Abstract
Background:
Atrial fibrillation (AF) is the most common cardiac arrhythmia. Thus, the aim of our study was to evaluate the smartphone-based electrocardiogram (ECG) recordings aimed at AF screening at Polish pharmacies.
Methods:
Prospective AF screening among patients aged ≥65 years was conducted at 10 pharmacies using Kardia Mobile with a dedicated application (Kardia app). Prior AF was a study exclusion criterion. CHA2DS2-VASc score (congestive heart failure, hypertension, age, diabetes mellitus, previous stroke/transient ischemic attack, female sex, and vascular disease) has been collected from every patient. A single-lead ECG has been acquired by the placement of fingers from each hand on the pads. Kardia app diagnosis has been evaluated by the cardiologist.
Results:
A total of 525 ECGs were performed. Kardia app diagnosis was provided in 490 cases. In 437 (89.18%) cases, it was “normal” rhythm, in 17 (3.47%) recordings “possible AF,” in 23 (4.69%) ECGs “unreadable,” and in 13 (2.65%) “unclassified”. After the cardiologist reevaluation, the new AF was identified in 7 (1.33%) patients. Sensitivity and specificity of Kardia app in detecting AF was 100% (95% confidence interval [CI]: 71.5%-100%) and 98.7% (95% CI: 97.3%-99.5%), respectively. The positive predictive value was 64.7% (95% CI: 38.3%-85.7%) and the negative predictive value was 100% (95% CI: 99.2%-100%). CHA2DS2-VASc score was 2.14 ± 0.69 for those with new AF and 3.33 ± 1.26 in the non-AF group.
Conclusion:
Kardia app is capable of fast screening and detecting AF with high sensitivity and specificity. The possible diagnosis of AF deserves additional cardiological evaluation. The results obtained in patients with low CHA2DS2-VASc score and “silent” AF confirm the importance of routine AF screening. Cardiovascular screening with the use of mobile health technology is feasible at pharmacies.
Introduction
Atrial fibrillation (AF) as the most common cardiac arrhythmia is associated with about 1/3 of all strokes. 1 –3 Furthermore, strokes related to AF are considered more severe or even fatal compared with strokes caused by other factors, and also entail high costs. 2 –4 Importantly, AF is commonly asymptomatic or patients may experience short-lived episodes related to nonspecific symptoms such as fatigue. 2,5 Thus, many patients with undetected AF have an increased risk of a stroke 2 and approximately 25% of them have AF diagnosed only after the event. 3
The world’s population is aging quickly. 4,6 Considering this and the fact that the incidence of AF sagaciously increases with age, the prevalence of AF is expected to double by 2060. 5,7 Therefore, the European Society of Cardiology (ESC) 8 recommends opportunistic AF screening among patients aged ≥65 years, but this procedure is not routinely performed. 1,3 If AF is diagnosed early and proper anticoagulant treatment is prescribed simultaneously, 64% of AF-related strokes might be prevented. 1 Hence early detection of AF is a major public health concern, 6,9 which should encourage cooperation of multidisciplinary teams and use of dedicated technologies. 8
Nowadays, one of the most interesting and state-of-the-art methods of AF detection is mobile health technology based on mobile and wireless devices such as smartphones. It can provide an inexpensive, convenient, and effective way for community-based AF screening. 1,4,10,11 Although it is still a relatively new field of health care, the World Health Organization claims that incorporation of mobile health technology into everyday practice may significantly help in the achievement of several health objectives. 12 The rapid growth of the number of smartphone users 5,10,11 makes the method available and more affordable than before. 1,10,13 It also opens several opportunities for delivery of health care and for the communication that has to date been unavailable. 10 Hence the importance of public places might be an effective venue for AF detection, leading to the development of multidisciplinary cooperation of health-care professionals. 1,2,8 Feasibility of AF detection by a smartphone application at pharmacies has been evaluated in Australia. This study documented that up to 1.5% patients had a previously undiagnosed AF. 1
Smartphone applications rely on single-lead electrocardiogram (ECG). Electrocardiogram’s electrical signals are converted to an ultrasonic frequency modulation (FM) sound signal and then transmitted to a smartphone with the application installed. 14 Therefore, it can be sensitive to environmental noise. This is why the value of this application should be explored at several public venues.
