels. 2 –4 This phenomenon, termed “no reflow” (NR) phenomenon, can be defined as an incomplete and non-uniform reperfusion of the microvascular bed post-ischemia, despite adequate restoration of epicardial blood flow. 5 The NR phenomenon is associated with poor prognosis (left ventricular remodeling, adverse clinical events, and death) 2,6 and has been the subject of intensive research effort. 7 While the pathophysiology of NR is complex and multifactorial, its hallmark is endothelial cell damage resulting in microvascular obstruction. 5,8 Considering that endothelium-dependent vasodilation of coronary vasculature is severely compromised in the reperfused ischemic heart 9,10 and perfusion pressure is the main determinant of coronary flow after an ischemic insult, 11,12 we hypothesized that increased reperfusion pressure post-MI may attenuate the NR phenomenon. Thus, we aimed to investigate the effect of modestly elevated blood pressure during reperfusion on the extent of NR area and infarct size in a porcine model of ischemia–reperfusion.
Methods
Experimental Protocol
All animals used in this study received humane care in compliance with the Guide for the Care and Use of Laboratory Animals from the Institute for Laboratory Animal Research. The protocol was approved by the Directorate of Agricultural & Veterinary Policy of the Region of Attica, in accordance to national and EU legislation.
Eighteen farm pigs weighing ∼35 kg were studied. Animals were premedicated with ketamine hydrochloride (15 mg/kg intramuscularly [IM]) and midazolam (0.5 mg/kg IM) and anesthetized with thiopental sodium (9 mg/kg intravenously [IV] bolus) and fentanyl citrate (0.5 mg IV bolus), followed by continuous IV infusion of thiopental sodium (1 mg/min), fentanyl citrate (4 mg/min), and pancuronium bromide (0.25 mg/min). After intubation, animals were mechanically ventilated with a Soxitronic (Soxil, S.P.A.; Segrate, Italy) volume ventilator. Midline sternotomy was performed and the heart was suspended in a pericardial cradle. A Millar pressure-tip catheter (Millar Inc, Houston, Texas) was placed in the ascending aorta via the right carotid artery, a Swan-Ganz catheter was placed in the left atrium through the left atrial appendage and a 6F catheter was placed in the right internal jugular vein. In a subset of animals (n = 8), a ring-shaped Doppler flow probe (MediStim Inc, PS100022) was placed around the mid-left anterior descending artery (LAD) to measure blood flow.
Experimental animals underwent myocardial ischemia by occlusion of the mid-LAD (between the first and second diagonal branch) for 1 hour, followed by 2 hours of reperfusion. Just prior to reperfusion, animals were randomized into 2 groups: in group 1 (control group, n = 9), no intervention was performed during the reperfusion period. In group 2 (n = 9), aortic pressure was increased by 20% (compared to ischemia) by partial clamping of the ascending aorta, distal to the origin of coronary vessels and to the tip of the Millar catheter. The ascending aorta was partially clamped just prior to reperfusion (after 1 hour of ischemia) and remained partially clamped during the 2 hours of the reperfusion period. Electrocardiogram, aortic blood pressure, central venous and wedge pressures were continuously monitored during the experiment. Recordings of aortic pressure (measured in the ascending aorta proximal to the clamping site), wedge pressure and right atrial pressure were obtained at baseline (prior to LAD occlusion) and every 15 minutes during ischemia and reperfusion. Recordings of LAD blood flow (obtained in only 8 animals due to technical issues) were obtained at baseline (prior to LAD occlusion) and at 15, 30, 60, and 120 minutes of reperfusion. Ventricular arrhythmias were treated with lidocaine. Body temperature was maintained at 37°C ± 0.5°C with a heating blanket throughout the experiment.
After 2 hours of reperfusion, thioflavin S (2%, 1 mg/kg) was infused through the Swan-Ganz catheter into the left atrium. Thioflavin S is a fluorescent dye, which stains endothelium receiving optimal blood flow. Areas that are hypoperfused (receiving ≤50% of normal blood flow) are not stained and appear as nonfluorescent regions within the infarct zone 5,13,14 ; these hypoperfused regions represent areas of NR. Subsequently, the LAD was reoccluded and gentian violet (1%) was injected into the left atrium. Gentian violet stains the nonischemic zone of the left ventricle (LV) blue; the region of the heart that is not perfused by Gentian violet (ie, does not stain blue) represents the ischemic area (area at risk). Once dye infusions were performed, animals were euthanized (by electrical fibrillation of the heart) and the heart was explanted. The LV, including the septum, was isolated (after removal of the atria and the right ventricular free wall) and cut into 1-cm thick sections perpendicular to the apex–base axis.
