Abstract
Aim:
To evaluate outcomes related to antiplatelet therapy in patients with ST-elevation myocardial infarction (STEMI) admitted to the San Gerardo Hospital in Monza, an extracorporeal membrane oxygenation (ECMO) reference center in the Monza-Brianza area.
Methods:
This retrospective study enrolled patients with STEMI hospitalized between 2013 and 2017.
Results:
This study included 653 patients (mean age: 67.5 years, 71% male). Across the study period, ticagrelor use showed consistent increases, from 22% of patients during 2013 to 85% in 2017. Cardiac arrest prehospitalization occurred in 100 patients (15.3%), either at home (n = 85, 13.0%) or during transfer (n = 15, 2.3%); 46 patients underwent ECMO for refractory cardiac arrest. Rates of 90-day survival (hazard ratio [HR]: 2.4, 95% confidence interval [CI]: 1.3-4.4, P = .004) and ST resolution (odds ratio [OR]: 2.5, 95% CI: 1.6-4.1, P = .000) were higher with ticagrelor than with other antiplatelet agents. When analyzed by each agent, patients on ticagrelor had longer survival (HR: 0.4, 95% CI: 0.2-0.8, P = .008) than patients on clopidogrel and more frequent ST resolution than those on clopidogrel or prasugrel (OR: 0.4, 95% CI: 0.2-0.7, P = .002 and OR: 0.4, 95% CI: 0.2-0.7, P = .006). There was no difference in mortality between ticagrelor and prasugrel.
Conclusions:
Changes in the treatment of high-risk patients with STEMI over time are in line with changes in treatment guidelines. In these patients, ticagrelor is associated with significantly improved 90-day mortality compared with clopidogrel.
Introduction
Cardiovascular disease is responsible for 3.9 million deaths a year in Europe, accounting for 45% of all deaths in this area. 1 In Europe, the incidence of ST-elevation myocardial infarction (STEMI) has been reported to be 43 to 144 per 100 000 people per year. 2,3 Studies investigating trends in mortality have shown that for myocardial infarction in general, and STEMI in particular, mortality rates have fallen over time. 4 -9 Despite this decrease, in-hospital mortality in patients with STEMI is still high, with overall rates reported to be 4% to 12%, 10 and 1-year mortality being approximately 10%. 11,12
Factors related to mortality in patients with STEMI include patient-, disease-, and treatment-related characteristics. Patient-related characteristics include age, sex, diabetes mellitus, and history of cardiovascular disease. 4,13 -15 Disease-related factors include Killip class and cardiac function. 13,15 Treatment-related changes include increased use of reperfusion therapy and primary angioplasty, development and use of new antithrombotic therapies, the increasing use of mechanical circulatory support (such as extracorporeal membrane oxygenation [ECMO] or Impella ventricular assist devices), and secondary prevention strategies. 4,8
The San Gerardo Hospital in Monza is the reference center in the Monza-Brianza area for the treatment of cardiac arrest using ECMO, which is a procedure that can be used to achieve revascularization and circulatory support in patients with cardiac arrest and STEMI who do not respond to standard resuscitation procedures. The aim of this study was to retrospectively determine outcomes related to antiplatelet therapy in the whole population of patients with STEMI admitted to the San Gerardo Hospital.
Methods
Study Design and Participants
Data from patients with STEMI hospitalized from January 1, 2013, to March 18, 2017, at the San Gerardo di Monza Hospital were collected and included in this retrospective analysis. All patients admitted with a diagnosis of STEMI for whom it was possible to collect data from the computerized medical records of the hospital were included in the full analysis, while only patients revascularized with percutaneous coronary intervention (PCI) and stent implantation were included in the primary end point analysis. Data were collected from medical records and entered into an anonymous database for statistical analysis.
The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines as well as all applicable Italian laws. Personal details of patients were collected anonymously and processed in compliance with Italian laws. Due to the retrospective nature of the study, no written informed consent was required.