The aim of our prospective study was to evaluate the smartphone-based (Kardia Mobile with a dedicated smartphone application) ECG recording aimed at AF screening at pharmacies. We intended to evaluate the sensitivity and specificity of Kardia Mobile in detecting AF in a population without a previous history of arrhythmia. Another aim was to test whether pharmacies can be considered for routine AF screening. Besides, to our knowledge, this is the first study presenting data regarding the incidence of new AF detected by smartphone-based screening at Polish pharmacies.
Methods and Materials
The prospective AF screening was conducted from December 2017 to November 2018 at 10 pharmacies (in 5 cities and 1 village) from 3 regions of Poland. The study was approved by the local Bioethics Committee (Poznan University of Medical Sciences) and all pharmacy owners consented to carry out the test.
The research team included cardiologists, pharmacists, and students from Student Scientific Societies of Pharmacoeconomics and Cardiology. Each screening test carried out by students was supervised by a trained pharmacist. Nonphysician researchers attended a course on the use of Kardia Mobile, the basics of ECG, and assessment of thromboembolic risk based on CHA2DS2-VASc score (congestive heart failure, hypertension, age, diabetes mellitus, previous stroke/transient ischemic attack, female sex, and vascular disease). 8
The device used was Kardia Mobile with a dedicated application (Kardia Phone application, AliveCor Inc, Mountain View, California) for smartphones (iOS or Android). Kardia Mobile is Food and Drug Administration approved and Conformité Européenne (CE) marked equipment, clinically validated for recording, storing, and transferring single-channel lead I ECGs. 2,3 We used the most recent update of the Kardia Phone application (Kardia app) version for the AF screening.
Single-lead ECG was acquired by the placement of at least 2 fingers of both left and right hand on the pads for 30 seconds. Cardiac electrical activity was transferred to the smartphone by FM of an ultrasound signal using a 19 kHz center frequency and a modulation index of 200 Hz/mV (FMECG). The ultrasonic FMECG received by the smartphone’s microphone was digitized at 44.1 kHz with 24-bit resolution. The smartphone application would demodulate the signal to a digital ECG trace (300 samples/s, 16-bit resolution) viewed in real-time, stored, and transmitted to a secure server. 15
After recording a 30-second tracing, the device would provide the first interpretation with an automated algorithm. The application displayed 4 potential findings: “possible AF,” “normal” rhythm, “unclassified,” and “unreadable.” “Possible AF” meant that the AF was detected on the basis of P-wave absence and R-R interval irregularity. 15,16 The diagnosis, however, should be confirmed by a physician. The “normal” rhythm finding indicated that the heart rate was regular and between 50 and 100 bpm, with shape, timing, and duration of every beat considered normal. 16 “Unclassified” meant that the quality of tracing was good but the Kardia Mobile software was not able to classify ECG recording as either “normal” or “possible-AF” (eg, sinus tachycardia > 100 bpm, irregular rhythm due to premature complexes). 16,17 “Unreadable” diagnosis suggested that ECG was not good enough for analysis due to sound or electrical interferences on the recording. 16 Apart from that, older versions of the Kardia app could provide no classification if the ECG was noninterpretable. There was also a possibility of “no analysis” notification displayed, when the ECG was noninterpretable (because of noise on the signal, for instance).
Every patient entering the pharmacy who looked to be aged ≥65 years was asked to join the study. Patients with a history of AF were excluded from the analysis. Once the patient agreed to join the study, written informed consent, the patient’s phone number, and CHA2DS2-VASc score were collected. Finally, the ECG was performed. Recordings protected with a password were saved and sent via e-mail to 2 cardiologists, one of whom interpreted the ECG within 2 days’ time. The cardiologist analyzed the recordings and gave the diagnosis of AF, normal sinus rhythm (SR), or SR with premature supraventricular (PAC) or premature ventricular (PVC) complexes (both classified as SR+). We have found 1 ECG with atrial stimulation and it was classified as SR+. When it was noisy and artifacts were present, the recording was evaluated as noninterpretable. ECG with “no analysis” notification (1 case) was considered noninterpretable as well. When an SR was present with or without premature complexes (or atrial stimulation), the recordings were classified into “non-AF group.”