The LV area at risk was identified by the absence of gentian violet dye. The borders of the area at risk were traced with sinice dye on heart slices. Afterward, sections were examined in a dark room under ultraviolet light (365 nm wave length, no barrier filter used) and the area of NR was traced as the nonfluorescent area (not stained by thioflavin) within the area at risk. 5,13 –15 Finally, sections were incubated with 1% triphenyl-tetrazolium chloride at 37°C for 20 minutes and the infarcted area, which remains unstained due to lack of nicotinamide-adenine dinucleotide in nonviable myocardial cells, was traced. 16 Tracings of the epicardium/endocardium and of the areas demarcated by gentian violet, thioflavin, and triphenyl-terazolium staining of both aspects (basal and apical) of each ventricular section were manually drawn on transparent plastic sheets (placed on top of the ventricular slices) by a researcher blinded to treatment assignment. Afterward, the plastic sheets were digitally photographed and the demarcated areas (ie, the area at risk, the NR area and the infarcted area) were measured by computer-based planimetry by a researcher blinded to treatment assignment. The myocardial area at risk was calculated as a percentage of the LV. The NR area and the infarcted area were calculated as a percentage of the area at risk.
Statistical Analysis
Continuous measures for each group were expressed as mean ± standard deviation. Normality of data was tested using the Shapiro-Wilk test. If normality was established, differences of morphometric measurements between groups were tested using independent samples
Results
Adverse Events and Mortality
One animal in group 1 and 1 animal in group 2 died during myocardial ischemia, due to arrhythmia and cardiogenic shock respectively. Eight animals in each group completed the experimental protocol and were included in the analysis.
Hemodynamic Measurements
At baseline and during ischemia, heart rate, aortic pressure, right atrial pressure and pulmonary capillary wedge pressure did not differ significantly between groups (
HR, SBP, DBP, CVP and PCWP During Ischemia and Reperfusion.a

Abbreviations: HR, heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; CVP, central venous pressure; PCWP, pulmonary capillary wedge pressure; SD, standard deviation; ns, not significant.
aHeart Rate, CVP, and PCWP did not differ between groups (

Systolic blood pressure at baseline, during ischemia and during reperfusion. Data are presented as mean ± standard deviation (SD).

Diastolic blood pressure at baseline, during ischemia and during reperfusion. Data are presented as mean ± standard deviation (SD).
Morphometric Measurements
The area at risk did not differ between the 2 groups (group 1: 28% ± 9% of LV vs group 2: 24% ± 8% of LV,

Assessment of “no reflow” area. Elevated blood pressure (group 2) during reperfusion resulted in significantly increased area of “no reflow” compared to control animals (group 1). Data are presented as mean ± standard deviation (SD).
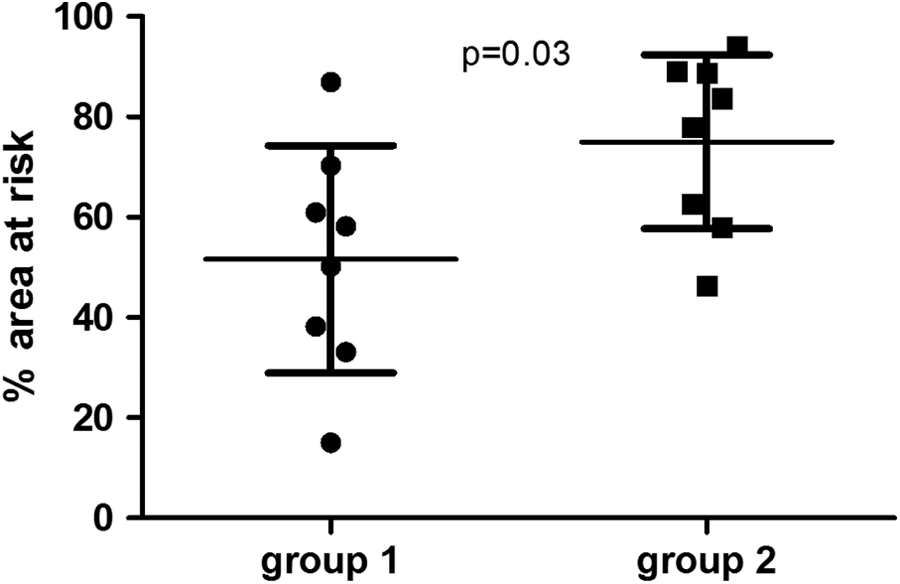
Assessment of infarcted area. Elevated blood pressure during reperfusion (group 2) resulted in significantly increased infarcted area compared to control animals (group 1). Data are presented as mean ± standard deviation (SD).
