Abstract
Vascular complications are a leading cause of mortality and morbidity in diabetic patients. Herbal drugs are increasingly being used in the treatment of cardiovascular disorders. The present study was designed to examine the therapeutic potential of Terminalia arjuna bark extract in improving myocardial function in streptozotocin (STZ)-induced diabetic rats. After 8 weeks of STZ administration, rats showed a decline in left ventricular pressure (LVP), maximal rate of rise and fall in LVP (LV [dP/dt] max and LV [dP/dt] min), cardiac contractility index (LV [dP/dt] max/LVP), and rise in LV end-diastolic pressure. Altered lipid profile, oxidative stress, and increased levels of endothelin 1 (ET-1), tumor necrosis factor-α (TNF-α), and interleukin 6 (IL-6) along with histological changes in heart and pancreas were observed in diabetic rats. T arjuna significantly attenuated cardiac dysfunction and myocardial injury in diabetic rats. It also reduced oxidative stress, ET-1, and inflammatory cytokine levels. The decreased body weight, heart rate blood pressure, and raised blood sugar in diabetic rats did not improve after T arjuna therapy. Results suggest that T arjuna bark extract improves the altered myocardial function in diabetic rats possibly through maintaining endogenous antioxidant enzyme activities, decreasing ET-1 and cytokine levels.
Introduction
Diabetic individuals have a significantly increased likelihood of developing cardiovascular disease. Diabetes inflicts a direct insult to the myocardium, manifesting as cellular, structural, and functional changes. 1,2 Diabetic cardiomyopathy is a clinical condition diagnosed when ventricular dysfunction develops in patients with diabetes in the absence of coronary atherosclerosis and hypertension. This diabetic cardiomyopathy has been postulated to contribute to both the higher incidence of congestive heart failure and the long-term mortality after myocardial infarction in diabetic patients. 3,4 Diabetic cardiovascular complications result from imbalances caused by increases in the toxic effects of systemic metabolic abnormalities such as hyperglycemia, dyslipidemia, formation of oxidants, endothelins, and reductions in the regenerative effects of endogenous protective factors such as insulin, nitric oxide (NO), and antioxidant enzymes. 1 New therapeutic approaches for diabetic cardiovascular complications need to focus on decreasing the toxic factors of diabetes as well as increasing the action of protective factors.
Diabetes-induced cadiomyopathies due to hyperlipidemia are usually treated by hypolipidemic drugs like statins. 5 Statins or hydroxymethylglutaryl CoA reductase inhibitor use is associated with reduced all-cause mortality in heart failure and in patients with nonischemic and ischemic cardiomyopathy. Statins exert additional beneficial pleiotropic effects independent of their lipid-lowering action, such as free radical-scavenging activity, anti-inflammatory, antithrombotic, and antioxidant actions, and improved endothelial function. 6,7 However, there is evidence suggesting adverse effects of the long-term use of statins. 8 Concurrently phytochemicals identified from traditional medicinal plants showed multiple beneficial effects in combating diabetes and the related complications without causing known side effects.
Terminalia arjuna (T arjuna, Combretaceae) is an important medicinal plant widely used in medicinal formulations for several ailments. Various bioactive compounds, like triterpinoids, tannins, flavonoids, and minerals have been isolated from the stem bark. 9 A number of clinical studies have demonstrated its cardioprotective properties, ranging from positive inotropic, hypolipdemic, coronary vasodilatory, and antioxidant effects. 10 -13 However, no evidence is available on the ability of T arjuna bark to improve myocardial function in diabetes-induced cardiomyopathy. Hence, the present study was designed to evaluate the therapeutic efficacy of T arjuna on myocardial function in animal model of STZ-induced diabetic cardiomyopathy and to elucidate the possible mechanisms by which it acts.
Materials and Methods
Animals
Healthy male Wistar Albino rats, weighing between 250 and 300 g, were obtained from the animal house of University College of Medical Sciences (UCMS), Delhi. They were housed in polyethylene cages and kept at room temperature maintained at 25°C ± 2°C with a 12-hour light–dark cycle and food and water given ad libitum. Experiments were performed according to the guidelines of the Committee for the purpose of control and supervision of experiments on Animals (CPCSEA), India, after approval by Institutional animal ethical committee, UCMS, Delhi, India.