Whenever the cardiologist diagnosed AF, the physicians contacted the patient on the phone and advised them to collect the ECG printout from the pharmacy where it had been performed and contact his or her GP for further evaluation.
Statistical Analysis
The data are shown as mean values ± standard deviation, median, and percentages. Comparison of 2 groups was performed by Mann-Whitney test since data did not follow a normal distribution (Shapiro-Wilk test). Comparison of more than 2 groups was performed by Kruskal-Wallis test. Comparison of nominal scale data was performed by χ2 test of independence. For statistical purposes, CHA2DS2-VASc score was evaluated on 2 levels (1 or ≥2 points). Calculations were performed using Statistica 12 and PQStat software. A critical P-value of .05 was used for significance in all analyses.
Results
Out of the 1064 patients asked to join the study, 290 refused and 132 patients were aged <65 years. About 117 patients were excluded due to their inability to fulfill the CHA2DS2-VASc protocol, double recruitment, and previously detected AF. Eventually, 525 patients were included in the study.
There were 358 (68.19%) female and 167 (31.81%) male participants. The average age of the study group was 73.72 ± 6.49; median: 73 (women: mean 73.54 ± 6.19; median 72 and men: mean 74.11 ± 7.12; median 73; P = .3529). The structure of the study group including sex, age, CHA2DS2-VASc score, and heart rate is presented in Table 1.
The Structure of the Study Group After the Over-Read by Cardiologists.

Abbreviations: AF, atrial fibrillation; bpm, beats per minute; the CHA2DS2VASc, congestive heart failure, hypertension, age, diabetes mellitus, previous stroke/transient ischemic attack, female sex, and vascular disease; M, men; SD, standard deviation; W, women.
a All patients included in the study.
b Including newly identified patients and those with previously known diagnosis and confirmed by cardiologists after the phone contact.
The Kardia app provided a diagnosis in 490 cases. There were 17 (3.47%) patients with “possible AF” finding. The cardiologist confirmed AF in 11 cases, while 2 recordings were reclassified to SR+ due to PACs, 1 recorded as SR with atrial stimulation (SR+), and 3 classified as noninterpretable due to artifacts and poor quality of ECG tracing. “Normal” rhythm was revealed in 437 (89.19%) Kardia app’s recordings, of which one was classified by the cardiologist as noninterpretable due to isoelectric line artifacts.
We found 13 (2.65%) recordings evaluated by the device as “unclassified,” of which 10 were diagnosed by the cardiologist as non-AF (2 cases of sinus tachycardia >100 bpm, 3 ECGs with SR and wide QRS complexes, 3 ECGs with SR and isolated artifacts that Kardia app marked as premature complexes, 2 cases of premature QRS complexes) and 3 as noninterpretable.
There were 23 (4.69%) recordings classified by the device as “unreadable,” of which 7 were qualified by the cardiologist as SR (6 ECGs with temporary artifacts and 1 ECG with low amplitude of QRS complexes), 1 ECG as SR+ (PVC with temporary artifacts of isoelectric line), and 15 were confirmed as noninterpretable (including 1 case with “no analysis” notification).
Out of 35 ECG tracings for which Kardia app failed to provide a potential diagnosis, there was 1 patient with AF. In 24 ECG tracings without potential findings, the cardiologist diagnosed SR (11 cases of sinus tachycardia >100 bpm, 9 cases of wide QRS complexes, and 3 ECGs with temporary isoelectric line artifacts), while in 8 cases, the cardiologist diagnosed PACs (5 ECGs) and PVCs (3 ECGs). Two ECG tracings were diagnosed as noninterpretable.
Finally, the cardiologist diagnosed AF in 12 patients (2.29% of participants). Those patients were contacted over the phone by a physician inquiring about any symptoms of arrhythmia and medicines used (brand and/or international name of anticoagulants). The interview helped to exclude 5 (0.96%) patients from the group of patients with a newly identified AF which eventually included 7 (1.33%) patients.
Examples of ECG recordings classified by the device as “possible AF,” “normal,” and evaluated by a cardiologist as false-positive AF are presented in Figures 1, 2, and 3, respectively.

The example of “possible AF” Kardia app finding.
