Abstract
Background:
Diabetic cardiomyopathy (DCM) is accompanied by microvascular complications that lead to myocardial dysfunction and heart failure. Most conventional therapies cannot ameliorate the microvascular insufficiency in DCM. In this study, we tested the hypothesis that undercarboxylated osteocalcin (ucOC) may be a new adjuvant therapy against the progression of DCM and its underlying microvascular pathology.
Materials and Methods:
Diabetes was induced in Wistar rats with a high-fat diet combined with streptozotocin injections, and ucOC was upregulated after warfarin administration in the treated group. After 8 weeks, cardiac functions were assessed using a Langendorff apparatus. Cardiac tissue samples were also extracted to assess the ucOC receptor and vascular endothelial growth factor (VEGF) for histopathological studies.
Results:
Both the systolic and the diastolic dysfunction observed in the DCM group were significantly improved after the increase in ucOC blood levels. Significant improvement in VEGF and CD31 expression after warfarin injection was associated with increased capillary density, neovascularization, and decreased myocardial fibrosis together with the reestablishment of myocardial structural and ultrastructural patterns.
Conclusion:
Undercarboxylated osteocalcin may have a promising effect in improving microvascular insufficiency and myocardial dysfunction in DCM.
Keywords
Introduction
Cardiovascular complications associated with diabetes cause death in more than 75% of patients with diabetes worldwide. 1 Diabetic cardiomyopathy (DCM) is a crucial diabetes-associated microvascular disease characterized by microvascular insufficiency, 2 impaired angiogenic response to chronic ischemia, 3 suppression of angiogenic growth factors, and extensive myocardial fibrosis. 4,5 These heterogeneous disorders have been related to structural changes in the myocardium that lead to left ventricular hypertrophy and/or systolic dysfunction. 6 -8
Vascular endothelial growth factor (VEGF) is a major regulator of angiogenesis in physiological and pathological conditions, leading to new blood vessel formation to meet the metabolic demands of the tissues. 9 Reduced VEGF expression and capillary density in diabetic patients has been incriminated in the pathogenesis of diabetic microvascular complications that eventually lead to cardiac dysfunction and heart failure. 10 -12
Osteocacin (OC), also known as bone γ-carboxyglutamic acid-containing protein, is a polypeptide that is posttranslationally modified to carry dicarboxylic glutamyl (Gla) residues. The fraction of OC that undergoes imperfect γ-carboxylation is referred to as under-carboxylated osteocalcin (ucOC). 13 -15 The fully carboxylated and undercarboxylated forms of OC are both found in bone and serum, but a higher proportion of ucOC is found in the circulation. 16 Administration of warfarin as an anticoagulant prevents posttranslational carboxylation of OC, resulting in increased levels of ucOC. 17 Osteocalcin could exert a protective effect on cardiovascular risk in diabetic patients by improving glucose tolerance and hyperlipidaemia. 18 -20
While our previous studies have illustrated the antioxidative and antiapoptotic role of undercarboxylated osteocalcin (ucOC) in protecting the diabetic heart from developing DCM, 18 the present study is the first, to the best of our knowledge, to investigate the potential role of ucOC as a therapeutic agent for DCM and to document the relationships among increased ucOC, upregulation of VEGF expression, improvement in myocardial microcirculation, and its subsequent cardiac functional and histopathological changes.
Materials and Methods
Diabetic Cardiomyopathy Rat Model
Fifty-six male Wistar rats (100-120 g) were purchased from the experimental animal care unit of Cairo Medical University. All animal procedures were performed in accordance with the guidelines of the National Institutes of Health and approved by the Institutional Animal Care and Use Committee. Type 2 diabetes was induced by feeding rats a high-fat diet (60% kcal fat) for 2 weeks, followed by 2 intraperitoneal (ip) injections of streptozotocin (STZ, 30 mg/kg/d ip in 0.01 M citrate buffer, pH 4.3; Sigma, St Louis, Missouri). 21 After STZ injection, fasting serum glucose and insulin levels were measured to confirm diabetes. Then, diabetic rats were maintained for 8 weeks and allowed to develop DCM.
Warfarin Administration
Warfarin powder (Sigma) was dissolved in alkaline aqueous solution and then injected at a dose of 0.25 mg/kg on 3 alternating days/week 22 as a therapeutic regimen after the development of DCM for 8 weeks. Rats were housed in individual cages to avoid any injuries.
Study Design
At the beginning of the study, rats were divided equally into 4 groups of 14 rats each. The first group was considered the control group, in which rats were fed standard rat chow, followed by ip injection of citrate buffer at a dose of 0.5 mL after 2 weeks. Rats were left without interference for 8 weeks. Then, 0.5 mL of alkaline aqueous solution was injected subcutaneously for 3 alternating days/week for 8 weeks. In the second (DCM) and third (DCM + warfarin) groups, development of DCM was established 8 weeks after the induction of diabetes according to the protocol described earlier. The untreated DCM group was left without interference for another 8 weeks, and the DCM-treated group received warfarin injection as described earlier for 8 weeks. The fourth group (warfarin control) was treated as a control group, except the rats received warfarin during the last 8 weeks of the study. All rats were euthanized 18 weeks after the beginning of the experiment, which was the duration of the study (Figure.1).