Chemicals
Streptozotocin (STZ) and urethane were purchased from Sigma Chemical Co, St Louis, Missouri, USA. Rosuvastatin was procured from Ranbaxy Pharmaceuticals, India. All other chemicals and reagents used in the study were of analytical grade.
Bark Extract of T Arjuna
Bark extract was procured from Jamia Hamdard, Delhi, India. The bark of T arjuna was used in the form of 50% aqueous ethanol extract. The shade-dried bark was coarsely powdered and extracted with ethanol (50%) using soxhlet apparatus. The extract was then filtered and concentrated to obtain the solid residue. The yield of the total aqueous ethanolic extract was 28.76%. Primary phytochemical screening of the ethanolic extract of T arjuna bark revealed the presence of glycosides, triterpenoids, phenols, flavonoids, tannins, and saponins.
Induction of Diabetes With STZ
Diabetes was induced by a single intraperitoneal (ip) injection of STZ (65 mg/kg; Sigma Chemical Co) dissolved in citrate buffer. 14 This dose induces type 1 diabetes mellitus in animals. The control animals were injected with equal volume of vehicle. After 7 days of STZ administration, blood was collected and serum samples were analyzed for blood glucose with Accu-chek glucometer (Roche Diagnostics, India). Animals showing a blood glucose level higher than 250 mg/dL were considered as diabetic rats and were used for the study.
Grouping of Animals
Animals were randomly divided into 7 groups, each having 7 rats. Group 1: normal control rats injected with vehicle (citrate buffer) and fed normal pellet diet (control group); group 2: rats were injected STZ ip and kept for 8 weeks (diabetic group); group 3: nondiabetic rats given rosuvastatin (20 mg/kg) by gavage for 30 days (rosuvastatin control); group 4, STZ-diabetic rats treated with rosuvastatin (20 mg/kg) for 30 days after 8 weeks of STZ administration (therapeutic rosuvastatin); group 5: nondiabetic rats given extract of T arjuna bark (500 mg/kg body weight) by gavage for 30 days (T arjuna control); group 6: STZ-diabetic rats treated with extracts of T arjuna bark (500 mg/kg body weight) by gavage for 30 days after 8 weeks of STZ administration (herapeutic T arjuna); and group 7: STZ-diabetic rats treated with Neutral protamine hagedorn (NPH, Ranbaxy pharmaceuticals, India) 6 to 8 IU/d subcutaneously for 30 days after 8 weeks of STZ administration (therapeutic insulin). Initial doses of insulin (daily) were started with 2 IU and depending on the extent of glycemic control, the dosage was adjusted for each rat. The animals were weighed weekly.
Hemodynamic Measurements
Rats were anesthetized with urethane dissolved in distilled water and injected ip at a dose of 1 gm/kg body weight. Disappearance of pedal reflexes indicated adequate anesthesia. To allow free air breathing, tracheostomy was done. Body temperature of the rat was maintained at 37°C to 38°C. Femoral artery of one side was exposed and a polyethylene catheter filled with heparin solution (500 IU/mL, v/v) was inserted into the artery through a small incision, for recording arterial blood pressure (ABP). The catheter was attached to a 23-gauge needle connected via a 3-way stopcock to a pressure transducer (Statham-P23D, Oxnard, California). Femoral vein of the other limb was cannulated to maintain fluid volume and take blood samples. Prior to recording ABP, the catheter was flushed with heparinized saline solution (500 IU/mL, v/v) to prevent the formation of any blood clot that might interfere with normal recording of ABP. The pressure recording system was calibrated with a mercury manometer before each experiment. Arterial BP was measured after 20 minutes of stabilization. Systolic, diastolic, mean arterial pressures, and heart rate were displayed and recorded on Power Lab data-acquisition system (4SP, AD Instruments, Australia) with a computerized analysis program (Lab Chart 7, AD Instruments).
To evaluate cardiac left ventricular function, a micromanometer-tipped catheter (Millar Instruments, Houston, Texas) was placed in the left ventricle via the right carotid artery to record the left ventricular pressure (LVP). The maximum and minimum rates of developed LVP (LV [dP/dt] max and LV [dP/dt] min) were recorded online by differentiating LVP. Left ventricular end-diastolic pressure (LVEDP) and maximal rate of rise of LVP divided by LVP (LV [dP/dt] max/LVP) were measured offline from the LVP data.
