Abstract
Hypothyroidism is considered a cardiac risk factor, but there is controversial evidence about its effects on coronary disease. The aim of this work was to evaluate the influence of hypothyroidism in rat hearts exposed to 2 degrees of stunning due to ischemia and reperfusion (I/R) as well as the underlying mechanisms. Hypothyroid (HypoT) rats were obtained by drinking 0.02% methimazole during 15 days. Isolated hearts were perfused and introduced in a flow calorimeter to measure contractile performance (P), total heat rate (Ht), and muscle economy (P/Ht). Hearts were exposed to 2 models of I/R, moderate and severe (respectively 20 or 30 minutes I/45 minutes R). Moreover, free cytosolic and mitochondrial calcium changes were measured by confocal fluorometry on cardiomyocytes. Comparison to euthyroid (EuT) hearts was done. Hypothyroidism was cardioprotective, but HypoT hearts were more sensitive than EuT hearts to the preischemic blockade of mitochondrial transporters mNCX and mKATP channels. Moreover, the postischemic recovery of P and P/Ht in HypoT hearts was strongly reduced by inhibition of the cellular pathways of PI3K/Akt and protein kinase C (PKC), and it was increased by nitric oxide synthase (NOS) inhibition. However, physiological concentrations of adrenaline reduced the cardioprotection of HypoT, but oral treatment with 20 mg/kg/day carvedilol prevented it. Results show that hypothyroidism reduces the mitochondrial Ca2+ overload during I/R by mKATP channel activation and Ca2+ extrusion through mNCX, while the PI3K/Akt and PKC pathways are involved in that cardioprotection. Contrarily, NOS activation and adrenaline blunt such cardioprotection, but carvedilol prevented the adrenergic dysfunction. These results would explain why hypothyroidism is a clinical risk factor in angor patients under adrenergic exacerbation but reduced the incidence of acute episodes of coronary syndrome in hospitalized patients. Results suggest that a treatment with carvedilol could be a potential therapeutic agent to prevent cardiac postischemic dysfunction in hypothyroid patients.
Introduction
Hypothyroidism affects between 4% and 10% of the population, and it is associated with decreased cardiac output, bradycardia, diastolic dysfunction, low basal metabolism, and fatigue. 1,2 Part of the risk was explained by the reduction in catabolism, contribution to hypercholesterolemia, risk of coronary thrombosis, and prolongation of QT interval. 3 Moreover, a reduced T3 plasmatic level was frequently found in patients with myocardial infarct, in a way that it was suggested a causal relationship among them. 4 Even when subclinical hypothyroidism increased the mortality of cardiac patients, levothyroxine administration could control it. 5 Considering the myocardial consequences of hypothyroidism, it was reported that the functioning of sarcorreticular Ca2+ (SERCA2) transporters was attenuated. 6,7 Recently, it was described that Ca2+ transients are blunted due to a reduced SERCA to phospholamban ratio, which causes a low sarcoreticular calcium load and disruption of the systolic RyR2 synchronization, and contractility deterioration. 8 However, other reports described that hearts from hypothyroid (HypoT) rats have a reduced susceptibility to ischemia–reperfusion (I/R) such as the protection against arrhythmia. 9,10 Moreover, HypoT hearts exhibited less ischemic diastolic contracture and infarct than euthyroid rats and higher messenger RNA expression of superoxide dismutase-1 (SOD1) and glutathione peroxidase-1 (GPX1), as antioxidant enzymes. 11 Those results partially explain the low incidence of acute episodes of coronary syndrome in hospitalized HypoT patients with angor. 12 Nevertheless, it is not completely known which mechanisms determine the resistance of HypoT hearts to I/R. 13 It is known that ischemia followed by reperfusion negatively affects mitochondrial function, and this dysfunction could be potentiated by other conditions that affect Ca2+ homeostasis, such as adrenergic stimulation and thyroid diseases. Moreover, clinical treatment of coronary diseases includes a β-blocker such as carvedilol in order to reduce cardiac demand. Additionally, carvedilol demonstrated to reduce the myocardial infarct in dogs and rats. 14,15 There were described other pleiotropic effects with this α, β-adrenergic antagonist, such as antioxidant activity, with beneficial effects on cardiac mitochondria in several ex vivo and in vivo models of cardiac ischemia. 16 The cardioprotection of carvedilol in myocardial infarct was also attributed to inhibition of the nuclear factor κB and Toll-like receptor 4 expression as well as the induction of β-arrestin 2 expression. 17 However, there are no reported studies about the effects of carvedilol on cardiac ischemia in HypoT individuals.
The prevention of an ischemic injury consequent to coronary disease can be studied with models of I/R that develop cardiac stunning, a condition characterized by reduced contractile and metabolic recoveries without significant infarct. The calorimetrical methodology is used to study cardiac energetic during well-oxygenated, hypoxic, or ischemic conditions, and this is the main advantage over the methodology of oxygen consumption. 18 Total heat rate is associated with the exothermic consumption of adenosine triphosphate (ATP) for obtaining energy during both resting (ionic pumps) and active (actomyosin and Ca2+ removal during the contractile cycle) states. It also includes the exothermic reactions of the mitochondrial metabolism coupled or not to the ATP resynthesis and calcium movements. 18 -21
Therefore, the aim of this work was to explore the myocardial mechanisms to maintain Ca2+ homeostasis that underlies the behavior of HypoT hearts under ischemia and reperfusion as well as the influence of adrenergic stimulation and carvedilol treatment.
Methods
Animals
The experimental procedures were done following the international rules and principles recommended in the Guide for Care and Use of Animals (NIH Nro publication # 85-23 revised in 1985 and 1996; National Academy Press, Washington, District of Columbia), and the Resolution 1047 anexo II of Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) de la República Argentina, 2005. The protocols were approved by the Laboratory Animals Care Committee of Facultad de Ciencias Exactas, Universidad Nacional de La Plata (Nr. 015-2015).
Treatments
Adult Wistar rats of both sexes (250-280 g weight) were obtained from the Biotery of the Institute of Cardiological Research and housed in the Biotery of Pharmacology of Facultad de Ciencias Exactas with up to 4 rats per cage until the day of the experiment, under uniform room temperature and artificial light cycle (12–12 hour) and fed with a standard pellet diet and water ad libitum. Animals were randomly assigned to 1 of 2 groups: euthyroid (EuT) and HypoT. Hypothyroidism was induced by drinking methimazole (Fluka; Sigma-Aldrich, St Louis, Missouri) 0.02% wt/vol in water for 2 weeks. 22 Hypothyroidism was tested by measuring the free T4 levels in blood of some of the rats before the ex vivo experiment, and they were determined by enzyme-linked fluorescent assay technique in an automated quantitative test used for humans (Vidas instruments; BioMérieux, Mary l´Etoile, France). Table 1 shows plasmatic free T4 levels and heart and body weight ratio from HypoT and EuT rats, which agree with those reported. 11
Basal Parameters of Euthyroid (EuT) and Hypothyroid (HypoT) Rats Obtained Before the Heart Isolation and Experiment of I/R.a

Abbreviations: BW, body heart; EuT, euthyroid hearts; HW, heart weight; HypoT, hypothyroid; I/R, ischemia/reperfusion; T4, levo-thyroxine plasmatic level; SEM, standard error of mean.
a Mean ± SEM (n).
b P < .01 by t test.
