Abstract
The role of oral anticoagulants (OAC) in atrial fibrillation (AF) is well established. However, none of the randomized controlled trials included patients with end-stage renal disease (ESRD) leaving a lack of evidence in this large, challenging and unique patient group. Patients on hemodialysis (HD) with AF have additional risk factors for stroke due to vascular comorbidities, HD treatment, age, and diabetes. Conversely, they are also at increased risk of major bleeding due to uremic platelet impairment. Anticoagulants increase bleeding risk in patients with ESRD and HD up to 10-fold compared with non chronic kidney disease (CKD) patients on warfarin. There are conflicting data and recommendations regarding use of OACs in ESRD which will be reviewed in this article. We conclude by proposing a modified strategy for OAC use in ESRD based on the latest evidence.
Introduction
End-stage renal disease (ESRD) is defined as a glomerular filtration rate (GFR) of <15 mL/min/1.73 m2 or requiring hemodialysis (HD). The unadjusted ESRD prevalence is 0.21% of the US population, affecting over 700 000 Americans in 2015, with over 40 000 having atrial fibrillation (AF). 1 Although many patients with ESRD and AF are at elevated risk of stroke, less than half receive oral anticoagulants (OACs). 1 In one reported study of patients, only 33.9% of dialysis patients with AF were treated with warfarin and 5.7% with direct OACs. 1 Two-year mortality in ESRD increases from 21% to 37% with concomitant AF. There is increasing recognition that AF is a marker of disease in addition to its well-documented role as a prothromboembolic state, as reflected in studies showing higher mortality other than from ischemic stroke. 2
The reasons for the higher mortality in patients with ESRD and AF can be divided into thromboembolic and hemorrhagic. The thromboembolic risk of ESRD is almost double that of patients with AF without ESRD and approaches 7 events per 100 patient-years (Table 1). A large meta-analysis found an inverse relation between GFR and stroke risk, with a risk of stroke increasing by 7% for every 10 mL/min/1.73 m2 fall in GFR below 60 mL/min/1.73 m2. 3
Epidemiology of Thromboembolism in Patients With AF With ESRD.a

Abbreviations: AF, atrial fibrillation; CI, confidence interval; ESRD, end-stage renal disease; GFR, glomerular filtration rate; HR, hazard ratio; RRT, renal replacement therapy.
aModified from the study by Lau et al 7 with permission.
Whilst stroke and intracranial hemorrhage are devastating manifestations of thromboembolism and hemorrhage in patients with ESRD, respectively (Table 2), there are many extracranial sources of hemorrhagic complications. Both forms of dialysis (peritoneal and intravascular) carry a 12-fold risk of gastrointestinal bleeding. 8 In a registry of over 300 000 Swedish health records, the risk of any bleeding was 30.5% in renal failure versus 14.1% in those without renal failure, with the difference exclusively in nonintracranial sources. 9
Clinical Presentations of Thrombosis and Hemorrhage in Patients With ESRD.a

Abbreviations: ESRD, end-stage renal disease; HD, hemodialysis.
aFrom the study by Lutz et al 10 with permission.
Despite many shared pathophysiological mechanisms, it is unclear which patients with ESRD will develop bleeding problems in contrast to those that will develop thrombosis. It is difficult to ascertain a dominant pathogenic factor responsible for a bleeding or clotting tendency in patients with renal failure. Factors beyond the clotting system such as high blood pressure together with thrombocytopenia and the administration of anticoagulants can result in an increased risk of bleeding. Conversely, stenosis of dialysis access fistulas and central veins increase risk of thrombus formation.
Guidelines for OAC Prophylaxis
Current guidelines from major international societies and food and drug administration (FDA) labeling are summarized below.
As illustrated in Table 3, there are conflicting and contradictory recommendations in the guidelines from major cardiac and renal societies and with FDA labeling. Despite multiple literature reviews focusing on specific drugs classes and patient populations, 13,14 there is no consensus as to whether an OAC is even warranted in ESRD, let alone the choice of agent or the dose. This stems fundamentally from the lack of any randomized trial data in these groups. We will now explore each of these oral agents in turn, specific to patients with ESRD.
