Abstract
Dual antiplatelet therapy with aspirin and a P2Y12 receptor antagonist remains a mainstay in the prevention of ischemic events following coronary stent placement. Significant controversy exists regarding the optimal management of high platelet reactivity despite antiplatelet therapy; however this finding has been consistently associated with poor clinical outcomes including greater risk of stent thrombosis and myocardial infarction. Variability in antiplatelet effects of clopidogrel and prasugrel has been linked to genetic polymorphisms and potential drug–drug interactions. Both of these factors have significant influence on the cytochrome P-450 enzyme system activity of the liver responsible for their biotransformation to the active form of both drugs. Very little has been publicized regarding differences in antiplatelet effects which may be associated with conditions in which the functional capacity of the liver may be temporarily compromised. Patients who present with cardiogenic shock due to acute coronary syndromes have evidence of multiorgan dysfunction including liver dysfunction that may affect the activity of these drugs. This review aims to explore existing evidence and propose additional considerations to the selection of antiplatelet therapy in patients with cardiogenic shock who receive catheter-based revascularization and stent placement.
Introduction
Ischemic coronary artery disease continues to be a cause of significant morbidity and mortality, with the overall prevalence increasing in the past decades. Cardiogenic shock as a complication of myocardial infarction occurs in about 5% to 10% of cases and is associated with a short-term mortality that has been reported between 40% and 70%. 1 –5 Many of these patients are managed with urgent or emergent revascularization in the cardiac catheterization laboratory and subsequently are initiated on guideline-based dual antiplatelet therapy with aspirin and a P2Y12 antagonist following percutaneous coronary intervention (PCI) with coronary stent placement. 6,7 Available P2Y12 receptor antagonists include clopidogrel, prasugrel, and ticagrelor, each with its own unique pharmacologic and pharmacokinetic (PK) characteristics. Although the American guidelines for ST-segment elevation myocardial infarction (STEMI) managed with PCI equally recommend any of the 3 available P2Y12 receptor antagonists, the European guidelines published by the European Society of Cardiology favor prasugrel or ticagrelor in this patient population. 6 –8 Both clopidogrel and prasugrel are prodrugs, requiring biotransformation to reach their respective active forms (Figure 1). For clopidogrel, 2 necessary biotransformation steps are mediated by the cytochrome P450 isoenzyme system in the liver while prasugrel undergoes initial esterase-mediated hydrolysis before undergoing a necessary second activation step within the liver. 9 Ticagrelor is a nonthienopyridine P2Y12 receptor antagonist which is already in its active form as the administered drug. In patients having cardiogenic shock, there is a theoretical risk that the metabolic activation of prodrugs may become impaired, leading to treatment failures. 10 Additionally, it is also likely that in a state of cardiogenic shock, absorption of medications may be compromised. 10 The following review aims to explore available evidence of this potential drug–disease interaction and to guide the optimal selection of antiplatelet therapy in patients having cardiogenic shock following PCI.

Bioactivation processes of P2Y12 receptor antagonists (reproduced with copyright permission from Schattauer Publishers).