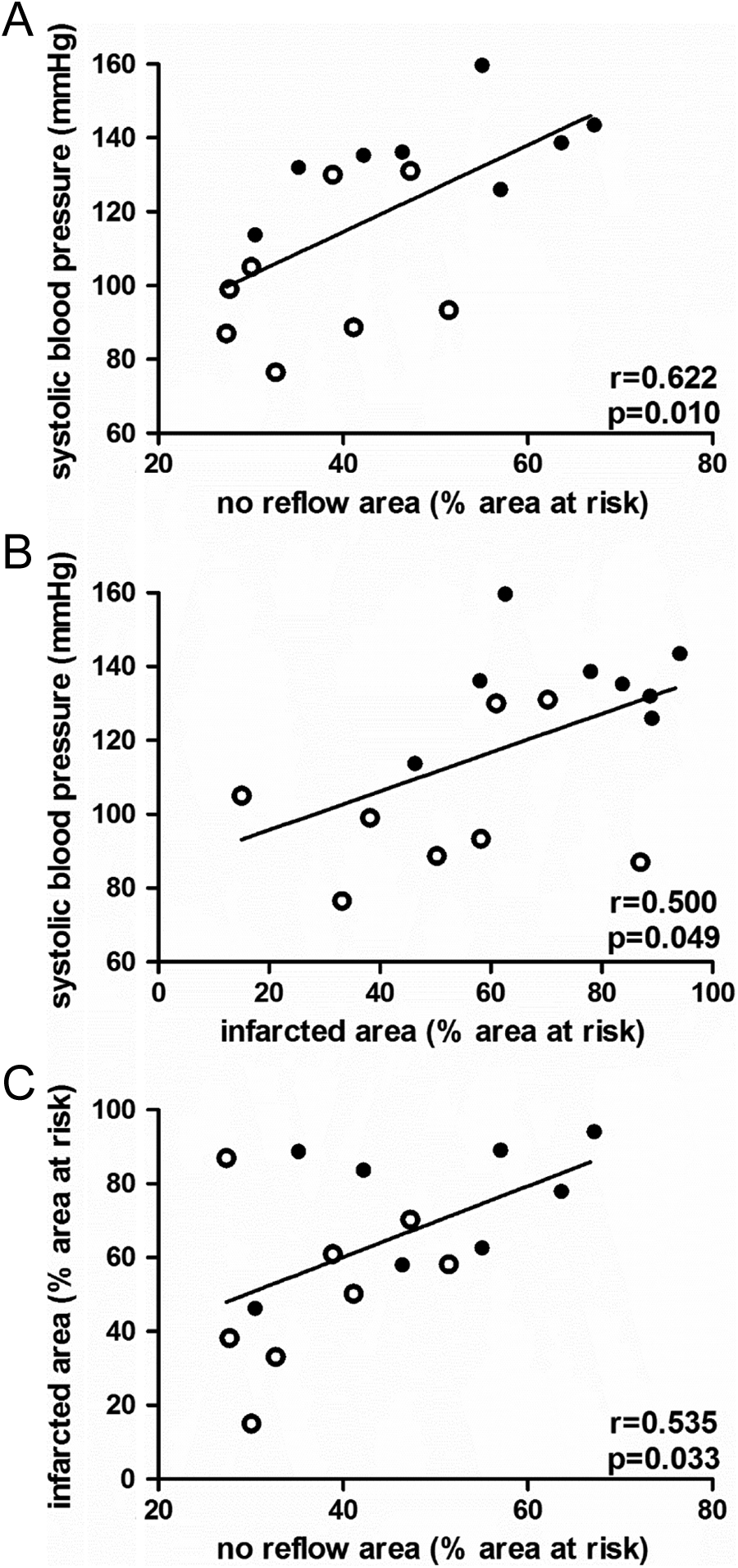
Cardiac afterload post-reperfusion correlates positively with infarct size and area of “no reflow.” A, Correlation between systolic blood pressure during reperfusion and area of “no reflow”. B, Correlation between systolic blood pressure during reperfusion and infarcted area. C, Correlation between infarcted area and area of “no reflow.” Open circles denote animals randomized to the control group; full circles denote animals randomized to undergo elevation of blood pressure during reperfusion.
Discussion
The area of NR increases 3-fold between 2 minutes and 3.5 hours of reperfusion, 17 with most of the expansion occurring within the first hour of reperfusion. 18 Therefore, it is reasonable to assume that therapeutic interventions aiming at attenuating the extent of NR must be applied during the early reperfusion phase.