Summary of Recommendations for OAC Use in AF Based on Renal Function.

Abbreviations: ACC, American College of Cardiology; AF, atrial fibrillation; AHA, American Heart Association; CCS, Canadian Cardiovascular Society; EHRA, European Heart Rhythm Association; ESRD, end-stage renal disease; GFR, glomerular filtration rate; HD, hemodialysis; HRS, Heart Rhythm Society; INR, international normalized ratio; KDIGO, Kidney Disease Improving Global Outcomes; OAC, oral anticoagulant.
aA 2.5-mg twice daily dose recommended when 2 or more of body weight <60 kg, age > 80 years, creatinine >1.5 mg present. For ESRD and HD subgroups, text is directly quoted.
Warfarin
Warfarin (Wisconsin Alumni Research Foundation coumARIN) is completely absorbed after its oral administration, 15 reaching a maximum plasma concentration within 6 hours. It distributes into a small volume of distribution and is eliminated by hepatic metabolism. 15 There is no renal excretion, but other factors such as drug interactions can impact the clinical efficacy of the drug in patients with ESRD. This is reflected by the lower doses required in pharmacodynamic studies of warfarin which show 19% overall dose reduction to achieve the same international normalized ratio (INR) in patients with GFR < 30 mL/min/1.73 m2.
A major shortcoming of warfarin is the variability in its therapeutic range between patients and within the same patient. It is the classic example of pharmacogenomic interactions, with warfarin metabolic enzyme CYP2C9 and warfarin target enzyme, vitamin K epoxide reductase complex 1 VKORC1 influencing warfarin maintenance dose. Other CYP enzymes are involved in the metabolism of warfarin, but the majority of biological variability is attributable to these interactions, 22 which can result in a 10-fold variation in maintenance doses required for a therapeutic INR. 16
This heterogeneity is captured in clinical studies by the “time in therapeutic range” (TTR), which is a large determinant of clinical outcome. Studies dichotomizing at 60% TTR confirm outcomes segregate either side of this threshold, 17 but there may be a predominant effect on improving safety (ie, lessening bleeding) rather than by increasing efficacy (preventing stroke). 18
The clinical evidence for stroke primary prevention with warfarin in AF is summarized in Table 4.
Clinical Evidence for Stroke Primary Prevention with Warfarin in AF.
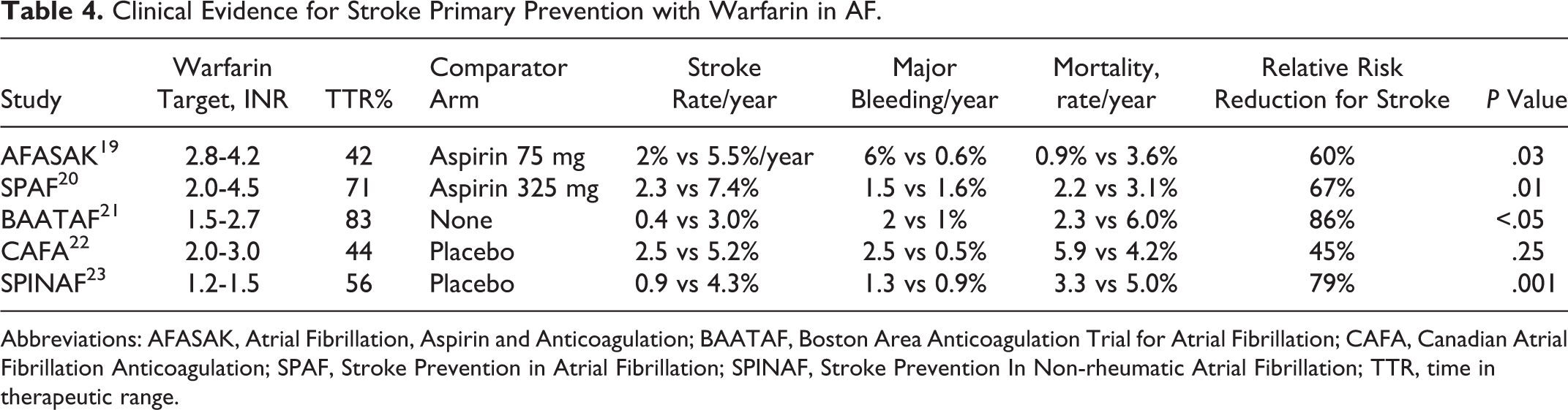
Abbreviations: AFASAK, Atrial Fibrillation, Aspirin and Anticoagulation; BAATAF, Boston Area Anticoagulation Trial for Atrial Fibrillation; CAFA, Canadian Atrial Fibrillation Anticoagulation; SPAF, Stroke Prevention in Atrial Fibrillation; SPINAF, Stroke Prevention In Non-rheumatic Atrial Fibrillation; TTR, time in therapeutic range.