Biochemical Estimations
After completion of hemodynamic studies, blood samples were collected. Serum was subsequently harvested by centrifuging and stored at −20°C for biochemical assays. Lipid profile—triglyceride, cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), lipid peroxidation marker malondialdehyde (MDA), reduced glutathione (GSH), superoxide dismutase (SOD), catalase and inflammatory marker tumor necrosis factor-α (TNF-α), interleukin 6 (IL-6), and endothelin 1 (ET-1) were measured in serum using appropriate kits.
Histopathological Examination
The tissues (heart, aorta, and pancreas) were fixed in 10% formalin solution for histopathological analysis. Formalin-fixed tissues were embedded in paraffin, sectioned at 4µm and stained with hematoxylin and eosin (H&E). The sections were examined under light microscope and photomicrographs were taken.
Statistical Analysis
Analysis was done on SPSS 20.0 statistical package. The data are presented as mean ± standard error of the mean (SEM). The groups were compared by 1-way analysis of variance (ANOVA) with Tukey test at 5% level of significance. P value of less than .05 was considered to be significant.
Results
All the rats that received STZ became diabetic. The mortality rate was 20% in diabetic rats. The rats died within 1 week of STZ administration.
Body Weight and Blood Sugar
Body weights (253.57 ± 4.52 g) and blood sugar (84.8 ± 6.5 mg%) were similar in all the groups at baseline. The body weights of 8-week diabetic rats were significantly lower (185.71 ± 5.91 g) and blood sugar (550.57 ± 23.65 mg%) was significantly higher than that of nondiabetic controls. There was no improvement in body weight and blood sugar levels after 1 month of T arjuna or rosuvastatin treatment. While body weight (252.14 ± 4.61 g) and blood sugar (76.43 ± 7.25 mg%) reached their baseline levels after 1 month of insulin therapy.
Hemodynamic Analysis
Arterial BP and Heart Rate
Diabetic rats had significantly lower systolic BP (105.85 ± 3.14 vs 118.85 ± 2.78 mm Hg), diastolic BP (67.57 ± 2.57 vs 80.28 ± 2.03 mm Hg), mean BP (80.33 ± 2.71 vs 93.14 ± 2.04 mm Hg), and heart rate (292 ± 13.31 vs 345.42 ± 12.56 beats/min) as compared to normal controls. The systolic BP (116.85 ± 2.26), diastolic BP (77.71 ± 2.11), mean BP (90.76 ± 2.13), and heart rate (339.85 ± 6.33) increased significantly after insulin therapy and was similar to its values in normal controls. These parameters did not change after T arjuna or rosuvastatin therapy.
Left Ventricular Function
In comparison to the control group, STZ treatment resulted in left ventricular dysfunction as indicated by a significant (P < .05) rise in LVEDP, fall in values of maximum and minimum rates of developed LVP (LV [dP/dt] max and LV [dP/dt] min) and reduced myocardial contractility index LV (dP/dt) max/LVP (Figure 1). Therapeutic treatment with T arjuna significantly improved (P < .05) the left ventricle function as compared to STZ group. Similar effect was seen with rosuvastatin and insulin treatments. Treatment with T arjuna or rosuvastatin alone did not have any significant effect on left ventricular function (Figure 1). No significant differences in left ventricular function were observed between T arjuna and rosuvastatin treatments.
