Abstract
Background:
The duration of randomized controlled clinical trials usually is approximately 3 to 5 years although hypercholesterolemia and other risk factors for atherosclerotic cardiovascular disease (ASCVD) are lifelong conditions.
Objectives:
The legacy effect, defined as the persistence of benefit of pharmacologic interventions in clinical trials after the end of the randomized phase when all participants receive active therapy, is used to examine the long-term benefit. We summarize the evidence for the existence of the legacy effect as it pertains to hypercholesterolemia, describe underlying mechanisms, and discuss its relevance to clinical practice.
Methods:
We examined all published (n = 13) randomized clinical trials of lipid-lowering agents compared to placebo or usual care with follow-up after the randomized phase for the presence or absence of a legacy effect.
Results:
A legacy effect was demonstrated in all studies. The current US and European guidelines recommend treatment with high-intensity statins for patients with manifest ASCVD and that individualized approach be used for primary prevention.
Conclusion:
The legacy effect results in significant long-term clinical benefits by preventing fatal and nonfatal events. This implies that early therapy would result in lower event rates. Long-term follow-up should be a part of clinical trial design in order to evaluate the presence or absence of a legacy effect.
Introduction and Definition
The usual duration of randomized controlled clinical trials is approximately 3 to 5 years although hypercholesterolemia and other risk factors for atherosclerotic cardiovascular disease (ASCVD) are lifelong conditions. To address this issue, a number of investigators have published post-trial outcomes that are usually categorized under the rubric “legacy effect.” The legacy effect is defined as the persistence of the benefit of pharmacologic interventions in clinical trials after the cessation of the randomized phase when all participants, those initially randomized to active therapy and those randomized to placebo, receive active therapy. 1 Information on the long-term benefits or harms of treatment among patients who continue active therapy and those who discontinue therapy may be important in the evaluation of the overall effects of the specific treatments and in the design of new clinical trials and meta-analyses.
In this review, we summarize the evidence for the existence of the legacy effect as it pertains to hypercholesterolemia, describe underlying mechanisms, and discuss its relevance to clinical practice. Most of the information on the legacy effect was derived from retrospective analyses of randomized clinical trials in patients with hypercholesterolemia. The only prospective study to evaluate the legacy effect is the Adjuvant Tamoxifen: Longer Against Shorter (ATLAS) trial. In this study, women with estrogen-positive breast cancer were randomized to receive tamoxifen for 10 or 5 years (standard of care at the time). Compared to the 5-year treatment group, patients in the 10-year treatment group had lower all-cause mortality at 15 years. 2 In this analysis, breast cancer mortality was 15% in the 5-year tamoxifen group versus 12.2% in the 10-year treatment group. Thus, 36 women needed to be treated for 10 years compared to 5 years to prevent one death due to breast cancer.
The legacy effect in cardiology was recognized initially with the publication of the Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications (EDIC), and the UK Prospective Diabetes Study (UKPDS) trials. 3 -5 These studies compared intensive versus conventional therapies in the management of diabetes, DCCT/EDIC in type 1 diabetic patients, and UKPDS in type 2 diabetes. Participants randomized to the intensive glucose control groups had lower microvascular complications and CV deaths up to an average long-term follow-up of 17 years. In the UKPDS, the intensive treatment cohort had a lower rate of microvascular complications and myocardial infarction (MI) and death from any cause. These benefits were attributed to the phenomenon of “metabolic memory” and decreased formation of advanced glycemic end products. 3 -5
Cholesterol and Atherosclerosis
Cholesterol crystals were first isolated from gallstones in 1784 by the French physician-chemist François Poulletier. 6 A century later, Nikolai Anitschkow described the link between cholesterol and atherosclerosis and laid the foundation for the “lipid hypothesis.” He induced atherosclerotic changes in the vessels of rabbits by feeding them purified cholesterol and demonstrated that the extent of atherosclerotic lesions depended on the amount and duration of exposure. 7 Since that time, a wealth of basic science, biochemistry, epidemiology, and controlled clinical trial data have proven the benefit of cholesterol lowering, usually with statin therapy, in decreasing fatal and nonfatal events in patients with coronary artery disease. Currently, the lowering of low-density lipoprotein cholesterol (LDL-C) is one of the best evidenced interventions in medicine to prevent major CV events. 8
Guidelines
The 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA (multisociety) guideline on the management of blood cholesterol and the 2019 ESC/EAS guidelines for the management of dyslipidemias include recommendations for pharmacologic treatment in primary and secondary prevention. 9,10 Both guidelines recommend a graded approach to the treatment of hypercholesterolemia based on the 10-year risk of ASCVD and comorbidities. Patients are classified into 4 categories: low, moderate, high, and very high risk. Although there is variation between the US multisociety and the European guidelines in the methods of estimating risk and in the exact recommendations for treatment, both guidelines agree that high-intensity statin therapy should be used in patients with manifest ASCVD. For primary prevention, individualized risk-based approach possibly with a shared decision-making by the physician and the patient is recommended.