There is no randomized data for warfarin efficacy in ESRD. Most analyses show increased bleeding risk outweighs stroke reduction in dialysis patients with AF. 24 –27 There are only sparse data which have showed reduced mortality in trials of warfarin use in HD patients. 17,28 It is also possible that the different outcomes in the use of peritoneal dialysis versus HD may explain some of the vitamin K antagonists (VKA) discrepancy seen in clinical trial data, as peritoneal dialysis has fewer bleeding complications and effect on INR stability (Figure 1). 29,30

Overall data for stroke reduction with warfarin use in patients with end-stage renal disease and atrial fibrillation. The dialysis method used may contribute to the variability of outcomes seen. HD indicates hemodialysis; PD, peritoneal dialysis; RRT, renal replacement therapy; GFR, glomerular filtration rate. From the study by Tan et al 25 with permission.
Moreover, warfarin use poses several problems in patients with ESRD. 31 In particular, the deleterious effects of warfarin on patients undergoing dialysis include effects on calcium metabolism 32 leading to calciphylaxis, tender skin lesions due to thrombosis/calcification of skin arteries. 33 The mechanism is thought to involve inhibition of the vitamin K–dependent activation of matrix proteins 34 and may also contribute to development of arteriovenous malformations in the brain. 35 Hence, it is possible that the increased risk of intracerebral hemorrhage may be unique to warfarin and not only due to an anticoagulated state. 36 Meta-analysis data suggest that even when in the therapeutic range there is an increased risk of major bleeding with warfarin. 37 Furthermore, the interaction with heparin given peri-dialysis makes it challenging to stay within therapeutic INR for patients with ESRD. 38
In summary, the available data are weak and conflicting regarding the need for routine use of warfarin. However, it is the best agent to use when a stable INR can be maintained and the bleeding risk is modest. Also, a lower INR of 1.8-2.5 in patients with ESRD does cause less bleeding complications, which may represent an easier therapeutic window to target. 39
Non–Vitamin K Oral Anticoagulants in ESRD
Non–vitamin K oral anticoagulants (NOACs) were developed in the last decade and have shown clinical noninferiority or superiority compared to warfarin in stroke prevention in patients with AF. 40 This section will review the 4 main agents (apixaban, rivaroxaban, dabigatran, edoxaban). It is important to note at the outset that none of these agents have any randomized data available from patients with ESRD. Despite this, apixaban has received FDA approval for its use in patients with ESRD based primarily on pharmacokinetic data from a small number of patients. Real-world prescribing practices reflect approximately 10% to 20% use of NOACs in ESRD. 1 See Figure 2 for a summary of their metabolism and excretion.

Pharmacokinetics of (A) warfarin, (B) apixaban, (C) rivaroxaban, (D) dabigatran, and (E) edoxaban. from the study by Chan et al 41 with permission.
Apixaban
This was the first NOAC to receive FDA labeling in January 2014 for patients with severe renal impairment, including those on HD. Even for HD patients, the dose was the same unless patients were over 80 years or weighed less than 60 kg, in which case the recommended dose was 2.5 mg twice daily. Of note, no international society guideline has endorsed this recommendation (Table 2). The FDA’s recommendation was based on a pharmacokinetic study of a single dose in 8 patients matched to 8 with normal renal function. This initial pharmacokinetic study of apixaban in healthy participants achieved steady-state within 2 to 3 INR of the first dose, with modest accumulation. 42 There was little pharmacokinetic variability with doses proportional over a 10-fold (2.5-25 mg) range. In this study, Wang et al 43 found 36% higher total apixaban exposure in participants with ESRD given 5-mg apixaban following a HD session compared with healthy participants. For the study protocol, HD sessions were performed heparin free, which differ from real-world ESRD practice. This formed the basis of the FDA recommendation that apixaban be used without dose modification in patients with ESRD on HD.