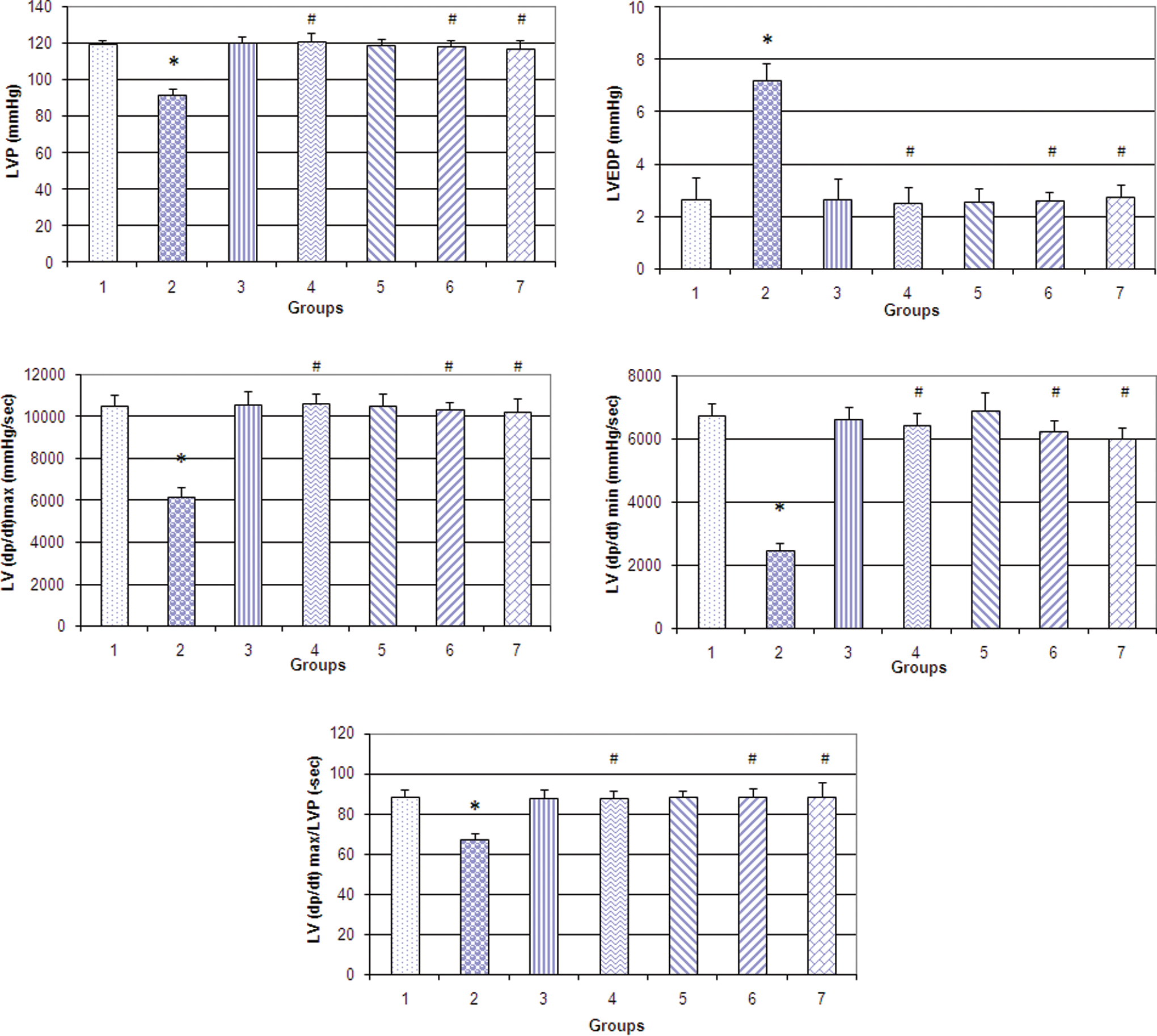
Left ventricular functions. LVP (A), LVEDP (B), LV (dP/dt) max maximal rate of rise of left ventricular pressure (C), LV (dP/dt) min maximal rate of fall of LVP (D), LV (dP/dt) max/LVP maximal rate of rise of LVP divided by LVP (E) in control (group 1), diabetic (group 2), rosuvastatin control (group 3), therapeutic rosuvastatin (group 4), Terminalia arjuna control (group 5), therapeutic T arjuna (group 6), and therapeutic insulin (group 7). *P < .05 vs group 1. #P < .05 vs group 2. LVP indicates left ventricular pressure; LVEDP, left ventricular end-diastolic pressure.
Biochemical Estimations
A significant (P < .001) rise in the content of MDA as well as a significant decline in the activity of GSH (P < .004), SOD (P < .001), and catalase (P < .006) was observed in the serum of STZ-treated rats compared with control (Table 1). Administration of T arjuna, rosuvastatin, or insulin markedly prevented all the STZ-induced alterations in antioxidant enzymes and maintained the rats at near normal status. Administration of T arjuna or rosuvastatin alone did not demonstrate any significant changes in the biochemical parameters and antioxidant enzymes when compared with the control group, indicating T arjuna and rosuvastatin did not have any significant effect.