Implications of High On-Treatment Platelet Reactivity
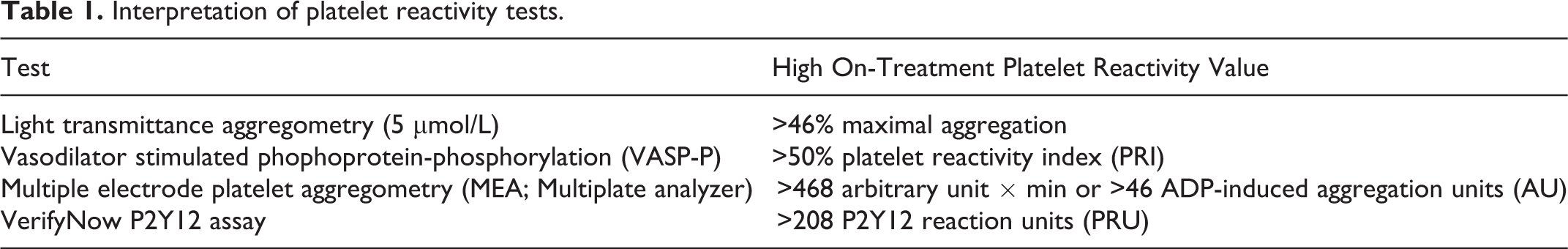
Numerous pharmacodynamic laboratory assessment methods are available to quantify the antiplatelet effects of P2Y12 receptor antagonists. Studies of these laboratory tests demonstrate a consistent association with negative clinical outcomes including stent thrombosis and myocardial infarction when high on-treatment platelet reactivity is detected. 11 –16 An expert consensus published in the Journal of American College of Cardiology describes the available platelet reactivity tests and establishes interpretative ranges for several of the studies including the 5µmol/L light transmittance aggregometry, Vasodilator stimulated phophoprotein-phosphorylation (VASP-P) analysis, Multiplate analyzer, and the VerifyNow P2Y12 assay. 16 The thresholds above which patients would be considered to have high on-treatment platelet reactivity are displayed in Table 1. The controversy surrounding any of these tests, which is beyond the scope of this review, has not been whether their results are correlated to outcomes, but rather how a clinician may modify antiplatelet therapy once a result is realized to improve the outcomes of patients with high on-treatment platelet reactivity.
Interpretation of platelet reactivity tests.
Recently, major factors affecting absorption and drug metabolism of clopidogrel have raised significant concern of clinicians faced with the challenge of choosing optimal antiplatelet therapy for patients undergoing PCI. Clinical characteristics of patients including large body mass index, advanced age, and presence of diabetes mellitus have been associated with suboptimal platelet inhibition by clopidogrel. 17 Significant controversy emerged regarding the use of proton pump inhibitors concomitantly with clopidogrel. Concurrent proton pump inhibitor therapy has been associated with less platelet inhibition by clopidogrel, possibly due to inhibition of the CYP2C19 isoenzyme system; however, there was not an increased cardiovascular event rate observed when omeprazole was studied compared to placebo in clopidogrel-treated patients with recent acute coronary syndrome (ACS) or PCI. 18 –21 A consensus on the matter issued by the American College of Cardiology Foundation (ACCF)/American College of Gastroenterology (ACG)/American Heart Association (AHA) in 2010 acknowledges this available evidence and suggests that the clinician choose to use proton pump inhibitor therapy in patients with clopidogrel based on assessment of the benefits of reducing gastrointestinal (GI) bleed risk to the theoretical risks of reducing the efficacy of antiplatelet therapy. 22 Genetic factors have also been found to play a role in high on-treatment platelet reactivity in patients treated with clopidogrel. Genetic polymorphisms to genes coding for the CYP 2C19 isoenzyme system, including the CYP 2C19*2 polymorphism, have repeatedly been linked to an increased risk of clopidogrel hyporesponsiveness and an increased incidence of cardiovascular events. 23 –25 Additionally the genetic coding related to variability of the ABCB1 genotype was identified to affect the activity of the p-glycoprotein transporter in the intestines, conferring a possible reduction to the absorption of clopidogrel. 25 This association was further supported by a study identifying genetic variants as risk factors for cardiovascular events in patients taking clopidogrel. 25
The association of high on-treatment platelet reactivity with negative clinical outcomes has led investigators to explore methods of modifying antiplatelet therapies in response to these laboratory results, of which the most robust studies have been unsuccessful to date. 26 –28 It remains clear that high on-treatment platelet reactivity persists as a risk factor for events including stent thrombosis; however, waiting to assess platelet reactivity may delay appropriate therapy and put patients at risk of worse clinical outcomes. Both prasugrel and ticagrelor when compared to clopidogrel were shown to reduce cardiovascular events in patients with ACS who undergo PCI, and both have demonstrated a more potent degree of platelet inhibition when studied in a randomized trial setting. 29 –32
Physiologic Changes in Patients with Cardiogenic Shock
It is currently unknown how the effects of cardiogenic shock with subsequent vital organ derangement may impact individual PKs and pharmacodynamics. The PK parameters most likely affected by impaired organs include absorption, distribution, metabolism, and excretion.