Primary Prevention
The ESC/EAS guidelines recommend that for individuals at low risk, an LDL-C target of 116 mg/dL is appropriate and a target of 100 mg/dL for those at moderate risk. In cases where the indication for use of statins is not clear, discussion with the patient and measurement of coronary calcium score are recommended. 10 However, when using the above approach, a significant number of CV events may occur among patients at lower or borderline CV risk (residual risk). Kohli-Lynch and associates in an in silico study of 100 simulated cohorts, each with 1 million patients, found that statin use in patients with borderline CV risk in primary prevention resulted in a gain of lifetime quality-adjusted life years (QALYs) and that it was cost-effective ($33 558/QALY). 11 The effect of statin use in primary prevention of ASCVD according to 5 major guidelines for sensitivity, specificity, and number needed to treat is indicated in Table 1. 12
Primary Prevention of Atherosclerotic Cardiovascular Disease According to 5 Major Guidelines for Sensitivity, Specificity, and Number Needed to Treat.a

Abbreviations: ACC, American College of Cardiology; AHA, American Heart Association; CCS, Canadian Cardiovascular Society; EAS, European Atherosclerosis Society; ESC, European Society of Cardiology; HDL, high-density lipoprotein; IQR, interquartile range; LDL, low-density lipoprotein; NICE, National Institute for Health and Care Excellence; PCE, pooled cohort equations; USPSTF, US Preventive Services Task Force.
a Reprinted from reference 12.
Secondary Prevention
In secondary prevention, statin therapy is recommended by both guidelines. Targets lower than 70 mg/dL are recommended for patients at high and very high risk. In addition, ezetimibe and/or PCSK9 antibodies are recommended in very high-risk ASCVD as needed. Maintenance of a heart-healthy lifestyle as the cornerstone for ASCVD risk reduction was reinforced and assessment of the response to therapy and patient adherence are important. 9 -11,13