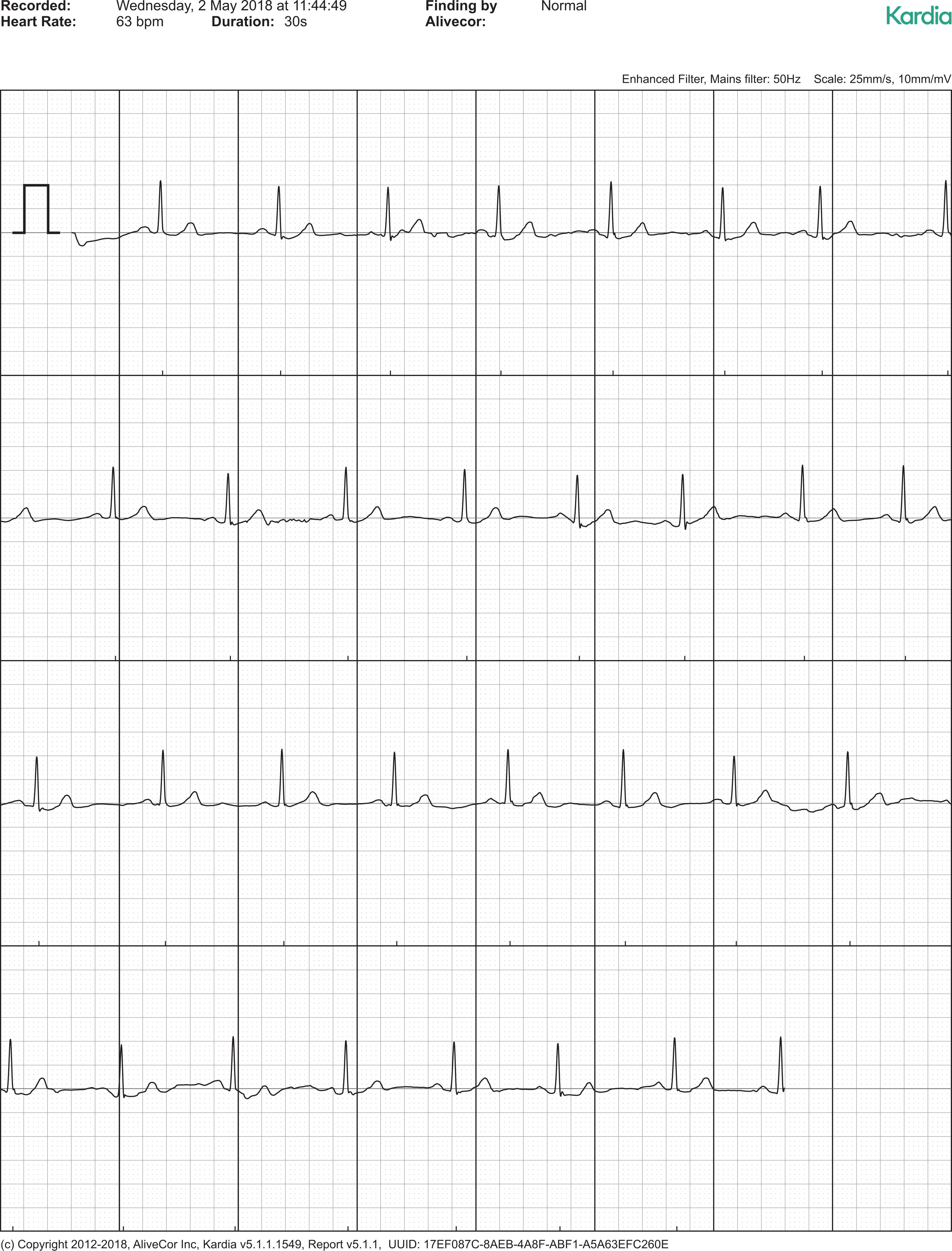
The example of “normal” Kardia app finding.

The example of false positive AF diagnosis.
In the group of patients with newly detected AF, there was 1 male with CHA2DS2-VASc score 1 and 3 men with score 2, as well as 2 men with score 3. In the case of 1 newly diagnosed AF in a woman, CHA2DS2-VASc score was 2. The exact distribution of CHA2DS2-VASc scores is presented in Table 2.
The Exact Distribution of the CHA2DS2VASc Score Domains.