Outcomes
Patient demographics and treatment patterns were assessed in all patients. The primary end point of the analysis was to compare mortality at 90 days between patients treated with ticagrelor and patients given another P2Y12 inhibitor (clopidogrel or prasugrel). A secondary end point was to compare the number of patients with ST resolution (described as 50% ST-segment resolution at 90 minutes after PCI execution) with ticagrelor compared with patients given another P2Y12 inhibitor. Finally, an exploratory data analysis was conducted to compare mortality and ST resolution rates among patients treated with ticagrelor compared with patients who received clopidogrel and patients who received prasugrel.
Statistical Analysis
All statistical analyses were performed using SPSS Statistics software (IBM, Armonk, New York). Descriptive statistics were calculated for continuous variables and included number of patients, mean, standard deviation (SD), and 95% confidence intervals (CIs). Frequencies and percentages were calculated for noncontinuous (categorical) variables. To assess population characteristics, χ2 tests and independent samples t tests were used for noncontinuous and continuous variables, respectively. Multivariate linear regression models and logistic regression were performed to assess any correlations among population characteristics and in-hospital mortality, while Cox regression was performed to check the variables related to 90-day mortality; significance was defined as P < .05. A propensity score analysis was also conducted using multivariate logistic regression analysis with baseline covariates as the predictors and ticagrelor versus no ticagrelor as the dependent variable. The 21 baseline covariates used were age, gender, Killip class, systolic blood pressure, ejection fraction, prior cardiac arrest, time to reperfusion (defined as door to balloon time), the left main coronary artery as the culprit vessel, diabetes status, dyslipidemia status, hypertension status, smoking habits, cardiovascular disease status, number of vessels affected, baseline creatinine, heparin treatment, bivalirudin treatment, glycoprotein IIb/IIIa inhibitor treatment, ST resolution, Thrombolysis in Myocardial Infarction (TIMI) flow status post-PCI, and treatment with a drug-eluting stent (DES) or bare metal stent. Thrombolysis in Myocardial Infarction flow status post-PCI was determined from patient angiogram data, which were analyzed by 2 reviewers in a blinded fashion.
Results
Patient Demographics
A total of 653 patients were included in the retrospective analysis between January 2013 and March 2017. The whole population had a mean ± SD age of 67.5 ± 12.8 years, and most patients (71%) were male (Table 1). One hundred patients (15.3%) had experienced a cardiac arrest prehospitalization, either at home (n = 85, 13.0%) or during transfer (n = 15, 2.3%); 46 patients were supported with ECMO for refractory cardiac arrest. In the overall population, 35% of patients had a Killip class >I at baseline (class II, n = 81; class III, n = 53; class IV, n = 97; Table 1).
Baseline Characteristics.

Abbreviations: EF, ejection fraction; HR, heart rate; PCI, percutaneous coronary intervention; SBP, systolic blood pressure.
a Values are presented as mean ± standard deviation unless otherwise stated.
b n = 638.
c n = 635.
d n = 641.
In total, 532 patients who were treated with PCI and with complete data available were considered for the outcomes analysis, 292 in the ticagrelor treatment group and 240 in the other P2Y12 group. Among the 240 patients who were treated with other P2Y12 inhibitors, 141 (58%) patients received clopidogrel and 99 (42%) patients received prasugrel.
Patients Characteristics by Treatment Group
Pre-PCI heparin, bivalirudin, or glycoprotein IIb/IIIa inhibitor use was significantly less common in patients treated with ticagrelor than in those who received clopidogrel or prasugrel, as significantly fewer patients had hypertension (60.6% vs 71.3%, P = .010; Supplementary Table 1). Pre-PCI TIMI scores were similar between patients treated with ticagrelor and those treated with other P2Y12 inhibitors; however, post-PCI TIMI 3 scores were reported significantly more often with ticagrelor than clopidogrel or prasugrel treatment (P < .001; Supplementary Table 1).
Ticagrelor use was associated with significantly higher rates of post-PCI ST resolution than clopidogrel or prasugrel (P = .006; Supplementary Table 1). Among patients who received ticagrelor, significantly more patients had a pre-PCI cardiac arrest compared with patients who received clopidogrel or prasugrel (13.0% vs 7.5%, P = .039). In the ECMO group, 40 patients were treated with successful PCI with stent deployment: 20 patients in the ticagrelor group and 20 in the other P2Y12 inhibitor group.