Several studies have shown that during states of hypotension or shock, the body will naturally shunt blood to vital organs and limit blood flow to the GI tract which is important for drug absorption. 33,34 Unfortunately, studies validating these principles are limited and conflicting. Tarling et al was able to demonstrate increased paracetamol absorption for patients not receiving dopamine compared to those receiving dopamine based on area under the curve (AUC) analysis. 35 However, Ariano et al showed there was no difference in enteric absorption and PK of oseltamivir in critically ill patients with suspected or confirmed influenza A in 2009. 36 Rate and extent of drug absorption may be impaired by GI mucosal edema associated with elevated venous pressures. Certain chemical characteristics of drugs exist to optimize the rate and extent of drug absorption including molecular size, solubility, and lipophilicity which remain relatively unaltered in varying disease states. 33,37 The changing entity which alters drug absorption lies in the various environmental factors (eg, pH, blood flow, surface area, and GI motility) which can significantly alter the balance that has been established in healthier individuals. 33,37 A review of the effects of cardiac surgery on drug absorption by Pea et al demonstrated that drug absorption may be erratic and unpredictable during the early postoperative period. 38 Although this was a different patient population than described in this review, the 2 groups share similar characteristics which both may alter drug absorption.
Following absorption across the GI tract, medications are distributed to the appropriate body tissues and receptors to elicit their pharmacologic activities. Distribution is a complex process influenced by several different factors including blood flow to tissues, protein binding of the medications, solubility of the drug, environmental pH, and permeability into the tissues. 33 Alterations in receptors and transporters may be expected due to physiologic changes as seen with decreased responses of catecholamines to their receptors in an acidic environment.
The liver is the primary organ responsible for the metabolism and effective clearance of many drugs introduced into the body. Biologic activation or deactivation processes are the foundation for drug metabolism, with the cytochrome P-450 enzyme system being the predominate mediator. Even in healthy individuals, genetic alterations arising from cytochrome P-450 enzymes can lead to a wide array of pharmacodynamic responses to medications. When these influences are compounded by impaired liver function due to hepatic congestion and hypoperfusion, as seen in patients with cardiogenic shock, drug metabolism becomes less predictable, resulting in potential serious adverse events due to overexposure or underexposure to effective drug.
Acute cardiogenic liver injury (ACLI) has been commonly attributed to hypoperfusion alone leading to hepatocyte atrophy. Several reports have countered this theory, indicating that hypotension alone does not induce acute liver injury. 39,40 Several studies evaluating the etiology of ACLI indicate that a majority of cases do not result from a single hemodynamic insult but are likely associated with a combination of hepatic congestion due to elevated venous pressures and impaired perfusion. 39,41,42 It is unknown the significance of ACLI on liver metabolism but several alterations to normal function would be expected including decreased hepatocyte function (activity of metabolic enzymes and transporters), alterations in blood supplied to the liver, and reduced synthesis of plasma protein involved in the protein binding of drugs. 43,44
Following drug metabolism, medications are excreted from the body in their active or inactive form based on individual PK properties. Although the liver helps clear some medications through metabolic inactivation, the majority of medications are eliminated through the kidneys. Clearance of medications with low hepatic extraction is not influenced by blood flow or amount of medication transported to the liver but by the ability of metabolizing enzyme. Inability of the enzymes to effectively metabolize medications will result in an increased AUC as well as a prolongation of the drug half-life (t1/2). High extraction medications are cleared independent of protein binding and almost solely based on hepatic blood flow. Even if medications are not bound to proteins due to hypoproteinemia, the liver will be able to effectively clear the medication. 44 Impaired kidney function becomes vitally important for medications that are eliminated in their active form, or if metabolites retain metabolic activity. Significant adverse effects can result with these medications if they are not appropriately dose adjusted.
As hepatic drug metabolism is based on blood flow, protein binding, and enzyme activity, it becomes evident how ACLI may influence this in patients with cardiogenic shock. With these things considered, patients with cardiogenic shock may not demonstrate the same response to medications as would be expected in hemodynamically stable patients with ACS. Of particular interest to the variability of oral P2Y12 receptor antagonist effects are the possible alterations in gastrointestinal absorption, variability in cytochrome P-450 enzyme metabolism, and alterations in platelet activation caused by other biological processes.