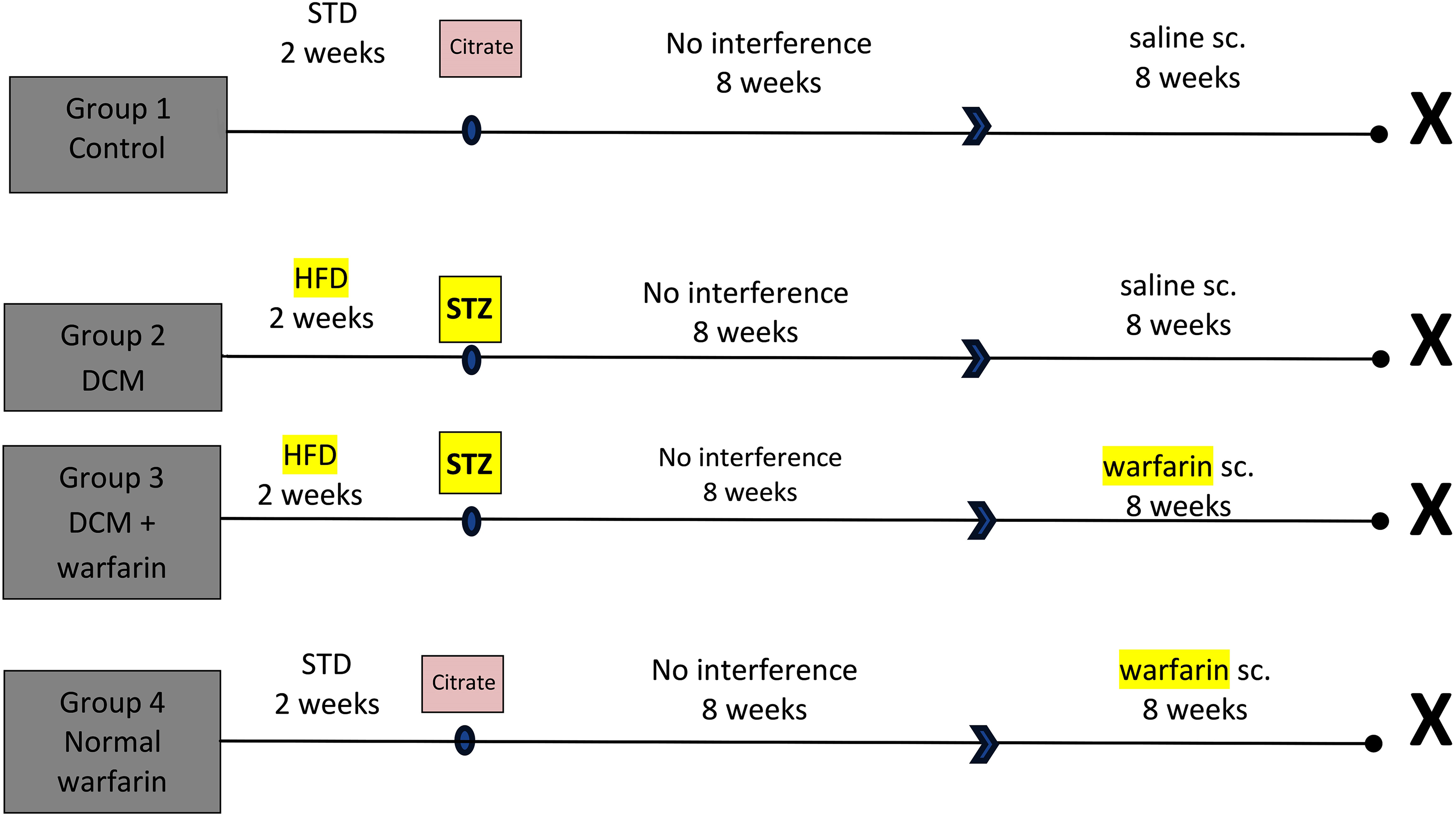
Schematic presentation of the experimental protocol.
At the end of the study, blood samples were obtained for biochemical measurements of fasting serum ucOC, glucose, and insulin levels. Homeostasis model assessment-insulin resistance (HOMA-IR) was calculated. Cardiac functions were assessed using the Langendorff apparatus and Powerlab data acquisition system. Finally, cardiac tissue samples were collected for histopathological examination, immunohistochemical evaluation of VEGF and CD31, and assessment of gene expression of VEGF and the OC receptor (GPrc6A).
Assessment of Cardiac Function by Langendorff Apparatus
Rats were anaesthetized using ketamine hydrochloride (25 mg/kg, ip) followed by heparin injection ip (1000 IU), and the hearts were rapidly extracted, cannulated, and connected to a constant flow Langendorff apparatus (Radnoti; Harvard Apparatus, USA). Constant perfusion was maintained at a flow of 16 mL/min at 37°C with Krebs–Henseleit (KH) buffer continuously gassed with 95% O2/5% CO2 (pH 7.4).
Cardiac function was assessed using a saline-filled latex balloon inserted into the left ventricle (LV) through an incision in the left atrial appendage. Hearts were placed in a water-jacketed heart chamber (Radnoti) at 37°C for 30 minutes until stabilization. End-diastolic pressure was adjusted at 10 mm Hg by filling the balloon with saline, and the balloon was then connected to a pressure transducer (MLT 844, AD instruments, Australia) and a Powerlab system (AD Instruments, Sydney, Australia) for analysis of recorded waves. The LV function was investigated by 3 parameters: first, left ventricular developed pressure (LVDP) (peak systolic pressure − end-diastolic pressure); second, contractility index (maximum rate of pressure increase [ΔP/ΔT]); and last, diastolic function index (maximum rate of pressure decline [−ΔP/ΔT]). Measurement continued for 15 minutes. 23
Biochemical Assessment
Fasting serum glucose (Diamond Diagnostics, Holliston, USA), 24 fasting serum insulin (enzyme-linked immunosorbent assay [ELISA]; Linco research, Missouri, USA) and serum ucOC (ELISA; MyBioSource, California, USA) were evaluated. Homeostasis model assessment-insulin resistance was assessed from the following equation: HOMA = glucose (mmol/dL) × insulin (µIU/mL)/22.5. Insulin resistance was considered if the index was larger than 2.0. 25
Real-time Polymerase Chain Reaction for Detection of GPRC6A and VEGF Gene Expression
TRIzol reagent (Invitrogen, California, USA) was used to isolate total RNA, followed by measuring its purity and concentration by an ultraviolet spectrophotometer. Reverse transcription of total RNA to complementary DNA was carried out with a reverse transcription reaction using Moloney murine leukemia virus (M-MLV) reverse transcriptase (Promega, Wisconsin, USA).