Patterns in Treatment Over Time
Across the study period, the use of ticagrelor showed consistent increases, from 22% of patients during 2013 to 85% of patients in 2017 (Figure 1). In contrast, the use of both clopidogrel and prasugrel steadily decreased between 2013 and 2017 and the use of glycoprotein IIb/IIIa inhibitor therapy remained generally consistent during the study period. Irrespective of the treatment received, the mean time to reperfusion increased between 2013 and 2015 hospitalizations but significantly decreased in 2016 and 2017 hospitalizations (Figure 2).
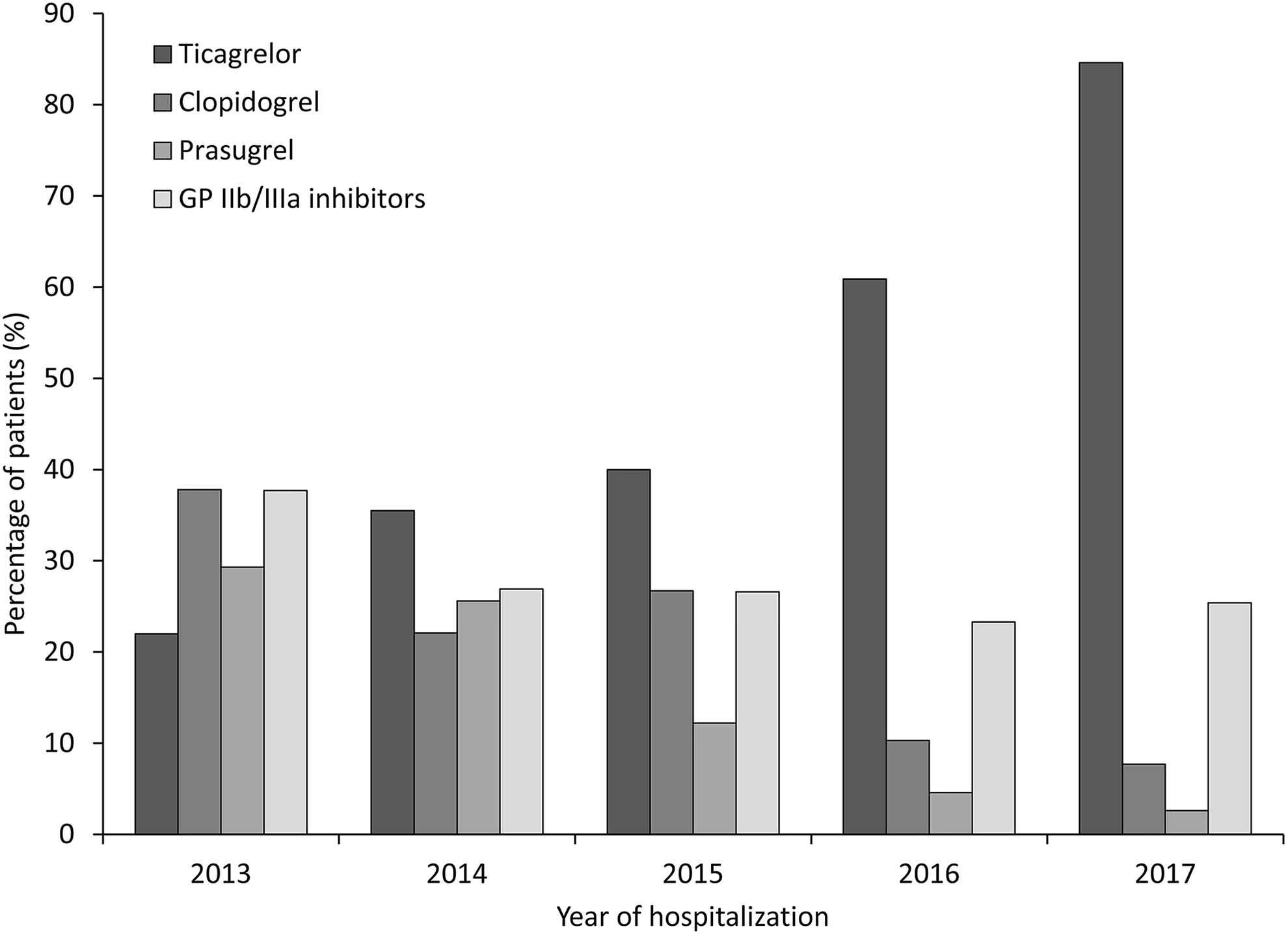
Use of ticagrelor, clopidogrel, prasugrel, and glycoprotein (GP) IIb/IIIa inhibitors by year of hospitalization.