Abbreviations: AF, atrial fibrillation; C, congestive heart failure; H, hypertension; A2, for age ≥ 75 years; D, diabetes; S2, stroke; V, vascular disease.
a The number of patients between 65 and 74 years was not presented—it results from the total number of patients and the number of patients ≥75 years (A2).
b Including newly identified patients and those with previously known diagnosis and confirmed by cardiologists after the phone contact.
Sensitivity and specificity of the Kardia app in detecting AF were 100% (95% confidence interval [CI]: 71.5%-100%) and 98.7% (95% CI: 97.3%-99.5%), respectively. Positive predictive value (PPV) and negative predictive value (NPV) was 64.7% (95% CI: 38.3%-85.7%) and 100% (95% CI: 99.2%-100%), accordingly. There were no-false AF negative diagnoses.
Over-read of all ECG tracings by cardiologists revealed that the Kardia app findings were convergent with the cardiologists’ evaluation in 462 (94.29%) cases. The discrepancy between the device and the cardiologist resulted from reclassification of Kardia app’s recordings into diagnoses of SR/SR+ or artifacts (Table 3). The agreement in the detection of the “normal” rhythm between Kardia app and the diagnoses of non-AF recordings (SR and SR+) by the cardiologist was 99.77%.
The Exact Distribution of Kardia App Possible Diagnosis and Cardiologists’ Evaluation (n = 490).
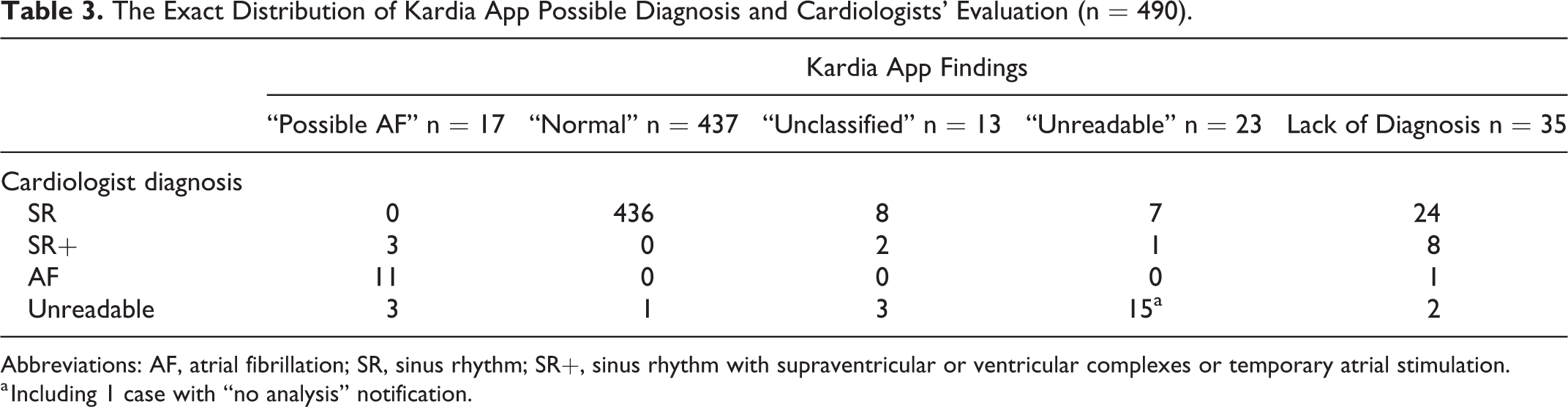
Abbreviations: AF, atrial fibrillation; SR, sinus rhythm; SR+, sinus rhythm with supraventricular or ventricular complexes or temporary atrial stimulation.
a Including 1 case with “no analysis” notification.
Statistically, significant correlation was observed between the diagnosis of AF and the patients’ sex (P = .004). There was no correlation either between the detected AF and age (P = .1647) or AF and CHA2DS2-VASc score (P = .0655).
Discussion
It was, to our knowledge, the first study in Poland aimed at smartphone-based AF screening. The most important findings of our study were that new AF was identified in 1.33% of opportunistically screened visitors of pharmacies with a high level of sensitivity and specificity. Contrary to PPV, NPV was high. Considering this, AF screening in patients aged ≥65 years might be routinely performed at pharmacies.