Real-time polymerase chain reaction (PCR) was carried out using an ABI 7500 Real-Time PCR System (Applied Biosystems, California, USA). The reaction contained SYBR Green Master Mix (Applied Biosystems). Data were analyzed with ABI Prism sequence detection system software and calculated using version 1·7 Sequence Detection Software (PE Biosystems, Massachusetts, USA). The relative expression of studied genes was calculated using the comparative threshold cycle method. The expression of β-actin messenger RNA was used as an internal control in all samples.
The sequences of the primers used for real-time PCR were OC receptor (GPRC6A) forward primer: 5′-CCAGTAGGGAGAGAATCAGGAGTAGGATGG3-′ and reverse primer: 5′-AGAGTTAAGGCTGTCATAGGTTCTGGGTACTCAG3-′; VEGF forward primer: 5′-ACAAAACGCTCGCTCAGATT-3′ and reverse primer: 5′-GTCCATGGTCAGAACGGACT-3′.
Histopathological Examination
After routine paraffin histological preparation, the LV specimen sections were subjected to staining with hematoxylin and eosin, and Masson Trichrome for evaluating tissue morphology and fibrosis and immunohistochemical evaluation of VEGF (1:50, rabbit polyclonal anti-VEGF primary antibody; Santa Cruz Biotechnology, Santa Cruz, California), CD31 (1:200, goat polyclonal anti-CD31 Ab; Abcam, Cambridge, Massachusetts), and caspase 3 (1:200, goat polyclonal anti-caspase 3 Ab; Abcam) as previously described. 26 Mouse spleen tissue served as a positive control. The negative controls were obtained in the automated staining protocol through omission of the primary antibody. All the histomorphometric parameters, including the number of foci of myocyte disarray, the percentage area of collagen fibers in Masson Trichrome-stained sections, the percentage area of VEGF, caspase 3 immunoreactivity. and the count of CD31 immunohistochemically positive blood capillaries were estimated via the Leica Imaging analyzer software system (Leica Qwin 500C; Leica, Cambridge, England) 27 with a standard measuring frame of 85 550 µm 28 in 10 randomly selected, nonoverlapping fields from each animal in the different study groups.
Electron Microscopic Examination
Specimens from the LV were cut into small slices, fixed in 4% glutaraldehyde, and post-fixed in 1% osmium tetroxide. Toluidine blue–stained 0.5-µm semithin sections were prepared. 29 Sections were examined and photographed using a JEOL JEM 1010 transmission electron microscope (JEOL, Tokyo, Japan).
Statistical Method
Data were coded and analyzed with SPSS version 23. Data are displayed as the mean and standard deviation. Comparisons between groups were carried out using analysis of variance with multiple comparisons post hoc testing. Correlation studies were performed using the Pearson correlation coefficient. 30 P values less than .05 were considered statistically significant.
Results
Upregulation of Serum ucOC and OC Receptor (GPRC6A) Gene Expression After Warfarin Administration
The development of DCM was accompanied by a significant reduction in serum ucOC and GPRC6A gene expression. Warfarin supplementation resulted in attenuation of the carboxylation of OC with a significant increase in the gene expression of ucOC and its receptor (GPRC6A). A similar effect was observed in the nondiabetic rats treated with warfarin relative to their nonwarfarin-treated control counterparts (Figure 2).
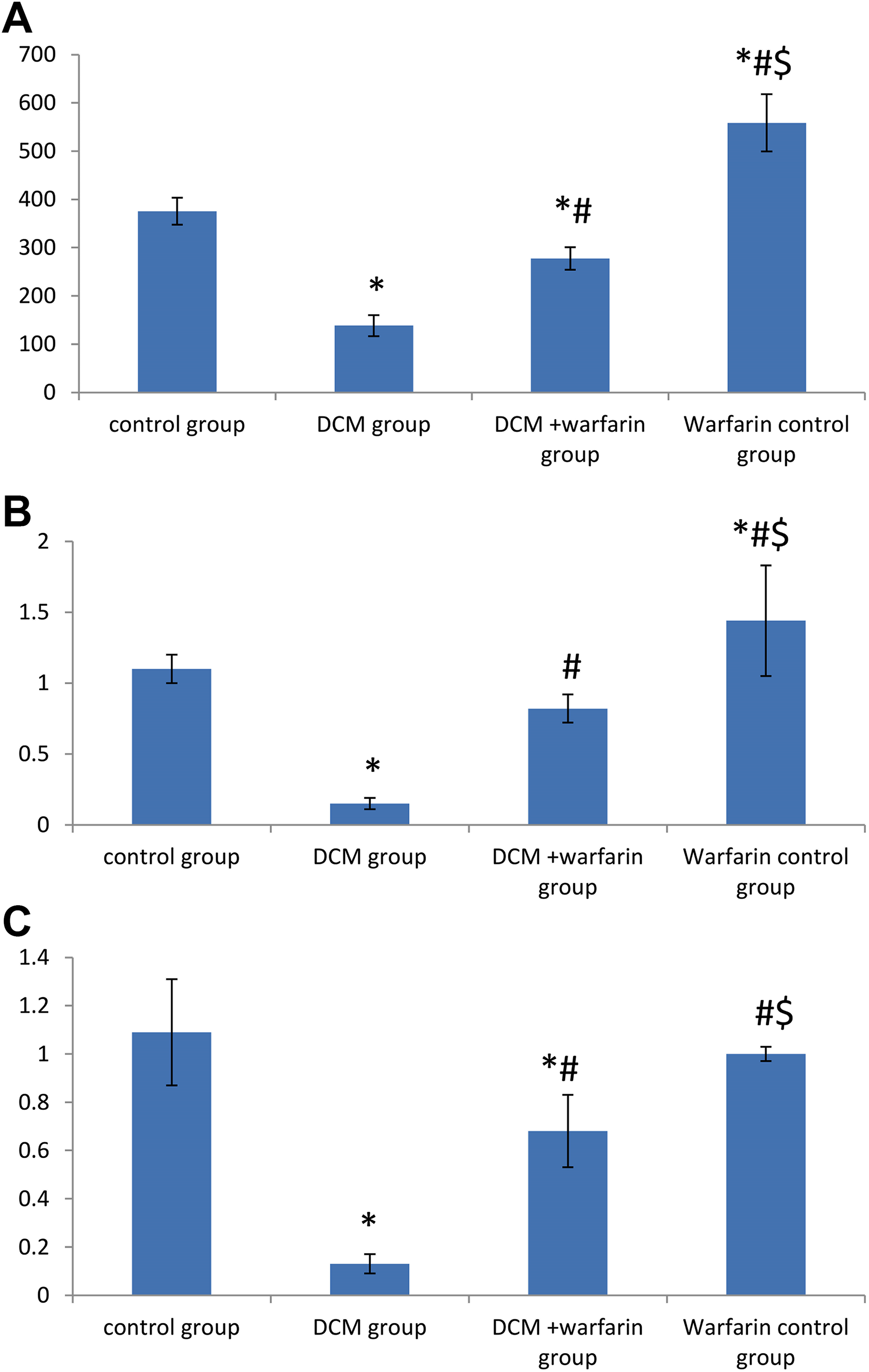