However, more recent data using multiple doses of apixaban in HD patients challenge the findings from the Wang et al. study. In a similar sized pharmacokinetic study of 7 patients with ESRD, apixaban 2.5 mg twice daily led to significant accumulation with only 4% removal by dialysis, again with no heparin used during dialysis. They also found that the 5-mg dose given twice daily led to supratherapeutic drug exposure, increasing the risk of bleeding. 44
Clinically, there is sparse data comparing apixaban to warfarin in patients with renal failure. One study is a retrospective matched cohort study of 143 patients. 45 The study was powered for safety, and found no significant difference in major bleeding between warfarin and apixaban (17.8% vs 9.6%, P = .149) or composite bleeding (27.4% vs 21.9%, P = .442). The population was high risk, with average CHADSVASc score of 6, HASBLED of 3, and stroke rate of 7.5% (P = NS between groups). Apixaban users received mainly 2.5 mg twice a day (61.6%) and were followed for 369 days. There were 9.5 major bleeding events per 100 person-years, 3.5 times greater than the rate in the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation trial where most patients received 5 mg twice daily. 46
A second similar sized retrospective study compared 120 HD patients receiving warfarin to 40 patients receiving apixaban 2.5 mg (n = 23) or 5 mg (n = 17) twice daily. Seven major bleeding events occurred in the warfarin arm compared with none on apixaban (P = .34). Rates of clinically relevant nonmajor bleeding events and minor bleeding were similar. The study was not powered to detect clinical efficacy or major bleeding. 47
A recent study with over 25 000 patients with ESRD and AF found similar rates of stroke/systemic embolism between apixaban and warfarin (hazard ratio [HR]: 0.88, 95% confidence interval [CI]: 0.69 -1.12; P = .29), but apixaban had a lower risk of major bleeding (HR: 0.72, 95% CI: 0.59-0.87; P < .001). Notably, standard dose apixaban (5 mg twice daily; n = 1034) was associated with significantly lower risks of stroke/systemic embolism and death compared to either reduced dose apixaban (2.5 mg twice daily; n = 1317; HR: 0.61, 95% CI: 0.37-0.98, P = .04 for stroke/systemic embolism; and HR: 0.64, 95% CI: 0.45-0.92, P = 0.01 for death) or warfarin (HR: 0.64, 95% CI: 0.42-0.97, P = 04 for stroke/systemic embolism; and HR: 0.63, 95% CI: 0.46-0.85, P = .003 for death). 48
Given the paucity of data, no firm recommendation can be made regarding apixaban. The routine use of 5 mg twice daily perhaps may not be advisable in all patients, even with the current body weight, age, and creatinine caveats.