Pharmacodynamic Assessment of Antiplatelet Agents in Cardiogenic Shock
Several small studies and case reports have been published using different methods, but all seem to suggest the possibility of cardiogenic shock as a negative influence on the antiplatelet effects of clopidogrel. 45 –53 Limitations of these studies include differing definitions of cardiogenic shock or hemodynamic instability, confounding causes of clopidogrel ineffectiveness including therapeutic hypothermia, different platelet function tests utilized, varying study methods which are observational or retrospective in nature, and limited sample size.
A study in 2010 evaluated the platelet reactivity index (PRI) by VASP-P analysis in hemodynamically unstable patients with STEMI treated with clopidogrel compared to stable patients. 45 In this study, the unstable patients were required to be both mechanically ventilated and on catecholamine agents for hemodynamic support. Platelet reactivity index was assessed prospectively at 4 hours, 24 hours, and 2 to 4 days after clopidogrel initiation which included an initial loading dose of 600 mg. A patient with a PRI greater than 53% at 24 hours was considered to be a nonresponder. Platelet reactivity index did not differ between the groups at baseline or 4 hours postinitiation of clopidogrel; however, PRI showed significantly less platelet inhibition in the unstable group at 24 hours and 2 to 4 day assessment times. A PRI value above 53% was detected in 7 (35%) of 20 stable patients and 17 (85%) of 20 unstable patients (P = .003). Clinical outcomes were followed up to 6 months postenrollment. Mortality was expectedly higher in the unstable group with 9 (42%) of 20 expiring within the evaluation period. Of these 20 unstable patients, 2 (10%) deaths were attributed to stent thrombosis (one definite and one probable according to authors based on the Academic Research Consortium definition for stent thrombosis). There were no deaths or stent thrombosis reported in the stable patient group. This study did not report bleeding outcomes in either stable or unstable patients. This study was the earliest report describing a possible difference in antiplatelet effects of clopidogrel in a hemodynamically unstable or cardiogenic shock population to that of a stable patient with myocardial infarction.
Results of a study presented at the Transcatheter Cardiovascular Therapeutics conference in 2011 evaluated the antiplatelet effects of clopidogrel in 237 patients with STEMI, 30 of which were considered by the investigators to be having cardiogenic shock. 46 Their definition for cardiogenic shock was hemodynamic instability with the need for catecholamine drug administration or mechanical ventilation. Patients routinely received 600 mg loading doses of clopidogrel. The investigators used the Multiplate analyzer as their test for platelet reactivity and showed a significant difference in median responses of 643 AU*min in the cardiogenic shock group compared to 492 AU*min in the noncardiogenic shock group (P = .04). They also suggest that platelet inhibition after switching to prasugrel in these patients was highly effective, although they did not release these specific results in the published abstract. There were no clinical outcomes reported within the abstract.
Two separate case reports were published, highlighting specific cases where clopidogrel and prasugrel antiplatelet effects were limited or delayed in patients with cardiogenic shock. 47,48 The first case reviewed a patient with STEMI who had ventricular fibrillation arrest and subsequent cardiogenic shock. 47 The patient received clopidogrel 600 mg and subsequently received prasugrel 60 mg prior to PCI. Multiplate analyzer values 4 and 6 hours later were reported to be 921 and 661 AU*min, respectively. The patient was given an additional 60 mg prasugrel and finally achieved an acceptable Multiplate analyzer value of 196 AU*min over 8 hours after initial thienopyridine loading. The other case report also involved a patient with STEMI who had a cardiac arrest, was brought to the catheterization laboratory after successful resuscitation, and was found to have a subtotal occlusion of the left anterior descending coronary artery. 48 According to their report, the patient received a loading dose of 600 mg clopidogrel prior to stent placement and intra-aortic balloon pump insertion. Multiplate analyzer 1 hour after receiving the loading dose showed a value of 1478 AU*min. This patient was subsequently loaded with 60 mg of prasugrel and the Multiplate analyzer result decreased to 443 AU*min. The report goes on to describe this patient later having an episode of acute stent thrombosis, which required thrombus aspiration and the administration of intravenous abciximab and conversion of prasugrel to ticagrelor maintenance antiplatelet therapy. Multiplate analyzer results 6 and 9 days after the ticagrelor initiation showed platelet activity of 242 and 278 AU*min indicating a much more adequate antiplatelet effect. Both of these cases highlight profound clopidogrel ineffectiveness and questionable prasugrel effectiveness in the patient with cardiogenic shock. The second case describes a possible benefit of ticagrelor in this setting, but it should be mentioned that platelet function studies were assessed much later and also after the patient had received the GP IIb/IIIa inhibitor abciximab. Because case reports represent such small numbers or individual responses, caution should be taken in the interpretation of these reports on this subject.