Isolation of Ventricles and Contractile and Calorimetrical Measurements
The method was previously described. 23 -25 Rats received nonfractioned heparin (2000 IU), anesthesia with pentobarbital overdose (60 mg/kg intraperitonelly), and analgesia with tramadol (10-20 mg/kg subcutaneously). Hearts were quickly excised and perfused by the Langendorff technique through coronaries with control Krebs solution (C) at flow rate of 7 mL/min/g regulated with a peristaltic pump (Gilson Minipuls 3; Villiers Le Bel, France). Perfusion flow was calculated by the equation CF = 7.43 × HW0.56 (where CF is coronary flow and HW is the heart weight), recommended to prevent myocardial edema under saline perfusion at high flow rate. 26 This flow induced an optimal developed pressure as it was previously described. 23 After perfusion, atria were removed as well as focus of spontaneous beating, a latex regulable-volume balloon filled with water, was connected by a flexible cannula to a Bentley DEL900 (Bentley, Nevada) or a Statham P23db (Oxnard, California) pressure transducer. The perfused ventricle was introduced into the calorimetrical chamber, and this one was submerged in the water bath at temperature of 37.0°C ± 0.01°C. Inside, the heart was electrically stimulated with 5 V for 5 milliseconds at 3 Hz by means of an stimulator (Letica LE12406; Letica, Barcelona, Spain or Grass SD9; Braintree, Massachusetts). The left intraventricular pressure (LVP, calibrated in mm Hg) and the calorimetrical signal were continuously recorded at optimal volume in a PowerLab 2/26 2-channels digital acquisition system (AD Instruments, New South Wales, Australia) or in a Grass polygraph of 8 channels (Grass Instruments, Quincy, Massachusetts) with A/D acquisition (TL-1 DMA Axon Instruments Inc, Foster City, California). The maximal pressure developed in contraction (P) was calculated from the difference between the peak LVP and the end-diastolic pressure (LVEDP). The diastolic contracture during I and R was calculated as the increase over the preischemic level (ΔLVEDP). Moreover, P was expressed as a percentage of the basal preischemic value.
The copper calorimeter was described in previous works. 18,20,23,24 It is composed by a cylindric copper mass to a quick heat conduction, containing an internal chamber where a small heart is adjusted with its perfusion and the cannula to measure LVP. The internal chamber has 2 ceramic modules with 127 thermosensitive units each one (Melchor Thermoelectrics, Trenton, New Jersey) connected in series. Both units detect the temperature changes between the calorimeter inside (heart) and outside (water bath), in microvolts, which were then amplified and digitized simultaneously with the LVP signal. A baseline of calorimetrical signal was obtained before introducing the heart and after taking it away both in the presence and in the absence of perfusion. The total heat rate (Ht) was calculated at any time from the difference between the cardiac signal and the respective baseline (with or without perfusion). It was expressed in mW/g of wet weight using a calibration factor obtained at the end of the experiment by applying a constant and known electrical power on ventricles as previously described. 18,23 It was a 2-mW potency of alternate current (2 kHz) and low voltage (1 v) which was unable to excite the heart, which acts only as a resistance. The total muscle economy was calculated as the P–Ht ratio (in mm Hg.g/mW).
Experimental Protocols in Hearts
After a stabilization in Krebs-C, some groups of hearts were exposed to certain treatments for 20 minutes, followed by a period of 20 minutes of no-flow ischemia (I) and to 45 minutes of reperfusion (R; moderate stunning, mI/R), as previously described. 23 Other groups of hearts were exposed to a period of 30 minutes of I and 45 minutes of R (severe stunning model [sI/R]). At the heart rate of 3 Hz and the wet atmosphere of the calorimetrical chamber, neither of the 2 models developed a significative infarct area (Figure 1B). The reperfusion was done with control Krebs, except when perfusing cyclosporine-A (Cys-A) which was present during both I and R as suggested previously. 25,27 Table 2 shows the initial absolute values of P and Ht in each group before exposing to the following protocols, either in HypoT or in EuT rat hearts. Protocols are shown in Figure 1A.

A, Schematic representation of perfusion protocols done with isolated rat hearts exposed to no-flow ischemia (shadow bar) and reperfusion done on euthyroid (EuT) and hypothyroid (HypoT) rat hearts. Control Krebs (C), Krebs-10 mM caffeine–36 mM Na+ (C-caff-low Na), 100 µmol/L 5-hydroxydecanoate (5-HD), 10 µmol/L clonazepam (Clzp), 0.2 µmol/L cyclosporine A (Cys-A), 30 µmol/L L-NAME (L-NAME), 10 µmol/L nitroprussiate (Nitrop), 100 µmol/L wortmannin (Wrt), 100 µmol/L chelerythrine (Che), 30 nmol/L adrenaline (Adre). On the top is indicated the time of any perfusion change in minutes. See Protocols in Methods section for details. B, Typical images of infarct obtained after 45 minutes reperfusion in hearts exposed to moderate ischemia (mI/R) and severe ischemia (sI/R) from EuT and HypoT rats.
Basal Mechanocalorimetrical Results Obtained Before Treating Hearts With the Respective Drug in the Several Experimental Groups (mean ± SEM).a
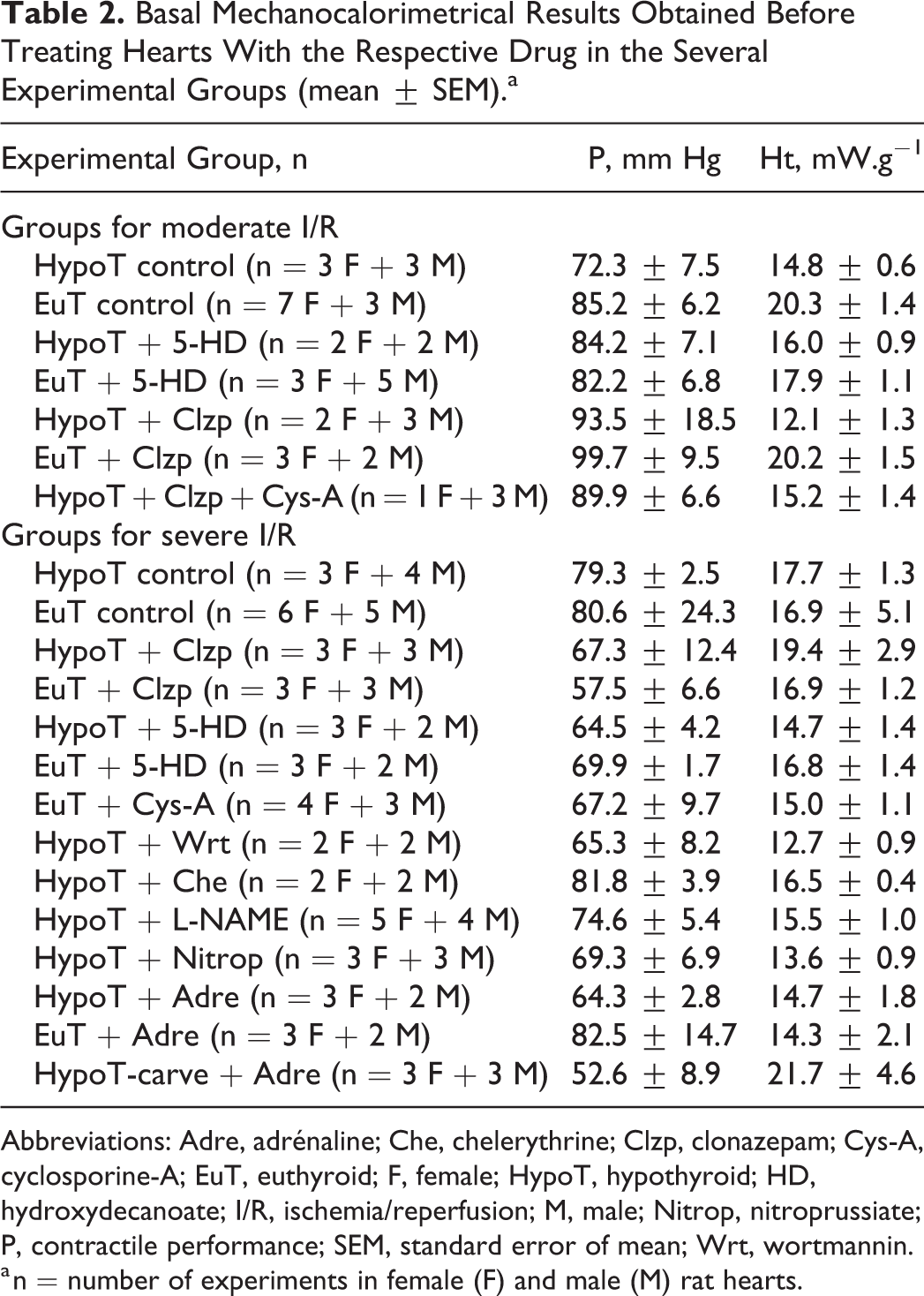
Abbreviations: Adre, adrénaline; Che, chelerythrine; Clzp, clonazepam; Cys-A, cyclosporine-A; EuT, euthyroid; F, female; HypoT, hypothyroid; HD, hydroxydecanoate; I/R, ischemia/reperfusion; M, male; Nitrop, nitroprussiate; P, contractile performance; SEM, standard error of mean; Wrt, wortmannin.