Meta-Analyses
A Meta-Analysis of the Legacy Effect of Intensive Glucose Control
Schernthaner reviewed studies on the effect of intensive glucose control. He reported that the reduction in CV mortality with blood pressure and lipid-lowering interventions were observed much earlier than the effect of intensive glucose lowering, that may take up to 10 years or more. 14
A Meta-Analysis of the Legacy Effect of Lipid-Lowering Agents
Recent meta-analyses have demonstrated the presence of the legacy effect of cholesterol-lowering therapy. Nayak and associates 15 reported that post-trial statin legacy effects on all-cause mortality were present in primary and secondary prevention trials. A meta-analysis of 44 255 patients across 8 trials studying lipid-lowering agents examined the presence of a legacy effect of these agents. During the first (randomized) phase of the trials, there was a reduction in all-cause mortality (0.84, 95% confidence interval [CI], 0.76-0.93; P = .0006) and CV mortality (0.72, 95% CI, 0.63-0.82; P = .0001). The legacy effect is implied by the persistence of this benefit in the follow-up phase as well (all-cause mortality: odds ratio [OR] 0.90, 95% CI, 0.84-0.97; P = .0035, and CV mortality: 0.82 95% CI, 0.73-0.93; P = .0014; Figures 1 and 2). 1
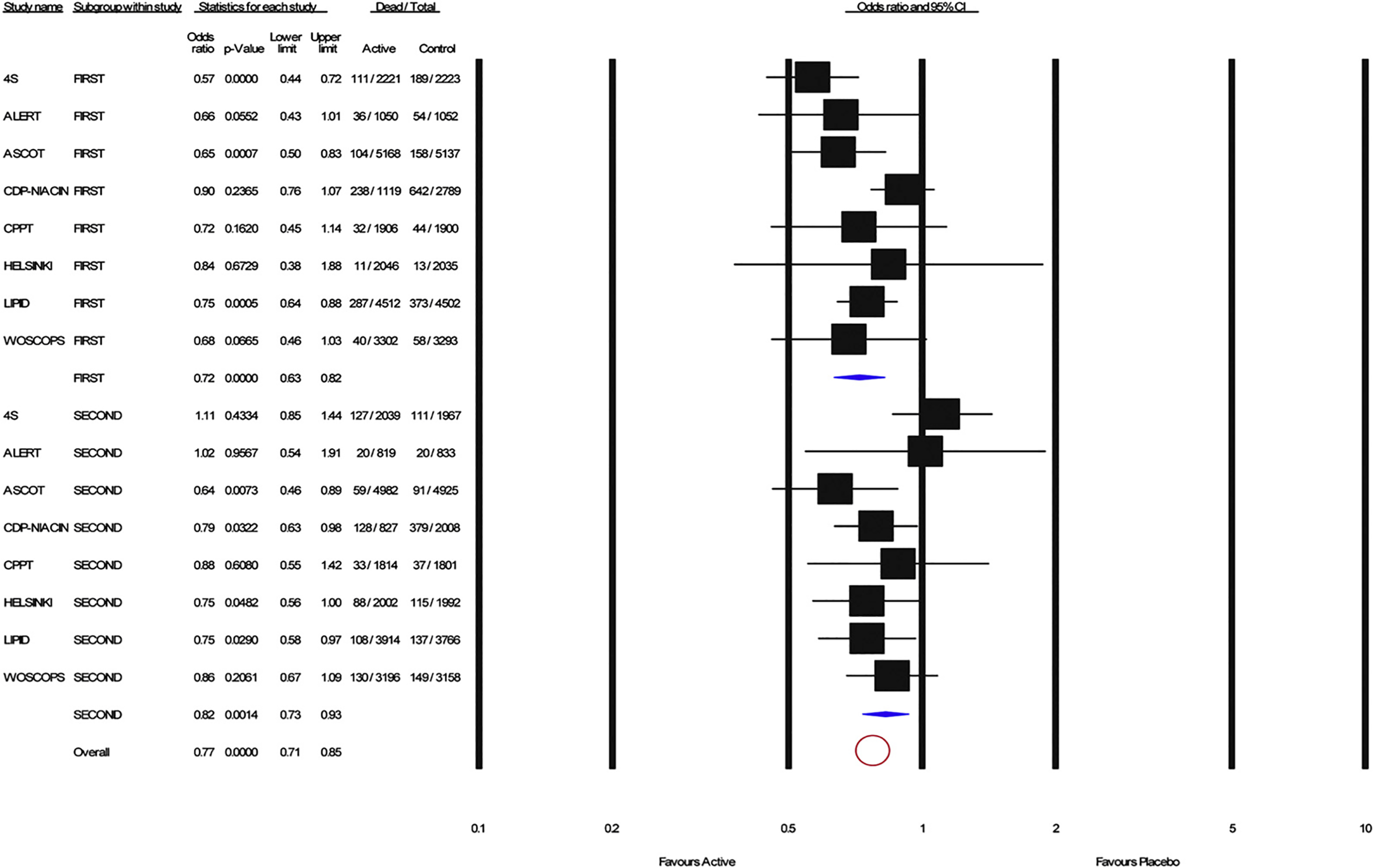
Forest plot for coronary heart disease mortality for the first phase (top) and the second phase (bottom). Solid squares represent the odds ratio in individual trials and have a size proportional to the inverse of the variants. Horizontal lines and diamonds denote the 95% confidence intervals for individual trials and summary statistics, respectively. Pooled estimates were computed from a random-effect model. Red circle denotes overall effect. Reprinted from reference 1.