Time to reperfusion by year of hospitalization. CI, confidence interval.
Outcomes
Based on binary logistic regression analysis, 90-day mortality was significantly correlated with age, Killip class III or IV, pre-PCI ejection fraction, pre-PCI cardiac arrest, and time to reperfusion. The left main artery as the culprit vessel was also significantly correlated with 90-day mortality. The year of hospitalization and pre-PCI systolic blood pressure were not significantly correlated with 90-day mortality rates.
Based on propensity score analysis, the 90-day survival rates were significantly higher with ticagrelor (n = 219) than with other antiplatelet agents (n = 200; hazard ratio [HR]: 2.4, 95% CI: 1.3-4.4, P = .004; Figure 3). This pattern of improved survival with ticagrelor versus other antiplatelet agents was consistent across all propensity score quintiles (Supplementary Table 2). In a Cox regression landmark analysis of in-hospital mortality, survival rates were significantly higher with ticagrelor versus other antiplatelet agents after 10 days of hospitalization (HR: 2.4, 95% CI: 1.2-4.7, P = .009; Supplementary Figure 1A) and 30 days of hospitalization (HR: 2.6, 95% CI: 1.4-4.9, P = .003; Supplementary Figure 1B). Similarly, based on binary logistic regression analysis, ST resolution was also more frequent with ticagrelor than with the other antiplatelet agents (79.0% vs 63.5% of patients, odds ratio [OR]: 2.5, 95% CI: 1.6-4.1, P = .000).

Kaplan-Meier curve of 90-day cumulative survival with ticagrelor (n = 219) versus no ticagrelor (n = 200), based on propensity score-matched data.
As far as the exploratory analysis is concerned, patients treated with ticagrelor (n = 217) had significantly longer survival (HR: 0.363, 95% CI: 0.172-0.767, P = .008; Figure 4A) and more frequent ST resolution (79.3% vs 60.0% of patients, OR: 0.387, 95% CI: 0.209-0.714, P = .002) than patients treated with clopidogrel (n = 80); in addition, patients treated with ticagrelor had more frequent ST resolution than patients treated with prasugrel (n = 75; 79.3% vs 69.3% of patients, OR: 0.367, 95% CI: 0.181-0.745, P = .006). There was no difference in mortality between the ticagrelor group and the prasugrel group (HR: 0.194, 95% CI: 0.025-1.480, P = .114; Figure 4B).
