Abstract
Introduction
Several antiarrhythmic drugs, as well as noncardiac drugs, block the rapid delayed rectifying potassium channel (IKr) and consequently prolong cardiac repolarization and increase the action potential (AP) duration.1–4 An increased AP duration can be observed as QT interval prolongation on the surface electrocardiogram (ECG). Drug-induced excessive prolongation of the heart rate-corrected QT interval (QTc) has been associated with torsade de pointes (TdP), a potentially lethal polymorphic ventricular tachycardia. Women are 2 to 3 times more likely to develop drug-induced TdP than are men.5–8 This disparity may be due to gender differences in cardiac repolarization. The QTc interval shortens after puberty in males but not in females, resulting in a longer QTc in women than in men. 9 Furthermore, females and males may have different dynamics of ventricular repolarization changes following exposure to a QTc prolonging drug.
Recently, a development program was undertaken to gain approval for an intravenous formulation of sotalol that could be administered to those patients who are unable to take sotalol orally. 10 The development program was based on a bioequivalence study where healthy participants were exposed to low sotalol doses. One of the unexpected findings of this study was that very low doses of sotalol resulted in a significant QTc prolongation in the entire study population. 11 This report aims to assess potential gender differences in QTc prolongation following intravenous sotalol administration that may provide an explanation for the greater incidence of TdP in females receiving QT prolonging drugs.
Methods
A bioequivalence study was undertaken comparing 80 mg oral sotalol and 75 mg intravenous sotalol administered over 2.5 hours to obtain US Food and Drug Administration (FDA) approval for intravenous sotalol. Electrocardiograms were closely monitored and systematically recorded in the study, providing sufficient data for the evaluation of potential drug-induced gender differences in cardiac repolarization. The methods of the original study have already been published in detail. 10
Study Population
The study was approved by the Institutional Review Board of the University of Miami, Miami, Florida, where the clinical part of the study was carried out. Sotalol-naive healthy participants 18 to 45 years of age with no clinically important findings on medical history, 12-lead ECG, physical examination, or laboratory evaluation were eligible for this study. A total of 18 volunteers were recruited, each was fully informed and gave written informed consent to participate in the study. Of the 18 participants, 16 completed the study according to the protocol, 1 was excluded due to noncompliance resulting in 15 volunteers for this analysis.
Intravenous Drug Administration
The intravenous administration consisted of a single dose of 75 mg sotalol administered over 2.5 hours with a constant infusion rate (sotalol hydrochloride injection, 15 mg/mL in an aqueous formulation, Lot number: API 021017; Academic Pharmaceuticals, Lake Bluff, Illinois).
Measurements of Sotalol Serum Concentrations
A total of 18 blood samples were collected from time 0 (before dose administration) to 48 hours following the start of the infusion in each participant. Serum levels of sotalol were measured using validated liquid chromatography–Tandem Mass Spectrometry (LC-MS/MS) technique at Rush Pharmacology Analytical Laboratory Services, Rush University, Chicago Illinois.
Electrocardiograph Measurements
A 12-lead ECG was recorded at baseline (time 0), and at 0.5, 1, 2, 3, 4, 5, and 48 hours following the start of the infusion in each participant. The QT and RR intervals were measured in each ECG lead by an experienced investigator who was blinded to the drug. Each QT was corrected for heart rate using the Fridericia formula12,13 (QTc = QT/ 3 √RR) and the Framingham formula 14 [QTc = QT + 0.154(1 − RR)]. For each 12-lead ECG, the average RR, QT, and QTc intervals were calculated and used for data analysis.
Data Analysis
Statistical analysis was performed using SPSS for Windows software package (version 10, SPSS Inc, Chicago, Illinois). Continuous variables were expressed as mean ± standard deviation, categorical variables were provided as absolute numbers. Differences between females and males in demographic and baseline characteristics, as well as in the magnitude of individual changes in ECG measurement were analyzed by independent sample t tests. Repeated measures of analysis of variance (ANOVA) were employed to assess gender differences in ECG measurements across the sampling time points. Linear regression analyses were performed and Pearson’s correlation coefficients were calculated to assess potential gender differences in correlations between sotalol blood levels and the ECG measurements. To evaluate predictors for the magnitude of QT/QTc changes, univariate analyses (General Linear Models) were performed. Differences were considered statistically significant with a P value of <.05 (2-sided test, α error <.05).