Reperfusion pressure post-AMI can be readily regulated in clinical practice through iatrogenic manipulation of arterial blood pressure. In a previous experimental study of our group, Nanas et al demonstrated that modestly reduced aortic pressure (mean pressure 60-75 mm Hg) during reperfusion (after 1 hour of ischemia) was associated with decreased coronary blood flow and increased infarct size. 19 This finding can be rationalized by the observation that coronary autoregulatory mechanisms, which in the normal heart maintain constant tissue perfusion despite changes in perfusion pressures, are severely compromised after an ischemic insult. 9,10 As a result, coronary flow and tissue perfusion in the ischemic heart are strongly dependent on the driving perfusion pressure. 11,12 In the study by Nanas et al, the investigators concluded that modestly reduced reperfusion pressures could not provide the necessary pressure gradient to perfuse NR areas, which contain a microvascular bed characterized by increased vascular resistance (due to endothelial damage and external compression due to tissue oedema). Based on these observations, we hypothesized that increased reperfusion pressure post-MI may attenuate the NR phenomenon, as it could provide adequate flow to overcome the high resistance of the microvasculature within the NR zone.
Contrary to our hypothesis, we found that modestly elevated reperfusion pressure was associated with an increase in NR area and in infarct size in a porcine model of ischemia–reperfusion. A similar afterload-induced exacerbation of infarct size has also been observed in Langendorff-perfused rat hearts. 20 Our findings could be explained by several pathophysiological mechanisms. First, elevated blood pressure (ie, increased afterload) increases left ventricular oxygen consumption in the reperfused heart through 2 distinct mechanisms: (1) increased afterload directly increases left ventricular mural tension and myocardial work and (2) increased myocardial perfusion (due to increased perfusion pressure), results in improved cardiac contractility and increased myocardial energy consumption (Gregg’s phenomenon). 21,22 Thus, elevated blood pressure may aggravate ischemia of reperfused myocardium (if not accompanied by comparable increases in tissue perfusion and oxygen supply), resulting in exacerbation of myocyte death (which naturally continues to occur in the risk area beyond the immediate postreperfusion period 23 ). This afterload-induced increase in infarcted area would naturally be associated with increased NR area, as the amount of myocardial tissue necrosis has been shown to be a major determinant of the extent of NR. 5,24,25 The aforementioned pathophysiological mechanism is supported by the fact that in our study we observed a significant positive correlation both between afterload and infarcted area (Figure 5B) and between infarcted area and NR area (Figure 5C). Taking the above into consideration, it is conceivable that interventions that increase reperfusion pressure and myocardial blood flow but concurrently decrease left ventricular afterload and myocardial oxygen consumption (like the intra-aortic balloon pump) 26,27 may result in attenuation of the NR phenomenon. 28,29 Second, increased afterload may directly affect NR, irrespective of changes in infarct size; elevations in blood pressure may result in increased resistance of the microvascular bed, due to higher extravascular resistive forces resulting from elevated left ventricular pressures (which increase in parallel with the aortic pressure). Third, it has been shown that in stenotic vessels, the augmentation of myocardial blood flow in response to increases in afterload and cardiac work is attenuated. 30 Thus, it is reasonable to assume that in the injured microvasculature of NR areas, where endothelial protrusions and blebs obstruct the vascular lumen, 5 the rise in blood pressure may not be accompanied by a respective increase of coronary flow. Fourth, increased reperfusion pressure may exacerbate NR by aggravating microembolization of the microvasculature by aggregates of red blood cells, neutrophils, and platelets (by pushing such aggregates distally into vessels of smaller diameter).
Taking the findings of the current and our previous experimental study 19 together, it could be argued that (1) a U-shaped curve association exists between reperfusion pressure (or aortic blood pressure during reperfusion) and NR area or infarct size, and (2) a mean aortic pressure between 80 and 100 mmHg during reperfusion is associated with reduced NR area and reduced infarct size. Of note, a similar U-shaped curve association has been observed in patients with acute coronary syndromes between arterial blood pressure and risk of future cardiovascular events. 31 Interestingly, a similar association between arterial blood pressure and clinical outcomes has also been observed in patients with acute stroke. 32
Study Limitations
Our study has important limitations. First, our model of iatrogenic AMI (which involves LAD ligation) cannot fully reproduce the pathophysiology of acute coronary syndromes (which involves plaque rupture and thrombus formation). Consequently, distal microembolization of thrombotic material, which contributes to development of NR in the clinical setting, cannot be simulated by our experimental model. 5,18,33 Nevertheless, there is evidence that mechanisms which contribute to the development of the NR phenomenon in experimental animals are also present in humans. 5,34 –36 Second, LAD blood flow measurements were obtained (due to technical issues) in only a subset of experimental animals (8 of 16, 4 in each group).
Footnotes
Author Contributions
Pantsios contributed to design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kapelios contributed to analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Vakrou contributed to analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Diakos contributed to analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Pozios contributed to analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Kontogiannis contributed to analysis, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Nanas contributed to conception and design, contributed to interpretation, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Malliaras contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