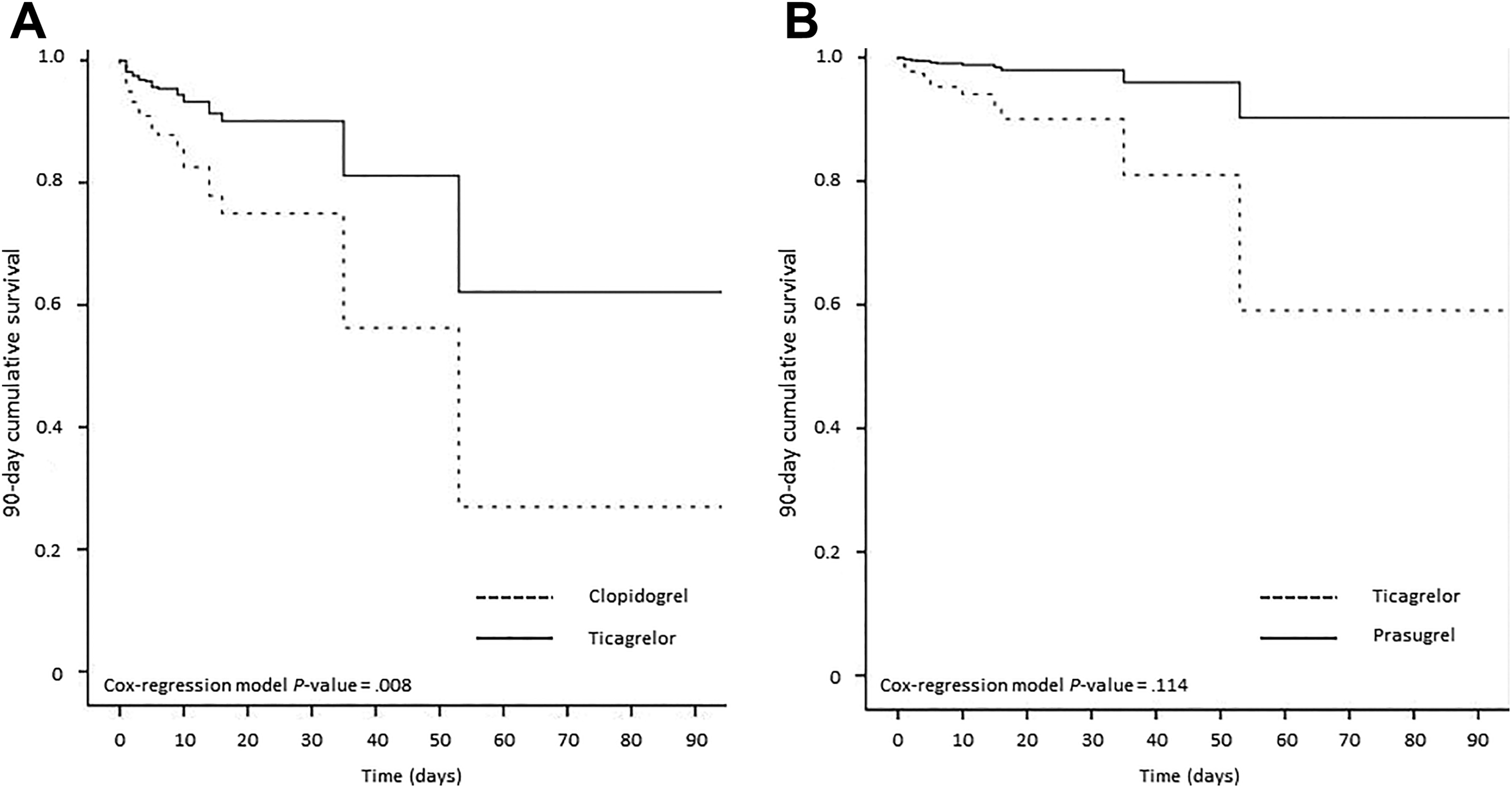
Kaplan-Meier curve of 90-day cumulative survival with (A) ticagrelor (n = 217) versus clopidogrel (n = 80) and (B) ticagrelor versus prasugrel (n = 75), based on propensity score-matched data.