Forest plot for all-cause mortality for the first phase (top) and the second phase(bottom) of the reviewed studies. Solid squares represent the odds ratio in individual trials and have a size proportional to the inverse of the variants. Horizontal lines and diamonds denote the 95% confidence intervals for individual trials and summary statistics, respectively. Pooled estimates were computed from a random-effect model. Red circle denotes overall effect. Reprinted from reference 1.
The Legacy Effect of Statins
Published in 1994, the Scandinavian Simvastatin Survival Study examined whether the addition of simvastatin improved all-cause mortality in patients with prior MI or angina pectoris and hypercholesterolemia (defined as serum cholesterol 212.7-309.3 mg/dL). 16 The trial was discontinued at 5.4 years of median follow-up since a 3.3% absolute risk reduction in all-cause mortality was seen in the simvastatin group (11.5% vs 8.2%; P = .0003). A reduction in multiple other end points including major coronary events, CV mortality, and coronary interventions was also observed in the simvastatin group. 16 Post hoc analysis demonstrated a reduction in the incidence of cerebrovascular disease. An observational, government registry-based study of mortality of the study participants over an additional 2-year follow-up was performed. Participants were sent a questionnaire 1 year after the cutoff date for the extended follow-up when most of the participants, irrespective of randomization group, were on a statin. Mean serum cholesterol levels were similar (199.5 and 197.6 mg/dL, respectively, in the groups originally randomized to placebo and simvastatin). During the follow-up phase of 10.4 years from randomization, 74 (3.6%) of the 2039 survivors originally randomized to the simvastatin group died as compared to 97 (4.9%) of the 1967 in the placebo group. Thus, the higher survival of the group originally randomized to simvastatin was attributable to the legacy effect. The combined outcome of death from definite coronary heart disease or definite nonfatal MI was lower in the pravastatin group from 7.9% to 5.5% (P <.001). 17 In summary, over the duration of the trial phase plus the follow-up phase, there were 353 (15.9%) deaths in the group originally randomized to placebo and 256 (11.5%) deaths in the group originally randomized to simvastatin, with a relative risk (RR) of 0.70 (95% CI, 0.60-0.82, P = .00002). 16
The West of Scotland Coronary Prevention Study (WOSCOPS) was a randomized clinical trial for primary prevention that compared the effects of pravastatin to placebo in men with hypercholesterolemia without a history of MI with an average follow-up of 5 years. 18 Ford et al reported that at a 20-year follow-up of the WOSCOPS after the trial completion, 35.2% of the placebo and 38.7% of the pravastatin groups were continued on statins. During this extended follow-up, overall mortality (hazard ratio [HR] 0.87, 95% CI, 0.80-0.94, P = .0007), CV mortality (HR 0.79, 95% CI, 0.69-0.90, P = .0004), and coronary mortality (HR 0.73, 95% CI, 0.62-0.86, P = .0002) were all lower in the group originally randomized to pravastatin compared to the placebo group. There was also a 35% lower rate of hospitalizations over the 20-year follow-up in the pravastatin arm. 19 These differences prove the legacy effect of treatment with pravastatin during the randomized phase of the study.