a n = number of experiments in female (F) and male (M) rat hearts.
Treatments in EuT rat hearts exposed to mI/R were (1) C (Krebs-C), (2) C/C+ 100 µmol/L 5-hydroxydecanoate (5-HD, to block the mKATP channels), 28 and (3) C/C+ 10 µmol/L clonazepam (Clzp, to block the mNCX). 24,29 Treatments in HypoT rat hearts exposed to mI/R were (1) C (Krebs-C), (2) C/C+ 100 µmol/L 5-HD, (3) C/C+ 10 µmol/L Clzp, and (4) C/C + 0.2 µmol/L Cys-A + 10 µmol/L Clzp (Cys-A + Clzp), reperfused also with C+ Cys-A (to block the mPTP). 25,27
The possibility that gender influences the response to I/R in EuT and HypoT hearts was evaluated by comparing P and P/Ht in those groups (Figures 2, 3 and 4). Since there were no found significant differences, the other treatments were done in mixed groups of both sexes as indicated in Table 2.

Mechanoenergetical performance of hearts isolated from hypothyroid (HypoT; A and B) and euthyroid (EuT; C and D) rats, separately for females (F) and males (M) and exposed to moderate ischemia/reperfusion (I/R). Mixed groups were previously perfused (drug period) with either 10 µmol/L clonazepam (Clzp) or 100 µmol/L 5-hydroxydecanoate sodium salt (5-HD) or treated with 0.2 µmol/L cyclosporin-A (Cys-A) before and all over the period Clzp-I-R. Upper panels show maximal pressure developed in contraction (P, as % of initial). Lower panels show muscle economy (P/Ht; in mm Hg.g/mW). Results are shown as mean ± standard error of mean (SEM; n). See 2-way analysis of variance (ANOVA) results in Table 5, *P < .05 versus the respective control group (HypoT or EuT, either F or M), # P < .05 versus HypoT-Clzp by post hoc tests.

Sarcorreticular Ca2+ release and cycling in hypothyroid (HypoT) and euthyroid (EuT) rat hearts and cardiomyocytes. Effects of reperfusing EuT (A) and HypoT (B) ischemic hearts with Krebs 10 mM and caffeine 36 mM Na+ (C-caff-low Na) on the changes in left ventricular pressure (ΔLVP) and heat rate (Ht). Changes in the cytosolic [Ca2+] measured by Fluo-4 ΔF/Fo (C) and of mitochondrial [Ca2+] measured by Rhod-2 ΔF/Fo (D) in isolated cardiomyocytes obtained from EuT and HypoT rats perfused with control Krebs (c) in the absence or the presence of 10 µmol/L clonazepam (Clzp) before treating with Krebs 10 mM and caffeine 36 mM Na+ (10 caff-36 Na). Results are mean ± standard error of mean (SEM; n). LVP, left intraventricular pressure.
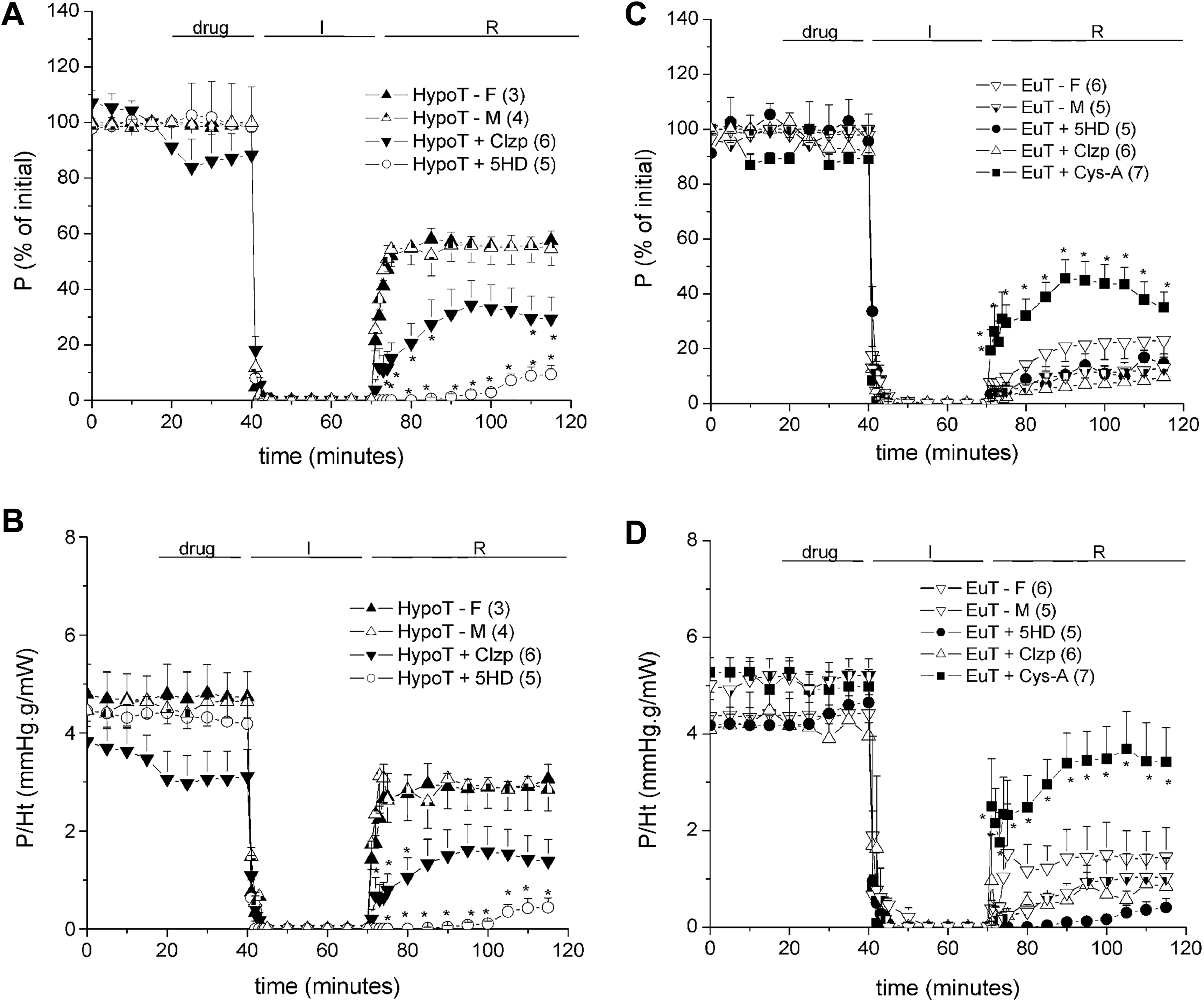
Mechanoenergetical performance of hearts isolated from hypothyroid (HypoT; A and B) and euthyroid (EuT; C and D) rats, separately for females (F) and males (M), and exposed to severe ischemia/reperfusion (I/R). Mixed groups were previously perfused (drug period) with either 10 µmol/L clonazepam (Clzp) or 100 µmol/L 5-hydroxydecanoate sodium salt (5-HD) or treated with 0.2 µmol/L cyclosporin-A (Cys-A) before and all over the period Clzp-I/R. Upper panels show maximal pressure developed in contraction (P, as % of initial). Lower panels show muscle economy (P/Ht; in mm Hg.g/mW). Results are shown as mean ± standard error of mean (SEM; n). See 2-way analysis of variance (ANOVA) results in Table 5, *P < .05 versus the respective control group (EuT or HypoT, either F or M) by post hoc tests.