Discussion
This retrospective analysis of a real-world population of patients with STEMI who presented at an ECMO center in Italy highlights the trends in the treatment and outcomes of these patients over the last 5 years. Dual antiplatelet therapy (DAPT) is the mainstay of treatment for patients with acute coronary syndrome (ACS) presenting with STEMI. Over the past 5 years, international guidelines have shifted toward using more potent P2Y12 inhibitors, such as prasugrel or ticagrelor rather than clopidogrel for DAPT. 16 -18 In line with these changes in guidelines, the use of clopidogrel at our center has decreased steadily over the years. The decrease in the use of prasugrel may have occurred because of contraindications (eg, age ≥75 years, weight <60 kg, and a history of stroke or transient ischemic attack) 19 that limit its use in the context of acute myocardial infarction when patient characteristics or history are not always known. The reduction in the use of clopidogrel and prasugrel was also associated with a decrease in the use of pre-PCI heparin, bivalirudin, or glycoprotein IIb/IIIa inhibitors. The current guidelines for STEMI do not highlight these treatments, so this reduction in use is not unexpected. 17,18
The change in guideline recommendations to use more potent P2Y12 inhibitors is based largely on the results of the ST-elevation ACS in the Platelet Inhibition and Patient Outcomes (PLATO) 20 and the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction (TRITON-TIMI) 38 studies. 19 The PLATO study assessed the use of pharmacotherapy alone or with coronary revascularization with PCI or bypass surgery in moderate-to-high risk patients with ACS and demonstrated that ticagrelor was associated with better outcomes than clopidogrel. 20 The TRITON-TIMI 38 study showed that prasugrel significantly reduced the risk of ischemic events in moderate-to-high risk patients with ACS undergoing PCI compared with clopidogrel, but with an increased major bleeding risk. 19 The results of our analysis are in line with those of the PLATO study and indicated that ticagrelor use in patients with STEMI reduced mortality. However, in our study, the results were seen in a real-world high-risk population, comprising 100 patients with cardiac arrest, 46 of them supported by venous-arterial ECMO when the resuscitation efforts were unsuccessful. This patient group is generally excluded from randomized controlled trials. Moreover, in this study, the use of ticagrelor was associated with more frequent ST resolution after PCI.
It is important to note that the optimal DAPT in high-risk patients with ACS who require PCI is the subject of ongoing discussion, mostly due to the results of recent real-world studies, such as CHANGE-DAPT 21 and other studies. 22 -26 The CHANGE-DAPT study showed that patients with ACS who were treated with a newer generation DES and DAPT with ticagrelor had a significantly higher risk for the composite outcome of all-cause death, any myocardial infarction, stroke, or major bleeding than patients receiving clopidogrel-based DAPT (7.8% vs 5.1%, P = .02). 21 While the results of the current analysis contradict CHANGE-DAPT, this could be due to at least 2 reasons. Firstly, CHANGE-DAPT included patients with all types of ACS, including those with non-STEMI or unstable angina. Secondly, there was a difference in the methods used between CHANGE-DAPT and in our analysis, mainly that CHANGE-DAPT assessed 2 consecutive treatment periods to compare clopidogrel (December 2012 to April 2014) and ticagrelor (May 2014 to August 2015) with a noteworthy overlap between the 2 periods. Indeed 13% of patients were treated with ticagrelor in the “clopidogrel period” and in the same way 15% of patient were treated with clopidogrel in the “ticagrelor period.” Finally, the end point used in CHANGE-DAPT included a bleeding component, which was not included in our analysis. Further research is warranted to determine what impact the results of CHANGE-DAPT will have on outcomes and prescribing patterns.
Moreover, in patients with STEMI and cardiac arrest, it is not clear which antiplatelet strategy is best. Although studies have shown that ticagrelor or prasugrel (but not clopidogrel) can provide effective platelet inhibition in patients with STEMI and cardiac arrest, 27,28 a meta-analysis has indicated that there is no difference between clopidogrel and ticagrelor or prasugrel in the risk of stent thrombosis or in-hospital mortality in these patients. 29
The novelty of this analysis was that we investigated outcomes according to antiplatelet therapy in a real-world group of patients with STEMI, including a considerable number of patients with cardiac arrest, as well as those with refractory cardiac arrest who were supported with ECMO—a patient population that has thus far been excluded from studies. Cardiac arrest and cardiogenic shock are known to alter the pharmacokinetics of therapies as these conditions alter gastric emptying, impact nasogastric suction, and can reduce drug absorption. 30,31 It is possible that ticagrelor, and to a lesser extent prasugrel, may be more effective in these patients due to their differing pharmacokinetic profiles from clopidogrel. 32 As clopidogrel undergoes 2 enzymatic steps for conversion to its active form, only 15% of the administered drug is activated. Furthermore, variability in enzymatic ability between patients results in inadequate antiplatelet response to clopidogrel in 30% of patients. 33 Prasugrel requires one enzymatic step for metabolic activation, which occurs at the time of absorption. 34 In contrast, ticagrelor is a directly active substance with rapid platelet inhibition observed immediately after intestinal absorption. 20,35 This may also be the reason for more frequent ST resolution with ticagrelor compared with other P2Y12 inhibitors observed in this analysis.