Oxidative Stress Parameters (Catalase, MDA, GSH, and SOD) and Inflammatory Cytokines (TNF-α, Interleukin 6) in Control (Group 1), Diabetic (Group 2), Rosuvastatin Control (Group 3), Therapeutic Rosuvastatin (Group 4), T arjuna Control (Group 5), Therapeutic T arjuna (Group 6), and Therapeutic Insulin (Group 7).a

Abbreviations: MDA, malondialdehyde; GSH, reduced glutathione; SOD, superoxide dismutase; SEM, standard error of the mean.
a All values are expressed as mean ± SEM (n = 7).
b P < .05 vs group 1.
c P < .05 vs group 2.
Compared with the control group, STZ-treated rats showed a significant rise in the serum TNF-α (P < .008) and IL-6 (P < .000) levels (Table 1). Therapeutic treatment with T arjuna, rosuvastatin, or insulin significantly reduced the serum TNF-α and IL-6 to near normal levels in STZ-treated rats. Administration of T arjuna or rosuvastatin alone did not show any effect on serum TNF-α and IL-6 levels.
The STZ-treated rats showed a significant rise in the serum ET-1 levels (P < .005; Figure 2). Therapeutic treatment with T arjuna, rosuvastatin, or insulin significantly reduced the serum ET-1 to near normal levels in STZ-treated rats. Administration of T arjuna or rosuvastatin alone did not show any effect on ET-1 levels.

Endothelin 1 levels in control (group1), diabetic (group 2), rosuvastatin control (group 3), therapeutic rosuvastatin (group 4), Terminalia arjuna control (group 5), therapeutic T arjuna (group 6), and therapeutic insulin (group 7). All values are expressed as mean ± standard error of the mean (n = 7). *P < .05 vs group 1. #P < .05 vs group 2.
Serum Lipid Levels
Compared with the control group, STZ-treated rats showed a significant (P < .05) increase in total cholesterol, triglycerides, and LDL-C and significantly (P < .05) decreased the level of HDL-C (Figure 3). Diabetic rats treated with T arjuna, rosuvastatin, or insulin showed a significant (P < .05) fall in the level of total cholesterol, triglycerides, and LDL-C and a significantly (P < .05) increased the level of HDL-C compared with the STZ-treated group (Figure 3). Administration of T arjuna or rosuvastatin alone to normal rats did not have any significant effect on serum lipid profile.

Lipid profile. Cholesterol (A), triglyceride (B), LDL-C (C), and HDL-C (D) in control (group 1), diabetic (group 2), rosuvastatin control (group 3), therapeutic rosuvastatin (group 4), Terminalia arjuna control (group 5), therapeutic T arjuna (group 6), and therapeutic insulin (group 7). All values are expressed as mean ± standard error of the mean (n = 7). *P < .05 vs group 1. #P < .05 vs group 2. LDL indicates low-density lipoprotein; HDL, high-density lipoprotein.
Histopathological Examination
Heart
Histopathological examination of heart tissues from STZ-treated rats showed widespread alterations in myocardial structure with subendocardial necrosis and vacuolation of the myocytes, indicating cardiomyopathy (Figure 4) in comparison with the control group where no such structural alterations were observed. Therapeutic treatment with T arjuna, rosuvastatin, or insulin demonstrated marked improvement in STZ-induced subendocardial necrosis and vacuolation of the myocytes. Treatment with T arjuna or rosuvastatin alone did not show any structural changes in the heart tissue.

Hematoxylin and eosin stained sections of left ventricle in different animal groups. A, Control group showing normal appearance of cardiac myofibers. B, Diabetic group showing alteration in myocardial structure with subendocardial necrosis and vacuolation of myocytes. C, Therapeutic rosuvastatin group showing mild myovacuolations. D, Therapeutic Terminalia arjuna group showing mild myovacuolations. and E, therapeutic insulin group showing slight necrosis and myovacuolations (H&E; ×40). H&E indicates hematoxylin and eosin.
Aorta
In all groups, the aortic wall showed a uniform thickness with no bulging in the lumen, and the endothelial lining was intact without any interruption. Also, elastic lamina and muscle fibers appeared normal. No atherosclerotic changes were observed in the aorta after STZ administration in any of the therapeutic groups.