To evaluate whether hypothyroidism had an effect on the SERCA2 release, the EuT and HypoT hearts were exposed to 20 minutes of ischemia and reperfused with Krebs containing 10 mmol/L caffeine and 36 mmol/L Na+ (R-caff-36 Na+). This intervention releases Ca2+ from the sarcoplasmic reticulum (SR) on the content, while the low Na+ minimizes the Ca2+ efflux through the sarcolemmal sodium/calcium exchanger (NCXSL). 24
Treatments in EuT rat hearts exposed to sI/R were (1) C (Krebs-C), (2) C/C + 100 µmol/L 5-HD, (3) C/C + 10 µmol/L Clzp, (4) C/C + 30 nmol/L adrenaline (Adre), and (5) C/C + 0.2 µmol/L Cys-A reperfused with C + Cys-A. Treatments in HypoT rat hearts exposed to sI/R were (1) C (Krebs-C), (2) C/C + 100 µmol/L 5-HD, (3) C/C + 10 µmol/L Clzp, (4) C/C + 30 µmol/L L-NAME (to block the nitric oxide synthases [NOS]), (5) C/C+ 10 µmol/L nitroprussiate (Nitrop, an endothelial NO donor), (6) C/C + 100 µmol/L wortmannin (Wrt, a selective inhibitor of the PI3K phosphorylation), (7) C/C+ 100 µmol/L chelerythrine (Che, a protein kinase C [PKC] inhibitor), and (8) C/C + 30 nmol/L Adre. Other group of HypoT rats was treated with carvedilol 20 mg/kg daily administered in drinking water for 7 days, 30 before the I/R experiment in the isolated heart with the following treatment: C/C + 30 nmol/L Adre and sI/R.
Measurement of Infarcted Area
To evaluate whether these models of I/R induce myocardial infarction, some of the reperfused hearts were freezed and then cut in 4 circular slices from apex to base. Sections were incubated for 20 minutes in 1% triphenyltetrazolium chloride (pH: 7.4, 37°C) and then scanned. With this technique, viable sections were stained red, and the infarct area remained white. Infarct area was calculated and expressed as percentage of the left ventricular area (Image-Pro Plus; Media Cybernetics, Rockville, Maryland).
Isolation of Ventricular Cardiomyocytes
Ventricular myocytes were isolated from HypoT and EuT rat hearts as described previously. 23,31 Briefly, the isolated heart was quickly perfused with a modified Krebs-24-HEPES solution without Ca2+ for 5 minutes, bubbled with O2 at 35°c to 37°c. Then, 50 µmol/L CaCl2, 0.1 mg/mL collagenase P (Roche), and 0.02 mg/mL protease XIV (Sigma-Aldrich) were added and perfused for 13 minutes. Finally, the enzymes were washed by perfusing a Krebs-24-HEPES-50 µmol/L Ca2+ solution for 5 minutes. Then, the ventricles were minced in pieces in a low-Ca2+ solution and then separated. The HEPES-buffered saline solution increased Ca2+ concentration by steps up to 1 mmol/L in which myocytes were stored until recording fluorometric signals.
Confocal Microscopy
Cardiomyocytes were loaded with 12 µmol/L Fluo-4AM (Molecular Probes/Invitrogen, Carlsbad, California) at room temperature for 15 minutes for measuring cytosolic free Ca2+. In other group, the mitochondrial compartment was loaded with 3 µmol/L Rhod-2AM (Molecular Probes/Invitrogen) for 1 hour at 4°C and washed for another 1 hour at 37°C. 32 As described previously, cells were placed in a perfusion chamber for a confocal microscope Leica SP5 (Leica Microsystems, Mannheim, Germany) and stabilized with Krebs-24 HEPES with 2 mmol/L Ca2+ (C). 23,31 Data were recorded every 20 seconds for 20 to 25 minutes of protocol by using the Leica LAS AF Lite version 2.2.1 software and expressed as the relative fluorescence intensity (F/Fo). The changes in baseline (ΔF/Fo) produced by the interventions were calculated after plotting points by the Origin 7.0 program (OriginLab Corporation, Northampton, Massachusetts), as described. 23 Cells with Fluo-4 were excited at 488 nm and detected at higher than 505 nm. Those with Rhod-2 were excited at 540 nm and detected at higher than 560 nm. It was measured in a selected region of interest (ROI) of the cell.
Cardiomyocytes from HypoT rats loaded with Rhod-2 or Fluo-4 were perfused with the following sequence of solutions in each protocol with 5 to 9 cells provenient from at least each of the 2 hearts: (1) C (5 minutes), C + 10 mmol/L Clzp (5 minutes), C-10 mmol/L caffeine-36 mmol/L Na+ +Clzp (10 minutes), and C (5 minutes) and (2) C (10 minutes), C-10 mmol/L caffeine-36 mmol/L Na+ (10 minutes), and C (5 minutes). Cardiomyocytes from EuT rats loaded with Rhod-2 or Fluo-4 were perfused with the protocol (2).
Solutions and Drugs
Control Krebs (Krebs-C) and Krebs-10 mmol/L caffeine and 36 mmol/L Na+ (Krebs-caff-36 Na) solutions to perfuse ventricles as well as the Krebs-24-HEPES solution to isolate and perfuse cardiomyocytes were prepared as described in previous works. 23,24,31
Clonazepam (Saporiti, Buenos Aires, Argentina) was prepared as an aqueous solution at 10 mmol/L and diluted to 10 µmol/L in Krebs-C for perfusion. Cyclosporine-A () was prepared in dimethyl sulfoxide (DMSO) at 0.2 mmol/L and diluted in Krebs-C to 0.2 µmol/L. Sodium 5-HD (ICN Biochemicals and Reagents, Costa Mesa, California) was prepared as a 100 mmol/L solution in DMSO and diluted to 100 µmol/L in Krebs-C. Wortmannin, Che, and L-NAME (Sigma-Aldrich) were prepared in DMSO and diluted 1:100 in Krebs-C. All the drugs were diluted from their stock solutions in Krebs-C at the moment of the experiment. Caffeine (ICN Biochemicals and Reagents) was directly dissolved in Krebs on the day of the experiment.