The Long-Term Intervention with Pravastatin in Ischemic Disease trial in primary prevention added to the growing body of evidence that statins were associated with lower mortality in those with established ASCVD. 20 The study was terminated early after a mean duration of 6.1 years when a prespecified limit for the overall difference in mortality had been reached. During the randomized phase of the trial, for every 1000 patients treated with pravastatin, death, MI, or stroke was prevented in 47 patients compared to placebo. The patients were then followed for 2 additional years using a questionnaire. During this extended follow-up phase, this number increased to 58. Also, during a longer follow-up of 10 years, the pravastatin group maintained a significantly lower risk of death from coronary heart disease (RR 0.89; 95% CI, 0.81-0.97; P = .009), from CV disease (RR, 0.88; 95% CI, 0.81-0.95; P = .002), and from any cause (RR, 0.91; 95% CI, 0.85-0.97; absolute risk reduction, 2.6%; P = .003). During the follow-up phase, about 85% from each group were on statins. Therefore, the lower mortality in the group initially randomized to pravastatin is due to a legacy effect. 20
The lipid-lowering arm (LLA) of the hypertension trial Anglo-Scandinavian Cardiac Outcomes Trial found that administration of 10 mg atorvastatin was associated with a 36% reduction in the composite end point of nonfatal MI and fatal coronary events after 3.3 years as compared to placebo. At that time, the atorvastatin limb of the study was discontinued and all patients were prescribed 10 mg atorvastatin daily. After discontinuation of the LLA of the trial, patients were followed for a mean of 11 years postrandomization and at that time there was a reduction in death from CV causes by 15% (HR [atorvastatin/placebo] 0.85, CI, 0.72-0.99, P = .0395) proving the existence of a legacy effect. 21,22
The Legacy Effect of Non-Statin Cholesterol-Lowering Therapies
The Coronary Drug Project (CDP) addressed the efficacy and safety of conjugated estrogens, clofibrate, dextrothyroxine, and niacin as compared to placebo in men aged 30 to 64 years with an electrocardiogram-documented MI. 23 The trial on 8341 male patients was conducted from 1966 to 1975, with a mean follow-up of 6.2 years. The estrogen arms and the dextrothyroxine arms were discontinued early due to the increased incidence of adverse events. At the end of the randomized phase of the trial, niacin was associated with a modest benefit in decreasing nonfatal recurrent MI, but had no statistically significant effect on total mortality. In 1980, the CDP coordinating center systematically began following patients who were still alive at the conclusion of the randomized phase of the trial. With a mean follow-up of 15 years (6.2 years during the randomized phase of the trial plus 8.8 years after study conclusion), the mortality in the niacin treatment group was 11% lower compared to placebo (52% vs 58%; P = .0004) despite the lower adherence to niacin during the randomized treatment phase (30% of patients took less than 60% of the protocol stipulated dose of 3 g/d). 23 This legacy effect was more pronounced among patients with a serum cholesterol level above 250 mg/dL prior to initiation of niacin therapy (Figure 3). 23

Survival curves for niacin and placebo treatment groups in the Coronary Drug Project: At mean follow-up of 15 years, nearly 9 years after termination of the trial, mortality in the niacin group was 11% lower than in the placebo group. This delayed benefit occurring after discontinuation of niacin may be due to the early favorable effect of niacin in decreasing nonfatal reinfarction or a result of its cholesterol-lowering effect or both. Reprinted from reference 23.
The Helsinki trial randomized primary prevention patients with non-high-density lipoprotein cholesterol above 200 mg/dL to gemfibrozil or placebo. During the clinical trial with a follow-up of 5 years, 14 (0.68%) of the 2051 patients randomized to gemfibrozil died due to MI compared to placebo where 19 (0.94%) of the 2030 patients died due to coronary disease. This difference in the rates (RR 0.73) was not statistically significant. The difference became statistically significant at the 18-year follow-up, when 99 of 2046 in the active group and 128 of 2035 in the placebo group died due to coronary disease (adjusted RR 0.76, 95% CI, 0.59-0.99). The continuation of the lower coronary disease mortality during the long-term follow-up when all patients were equally likely to receive gemfibrozil implies the presence of a legacy effect. 24
The discovery of protease proprotein convertase subtilisin-kexin type 9 (PCSK-9) is a promising development in the field of lipidology. Proprotein convertase subtilisin-kexin type 9 monoclonal antibodies, when added to statin therapy, can further lower LDL-C levels by 60% or more. The FOURIER trial with the use of evolocumab in patients with atherosclerosis and the ODYSSEY OUTCOMES trial with the use of alirocumab in patients with recent acute coronary syndrome showed promising results. Robinson and associates reported that ongoing studies with PCSK-9 antibodies may determine whether a “legacy effect” will be observed with this class of agents. 25 Long-term outcomes of the trial participants in studies with these agents and whether there exists persistence of benefit with time remain to be seen.