Our results are consistent with other studies. Using Kardia Mobile, Lowres et al 2 found 1.5% of patients with a previously undiagnosed AF, while Chan et al 3 and Halcox et al 18 revealed a new AF in 0.49% and 1.84% of individuals, respectively. Soni et al, 19 meanwhile, enrolled 2100 people in 50 villages in India and the prevalence of AF was 1.6% but among participants above 65 years of age, the incidence was 3.2%.
In our study, sensitivity and specificity of the Kardia app in detecting AF were found to be 100% and 98.7%, respectively. The sensitivity of 100% was also reported by Koshy et al 20 and was higher than in the studies conducted by Lau et al (98%), 15 William et al (96.6%), 21 and Chan et al (71.4%). 3 Specificity corroborated with the results presented by Chan et al (99.4%), Lau et al (97%), and William et al (94.1%). 3,15,21 Differentiation of sensitivity values between various studies may result from including or excluding “unreadable” and “unclassified” recordings. 20 Another explanation might be the version of Kardia app used and its different algorithm of detecting AF. 3
Positive predictive value obtained (64.7%) seems to be low in our study compared with the result (80%) found by Selder et al. 22 It is, however, consistent with the result (66.3%) presented by William et al 21 who examined diagnoses provided by Kardia app and interpretation of nearly simultaneously acquired 12-lead ECGs in patients with AF admitted for antiarrhythmic drug initiation.
In our study, 1 false-positive AF result was due to atrial stimulation. The manufacturer does not recommend performing ECG with the use of Kardia app 17 in patients with pacemakers as it has not been validated yet. It was our intention, however, to perform a screening test and reduce exclusion criteria. Fifty percent of false-positive AF ECGs (3 cases) were due to significant noise and were found by a cardiologist to be noninterpretable. It is possible that were the ECG recordings performed again, there would be no artifacts.
William et al 21 found 27.6% ECG recordings to be “unclassified,” while in fact, 7.5% of the Kardia app findings had significant noise. Probably “unclassified” and “unreadable” Kardia app potential diagnoses were mixed together in that study. These 2 potential diagnoses had different diagnostic criteria, however. Selder et al, 22 meanwhile, reported 17% of Kardia app ECG recordings as “unclassified” and 2% as “unreadable.” The rate of “unclassified” recordings higher than in our study (2.65%) could be related to their study population because patients presenting symptoms of cardiovascular disease might experience different forms of atrioventricular or intraventricular conduction delays, while Kardia app is not capable of detecting heart rhythm other than AF and SR.
In our study, 4.6% of ECG recordings were noninterpretable after cardiologist review which is consistent with the results obtained by William et al (4%) 21 and less than in the study by Selder et al (8%). 22 It could have resulted from the fact that interpretation of ECG tracing quality might be difficult for nonprofessionals. 23 When unsupervised, the presence of movement and contact artifacts may lead to more noninterpretable ECG recordings as well as false-positive diagnoses of AF. 23 Thus, it would be advisable in our study to repeat the ECG if tracing was evaluated as nonreadable. Some noise exposure level is inevitable, however. As disclosed in this study and by Lowres et al, 1,2 pharmacies might be considered suitable venues for conducting AF screening in contrast to noisy facilities such as shopping centers where artifacts would be common.
The occurrence of lack of diagnosis in Kardia app has not been reported so far. In our study, most of the recordings, although classified by the cardiologist as SR, presented sinus tachycardia > 100 bpm or SR with wide QRS complexes. Although the lack of diagnosis might be related to the version of Kardia app used, the explanation deserves further investigations.
In our study, CHA2DS2-VASc score was lower for patients with newly identified AF (2.14 ± 0.69) than in the non-AF group(3.33 ± 1.26). Conversely, Lowres et al 2 revealed CHA2DS2-VASc score of 3.7 ± 1.1 for newly identified AF and 3.2 ± 1.1 for the non-AF. Surprisingly, lower CHA2DS2-VASc score in newly identified AF patients (6 men and 1 woman) compared with those without AF in our study might have resulted from the fact that females get one additional point in the assessment of CHA2DS2-VASc score. In the study conducted by Lowres et al, 2 the ratio of male to female in newly identified AF was 2 to 1. As presented in our study, however, pharmacies are more likely to be visited by women 24 but AF is more common in men. 25 Men could also remain undiagnosed longer because women are more likely to experience AF symptoms, which deteriorate their quality of life. 26 Nevertheless, since women live longer, the total lifetime risk of AF is recognized as similar in both sexes. 27 Thus, significantly more frequent detection of previously undiagnosed AF among men, as in our study, merits further investigation.