Rivaroxaban
Rivaroxaban received FDA approval in April 2011 for prevention of stroke and systemic embolism in AF. In August 2016, the FDA labeling of rivaroxaban was updated to state that the 15-mg dose could be used in patients with ESRD. However, the label contained a caveat that no clinical effectiveness data have been published in such patients. 49
The data cited by the FDA 12 stem from the following pharmacokinetic studies. In 8 patients receiving HD, a 15-mg dose of rivaroxaban caused a 56% increase in post-dialysis area under the curve (AUC), compared with healthy participants. There was high inter-patient variability within levels, even among healthy participants. The authors commented this reflected decreased clearance in patients with ESRD, mirroring results in patients with moderate chronic kidney disease. 50 Another study of 18 patients on HD confirmed that dialysis did not remove rivaroxaban significantly. 51 This study also examined steady state levels achieved after 7 days of 10-mg rivaroxaban daily and demonstrated trough levels similar to those in patients receiving 15 mg daily with moderate CKD in the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET-AF) trial. 52,53 Finally, the steady state AUC with 10 mg of rivaroxaban in patients with ESRD was comparable to that seen in healthy participants receiving 20 mg. 54 This study has been criticized for large interpatient variability 41 in trough levels of rivaroxaban, possibly reflecting the small sample size (n = 6). 51
The largest clinical study by Chan et al utilized a database of dialysis patients comprising 29 977 HD patients with AF. 55 Of those, 244 patients were prescribed rivaroxaban for on average 106 days. This group was associated with a higher risk of hospitalization or death from bleeding when compared with warfarin (rate ratio, 1.38; 95% CI: 1.03-1.83; P = .04). Unadjusted event rates of major bleeding varied with dose used (42.6 events/patient year with rivaroxaban 20 mg, and 23.9 events/patient year with rivaroxaban 15 mg). However, their definition of major bleeding varied from that used by the International Society of Thrombosis and Haemostasis. In adjusted survival analyses risk of hemorrhagic death was greater with rivaroxaban (HR: 1.71; 95% CI: 0.94-3.12; P = .07) compared to warfarin. The study was underpowered to detect differences in stroke and arterial embolism, but notably, there were no strokes on rivaroxaban. Likely secondary to the higher dose of rivaroxaban in this high risk population, a major bleeding rate of 68.4 per 100 person-years was recorded in the study (more than 18-fold higher than the rate observed in patients with poor renal function during the ROCKET-AF trial 56 ).
With the 2019 AHA/ACC/HRS focused update on AF guidelines not recommending rivaroxaban in patients with ESRD or dialysis (Table 4), it is likely that there will be no role for this agent in the future.
Dabigatran
Dabigatran was the first NOAC approved by the FDA for use in AF in October 2010. In the United States, a 75-mg dose is recommended in patients with GFR of 15 to 30 mL/min/1.73 m2, whereas in Europe, a 110-mg dose is recommended for patients with GFR between 30 to 49 mL/min/1.73 m2 and high risk of bleeding (Table 2). The 75mg dose is not approved in Europe for patients with severe CKD. Approximately 80% to 85% of the drug is excreted by the kidneys mostly through glomerular filtration (Figure 2). The half-life of the drug is 9 hours but increases to 25 to 30 hours in individuals with a creatinine clearance <30 mL/min/1.73 m2. 57 Dabigatran is also the only OAC that is dialyzable, with 50% to 60% of the drug eliminated during a 4-hour treatment. 58
Dabigatran exposure negatively correlates with renal function, with a 6.3-fold higher AUC in patients with severe CKD after a single dose, compared with healthy participants. 59 Hence, 150- or 110-mg dabigatran are not indicated in Europe in patients with severe CKD or ESRD (Table 2). The FDA approved use of a 75-mg dose in patients with GFR between 15 and 30 mL/min/1.73 m2 based on pharmacokinetic studies but offers no dosing recommendations for ESRD. A simulation study suggested that a 75- or 110-mg dose taken once daily would result in therapeutic levels in HD patients. 60
The only clinical data comes from the aforementioned study by Chan et al, where 281 patients with ESRD received dabigatran for an average of 168 days. Major bleeding risk increased by 48% and fatal bleeding risk increased even more, by 88% compared to warfarin. 55 As dabigatran remains contraindicated in ESRD and is seldom used off-label in this population, it is unlikely that more population-based safety data on dabigatran in ESRD will emerge.
Edoxaban
Edoxaban was approved for use by the FDA in January 2015. The recommended dose in patients with GFR 15 to 50 mL/min/1.73 m2 is 30 mg daily with no recommendation for ESRD. In subgroup analyses of Effective Anticoagulation With Factor Xa Next Generation in Atrial Fibrillation–Thrombolysis In Myocardial Infarction 48 among patients with GFR between 30 and 50 mL/min/1.73 m2, edoxaban remained noninferior for stroke prevention compared with warfarin. 61 A prospective safety study of 93 Japanese patients found similar 3-month bleeding risk in patients with stage 4 CKD on 15 mg of edoxaban compared to 30 or 60 mg of edoxaban in patients with mean GFR of 70 mL/min/1.73 m2. 62
There is little pharmacokinetic data on edoxaban in ESRD, and as a result, its use is not recommended. 11
Ongoing Trials
A number of ongoing studies are underway to address the deficiencies in data in patients with ESRD.