Proposed Mechanisms of the Legacy Effect
The mechanism of how treatment with cholesterol-lowering therapy may provide long-term benefit is not well known. Statins have pleiotropic effects including improvement in endothelial dysfunction, antioxidant activity, increased bioavailability of nitric oxide, and stabilization of atherosclerotic plaques. 26,27
Metabolic Memory
Many gene variants have been associated with lower LDL-C levels, including loss-of-function mutations in the gene encoding PCSK-9. 28 Mendelian randomization studies have shown that these variants are associated with a correspondingly lower risk of coronary heart disease. This “metabolic memory” appears to confer a lifetime advantage. Prolonged exposure to lower LDL-C is associated with a 3 times greater reduction in the risk of CV disease per unit reduction in LDL-C, when compared to short-term LDL-C lowering after atherosclerosis has already developed. 29,30 This is expected since atherosclerosis is a chronic disease that begins early in life. 31
Effect of Telomeres
Stabilization of atherosclerotic plaques by statins may be related to their influence on telomere length. Telomeres determine cell senescence and apoptosis by shortening during each cell cycle until a particular threshold when cell death is triggered. The endothelium in atherosclerotic lesions exhibits senescence-associated phenotypes, 32 and shorter leukocyte telomeres are associated with a risk of coronary artery disease in the general population, independent of conventional risk factors. 33 Patients on statin therapy display higher telomere activity (P < .0001) and lower telomere erosion with aging compared to controls not on statins independent of multiple covariates including age, gender, smoking, inflammation, blood pressure, and lipid levels. 34 Among patients in the WOSCOPS study, pravastatin attenuated the decline of telomere length that correlates with a higher risk of coronary artery disease. 35 Whether this contributes to the legacy effect is yet to be proven.
Prevention of Nonfatal Events
The CDP investigators proposed 2 theories to explain the legacy effect of niacin. The first is that, since the incidence of nonfatal MI was higher in the placebo group during the randomized phase, there were more persons with impaired left ventricular function due to a prior MI in the placebo group indicating a legacy effect. During the randomized phase of the study, patients randomized to niacin were less likely to have a nonfatal MI, although there was no difference in mortality. However, at the 11-year follow-up, there was a lower rate of all-cause death in the niacin group.
Patients in the placebo group who sustained a nonfatal MI during the randomized phase were more likely to die from a second MI. We propose that this may be the reason for the higher mortality in the placebo group (Figure 3). 23
Prevention Slowing of Disease Progression
The results of the ATLAS trial imply that the rate of disease progression is a mechanism contributing to the legacy effect. The participants who received 10 years of treatment compared to 5 years of treatment for estrogen receptor-positive breast cancer with tamoxifen had lower rates of breast cancer recurrence, breast cancer mortality, and overall mortality. 2
Normalization of lipids with the use of statins has been shown to slow the progression of atherosclerosis. 36 Patients initially randomized to active therapy have a decreased accumulation of atheromatous burden, while atherosclerotic lesions in the placebo group may destabilize and cause an acute coronary event. Thus, it is expected that even when the rate of statin use becomes equivalent during follow-up, a long-term advantage is gained by those treated earlier. 19
There is a lag phase between the onset of pathophysiologic changes and the occurrence of clinical events. Thus, early administration of lipid-lowering agents may delay the formation and progression of atherosclerotic disease. 19 In the Atherosclerosis Risk in Communities study, the presence or absence of sequence variants in PCSK-9 that were associated with reduced plasma levels of LDL-C was associated with lower risk. The study showed that lifelong reduction in LDL-C resulted in a significant reduction in the incidence of coronary events. 37 As stated by Brown and Goldstein, 38 with respect to LDL lowering, it is not only how low it is but also for how long the cholesterol is lowered that is important.