Involvement of pharmacists in the AF screening is in line with ESC guidelines for AF management. 8 They recommend building teams combining multiple skills, education, and expertise both in medical conditions and medicines management. 8,28 Pharmacies may also facilitate a physician review for those with suspected AF. 2 Same reason is indicated in the case of AF screening by nurses in general practice, which offers the immediate over-read and action by the GP. 13
Apart from being available, technologies and solutions used in health care should be affordable, too. 29 The official price (US$99, April 2019) of Kardia Mobile 16 and multiple use options confirm the community AF screening has the potential to reduce socioeconomic disparities in access to ischemic stroke prevention and to alleviate the burden of cardiovascular disorders. Results presented in this study contribute to the feasibility of the method in common AF screening. 3 It is confirmed also by Lowres et al 1,2 who revealed cost-effectiveness of AF screening with an incremental cost-effectiveness ratio of €3142 per gained quality adjusted life years (QALY). This result is aligned with studies conducted by Aronsson et al 4 and Halcox et al 18 who concluded that screening for AF is cost-effective in older individuals. Besides, Aronsson et al 4 presented that screening for asymptomatic AF would result in about 270 fewer strokes and 420 more QALYs among Swedes aged 75 years. Desteghe et al 30 pointed out that Kardia app provided cost-effectiveness also in hospitalized patients. Apart from the pharmacoeconomic benefits, 1,2,4,18 this approach is highly acceptable among those who are ≥65 years of age 18 Therefore, dissemination of mobile health technologies for AF screening and stroke prevention should be considered as a practice which might be routinely performed and covered through government-funded incentives.
Limitations
Firstly, the equipment used is sensitive to noise, thus it would be advisable to measure the noise level at pharmacies and its effect on the number of unreadable ECG tracings. Repeated performance in the case of any interferences could reduce the number of noninterpretable ECGs which may result from inaccuracy of Kardia Mobile paramedical users. Moreover, giving the people the opportunity to be screened on repeated visits at their pharmacy may help identify more patients with paroxysmal AF. The sensitivity and specificity of Kardia app might be overestimated in our study due to the lack of Kardia app finding in 35 cases. It could also be worthwhile to ask patients about medicines used. It would be advisable to perform the study using a second device simultaneously to control the results obtained from Kardia app. The number of poor recordings might be reduced by turning the device to airplane mode temporarily. It might, however, extend the duration of each screening test. Someone might indicate the students’ participation in the study as another limitation. As has been pointed out by Young and Badowski, 31 however, academic centers can also use telemedicine for teaching and training students showing them that collaboration in research is effective and might involve state-of-the-art technologies.
Conclusions
Atrial fibrillation was identified in 1.33% of patients, ≥65 years of age, without a previous history of AF. This first study in Poland confirmed that Kardia app is capable of fast screening and detecting AF with high sensitivity, specificity, and NPV. High specificity and high NPV are crucial for a screening test. The possible diagnosis of AF deserves additional cardiological evaluation. Less than 5% of ECG recordings were noninterpretable. The results obtained in patients with a low CHA2DS2-VASc score and “silent” AF confirm the importance of routine AF screening and indicate that every patient should be considered individually in terms of pharmacotherapy. Cardiovascular screening using mobile-health technology is feasible and might be considered for common use for opportunistic AF screening at pharmacies.
Footnotes
Acknowledgments
The authors would like to thank all the participants and pharmacists for their willingness to join the project. Moreover, they would like to thank the pharmacy owners who disinterestedly allowed them to carry out the research. They would also like to thank David Albert, the Founder & Chief Medical Officer at AliveCor Inc and EM-MED Sp. z o.o. for their consultations on technical issues.
Author Contributions
TZ and JZ took part in each part of the study. AB contributed to the concept of the study, to the analysis and interpretation of the data and to the drafting and critical revision of the manuscript. AS, EN contributed to the concept of the study and critical revision of the manuscript. MM, RD, WT, PR, DK, DS, AS, MD contributed to the data acquisition and analysis, and to the critical revision of the manuscript. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