A, Serum undercarboxylated osteocalcin (ucOC; μg/mL); (B) osteocalcin receptor expression (GPrc6A); and (C) vascular endothelial growth factor (VEGF) gene expression in all studied groups. Values are represented as the mean ± SD (n = 8). *Statistically significant relative to the control group at P ≤ .05. #Statistically significant relative to diabetic cardiomyopathy (DCM) group at P ≤ .05.
Evaluation of Serum ucOC in Induced Diabetes
The elevation in serum levels of ucOC after warfarin injection was associated with a significant reduction in fasting glucose and insulin resistance and a significant increase in serum insulin. The same significant results occurred in normal rats receiving warfarin compared to the untreated control rats (Table 1).
Measurement of Fasting Serum Glucose, Serum Insulin, and HOMA-IR (Homeostasis Model Assessment-Insulin Resistance) in All Studied Groups.a

Abbreviations: DCM, diabetic cardiomyopathy; HOMA-IR, homeostasis model assessment-insulin resistance; SD, standard deviation.
a Values are represented as the mean ± SD (n = 8).
b Statistically significant relative to the control group at P ≤ .05.
c Statistically significant relative to the DCM group at P ≤ .05.
d Statistically significant relative to the DCM + warfarin group at P < .05.
Results of Cardiac Function Assessment
Measurement of LVDP, ΔP/ΔT, and −ΔP/ΔT with the Langendorff apparatus revealed deterioration in both systolic functions (presented by a significant reduction in LVDP and ΔP/ΔT) and diastolic function (presented by −ΔP/ΔT) in the DCM group, confirming the occurrence of DCM (Figure 3). Increased ucOC in the DCM-warfarin group was associated with significant elevation in LVDP, ΔP/ΔT, and −ΔP/ΔT. The warfarin control group showed nonsignificant differences in LVDP, ΔP/ΔT, and −ΔP/ΔT relative to the untreated control group.

Mean ± standard deviation (SD) of cardiac functions in all studied groups (A-C). A, Left ventricular developed pressure (LVDP; mm Hg); (B) maximum rate of pressure rise (ΔP/ΔT); (C) maximum rate of pressure decline (−ΔP/ΔT). Samples of Powerlab recordings (D-G): (D) a rat from the control group; (E) diabetic cardiomyopathy (DCM) group; (F) DCM + warfarin group; and (G) warfarin control group. The upper channel is the recording of intraventricular pressure. The lower channel shows the positive and negative rates of change per unit time (ΔP/ΔT). *Statistically significant relative to the control group at P ≤ .05. #Statistically significant relative to the DCM group at P ≤ .05.
Changes in VEGF Gene Expression in Cardiac Tissue
Induction of DCM resulted in a significant decrease in VEGF expression (Figure 2). A significant increase in VEGF expression was observed in the DCM-warfarin group compared to the untreated group. Warfarin control rats showed no significant change in VEGF expression relative to control rats. In addition, correlation studies showed that VEGF was positively correlated with serum ucOC (r = .771, P < .05; Figure 4). The VEGF was also positively correlated with cardiac function parameters: LVDP, ΔP/ΔT, and −ΔP/ΔT (r = .799, P < .05; r = .872, P < .05; r = .890, P < .05; respectively).

Correlation between (A) vascular endothelial growth factor (VEGF) gene expression in cardiac tissue and serum undercarboxylated osteocalcin (ucOC). B, VEGF expression in cardiac tissue and maximum rate of pressure increase (ΔP/ΔT). C, VEGF expression in cardiac tissue and maximum rate of pressure decline (−ΔP/ΔT). VEGF and left ventricular developed pressure (LVDP) in all studied groups.