Based in part on such studies, our group has proposed focusing on primordial prevention. This may provide a greater overall benefit although randomized trials are needed to assess the benefit compared to the risk of adverse and long-term medication side effects.
Disease Regression
Regression of the atheromatous plaque by lipid-lowering therapies among patients initially randomized to active therapy may also contribute to the persistence of benefit after the termination of the randomization phase. Plaque regression has been demonstrated to be an active, dynamic process. Administration of recombinant apoA-I Milano/1-palmitoyl-2-oleoyl phosphatidylcholine complexes (ETC-216) in rabbits showed a 6% reduction in magnetic resonance imaging and intravascular ultrasound measured plaque burden compared to placebo. 39 Similar mechanisms operating over a period of years may underlie the legacy effects observed with the use of statins.
Conclusion
To address concerns of a link between prolonged statin therapy and delayed onset of adverse events, investigators have followed participants after the end of the clinical trials. The need for early treatment with statins of patients with ASCVD and those at risk for this condition is supported by large clinical trials, meta-analyses, and clinical guidelines. The legacy effect has been observed in clinical trials of statins, niacin, and gemfibrozil. This review is based on randomized placebo-controlled clinical trials. They are essential for establishing the efficacy and safety of pharmacologic, and other, interventions, but have significant drawbacks. These include short-duration, selection bias, arbitrary noninferiority analyses, ambiguity in applying the overall findings to individual patients, the use of combined end points giving equal weight to events of different clinical importance (eg, death and angina), surrogate end points, and that they do not directly apply to the population for which the intervention will be used in practice. The end point of each study is usually decided after consideration of event rates, duration of the study, finances, availability of suitable patients, and other factors.
Primordial Prevention
The difficulty of physicians in deciding the appropriate course of action based on the results of clinical trials has been known for more than 2 millennia when Hippocrates stated in the Aphorisms, “Life is short, and art long, opportunity fleeting, experience misleading, and judgment difficult”(Ὁ βίος βραχύς, ἡ δὲ τέχνη μακρή, ὁ δὲ καιρὸς ὀξύς, ἡ δὲ πεῖρα σϕαλερή, ἡ δὲ κρίσις χαλεπή). 40 Hippocrates would have been helped in addressing this difficult judgment by Jean-Paul Sartre who in his lecture “Existentialism is a Humanism” (L’existentialisme est un humanism) stated that the existence precedes the essence (l’existence précède l’essence), and therefore we must decide our course of action (or inaction) in the presence of incomplete information. 41 In keeping with this concept, all those in charge of patients should act today and advise our patients appropriately. We should strive to prevent the occurrence of ASCVD rather than focus on its complications such as MI, angina, and sudden death. Poliomyelitis is not a feared disease at the present time because the cause is known and vaccines were developed. In the past, a complication of this disease (paralytic polio) was treated with iron lungs. The cause of ASCVD is also known. It does not occur when LDL-C is well controlled, for example, below 35 mg/dL from a young age. However, although the cause is known, instead of using primordial prevention, we insert iron stents. Neither iron lungs nor iron stents should be needed based on the current state of knowledge. In the absence of a clinical trial, testing the above hypothesis we should bring the cholesterol level of each patient as close to normal as possible considering individual circumstances, genetic, cultural, physiological, pathological, and psychological differences. The target of therapy should not be to postpone events, but to avoid the disease. Focusing on clinical events is not sufficient and investigators have advocated for novel methods of primordial prevention as early intervention may provide the greatest cumulative benefit. 42 As Brown and Goldstein 38 stated, “In lowering LDL levels, the appropriate consideration may not only be how low, but also how long.” Statin use from an early age to achieve very low LDL-C may confer long-term benefits if proven safe. 43
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