Statistical Analysis
Results were expressed as mean ± standard error of mean. Multiple comparisons by 2-way analysis of variance (ANOVA) for repeated measures were done for the respective groups of experiments (variables treatment and time). Post hoc Tukey tests between treatments are shown in the respective figures. For comparing only 2 samples, the unpaired Student t test was used. All statistical analyses were performed by using the Graph Pad Prism version 6.0 (San Diego, La Jolla, California), and significance was considered for P < .05.
Results
Effects of Hypothyroidism in Moderate Cardiac Stunning and Role of mNCX and mKATP
Before any experimental protocol, the maximal pressure developed in contraction (P) from HypoT rat hearts was similar to that of EuT rat hearts. Table 2 shows initial values of P and Ht in all experimental groups as well as the number of results by gender. After exposing isolated hearts from HypoT rats to 20 minutes I/45 minutes R (moderate stunning, mI/R), the postischemic contractile recovery (PICR) was higher than that obtained in the EuT hearts (Figure 2A and C) as well as it was the total muscle economy (P/Ht; Figure 2B and D). There was not a significative difference in the response to I/R between male and female rat hearts, either EuT or HypoT. Moreover, neither EuT nor HypoT hearts developed significant infarct areas after mI/R (5.2% ± 1.3%, n = 4, vs 3.05 ± 0.7%, n = 4, respectively, Figure 1B).
The possibility that the mKATP channels play a role avoiding the mitochondrial Ca2+ overload during the mI/R was evaluated by selectively blocking them with 100 µmol/L 5-HD perfusion before mI/R. This treatment raised the diastolic pressures (ΔLVEDP) during I/R in both, but more in EuT than in HypoT hearts (Table 3), although it did not change the recovery of P (%) during R (Figure 2A and C). Results suggest that mKATP channels only contribute to regulate the diastolic Ca2+ level.
Comparison of the Changes in Diastolic Pressure (ΔLVEDP, in mm Hg) in All Experimental Groups Exposed to Moderate I/R at 5 and 20 Minutes Ischemia and 5 and 45 Minutes Reperfusion.a

Abbreviations: Adre, adrénaline; ANOVA, analysis of variance; Che, chelerythrine; Clzp, clonazepam; Cys-A, cyclosporine-A; DFn, degrees of freedom; EuT, euthyroid; F, female; HD, hydroxydecanoate; HypoT, hypothyroid; I/R, ischemia/reperfusion; M, male; SEM, standard error of mean.
a Results are Shown as Mean ± SEM.
b P < .05 versus the respective control condition (C, in EuT or HypoT, respectively) by post hoc Tukey tests.
c P < .05 versus the respective drug-EuT condition.
When the mNCX was selectively blocked with 10 µmol/L Clzp, the HypoT hearts reduced P and P/Ht during R (Figure 2A and B) and developed a strong diastolic contracture, up to about 85 mm Hg over the preischemic level (Table 3). Contrarily, Clzp increased the contractile recovery in EuT hearts as well as the muscle economy (Figure 2C and D) without generating diastolic contracture (Table 3). Results suggest that hypothyroidism changes the role of mNCX. In order to evaluate whether the strong loss of contractility and diastolic contracture in the Clzp-perfused HypoT hearts was due to the opening of mPTP channels, a Clzp-treated HypoT group was perfused with 0.2 µmol/L Cys-A before I and reperfused with it to block the mPTP. Figure 2A and B shows that the recovery of P during R was increased from about 40% to 75% of initial, and diastolic contracture was reduced by Cys-A (Table 3). Results suggest that the Ca2+ extrusion through mNCX contributes to prevent the mPTP opening in HypoT hearts.
Effects of Hypothyroidism on Cellular Ca2+ Levels
In order to evaluate whether hypothyroidism affects the SERCA store and release, isolated HypoT and EuT hearts were exposed to moderate I (20 minutes I) and reperfused with 36 mmol/L Na+ and 10 mmol/L caffeine Krebs solution (Krebs-low Na-caff). This solution releases Ca2+ from the SR and prevents its efflux through the sarcolemmal NCX, becoming in contracture (ΔLVP), whose peak is proportional to the SR Ca2+ content. During this treatment, relaxation is a consequence of the mitochondrial Ca2+ uptake and the efflux through the sarcolemmal Ca-ATPases easily saturated. Figure 3A and B shows that HypoT hearts had a lower LVP peak than the EuT hearts, suggesting that they have lower SR Ca2+ content available to be released. However, the area under curve (AUC) of ΔLVP (1129.6 ± 108.8, n = 5) in HypoT was similar to that of EuT (1052.0 ± 91.3, n = 6) as well as the AUC-Ht (413.14 ± 25.2 vs 365.8 ± 39.3, respectively). This similarity in AUC-ΔLVP seems to be due to the lower rate of relaxation in HypoT (t1/2 of more than 52 minutes) than in EuT (t1/2 of about 8 minutes).
In nonischemic isolated cardiomyocytes perfused with Krebs-low Na-caff, the free cytosolic Ca2+ (Fluo-4 signal) was lower and more quickly removed in HypoT than in EuT hearts (Figure 3C). Accordingly, the free mitochondrial Ca2+ level (Rhod-2 signal) was more quickly increased in HypoT than in EuT but then easily lost (Figure 3D).
Effects of Hypothyroidism in Severe Cardiac Stunning and Role of mNCX and mKATP
When another group of HypoT hearts was exposed to a severe ischemia (sI/R, 30 minutes I/45 minutes R), the P and P/Ht recoveries during R were greatly improved with respect to the EuT hearts with a similar model (Figure 4A and B vs 4C and D). There were no differences between male and female rat hearts, neither in HypoT nor EuT rats (Figure 4). The diastolic contracture (ΔLVEDP) was less increased in HypoT than in EuT hearts during R (Table 4). Neither EuT nor HypoT hearts developed significant infarct areas after sI/R (2.3% ± 0.3%, n = 6, vs 1.8% ± 0.1%, n = 5, respectively; Figure 1B).