A recent study published in 2014 reporting an analysis of the ISAR-SHOCK (Intracoronary stenting and antithrombotic regiman–cardiogenic shock) registry included 145 patients with acute myocardial infarction having cardiogenic shock undergoing primary PCI. 49 This study was not randomized and included a significant portion of patients initially receiving clopidogrel (137 of 145, 94%) with the remaining initially receiving prasugrel (8 of 145, 6%). Multiplate analyzer results were reported following the initial thienopyridine loading doses, with a median of 5.8 hours between initial dosage and laboratory sample collection. High on-treatment platelet reactivity was observed in 58 (42.3%) of 137 clopidogrel-treated patients and 3 (37.5%) of 8 prasugrel-treated patients. Forty-two clopidogrel patients were then switched to prasugrel to join the 8 initially prasugrel-treated patients to form what would become the eventual prasugrel cohort of this study. The remaining 95 clopidogrel-treated patients would represent the clopidogrel cohort in the clinical end point analysis. Of the patients switched from clopidogrel to prasugrel, there was a significant reduction in platelet reactivity (median 619 [interquartile range 503-965] reduced to median 255,156-340 P < .0001). The clinical outcomes reported between the 2 cohorts (clopidogrel vs prasugrel) demonstrated a lower incidence of death at 30 days in patients treated with prasugrel (30% vs 50.5%, P = .025). There were no statistically significant differences detected in other clinical end points including myocardial infarction, stent thrombosis, ischemic stroke, or TIMI major or minor bleeding. Because these results are based on a nonrandomized patient population, they should be interpreted with caution, but the use of prasugrel in this population did prove to reduce the platelet reactivity in patients already treated with clopidogrel. Regarding the clinical end points, it seems favorable that greater platelet inhibition in this patient population was not associated with any reported excessive bleeding risk. This study suggests that as expected prasugrel retains significantly more reliable antiplatelet effects than clopidogrel in patients with cardiogenic shock, although it should be noted there were a portion of prasugrel-treated patients (9 of 50, 18%) with residual high on-treatment platelet reactivity.
An abstract of a study presented at the 2014 European Society of Cardiology Congress reviewed results from the IABP-SHOCK (Intra-aortic Balloon Pump–Cardiogenic Shock) II trial, comparing patient outcomes according to which antiplatelet therapy patients received. 50 This study included patients who had acute myocardial infarction, were in cardiogenic shock, and were to be randomized to receive IABP or no IABP to support their hemodynamics. The investigators compared outcomes of patients who received clopidogrel to those that received either ticagrelor or prasugrel. Unadjusted all-cause mortality was found to be significantly lower in patients receiving prasugrel or ticagrelor to those receiving clopidogrel (29.7% vs 41.9% at 30 day, P < .05, 38% vs 54.4% at 1 year, P < .01); however, this result was not statistically significant when assessed through a multiple logistic regression analysis. Unexpectedly, GUSTO bleeding outcomes combined were encountered less frequently in patients receiving ticagrelor or prasugrel than those receiving clopidogrel (18% vs 29.3%, P < .05). Multivariate adjusted bleeding outcomes were not reported in the abstract at the time of this publication. This abstract did not report any findings of platelet reactivity testing but did present some favorable results supporting use of potent P2Y12 receptor antagonists in the cardiogenic shock population. This study builds on previous studies suggesting possible improved clinical outcomes, albeit nonadjusted, without increasing the net clinical risks of bleeding with more potent antiplatelet therapy.