Results
Study Population
A total of 15 healthy volunteers, 9 females and 6 males, were included in this study. The demographic data and baseline characteristics of these participants are summarized in Table 1 . Male participants were significantly taller and had greater body weight and body surface area (BSA). The serum creatinine was higher in males but was within the normal range in both genders. The baseline QT intervals were practically identical in females and males, while there was a trend toward higher heart rate (shorter RR) and longer QTc in females compared to males.
Demographic and Baseline Characteristics of Female and Male Volunteers
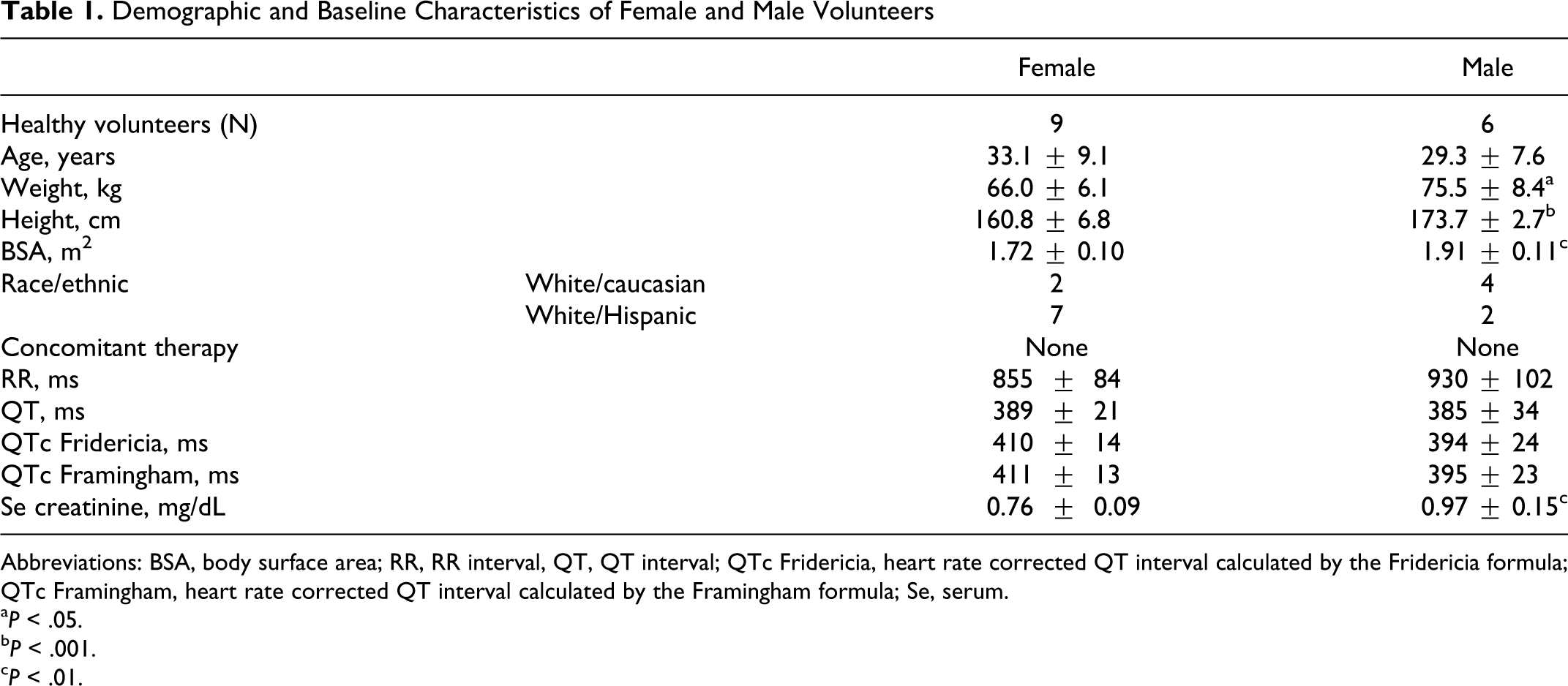
Abbreviations: BSA, body surface area; RR, RR interval, QT, QT interval; QTc Fridericia, heart rate corrected QT interval calculated by the Fridericia formula; QTc Framingham, heart rate corrected QT interval calculated by the Framingham formula; Se, serum.