Pathological Changes in Diabetic Rats
The myocardium of the DCM group has revealed marked myofibrillar degeneration with the presence of karyolitic nuclei and few intervening blood capillaries. This degenerative appearance was significantly mitigated in the DCM-warfarin group, where the cardiomyocytes displayed longitudinally arranged branching and anastomosing cardiomyocytes with only a few localized areas of myofibrillar loss. The cardiac muscle nuclei of this group appeared oval, vesicular, and centrally located. The myofibrils were seen separated by interfiber spaces containing thin elongated nuclei of fibroblasts and endothelial-lined blood capillaries. The warfarin control group displayed a histological structure comparable to that of the control group (Figure 5).

Histopathological changes in the cardiac muscle. A, Control group. B, Diabetic cardiomyopathy (DCM) group showing multiple foci of cardiomyocyte disarray (circles), karyolitic nuclei (white arrows), and few interfibrillar capillaries (arrow head). C, DCM + warfarin group showing branching and anastomosing cardiomyocytes with few localized areas of myofibrillar loss (circle) and multiple blood capillaries (arrow heads). D, Warfarin control group showing healthy cardiac muscle fibers with central, oval vesicular nuclei (black arrow). The myofibrils are seen separated by narrow interfiber spaces containing thin elongated nuclei of fibroblasts and endothelial-lined blood capillaries (arrow head). E, Number of foci of myocardial disarray (n = 6). F-I, Caspase 3-stained sections in the different groups displaying strong positive cytoplasmic caspase 3 immunoreactivity in the DCM group which is significantly ameliorated in the cardiomyocytes of the DCM + warfarin group. J, Area percentage of caspase 3 immunoreactivity (n = 6). *Statistically significant relative to the control group. #Statistically significant relative to the DCM group (scale bar 50 µm).
Examination of the Masson Trichrome-stained sections of the DCM group revealed a marked increase in collagen deposition, where the dense collagen bundles were seen intervening between the myofibrils and surrounding the blood vessels and the covering perimysium. The DCM-warfarin group, on the other hand, showed a statistically significant reduction in collagen deposition with scant collagen fibers in the perivascular and interfibrillar spaces. The warfarin-only group displayed collagen deposition akin to the control group (Figure 6).
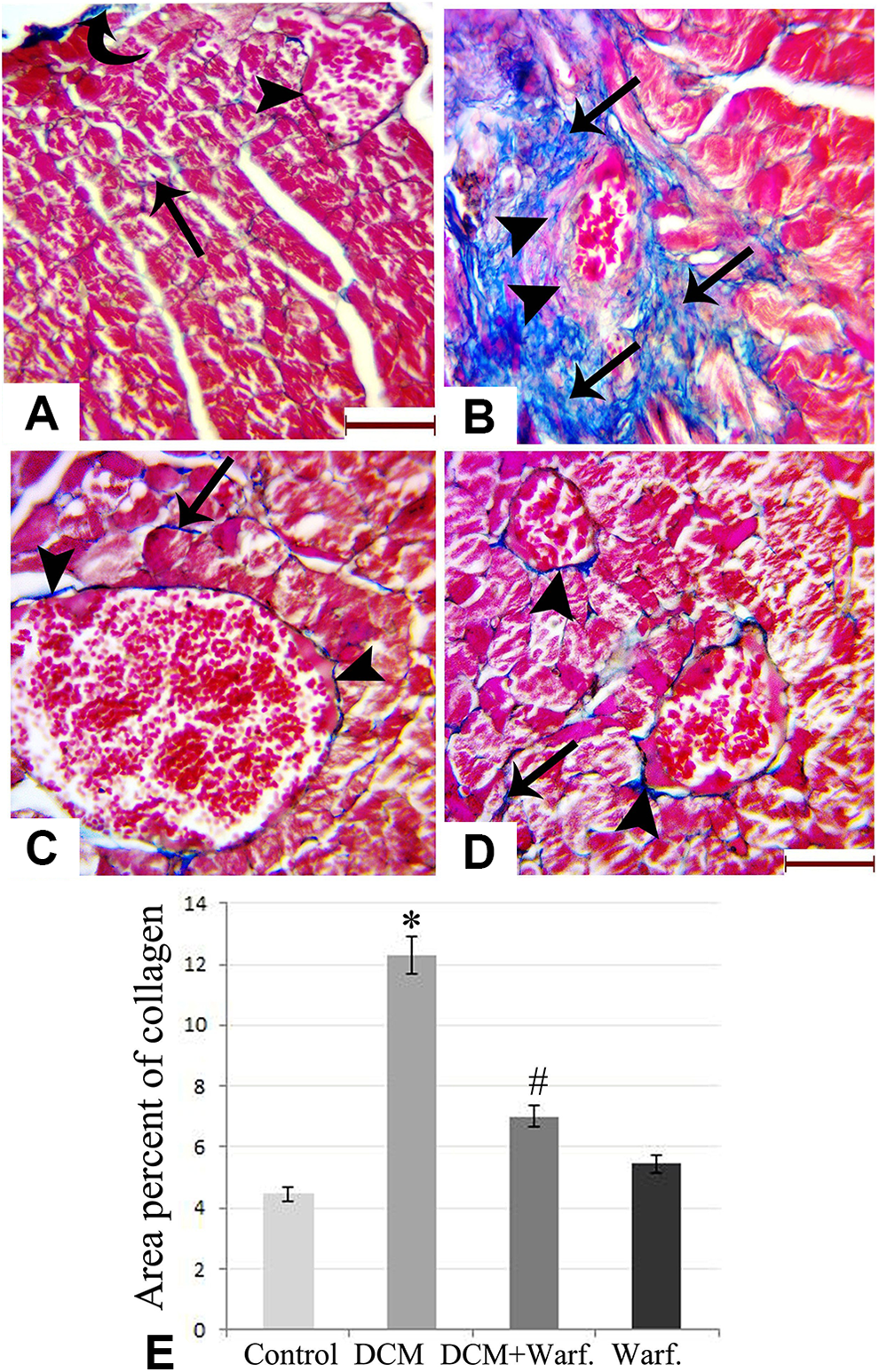
Masson Trichrome-stained sections of (A) control group; (B) diabetic cardiomyopathy (DCM) group showing extensive areas of blue-stained collagen bundles in the perivascular and interfibrillar spaces; (C) DCM + Warfarin group showing minimal collagen fibers surrounding the cardiomyocytes and the myocardial blood vessels; (D) warfarin control group; (e) area percentage of collagen in the different groups (n = 6) showing statistically significant increment in collagen deposition in the DCM group and significant amelioration in the DCM + Warfarin group. *Significant relative to the control group. #Significant relative to the DCM group (curved arrow: collagen in the perimysium; arrow heads: perivascular collagen; arrows: interfibrillar collagen; Scale bar 30 µm).