An observational case–-control study published in 2013 by Ibrahim et al evaluated platelet functions studies in 84 patients with ACSs who were initiated on therapeutic hypothermia following cardiac arrest. 51 Included patients may have been managed with or without stent implantation, and it was reported approximately 80% of each group underwent PCI with stent placement. This study included a group of more patients with stable ACS to serve as controls and used the VASP-P analysis to assess the antiplatelet activity of the P2Y12 receptor antagonists. Patients in this study could have received clopidogrel, prasugrel, or ticagrelor as their P2Y12 receptor antagonist. Although this study was primarily assessing the antiplatelet agents as they relate to hypothermia, baseline characteristics including left ventricular ejection fraction, blood pH on admission, serum lactic acid, glomerular filtration rate, and alanine aminotransferase, were all reported to be significantly worse suggesting the hypothermia group was expectedly much more critically ill, and likely more hemodynamically compromised. According to this study. significantly more patients in the hypothermia group were found to be nonresponders (22.5% vs 60.7%; P < .001). Analysis of patients by individual medications showed that clopidogrel-treated hypothermia patients were found to be nonresponders more frequently than clopidogrel-treated control patients (40 [82%] of 49 vs 11 [26%] of 42; P < .001). There was not a difference in nonresponder rates in the prasugrel or ticagrelor-treated patients between hypothermia and normothermia groups. Mean PRI was higher in the clopidogrel-treated hypothermia group (66.39% ± 19.1%) compared to the prasugrel-treated hypothermia group (41.5% ± 21.0%, P < .001) or the ticagrelor-treated hypothermia group (37.6% ± 25.0%, P = .003). There were not significant differences reported for clinical outcomes assessed other than in-hospital mortality which was expectedly greater in the hypothermia group compared to the control group (33% vs 5%). Stent thrombosis was seen in 5% of the normothermia and in 7.1% of the hypothermia group and major or minor bleeding was reported in 3.8% and 6%, respectively. Definitions for stent thrombosis and bleeding were not detailed. Clinical outcomes were not compared between the different antiplatelet agents selected within the hypothermia group. Based on the data within this study, both prasugrel and ticagrelor expectedly seem to retain more reliable platelet inhibition than clopidogrel in hemodynamically unstable patients with ACS requiring therapeutic hypothermia. The authors contributed the difference in antiplatelet effects observed within this study to the possible decreased activity of cytochrome P450 system in the liver, possibly due to temperature or other causes of liver dysfunction. No matter the contributing factor this study continues to add to the body of evidence mounting that hemodynamically unstable patients with ACS may demonstrate suboptimal response to clopidogrel.
Another observational case series was previously published describing the antiplatelet effect of clopidogrel in 25 patients treated with therapeutic hypothermia following successful cardiopulmonary resuscitation. 52 This report did not aim to include patients actively in cardiogenic shock, and the specific hemodynamic parameters of these patients were not reported in the analysis; however, it would be expected that a significant portion of these patients remained hemodynamically unstable. The mean simplified acute physiology score (SAPS) II score reported in this cohort was 59 ± 16 standard deviation. Of the 25 patients, 9 received a 300 mg clopidogrel loading dose and 16 received a 600 mg loading dose. All patients were maintained on 75 mg daily clopidogrel maintenance. The VASP-P analysis was assessed day 1 and day 3 (median 21 and 68 hours following initial loading). All day 1 results collected demonstrated PRI >50% and only 5 (31%) of the 16 patients who were evaluated at day 3 demonstrated PRI <50%. These results demonstrated a uniformly unacceptable degree of platelet inhibition within the first day in a therapeutic hypothermia population. Because of the multifactorial contributing factors which may be present in such a critically ill patient population, it may not be known if hemodynamic instability played any role to these suboptimal clopidogrel effects, but this study further supports the notion that therapeutic hypothermia may represent a significant contributor to reduced metabolic activation of clopidogrel.