Pancreas
The normal tissue section of the pancreas show lobules of exocrine acini, interlobular ducts, and occasional islets of Langerhans. The islets were not observed after STZ administration, indicating the induction of diabetes. The islets did not reappear after T arjuna, rosuvastatin, or insulin therapy. Administration of T arjuna or rosuvastatin alone to normal rats did not have any significant effect on the histopathology of the tissues.
Discussion
In the present study all rats that received STZ became diabetic. The high levels of blood glucose confirmed the efficacy of STZ in producing experimental chronic (8 weeks) diabetes in rats. Streptozotocin is widely used to induce experimental diabetes in animals. As a result of the STZ action, DNA damage occurs and the islets undergo destruction by necrosis. 14 Rats treated with STZ display many features seen in human beings with uncontrolled diabetes, including hyperglycemia, hypoinsulinemia, increased urinary glucose levels, and consequently polyurea as well as weight loss. It is known that diabetes is associated with caloric deficiency that results in marked reduction in body weight. 15 In the present study, there was progressive fall in body weight and rise in blood sugar after STZ administration. These parameters showed recovery after insulin therapy but not after T arjuna or rosuvastatin therapy. Terminalia arjuna has previously been shown to have an antidiabetic effect in 1 study. 16 Our data do not support this finding. However, T arjuna provided cardioprotection in diabetic rats of our study. In the earlier study, T arjuna therapy was started immediately after induction of diabetes, while we started its treatment after 8 weeks of STZ induction. This could be one of the reasons for difference in the control of blood glucose.
In the present study, diabetic rats had significantly lower systolic, diastolic, mean BP, and heart rate than that of the normal controls. These parameters increased significantly to their baseline values after insulin therapy but remained unchanged after T arjuna or rosuvastatin therapy. The reduction in arterial pressure may be attributed to decreased cardiac output in diabetic rats due to hypovolemia caused by hyperglycemic osmotic diuresis. There are conflicting reports of BP in experimental diabetes. 11,17 Differences in rat strain and duration of diabetes could explain the conflicting data.
Eight weeks after STZ administration, there was a significant increase in LVEDP and a fall in inotropic (LV [dP/dt]max, index of myocardial contraction) and lusitropic (LV [dP/dt]min, index of myocardial relaxation) states of the heart. Fall in LV (dP/dt)max/LVP was also observed in STZ-treated rats. The LV (dP/dt)max/LVP is the maximal rate of rise in LVP divided by LV systolic pressure and is a close index of myocardial contractility. 18 Increased LVEDP indicates an increased preload or incomplete emptying of the left ventricle and impaired left ventricular performance, such as seen in diabetic cardiomyopathy. 17 Terminalia arjuna, rosuvastatin, and insulin appeared to preserve left ventricular function as evidenced by significant restoration of LV (dP/dt)max and LV(dP/dt)min and restoration of elevated LVEDP due to STZ challenge. Increase in LV (dP/dt)max, LV(dP/dt)max/LVP, and LV(dP/dt)min reflects an overall enhancement of myocardial contractility and relaxation, suggesting improvement in left ventricular dysfunction caused by STZ. Reduction in LVEDP implies that there is an increase in blood flow through the subendocardial region of the heart that bears the maximum brunt of the ischemic insult. Under ischemic conditions, there is a disproportionate reduction in blood flow to the subendocardial regions of the heart, which is subjected to the greatest extravascular compression during systole. The bark extract of T arjuna thus may indirectly tend to restore normal blood flow in these regions by reducing the elevated LVEDP, as reported earlier. 11 Rosuvastatin and insulin treatments improved the left ventricular function and hypertrophy, owing to their cardioprotective properties as reported in earlier studies. 5,19 Treatment with T arjuna bark extract improved cardiac performance, reflecting a cardioprotective effect.