Changes in Myocardial Microvasculature and Capillary Density
The current study recorded a statistically significant reduction in VEGF and CD31 immunohistochemical expression in the DCM group. This defective neoangiogenesis was significantly improved with warfarin treatment (Figures 6 and 7).
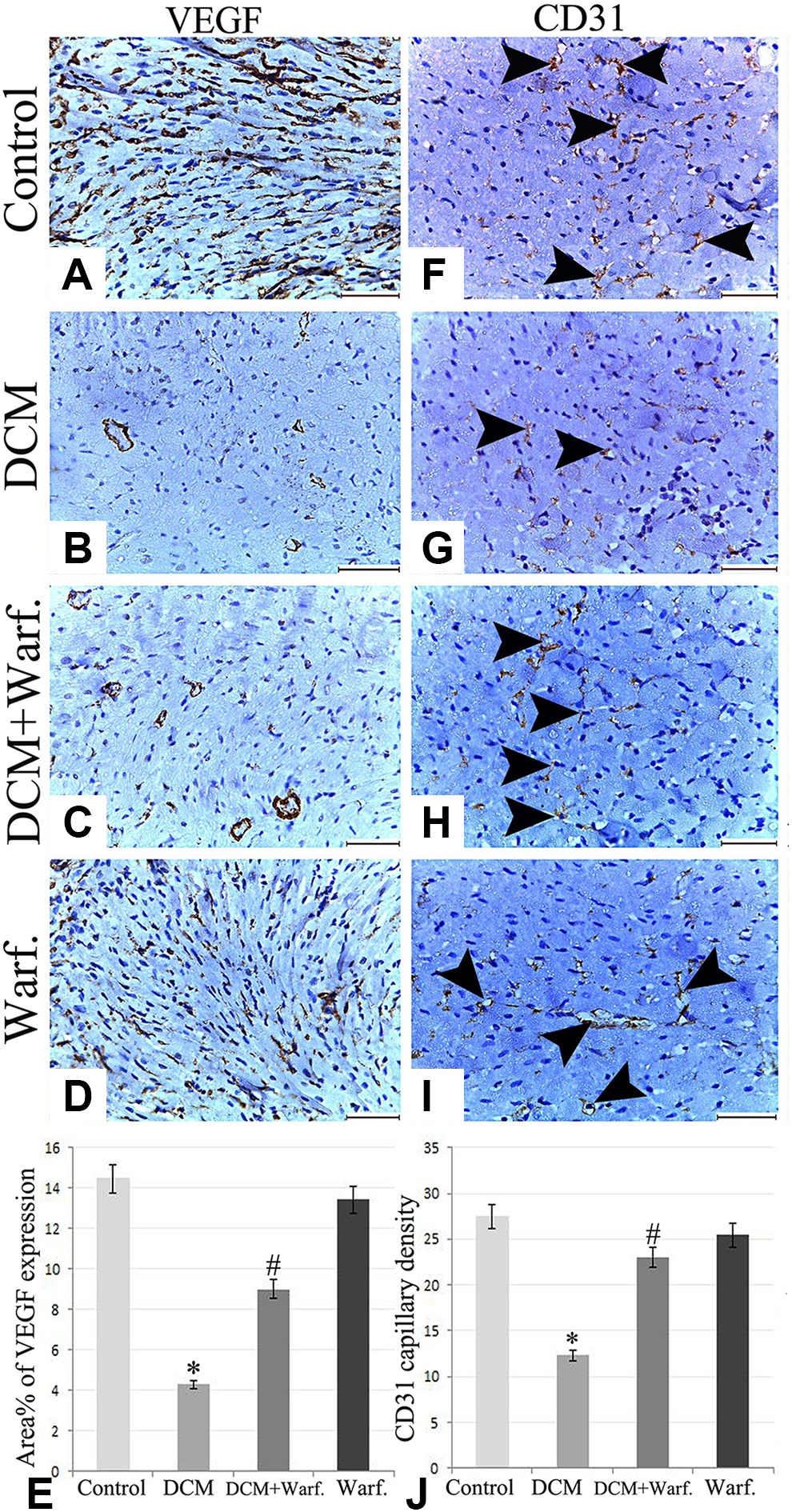
A-D, Immunohistochemical expression of vascular endothelial growth factor (VEGF) in the different groups showing obvious microvascular insufficiency with diabetic cardiomyopathy (DCM) and restoration of VEGF immunoreactive microvessels in the warfarin treatment (scale bar 50 µm); (E) area percent of VEGF in the different groups (n = 6); (F-I) Immunohistochemical expression of CD31 in the different groups (scale bar 50 µm; arrow heads: CD31 immunohistochemically stained capillaries); (J) CD31 capillary density measured by estimating the count of CD31 immunohistochemically positive capillaries in 8 rats at magnification ×400. *Statistically significant relative to the control group. #Statistically significant relative to the DCM group.