a P < .05.
b P < .001.
c P < .01.
Electrocardiographic Changes due to Intravenous Sotalol Administration
The changes in ECG measurements during and after sotalol infusion are summarized in Table 2 . Compared to baseline measurements obtained just before the start of drug administration (time 0), an early and significant increase in RR and QT intervals were observed in both females and males following the start of sotalol infusion. The RR intervals became significantly longer at 0.5 hours of the 2.5-hour infusion, showed a peak value at 1 hour, and then slowly shortened despite continuing drug administration. The changes in QT intervals are also shown in Figure 1 . The QT intervals were similar in females and males at baseline (389 ± 21 ms vs 385 ± 34 ms, NS) and then significantly increased by 1 hour in both females (426 ± 29 ms, P < .001) and males (407 ± 31 ms, P < .001). The peak QT was seen at 2 hours of infusion in both female and males (436 ± 28 ms and 413 ± 28 ms, respectively, P < .001 compared to baseline). The magnitude of individual changes from baseline were significantly greater (P < .05) in females than in males (Table 3 ). The QT remained longer in females than in males after the end of infusion until the end of the 5-hour study period (Figure 1, Table 2).
Effects of 75 mg Intravenous Sotalol Administered Over 2.5 hours on ECG measurements in Females and Males a

aQTc Fridericia and QTc Framingham is the heart rate corrected QT intervals calculated by the Fridericia, and Framingham formulas, respectively. Repeated Measures of analysis of variance (ANOVA) indicate that QTc Fridericia and QTc Framingham were significantly longer in females than in males (P < .05).
b P < .01.
c P < .05.
d P < .001 compared to baseline measurements that were obtained immediately before dosing (0 hour).

Gender differences in QT intervals following intravenous sotalol administration. The QT intervals were plotted as a function of time. Sotalol infusion was started at time 0 and was administered over 2.5 hours with a constant infusion. The QT was similar in females and males at baseline (time 0) and then significantly increased by 1 hour in both females and males (P < .001), with a greater magnitude in females. The peak value was seen at 2 hour infusion in both genders. After the end of infusion, the QT shortened in both males and females, however it was still significantly longer at 5 hours than at baseline in females (P < .001) but did not differ from baseline significantly in males.
Maximum Individual Changes From Baseline Measurements by Gender a

Abbreviations: QTc Fridericia, heart rate corrected QT interval calculated by the Fridericia formula; QTc Framingham, heart rate corrected QT interval calculated by the Framingham formula.
aThe magnitude of individual changes from baseline to maximum values in QT and QTc intervals were significantly greater in females than in males.
The QT intervals were corrected for heart rate by both the Fridericia and Framingham formulas. The 2 formulas resulted in virtually identical QTc intervals (Table 2). At 1 hour of infusion, QTc became significantly greater in females than it was at baseline (Framingham QTc 411 ± 13 ms vs 427 ± 18 ms, P < .001), while in males the increase was not significant (395 ± 23 ms vs 400 ± 26 ms). The longest QTc intervals were observed at 2 hours in both genders (Table 2, Figure 2 ). Compared to baseline, the increase was very significant in females (Framingham QTc baseline vs 2 hours: 411 ± 13 ms vs 438 ± 13 ms, P < .001), while it was less significant in males (395 ± 23 ms vs 413 + 27 ms, P < .05). The magnitude of individual changes from baseline were significantly greater (P < .05) in females than in males (Table 3). Furthermore, the QTc intervals in males returned to the baseline value after the end of infusion, while in females QTc remained significantly longer until the end of the 5-hour study period (Figure 2, Table 2). When the gender differences in sotalol-induced QTc changes were analyzed across each time point, the difference was statistically significant between females and males, with females having greater QTc prolongation (repeated measures of ANOVA: P < .05).