Hyperglycemia results in the production of reactive oxygen and nitrogen species, which leads to oxidative myocardial injury. 20 These alterations are believed to result from acute cardiac responses to suddenly increased glucose levels at the early stage of diabetes. Oxidative stress causes abnormal gene expression, altered signal transduction, and the activation of pathways leading to programmed myocardial cell death. The resulting myocardial cell loss thus plays a critical role in the development of diabetic cardiomyopathy. Various strategies for targeting the prevention of hyperglycemia-induced oxidative myocardial injury may be fruitful. A significant rise in the level of MDA was observed in the serum of STZ-induced diabetic rats, which decreased significantly after T arjuna treatment. Our results show that oxidative stress plays an important role as MDA levels were increased in diabetes rats but reduced after treatment. Decreased activity of the antioxidant system, that is, GSH, SOD, and catalase, was observed in the serum of STZ-induced diabetic rats, which is in agreement with previous studies. 11,21 The altered balance of the antioxidant enzymes caused by decrease in catalase, SOD, and GSH activities may be responsible for the inadequacy of the antioxidant defenses in combating free radical-mediated damage. These enzymes have been suggested as playing an important role in maintaining physiological levels of oxygen and hydrogen peroxide by hastening the dismutation of oxygen radicals and eliminating organic peroxides and hydroperoxides generated by STZ administration. Treatment with T arjuna increased the activity of antioxidant enzymes, as T arjuna has been reported to be rich in flavonoids, well-known antioxidants, which scavenge the free radicals generated during diabetes. 11
There is increasing evidence that inflammation is involved in the pathophysiology of heart failure and diabetes. 22 Increased circulating concentrations of IL-6 and TNF-α were found in type 2 diabetes and impaired glucose tolerance. Metabolic changes induced by hyperglycemia lead to dysregulation of cytokine control, increasing their levels by an oxidative mechanism. In the present study, TNF-α and IL-6 were observed to be significantly increased in diabetic rats, which reverted to normal after rosuvastatin, T arjuna, and insulin therapy. Statin treatment significantly decreased the TNF-α and IL-6 levels owing to their anti-inflammatory property, confirming earlier studies. 23 Terminalia arjuna bark extract treatment reduced the serum TNF-α and IL-6 levels of diabetic rats, suggesting anti-inflammatory activity of the bark extract.
Dysfunction of the vascular endothelium is an early finding in the development of cardiovascular disease. Endothelin 1 has also been demonstrated to be associated with increased oxidative stress and endothelial dysfunction in humans. 24 Under physiological conditions, ET-1 is produced in small amounts, mainly in endothelial cells, primarily acting as an autocrine/paracrine mediator. Under pathophysiological conditions, however, the production is stimulated in a large number of different cell types, including endothelial cells, vascular smooth muscle cells, cardiac myocytes, and inflammatory cells such as macrophages and leukocytes. 24 In the present study, ET-1 was observed to be significantly increased in diabetic rats, which reverted to normal after rosuvastatin, T arjuna, and insulin therapy. The biological effects of ET-1 are transduced by 2 pharmacologically distinguishable receptor subtypes, ETA and ETB receptors. 25 In the vasculature, the ETA receptor is mainly located on vascular smooth muscle cells and mediates potent vasoconstriction. The ETB receptor is primarily located on endothelial cells but may also be present on vascular smooth muscle cells. Stimulation of the endothelial ETB receptor results in the release of NO and prostacyclin that cause vasodilatation, whereas stimulation of the vascular smooth muscle cell ETB receptor results in vasoconstriction. Thus, the net effect produced by ET-1 is determined on the receptor localization and the balance between ETA and ETB receptors. 24 In the present study, ET1 might be acting via endothelial ETB receptors as we observed a fall in BP with increased levels of ET1 in diabetic group. Apart from its direct vasomotor activity, ET-1 has been implicated in inflammatory processes within the vascular wall. Specifically, ET-1 in subnanomolar concentrations has been demonstrated to activate macrophages, resulting in the release of proinflammatory and chemotactic mediators, including TNF-α, IL-1, IL-6, and IL-8 which are of importance in the atherosclerotic process. 26 Possibly, release of IL-6 may further increase oxidative stress. Previous in vitro studies indicate that lipid-lowering treatment suppresses the expression of ET-1 in endothelial cells, 27 thereby attenuating the negative effect of ET-1 on endothelial function. Thus, the cardioprotective effect of T arjuna observed in the present study may be attributed to its lipid-lowering and ET-1-suppressing effects similar to statins.
In animal models of diabetes, several functional and structural alterations of the heart or cardiac muscle have been documented. In the present study, histopathological findings of STZ-treated heart demonstrated widespread alterations in myocardial structure, subendocardial necrosis, and vacuolation of the myocytes, indicating cardiomyopathy as reported in other studies. 28 Our findings support the previous observations that diabetic rats do not have histologic evidence of fibrosis or increased collagen content. 17 Rosuvastatin and insulin treatment therapeutically attenuated the STZ-induced cardiac alterations, which is in consonance with previously reported studies. 11,17 Terminalia arjuna and rosuvastatin alone groups exhibited normal cardiac fibers without any pathological changes, indicating that T arjuna and rosuvastatin did not have any significant effect on myocardium. Oral administration of T arjuna bark extract significantly alleviated the STZ-induced cardiac injury, confirming its cardioprotective actions.