Myocardial Ultrastructural Results
Electron microscopic examination of the control group revealed regular strands of parallel longitudinal cylinders of myofibrils disposed into repeated contractile units of successive sarcomeres with clear cross-banding (alternating dark [A] and light [I] bands bisected by Z lines with the center of each A band occupied by a pale H zone crossed by a thin M line). Some T tubules were seen between the myofibrils near the Z lines. Regularly arranged rows of mitochondria with closely packed cristae were seen in between the myofibrils.
The cardiac muscles of the DCM group showed marked attenuation and separation of myofibrils, disrupted intercalated disks, and areas of fibrillolysis with indistinguishable A-bands, I-bands, and Z-lines. Markedly swollen mitochondria with degenerated cristae and ruptured membranes were frequently observed.
The warfarin-treated diabetic rats displayed a statistically significant reduction in the degenerated mitochondria with preservation of the architectural pattern of the cardiac myocytes and intercalated disks. The normal-warfarin group showed results comparable to those of the control group with healthy sarcomeres, mitochondria, and transverse and longitudinal parts of the intercalated disks (Figure 8).
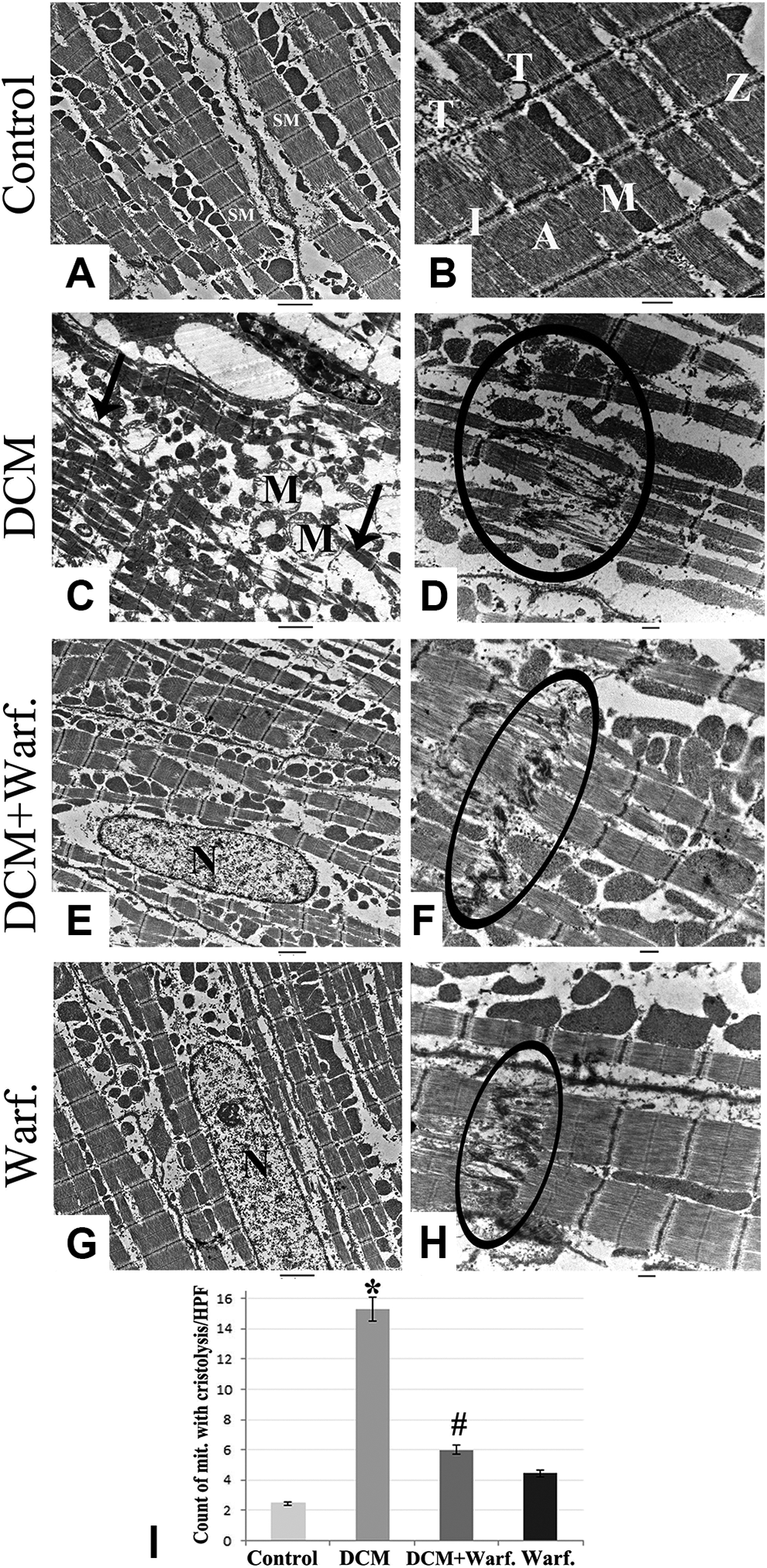
Electron microscopic picture of the different groups showing (C) degenerative changes in myocardium of the diabetic cardiomyopathy (DCM) group with marked attenuation of cardiac myofibrils (arrows) and vacuolated mitochondria (M). (E) DCM-Warfarin treated group showing restoration of the normal organization of the sarcomere, nuclei (N). G, Warfarin-only group showing normal arrangement of myofibrils with regular alignment of cross striations and healthy nuclei (N) and M. B, D, F, H, Demonstration of the intercalated disks (circles) in the different groups showing intercalated disk disruption with DCM and restoration of its regular architecture with warfarin treatment. I, Count of mitochondria with cristolysis per high-power field (HPF; n = 8) and disruption of intercalated disc (circle). *Statistically significant relative to the control group. #Statistically significant relative to the DCM group. (A, C, E, G: scale bar 2 µm; B, D, F, H: scale bar 500 nm). SM indicates sarcomere; A, A bands; I, I bands; Z, Z lines; T, Tubules.