Comparison of the Changes in Diastolic Pressure (ΔLVEDP, in mm Hg) in All Experimental Groups Exposed to the Severe I/R at 5 and 30 Minutes Ischemia and 5 and 45 Minutes Reperfusion.a
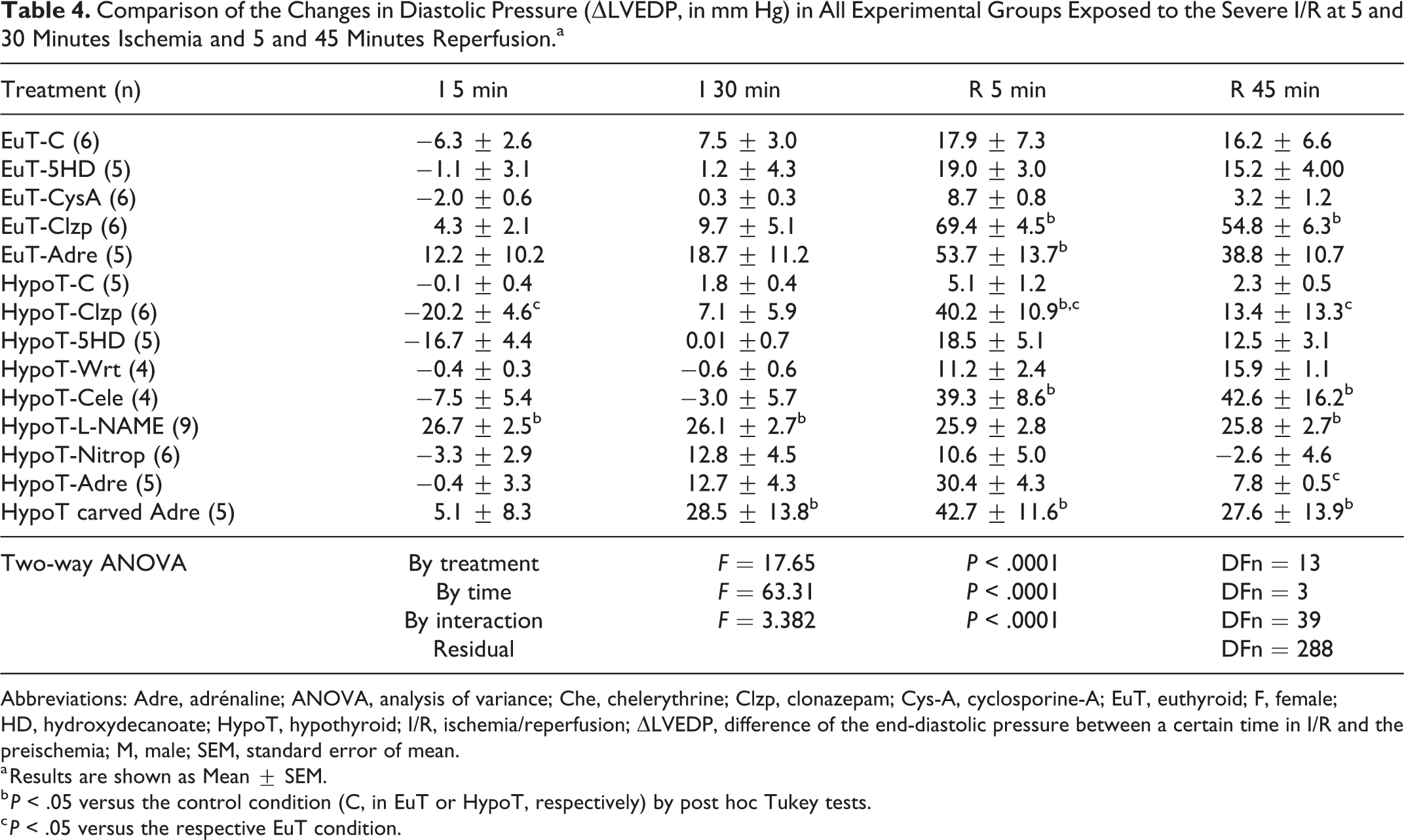
Abbreviations: Adre, adrénaline; ANOVA, analysis of variance; Che, chelerythrine; Clzp, clonazepam; Cys-A, cyclosporine-A; EuT, euthyroid; F, female; HD, hydroxydecanoate; HypoT, hypothyroid; I/R, ischemia/reperfusion; ΔLVEDP, difference of the end-diastolic pressure between a certain time in I/R and the preischemia; M, male; SEM, standard error of mean.
a Results are shown as Mean ± SEM.
b P < .05 versus the control condition (C, in EuT or HypoT, respectively) by post hoc Tukey tests.
c P < .05 versus the respective EuT condition.
In order to evaluate whether the extrusion by mNCX induces cardioprotection in HypoT hearts, 10 µmol/L Clzp was perfused before I. Consequently, the P recovery was reduced from about 54% to 29% of initial value at the end of R, while P/Ht was proportionally reduced (Figure 4A and B) and LVEDP was increased (Table 4). However, Clzp did not significantly change the P and P/Ht recoveries of EuT hearts (about 10% of the preischemic; Figure 4C and D).
The hypothesis about a role of mKATP channels in cardioprotection of hypothyroidism under sI/R was confirmed with the effect of perfusing the selective blocker 5-HD before I. In HypoT hearts, both P and P/Ht were strongly reduced (Figure 4A and B) with diastolic contracture (ΔLVEDP increased) during sI/R (Table 4). In EuT hearts, 5-HD did not modify the low P and P/Ht recoveries during R (Figure 4C and D) but increased ΔLVEDP (Table 4), showing the different role of mKATP channels.
Moreover, Cys-A was perfused to assess whether the dysfunction under sI/R in EuT hearts was due to activation of mPTP. The strong increase in the postischemic P and P/Ht recoveries (Figure 4C and D) and reduction in diastolic contracture (ΔLVEDP; Table 4) confirmed this hypothesis.
All these results suggest that the activation of mKATP channels and mNCX protect the HypoT hearts from mitochondrial Ca2+ overload during sI/R, while dysfunction of EuT hearts depends on mPTP activation.
Role of Cellular Pathways of PI3K, PKC, and NO in the Severe Stunning of HypoT Ventricles
To evaluate the role of the PI3K/Akt pathway in cardioprotection of HypoT hearts, the selective inhibitor 100 µmol/L Wrt was perfused before I. The important role of this pathway was demonstrated because the postischemic P was strongly reduced as well as P/Ht (Figure 5A and B), while ΔLVEDP was increased at the end of R (Table 4). Moreover, the role of PKC in HypoT hearts was evidenced by perfusing the selective inhibitor Che at 100 µmol/L, and Figure 5A and B shows that this drug strongly reduced P and P/Ht, while Table 4 shows a great diastolic contracture during R.