No atherosclerotic changes were observed in aorta after STZ administration in any of the therapeutic groups. The apparent lack of increased atherosclerosis in the diabetic rats may be explained by conflicting protective and proatherogenic effects. It is well known that the distribution of cholesterol between lipoprotein fractions may modulate lesion formation. In particular, it has been demonstrated that diabetes associated hypertriglyceridemia reduces atherosclerosis in rabbits. 29 Our diabetic rats had markedly increased triglyceride levels. Thus, in our diabetic rats, a lipid profile of reduced atherogenic potential could have counterbalanced the proatherogenic effects of diabetes. Moreover, there may be differences in the susceptibility to proatherogenic effects of diabetes between murine strains. Significant lipid-lowering effect of T arjuna observed in the present study is consistent with the earlier reports. 30
Insulin was used as a positive control in the present study for maintaining the blood glucose levels. Cardiovascular actions of insulin play an important physiological role in coupling metabolic and cardiovascular homeostasis under healthy conditions. Several studies demonstrated that insulin can prevent, or even reverse, the derangements caused by chronic diabetes. 2,17,31 The mechanism responsible for this protective effect is still unknown because diabetes is a long-standing metabolic disorder with several outcomes. It has been demonstrated in normal cardiac myocytes that insulin speeds the glucose transport into the cell. However, it has also been demonstrated that insulin promotes a positive inotropic effect independent of glucose uptake. 2 In the present study, since insulin is regulating oxidative stress, levels of inflammatory markers, and ET-1, it can be said that insulin is improving cardiac function by mechanisms apart from controlling hyperglycemia.
Statins prevent cardiovascular disease via improving endothelial function, modulating inflammatory responses, plaque-stabilizing action, and preventing thrombus formation. 5 They also improved plasma cytokines and neurohormonal imbalance along with cardiac function in patients with dilated cardiomyopathy. 23 Despite benefits, statins have been reported to cause adverse effects like muscle toxicity, including myopathy and rhabdomyolysis, and effects on liver enzymes. 8 Development of standardized, safe, and effective herbal formulations as multitarget therapeutics and prophylaxis could be a tenable approach for the future. Statins, the standard hypolipidemic drugs were used in the present study to compare their cardioprotective actions with T arjuna (unknown natural product).
Current scientific research has proved that T arjuna contains specific medically active constituents namely triterpine glycosides, arjunine, and arjunetein. 13 Its bark is rich in saponnins, natural antioxidants (flavonoids–arjunone, arjunolone, and leteilin), and minerals. It is rich in co-enzyme Q-10 that is known to prevent heart problems. Regular use of T arjuna improves the pumping activity of heart, improves the cardiac muscle strength and autonomic control, and decreases the LDL cholesterol levels. 9,10,13,32 Thus, combined or independent action of several constituents might be responsible for the beneficial effects of T arjuna in various complications of diabetes. It may be compared with statins as a potential agent having similar pharmacological benefits.
Conclusion
The present study has demonstrated for the first time that oral administration of T arjuna bark extract improved the myocardial function in rats having uncontrolled diabetes for 8 weeks. Since T arjuna is not able to control hyperglycemia in our rats, the improvement observed is probably due to an antioxidant mechanism. Terminalia arjuna reduced the serum proinflammatory cytokines TNF-α, IL-6 levels, and ET-1 levels; this effect might have been attributed to reduction in oxidative stress. The bark extract of T arjuna may serve as an alternative therapeutic agent for the treatment of diabetic cardiomyopthy.
Footnotes
Acknowledgments
The authors would like to acknowledge Professor M. S. Y. Khan, Jamia Hamdard (India) for providing Terminalia arjuna bark extract, Dr Rafat Ahmed, Department of Biochemistry, UCMS (India) for her expert advice, Ms Shagufta Yasmeen, Ms Mythily Subramaneyaan, and Mr Mohd Aslam for their assistance in handling animals and performing biochemical tests.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