The majority of these studies have not explored clinical outcomes in depth, and while there have not been robust prospective randomized controlled trials assessing clinical outcomes in this population, an analysis of a single center registry aimed at identifying incidence and predictive characteristics of stent thrombosis. 53 This retrospective study including 5833 patients undergoing PCI, of whom 87% were reported to be receiving dual antiplatelet therapy with aspirin and clopidogrel. Among the 112 (1.9%) patients who were identified as having cardiogenic shock at the time of PCI, 11 (10%) developed definite stent thrombosis and an additional 11 (10%) developed probable stent thrombosis within 30 days postprocedure. The univariate analysis of this registry revealed cardiogenic shock to be the strongest predictor of both definite stent thrombosis (hazards ratio [HR] 5.0; 95% CI 2.4-10.7) and definite or probable stent thrombosis (HR 8.3; 95% CI 5.0-14.0). This predictor was stronger than other more established risk factors including stent length >20 mm, not receiving dual antiplatelet therapy, STEMI, presence of diabetes mellitus, first generation drug-eluting stent (DES), stent diameter <3 mm, and 3-vessel PCI. Because this was a retrospective study design, there were no platelet aggregation studies to compare the pharmacodynamic activities of P2Y12 receptor antagonists in patients with and without cardiogenic shock, nor was the specific choice of antiplatelet agents reported in the cardiogenic shock cohort.
In all, there exists a growing body of evidence to indicate potentially suboptimal antiplatelet effects of clopidogrel in patients with cardiogenic shock. Although both ticagrelor and prasugrel are proven to be more potent antiplatelet agents than clopidogrel in a randomized population, their additional reliability of platelet inhibition in patients with hemodynamic instability may be of even greater clinical significance to reduce the subsequent risk of ischemic events. Stent thrombosis, which remains one of the most concerning complications following PCI, is reported commonly in the population with cardiogenic shock, indicating that even though these patients are more subject to bleeding complications, this thrombotic complication persists as a significant threat to the outcomes of these patients. Ticagrelor, being an agent that is already in its active form as administered, may be the ideal orally administered antiplatelet agent in this patient population as there was some suggestive observation in the high on-treatment platelet reactivity may still persist with prasugrel. 49 Cangrelor represents an intravenous P2Y12 receptor antagonist which has not been reported specifically in patients with cardiogenic shock, but may also serve as a potential alternative to agents with reduced activity in cardiogenic shock.
Based on the currently available retrospective studies demonstrating more potent platelet inhibition by ticagrelor or prasugrel in patients having cardiogenic shock, it would be expected that there may also be an increased risk of bleeding using these agents. Hemodynamically unstable patients are more likely to have coagulopathic derangements caused by shock, hence the risks of bleeding may be greater than that observed in the randomized controlled trial populations in which each agent was originally studied. It is not known at this time how the risks of bleeding would be balanced relative to the benefits of reduction of ischemic events including stent thrombosis in this specific patient population, although none of the studies reporting clinical outcomes reported statistically significant increases in bleeding events using more potent platelet inhibitors prasugrel or ticagrelor. At this present time, this is an area in need of further investigation through more robust prospective comparative studies.
Conclusions
Observational studies indicate the antiplatelet effects of clopidogrel may be diminished in the setting of cardiogenic shock. As the studies on this matter are retrospective, non-randomized and do not compare the different agents sufficiently to each other, additional studies should be undertaken to identify the ideal P2Y12 receptor antagonist in patients undergoing percutaneous revascularization who are hemodynamically compromised by cardiogenic shock. Because 2 of the studies reviewed focused primarily on therapeutic hypothermia patients, it must be noted that the evidence to date does not clearly indicate whether the hemodynamic instability of cardiogenic shock and the reduced body temperature of therapeutic hypothermia may play independent roles in the diminished antiplatelet effect of clopidogrel. Because of the lack of randomized comparative studies to date in this population, no definite conclusions can be determined at this time; however, in the meantime, the clinician should carefully consider all factors associated with patient’s baseline characteristics, drug interactions, and clinical conditions including cardiogenic shock to determine the most appropriate P2Y12 receptor antagonist following PCI. Prasugrel and ticagrelor may represent more reliable oral P2Y12 receptor antagonists in patients with cardiogenic shock, and although not available in the United States currently, the intravenous P2Y12 receptor antagonist cangrelor may also serve as a potential agent for future studies.
Footnotes
Author Contribution
Weeks contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Sieg contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Paruthi contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Rajapreyar contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