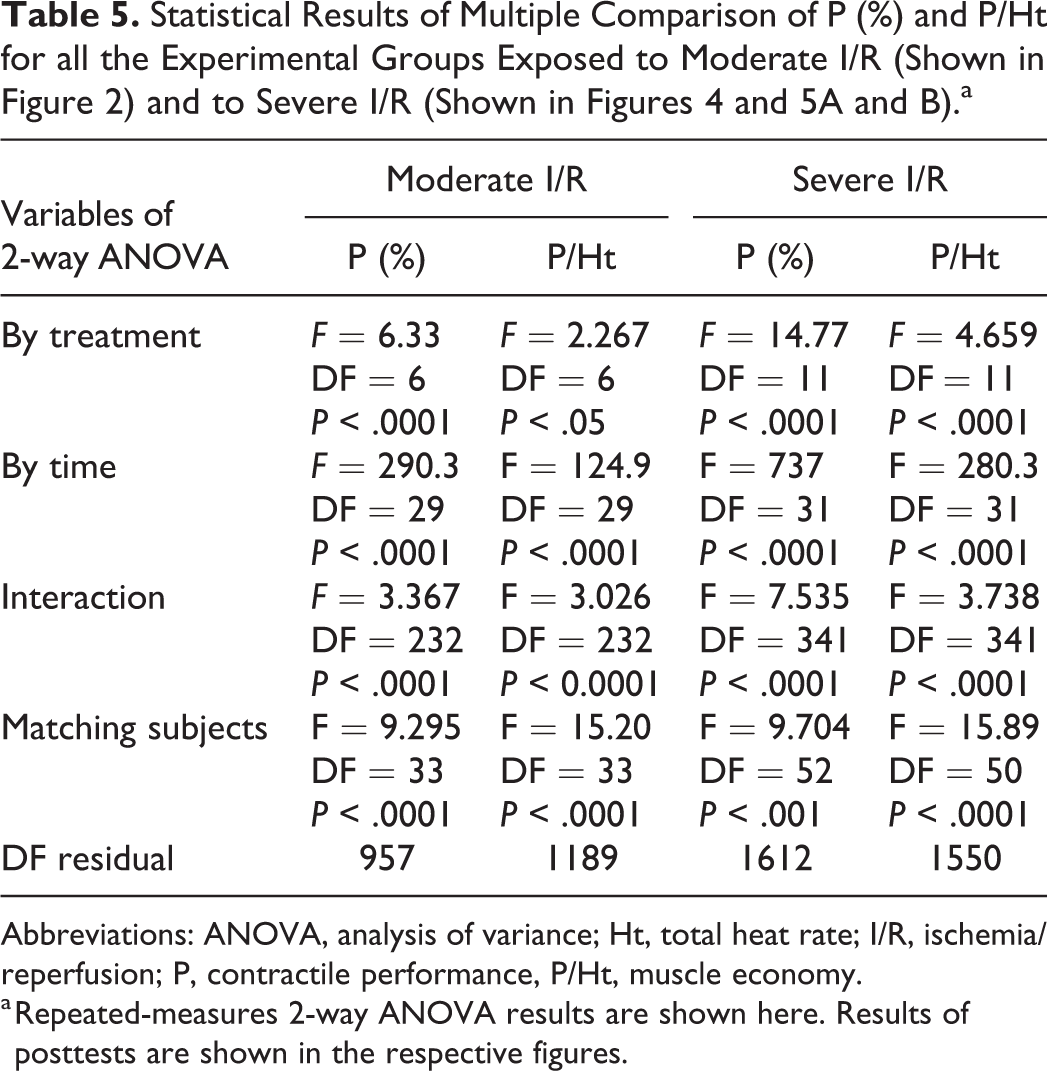
Role of PKC, PI3K/Akt, NOS, adrenaline, and carvedilol in the mechanoenergetical performance of isolated hypothyroid (HypoT) rat hearts exposed to severe ischemia/reperfusion (I/R). Upper panels show maximal pressure developed in contraction (P, as % of initial). Lower panels show muscle economy (P/Ht; in mm Hg.g/mW in B and as % of initial in C). The effects of previous perfusion of 1 µmol/L chelerythrine (Che), 100 µmol/L wortmannin (Wrt), 30 µmol/L L-NAME, and 10 µmol/L nitroprussiate (Nitrop) are shown in (A and B). The effects of perfusing 30 nmol/L adrenaline in HypoT and EuT hearts and the previous treatment with 20 mg/kg/d carvedilol during 1 week are shown in (C and D). Results are shown as mean ± standard error of mean (SEM; n). See 2-way analysis of variance (ANOVA) results in Table 5 for (A and B) post hoc tests: *P < .05 versus HypoT group. NOS indicates nitric oxide synthase.
Abbreviations: ANOVA, analysis of variance; Ht, total heat rate; I/R, ischemia/reperfusion; P, contractile performance, P/Ht, muscle economy.
a Repeated-measures 2-way ANOVA results are shown here. Results of posttests are shown in the respective figures.
In order to study whether the NO production participates in the cardioprotection of hypothyroidism, the NOS were blocked by 30 µmol/L L-NAME before I and up to the first 5 minutes of R. The postischemic P was improved without changes in the muscle economy (Figure 5A and B), but ΔLVEDP was also increased during sI/R (Table 4). Moreover, when the NO was released by 10 µmol/L nitroprussiate perfusion in another group of HypoT hearts, the recoveries of P and P/Ht (Figure 5A and B) and the diastolic contracture (ΔLVEDP; Table 4) were not significantly changed. Results suggest that NO production does not play an important role in HypoT cardioprotection.
Effects of Adrenaline and Carvedilol on HypoT Cardioprotection
Considering that our previous results do not explain the clinical risk of hypothyroidism, we evaluated whether the presence of adrenaline in similar concentrations to the plasmatic ones maintains the cardioprotection of HypoT rat hearts. Figure 5C and D show that Adre 30 nmol/L reduced the cardioprotection because HypoT hearts recovered low P and P/Ht while it increased the diastolic contracture (Table 4) similarly to the EuT hearts. Contrarily, oral treatment with 20 mg/kg carvedilol daily for 1 week before the experiment prevented the effect of Adre by contributing to restore the HypoT cardioprotection. This treatment improved the postischemic P and P/Ht recovery (Figure 5C: 2-way ANOVA by treatment: F = 4.01, P = .0438, by time: F = 164.0, P < .0001; Figure 5D: 2-way ANOVA by treatment: F = 3.87, P = .048, by time: F = 35.0, P < .0001). However, it maintained a high diastolic contracture during I/R (Table 4).
Discussion
This work shows that mitochondrial mechanisms such as the mKATP channels and the mNCX are very important to regulate the m[Ca2+] in HypoT rat hearts in a way that they prevent Ca2+ overload and severe dysfunction during I/R. However, in the presence of physiological concentrations of adrenaline, the cardioprotection of the hypothyroidism disappeared. Nevertheless, the oral treatment with the α, β-blocker carvedilol restored cardioprotection in HypoT hearts perfused with adrenaline. Results suggest that the cytosolic and mitochondrial Ca2+ overload characteristic of β-adrenergic stimulation would reduce the intrinsic HypoT cardioprotection and opens the potential utility of carvedilol as a preventive therapy in HypoT patients of angor.
A recent report showed that HypoT rat hearts blunted the Ca2+ transients with lower SERCA release. 8 Although initial contraction (P) was not significantly reduced in our well-perfused isolated HypoT hearts respect to the EuT group (Table 2), resting HypoT cardiomyocytes reduced their caffeine-dependent cytosolic Ca2+ release (Fluo-4 signal), accelerated the cytosolic removal, and further blunted the increase in mitochondrial free Ca2+ (Rhod-2 signal; Figure 3). This behavior suggests that mitochondrial Ca2+ transporters were functionally affected by hypothyroidism.
The simultaneous measurement of contractility and calorimetry allows to evaluate minimal changes in metabolism and mitochondrial function during both resting and active states. 18,20,23,33 Before ischemia, the total muscle economy (P/Ht) of beating HypoT and EuT ventricles was not different, but after the challenge of severe I/R it increased in HypoT hearts. These results suggest that HypoT hearts increased the postischemic contractile performance with less energetic consumption, which may be associated with a reduced metabolism. It is known that Ht is released by the myocardial exothermic processes such as actomyosin coupling, cytosolic Ca2+ uptake, other ionic pumps, and mitochondrial metabolism in basal and active conditions (this one proportional to oxygen-consumption). 19,20,34 Then, with the bigger postischeimc contractile recovery, the HypoT hearts have to release a higher tension-dependent heat than the EuT hearts. Consequently, the low Ht (relative to P) may be due to reduction in the tension-independent heat (a part of the active energy related to Ca2+-removal and mitochondrial metabolism). These results could be in agreement with reports that described a reduced metabolism and oxidative capacity in HypoT hearts. 35
The mitochondrial metabolism is mainly related to [Ca2+], which depends on the uptake by the uniporter (UCam) and the extrusion in exchange with Na+ (mNCX) in response to the cytosolic Ca2+ oscilations. 36 It is well-known that it is Ca2+ that indirectly adapts the mitochondrial metabolic activity to cardiac demand. 34 The present results demonstrate that after blocking the mNCX with Clzp in hearts exposed to a moderate stunning I/R model, the PICR was reduced in HypoT hearts but increased in EuT, with proportional changes in muscle economy (P/Ht). Moreover, the postischemic dysfunction induced by Clzp in HypoT hearts was attenuated by Cys-A, a known inhibitor of mPTP. It is known that these mitochondrial transition pores open in response to Ca2+ overload, with loss of mitochondrial content and dysfunction or apoptosis. 37 Therefore, our results demonstrate that mitochondrial Ca2+ extrusion through the mNCX in HypoT hearts prevented Ca2+ overload and their opening during I/R. Moreover, the low diastolic contracture (ΔLVEDP) and high P/Ht during I/R in HypoT hearts suggest that mitochondrial and cytosolic Ca2+ were efficiently removed by sarcolemmal (NCX and Ca-ATPases) and the sarcorreticular (SERCA2) transporters. This efficiency in Ca2+ removal contributes to improve the postischemic recovery of ATP synthesis with less energetical uncoupling associated with mitochondrial Ca2+ overload, and consequently higher sarcoreticular Ca2+ load and release and better contractile recovery in HypoT than in EuT hearts.