These include Renal Hemodialysis Patients Allocated Apixaban Versus Warfarin in Atrial Fibrillation (NCT02942407), an American, open-label, blinded end point clinical trial, recruiting patients since December 2016 (aiming for n = 720) with an estimated completion date in May 2019. Patients are randomized to 5 or 2.5 mg apixaban twice daily or to warfarin. 63 Primary outcome is time to major or clinically relevant bleeding.
The next is Compare Apixaban and Vitamin-K antagonists in Patients with Atrial Fibrillation and End-Stage Kidney Disease (NCT 02933697), a prospective parallel-group trial recruiting patients (aiming n = 222) since April 2017 and will end in April 2019. Compare Apixaban and Vitamin-K antagonists in Patients with Atrial Fibrillation and End-Stage Kidney Disease will assess the safety of apixaban 2.5 mg twice daily versus phenprocoumon (INR target 2-3) in patients on HD with AF. 64 The primary outcome is incidence of major and clinically relevant bleeding, as well as specific bleeding in dialysis patients.
The STOP- HARM (The Strategy to Prevent Hemorrhage Associated with Anticoagulation in Renal Disease Management Trial, NCT02885545) randomized trial will assess whether left atrial appendage occlusion with a Watchman device confers better protection from strokes than oral anticoagulation with VKA (INR target 2-3), rivaroxaban 15 mg daily or apixaban 2.5 mg twice daily in patients with dialysis-dependent CKD. Patient recruitment started in September 2016 and will continue until December 2021. 65 Primary outcome is time to major bleeding. Unfortunately, recruitment was withdrawn in March 2018 due to no patients being enrolled in the first year.
Notably, no trial has a third arm, namely, no oral anticoagulation or intervention.
Key Knowledge Gaps
Many gaps remain in our treatment of this high-risk challenging group of patients. Is no drug better than any drug? 66 Also, how do OACs interact with antiplatelets for patients with CAD, for example, post-PCI? 67 Furthermore, there is a growing debate over the role of device therapy in stroke patients with ESRD, with registry data showing similar efficacy and safety in patients with CKD. 68 There is a clear attractiveness in patients with ESRD, given the requirement for only short-term periprocedural warfarin, but there are also a lack of data from left atrial appendage occlusion trials in this group. The role of reversal agents in cases of overdose or major bleeding is also yet to be clarified in patients with ESRD. Only dabigatran has a specific reversal agent (idarucizamab) and is substantially removed with dialysis. The FDA has recently approved an agent (Andexanet) for reversal of apixaban and rivaroxaban, but the studies used to base this recommendation on did not include patients with ESRD. 69
Conclusions
Preventing stroke in patients with AF and ESRD provides unique challenges, and physicians currently lack clear guidance due to a shortage of clinical data. The randomized trials for approval of all NOACs excluded patients with ESRD, and hence, smaller pharmacokinetic studies form the base of a guideline update for patients with ESRD. The available clinical data are inconclusive and conflicting highlighting the difficulties in a “one-size-fits-all approach” to prescribing OACs. There is conflicting evidence for low-dose apixaban and rivaroxaban, but until definitive studies are published in patients with ESRD, this will be an area where risks and benefits must be clearly documented and discussed with the patient. Populations where low dose medications have been used with success require revalidation in a real-world population to ensure efficacy and safety. As for warfarin, the advent of pharmacogenomics holds promise to tailor therapy, but the ease and attraction of NOACs mean these will almost certainly be the agents investigated in future studies of stroke prevention in ESRD. Also, the role of LAA occlusion devices is currently under investigation in ESRD but likely to find an increasing role. Even once more data are available, dialysis patients and physicians will still have to work together to decide on the best approach for stroke prevention.
Footnotes
Author Contributions
J.Z. and A.B. contributed to conception, design, acquisition, analysis, and interpretation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