Discussion
The mechanisms implicated in ameliorating the DCM-induced microvascular insufficiency have attracted considerable attention 31,32 ; thus, the current study has been designed to elucidate the therapeutic potential of osteocalcin in treating the cardiac microvascular and pathological changes induced in DCM by increasing the ucOC level in blood with warfarin. Previously, different methods, such as transgenic mice (Esp−/−) or recombinant ucOC, and different pharmaceutical agents, including warfarin, β blockers, vitamin K inhibitors, and statins, were suggested to modulate the level of ucOC. 33 In our study, we used parenteral warfarin that directly targets carboxylase, followed by ucOC upregulation.
In a previous study, we concluded that the use of ucOC as a prophylactic agent can partially prevent the occurrence of DCM through its antioxidant and antiapoptotic properties via the adiponectin pathway. 18 The present study, on the other hand, illustrates the role of ucOC as a therapeutic agent in modulating microvascular insufficiency and the subsequent cardiac functional and histopathological changes occurring after the complete establishment of DCM.
The UcOC was shown to improve both systolic and diastolic cardiac dysfunctions associated with DCM. This improvement was manifested by significant reduction in LVDP, ΔP/ΔT, and −ΔP/ΔT and coincides with the hypoglycemic and insulin-sensitizing effects observed with warfarin administration in the current and previous studies, 34 -37 since hyperglycemia is responsible for major functional changes in the myocardium with a gradual reduction in cardiac mass over time. 38,39
The present work demonstrated that DCM-induced functional dysfunction was associated with various histopathological changes, including interstitial and perivascular collagen accumulation. This nonconducting fibrillar collagen network essentially leads to a progressive increase in ventricular stiffness, thereby compromising cardiac output and eventually causing substantial impairment of cardiac function. 40 -42 Moreover, the intricate interplay between myocardial fibrosis and microvasculature also arises from the fact that cardiac interstitial fibrosis acts as a barrier to a good blood supply to the cardiomyocytes, thereby diminishing oxygen diffusion, increasing local hypoxia, and finally decreasing the viability of collagen surrounding the cardiomyocytes. 43 -47
A comprehensive look at the histomorphological features of the current study elucidates that the upregulation of OC after warfarin administration has a potential role in the reconstitution of myocardial architecture together with microvasculature recruitment and amelioration of fibrosis. These data were supported by increased VEGF and CD31 expression in the myocardium. Data from the current study also illustrated a statistically significant reduction in capillary density and VEGF expression in diabetic myocardial tissue. This finding is in accordance with previous experimental and clinical studies elucidating that defective angiogenesis together with VEGF attenuation are seminal events in the pathogenesis of DCM, and the restoration of local VEGF in diabetic heart may result in improved capillary density and myocardial perfusion. 1,11,26
The improvement in neoangiogenesis with warfarin treatment noted in the present study, together with the positive correlation observed between VEGF and cardiac functional parameters, suggests that VEGF overexpression can be one of the mechanisms by which ucOC can improve cardiac functions. These observations are consistent with previous animal models and clinical studies illustrating the role of microcirculatory defects in the development of cardiomyopathy. 48
Ultrastructurally, the cardiomyocytes of the diabetic rats showed marked attenuation and separation of myofibrils, areas of myofibrillar fragmentation together with swelling, cristolysis, and disorganization of mitochondria. These findings could be attributed to the effect caused by the hyperglycemic state on progressive protein glycation and the formation of advanced nonenzymatic glycation end products in intracellular and extracellular proteins, lipids, and nucleic acids of the myocardium. 49,50 Moreover, hyperosmolarity accompanying hyperglycemia essentially results in shrinkage of ventricular cardiomyocytes and increased myofibrillar separation. 51 Therefore, the reconstitution of myocardial structure noticed with warfarin could thus be attributed to its improving effect on hyperglycemia, which is represented in our work by a significant decrease in fasting glucose and insulin resistance presented by HOMA-IR.
Finally, the relationship between overexpression of the ucOC receptor (GPRC6A) and its effect on microvascular improvement through targeting VEGF still warrants further investigation. Also, it is better to use ucOC instead of parenteral warfarin in further studies for more evident results.
Conclusion
Diabetic cardiomyopathy is essentially a combination of degenerative cardiomyocytes in the context of myocardial fibrosis and microvascular deficiency. The suggested impact of osteocalcinon myocardial replenishment in the current study may be through re-establishment of myocardial microvasculature, upregulation of VEGF expression at both the molecular and the tissue levels, recruitment of capillary density, improvement in myocardial functions, and attenuation of myocardial fibrosis together with reconstitution of myocardial structural and ultrastructural patterns. This finding adds evidence to a new strategy for treating the myocardial degeneration and microvascular insufficiency associated with DCM, thereby improving its complications.
Footnotes
Authors’ Note
The work was performed in the experimental animal care unit of Cairo Medical University.
Acknowledgments
The authors thank Mrs Afaf M. Afify and Mrs Azza Kamal for their technical support.
Author Contribution
Nermeen B.Sadek, shared in designing the protocol of the work, carried out statistical analysis with interpretation of data, wrote and revised the manuscript, Sarah M. Gamal, performed the experimental work, wrote and revised the manuscript. Basma E. Aboulhoda, performed the histopathological analysis with data interpretation, wrote and revised the manuscript. Laila A. Rashed, performed the biochemical analysis, wrote and revised the manuscript. Heba M. Shawky, carried out data analysis, wrote and revised the manuscript. Maha M. Gamal El-Din, designing the protocol of the work, wrote and revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