Present results also demonstrate that the activation of mKATP channels contribute to prevent the mitochondrial Ca2+ overload evoked by I/R in HypoT hearts. It is known that diazoxide opens the mKATP channels, and it reduces the mitochondrial Ca2+ load because of the fall in the internal membrane electrical gradient (ΔΨm) and the activation of mitochondrial respiration. 28,38 Figure 4 shows that 5-HD, a selective inhibitor of mKATP channels, strongly reduced the postischemic P in HypoT hearts exposed to sI/R but not in the EuT hearts. However, HypoT and EuT hearts exposed to moderate I/R 5-HD did not affect P or P/Ht (Figure 2). Therefore, the mKATP channel opening contributes to cardioprotection only in HypoT hearts under sI/R, preventing the mitochondrial Ca2+ overload. In contrast, under this severe I/R model, the EuT hearts suffered Ca2+ overload and mPTP opening, as it was demonstrated by the reversion of dysfunction induced by Cys-A (Figure 4).
On the other hand, the mitochondrial dysfunction after ischemia was associated with changes in cellular pathways such as low level of both phosphorylated Akt and redox-activated protein kinases as PKC∊-signaling pathways. 39 These changes indirectly contribute to mitochondrial mPTP opening, swelling, Ca2+ and reactive oxygen species (ROS) overload, and apoptosis. 39 Contrarily, the phosphorylation of PI3K/Akt pathway drives to cardioprotection, while good levels of PKC∊ stimulate the mKATP opening. 40 Other authors agree with the cardioprotective sequence of PI3K/Akt phosphorylation, NO production, and Ca2+-activated K+-channels in the ischemic hearts. 41 Moreover, phosphorylation of PI3K/Akt stimulates the eNOS and S-nitrosilation of proteins, which finally activate the mKATP channels. 42 In our results, both the blockade of the PI3K/Akt phosphorylation with Wrt and that of PKC pathway with Che strongly reduced the postischemic P and muscle economy of HypoT hearts exposed to severe I/R (Figure 5A and B) and also increased diastolic contracture (LVEDP). Therefore, results strongly suggest that these pathways contribute to cardioprotection in HypoT hearts. Accordingly, other study showed that PKC∊ is overexpressed in HypoT rat hearts. 10 Other protein pathways were involved in resistance of HypoT rat hearts to I/R injury, such as the reduction in mitogen-activated protein kinases (MAPK) and jun N-terminal kinase (JNK) phosphorylation. 43 Moreover, HypoT rat hearts showed increased expression of conexin-43 (Cx43) and its phosphorylated forms, as well as increased PKC∊ that phosphorylates Cx43, in comparison with EuT controls, in a way that this fact explained the resistance to malignant arrhythmias. 44 Another work related the HypoT cardioprotection to higher synthesis of antioxidant enzymes, such as SOD. 11 All these different pathways could contribute to activate mKATP channels and to protect mitochondria against Ca2+ overload and energetical uncoupling, as we found in our HypoT rat postischemic hearts.
Another important regulator of apoptosis is the NO, which has a bifunctional role in the ischemic dysfunction, depending on concentration. It was described that high NO levels produced by the inducible NOS (iNOS) promote apoptosis, while low NO levels catalyzed by the endothelial isoenzyme (eNOS) improve cardioprotection. 45 Therefore, we evaluated the role of NO production in HypoT hearts exposed to sI/R and found that blocking NOS with L-NAME improved the contractile recovery maintaining constant the total muscle economy (Figure 5A and B). These results suggest that HypoT promotes the NO production at a high level, which might not contribute to cardioprotection. Although in different degree, this conclusion is in agreement with a report showing that fetal HypoT followed by I/R in the adult rat hearts increased the expression of iNOS, the Bax–Bcl2 ratio, and apoptosis with respect to EuT hearts, and all of them were reverted by iNOS inhibition. 46 In addition, in our results, the endothelial NO donor nitroprussiate reduced P only in the first times of R, as a nonmirror effect to that of L-NAME, which suggests that NO effect is really different depending on its concentration and the cellular localization. Therefore, present results suggest that the iNOS activation is negative to the postischemic recovery of HypoT hearts, but its influence was minimized by other cardioprotective mechanisms, such as the previously discussed about prevention of mitochondrial Ca2+ overload and activation of PI3K/Akt and PKC pathways.
However, all those cardioprotective mechanisms were blunted in the presence of Adre in physiological concentrations. As it was described, the β-adrenergic stimulation of HypoT hearts increases both Ca2+ influx by L-channels and cytosolic removal by SR and mitochondria, increasing the Ca2+ overload typical of I/R. 8 With this high cytosolic Ca2+ level, the cardioprotective mitochondrial mechanisms of hypothyroidism would be minimized, therefore explaining that both PICR and P/Ht were reduced with strong diastolic contracture as well as it happened with EuT hearts (Figure 5C and D). The consequences obtained with physiological concentrations of Adre explain why hypothyroidism is a risk factor in patients with coronary disease. 2 However, it was described that HypoT reduces the incidence of acute episodes of coronary syndrome in hospitalized HypoT patients of angor. 12 The difference could be due to the clinical control of adrenergic stimulation by β-blockers. In fact, when we orally treated HypoT rats with carvedilol, the postischemic hearts improved contractile recovery with a proportional energetic output, despite maintaining the diastolic contracture. This effect of carvedilol could be associated to reduction in β-adrenergic pathways and to the antioxidant activity on cardiac mitochondria, previously described. 16 However, another study should be done to understand the mechanism of cardioprotection. Therefore, this work opens a window to study whether the β-blockers of last generation (carvedilol, bisoprolol or nebivolol) could help restore the mitochondrial cardioprotective mechanisms of hypothyroidism seen in the absence of adrenergic stimulation in rat hearts.
Conclusions
By using the mechanocalorimetrical methodology, it was demonstrated a functional cardioprotection of hypothyroidism on cardiac I/R, which was mainly associated with reduced mitochondrial Ca2+ overload (with the participation of the mNCX and mKATP channels, as well as the PI3K/Akt and PKC pathways). Moreover, it was shown the deleterious role of physiological adrenergic stimulation to undo the cardioprotection and the beneficial restoration elicited by oral treatment with carvedilol.
Footnotes
Authors’ Note
María Inés Ragone, Germán A. Colareda, and Patricia Bonazzola are from Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina. Matías Bayley Fellow from Universidad Nacional de La Plata.
Acknowledgments
Thanks to Dra Laura Delaplace and technicians of Laboratorio Central de la Facultad de Ciencias Exactas, UNLP, for measuring the T4 levels in rat blood.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National University of La Plata (UNLP) (Grant Numbers 11X-642 for 2013-2016, and 11X-795 for 2017-2020).