Because of the exploratory nature of this analysis and limited sample size between ticagrelor, clopidogrel, and prasugrel, we cannot draw definitive conclusions. Our data support the results of several randomized clinical trials that have shown the superiority of ticagrelor in both mortality and ST resolution compared with clopidogrel in patients with STEMI. 36 -39 However, although the increased use of ticagrelor in recent years was associated with improvements in survival, other factors, such as changes in clinical practice resulting in reductions in time to perfusion and improved support techniques, may have also contributed to the observed decrease in 90-day mortality rates over time. The more frequent ST resolution with ticagrelor compared with prasugrel in a high-risk STEMI population found in this analysis is also interesting in speculative terms. As ST resolution is an event observed immediately after PCI, this raises the question as to whether, in the more critical patients, there may be a pharmacokinetic characteristic of ticagrelor that has a positive effect on reperfusion during PCI compared with prasugrel. 40 Given the advantage observed with ticagrelor within a few weeks in our high-risk STEMI population, and considering that high-risk patients are the group of patients that may obtain the greater advantage of prolonged (more than 12 months) DAPT, such treatment can reasonably be considered as the first phase of a long-term therapeutic strategy. This topic warrants further investigation.
There are some limitations to this analysis that are inherent to any retrospective analysis of a heterogeneous patient population, most importantly selection bias; while propensity score-adjusted multivariate analyses were performed to adjust for potential confounders, residual confounding cannot be excluded. In addition, we decided not to analyze bleeding/safety data, but only mortality and ST resolution data. Despite this study limitation, the reduction in mortality correlated with ticagrelor use could mean that, irrespective of bleeding/safety profile, ticagrelor treatment positively impacts the mortality rate.
In conclusion, changes in the treatment of these high-risk patients with STEMI over the years at our treatment center are in line with changes in treatment guidelines. Taken together with the results of previous randomized controlled trials, the findings from our patient population indicate, despite the small number of patients and high incidence of cardiac arrest, that ticagrelor is associated with improved survival compared with other available antiplatelet therapies.
Supplemental Material
Supplemental Material, Righetti_et_al_Supplementary_materials - Outcomes Related to Antiplatelet Therapy in a High-Risk ST-Segment Elevation Myocardial Infarction Population: A Retrospective Real-World Analysis of an Italian ECMO Center
Supplemental Material, Righetti_et_al_Supplementary_materials for Outcomes Related to Antiplatelet Therapy in a High-Risk ST-Segment Elevation Myocardial Infarction Population: A Retrospective Real-World Analysis of an Italian ECMO Center by Stefano Righetti, Elisabetta Montemerlo, Federica Soffici, Davide Sala, Alessandro Bozzano, Andrea Mauro, Elena Maggioni, Leonello Avalli, Paola Camisasca, Virgilio Colombo, Felice Achilli, Maddalena Lettino and Ivan Calchera in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
The authors certify that all data generated are present in the manuscript. The study was conducted at The San Gerardo di Monza Hospital, Monza, Italy.
Acknowledgments
The authors would like to thank Simone Tait of Springer Healthcare Communications who wrote the outline and subsequent drafts of this manuscript; Sarah Greig, PhD, of Springer Healthcare Communications who assisted with manuscript revisions post submission; Samuele Zago for intellectual and writing assistance; and Giorgio Reggiardo of Medi Service Genova for statistical analysis.
Author Contributions
All authors were involved in and contributed equally to the study design, enrollment of patients, analysis of data, and the preparation of manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing assistance and statistical analysis were funded by AstraZeneca, Italy.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