Gender differences in QTc intervals following intravenous sotalol administration. The QT intervals were corrected for heart rate by the Framingham formula and plotted as a function of time. Sotalol infusion was started at time 0 and was administered over 2.5 hours with a constant infusion. There was a nonsignificant trend toward longer QTc in females at baseline. The longest QTc intervals were observed at 2 hours for both genders. The QTc intervals in males returned to the baseline value after the end of infusion, while in females QTc remained significantly longer until 5 hours. Females had greater QTc prolongation than males following sotalol administration (P < .05 with repeated measures of analysis of variance [ANOVA]).
Linear regression analysis was performed to assess the relation between sotalol serum concentrations and the length of QTc intervals. The result is shown in Figure 3 . The length of QTc intervals strongly correlated with serum sotalol concentration in both males and females (r = .93, P < .001). The equation that describe the relation between the QTc intervals and sotalol concentrations was QTc = 0.0383 × (sotalol concentration) + 412.49 in females and QTc = 0.0365 × (sotalol concentration) + 388.57 in males. The upward shift of the female’s trend line indicates that females have a 24 to 25 ms longer QTc than males at any serum sotalol concentration level in the concentration range studied.

Correlation between sotalol serum concentration and QTc intervals following intravenous sotalol administration. QTc intervals were plotted as the function of sotalol serum concentration. A linear relationship and very strong correlation can be seen between sotalol concentration and the length of QTc intervals in both females and males. Based on the trend lines, it is predictable that females have a 24 to 25 ms longer QTc at any serum sotalol concentration than males in the concentration range studied.
Compared to females, the greater body weight and BSA of males could have translated to lower serum sotalol concentration that may have influenced the observed gender differences in QT and QTc prolongation. However, body weight and BSA did not correlate with QTc prolongation. While there was a trend toward higher serum sotalol concentration in females than in males (maximum serum concentration: 702 ± 335 ng/mL vs 655 ± 411 ng/mL), this difference was not statistically significant (P = .813) with a large variability in each group. The area under the concentration-time curve (AUC) did not differ significantly between females and males. Furthermore, univariate analysis showed that neither the ethnicity nor the body weight or BSA had predictive value for the magnitude of QTc prolongation. The single factor that had predictive value was female gender (P < .05).
Discussion
In this study, females had a greater QTc prolongation than males following administration of identical doses of intravenous sotalol, and females had greater QTc than males at any sotalol blood level.
The relation between QTc prolongation and increased risk of fatal arrhythmias is well established. Women are 2 to 3 times more likely to develop drug-induced TdP than are men.5–8 The observed excess QTc prolongation in females seen in our study may partly explain the disparity in the incidence of drug-induced TdP between females and males.
Sotalol is an antiarrhythmic drug with Vaughan Williams class II (β-adrenergic receptor blocking) and class III (AP duration prolonging) properties.15–17 The class III effect of sotalol is especially important in its antiarrhythmic action.18,19 Sotalol and other antiarrhythmic agents with class III properties block IKr, the rapid delayed rectifying potassium channel, in a concentration-dependent manner.1–4,18,19 This results in a blockade of potassium outflow currents and prolongation of the plateau (phase II) of the AP that is seen as QT prolongation on the surface ECG. This action can be therapeutic since lengthening the refractory period can prevent re-entrant arrhythmias. However, when the AP is markedly prolonged, early after-depolarizations (EADs) may occur that are believed to be the basis for the clinical syndrome of TdP.1,20–22 Unfortunately, numerous noncardiac drugs can also block the Ikr channel, resulting in an unwanted drug-induced QT prolongation and increased risk of proarrhythmia and sudden cardiac death.2–4 Indeed, several drugs including cisapride, hismanal, and terfenadine were withdrawn from the market because of QT prolongation and fatal arrhythmias.
While the reason for the gender disparity in the incidence of drug-induced TdP have not been completely clarified, it is believed that gonadal steroids play a role. It has been reported that females and males have no differences in QTc intervals before the time of puberty. From puberty, males develop shorter QTc while there is no substantial changes in QTc in females, suggesting that testosterone is responsible for the shorter QTc in men than in women. 9 The resultant longer “baseline” QTc in females has been associated with the higher susceptibility to TdP, but the association between QTc prolongation, sex hormones, and TdP is probably far more complex.23,24
Experimental data indicates that progesterone acutely increases the slow delayed rectifier potassium current (IKs) and shortens the AP in a dose-dependent manner.
25
In a physiologic concentration range, acute effects of estradiol result in a dose-dependent reduction in Ikr current that lengthens the AP duration.
26
Testosterone regulates both IKs and the
While we do not know the mechanism for the greater sotalol-induced QTc prolongation in females than in males seen in our study, we assume that sex hormones play a substantial role.
In a prior publication, we have reported an unexpected finding regarding the dose of sotalol and a measurable class III effect. 11 It has been reported that significant QTc prolongation occurs only at daily oral sotalol doses of 160 mg and higher.15,16 Our finding in 15 healthy participant contradicted this report. Compared to baseline, a very significant QTc prolongation was observed at the time when 30 mg intravenous sotalol was administered indicating that a single very low dose of sotalol can result in significant AP prolongation. Additionally, we found a very strong correlation between serum sotalol concentration and QT prolongation, suggesting that the AP prolonging effect of sotalol is a function of sotalol serum concentration. 11
The current report extends these observations, identifying the differences between females and males. While significant QTc prolongation was seen with low sotalol dose in both genders, the dose of sotalol required to induce significant QTc prolongation was lower in females than in males. Females had significant QTc prolongation at 1 hour of the 2.5-hour infusion. By this time, 30 mg sotalol was administered. In males, significant QTc prolongation was seen at 2 hours of the infusion. By this time, 60 mg sotalol was administered. Furthermore, the QTc prolongation remained significant in females after the end of the infusion until the end of the 5-hour study period, while in males the QTc prolongation was short lasting and the QTc returned to the baseline by the end of the study period. These observations indicate a gender difference, namely females have earlier and longer lasting QT prolongation than males. At higher sotalol doses, this difference may make females more prone to excessive QT prolongation and TdP.
There was a trend toward longer QTc in females than in males before drug administration (410 ± 14 ms vs 394 ± 24 ms), a statistically nonsignificant 16-ms difference. Following sotalol administration, the difference significantly increased. When the correlation between serum sotalol concentration and QTc was assessed with linear regression analysis, a very strong correlation was seen in both females and males. Body weight and BSA did not correlate with sotalol blood level and QT prolongation. The single factor affecting the degree of QT prolongation was female gender.
Conclusion
Females had greater QT prolongation than males following sotalol administration. The single predictor for greater QTc prolongation by sotalol was female gender. A significant QTc prolongation was seen following a single low dose of intravenous sotalol. This early effect may indicate antiarrhythmic efficacy, as well as a potential for early toxicity, making ECG monitoring and frequent determination of QTc important when sotalol therapy is initiated.
Footnotes
The work for this study was done at the Miller School of Medicine, Division of Clinical Pharmacology, University of Miami, Miami, Florida, USA.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Doctor Somberg has equity holdings in Academic Pharmaceuticals. Doctor Ranade and Doctor Molnar are employees of Academic Pharmaceuticals. Doctor Preston and Doctor Cvetanovic have no conflict of interest.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Academic Pharmaceuticals, Lake Bluff, IL USA.
