Abstract
Nonvalvular atrial fibrillation (AF) confers an increased risk of thromboembolism, with a 5-fold higher risk of ischemic stroke. Oral anticoagulation (OAC) has shown to be highly effective in preventing stroke and mortality compared to placebo and is also used in patients without AF for both treatment and prophylaxis of venous thromboembolism. The OAC halts the coagulation by different mechanisms. Until recently, the only option was the vitamin K antagonists (VKAs), but their inherent limitations have promoted the development of novel oral anticoagulants (NOACs), which may offer efficacious and safer alternatives. Patients should be carefully selected to receive the most suitable treatment for each one. As the VKA efficacy and safety largely rely on the time the patient remains within the therapeutic range, this could be a useful selection criterion. Bleeding remains the main complication of all OACs. Although clinical trials of stroke prevention in AF have shown a significant reduction in hemorrhagic stroke and intracranial bleeding with the NOACs, as prescriptions are increasing, clinicians need to be prepared to accurate management of bleeding complications. Withholding the drug is usually enough for most cases of mild bleeding, but in patients with major, life-threatening bleeding, other measures, such as fluid replacement and blood transfusion, might be necessary while waiting for specific reversal agents that may reach the market soon. In case of acute bleeding, the accurate estimation of anticoagulant effect could also be needed, but the currently available coagulation tests only offer a qualitative measure. While awaiting long-term safety data, the choice between all these available therapies should be based on patient preferences, compliance, and ease of administration as well as on local factors affecting cost-effectiveness. But the increasing variety of therapeutic options when chronic OAC is needed can only improve the provided health care.
Introduction
Nonvalvular atrial fibrillation (AF) confers an increased risk of stroke and thromboembolism, with a 5-fold higher risk of ischemic stroke. When stroke occurs in association with AF, mortality rates as well as the functional deficits are higher, since strokes are more severe leading to long-term disability. 1,2
In AF, oral anticoagulation (OAC) is highly effective in preventing stroke and mortality compared to placebo/control. 3 The decision of thromboprophylaxis has classically been based on stroke risk, as assessed by various stroke risk stratification schemes. 4 Anticoagulation is also used in patients without AF for both treatment and prophylaxis of venous thromboembolism (VTE), a potentially life-threatening event affecting approximately 70 cases per 100 000 each year in the United States. 5
Anticoagulants interfere with the coagulation cascade by different mechanisms. Until recently, the only oral option for chronic OAC was the vitamin K antagonist (VKA) class of drugs that induce the synthesis of nonfunctioning coagulation factors by blocking the incorporation of vitamin K. When used as thromboprophylaxis for AF, treatment with VKAs results in a 64% stroke risk reduction when compared to placebo/control and a 37% stroke risk reduction compared to antiplatelet therapy. 3 Moreover, anticoagulation therapy reduces all-cause mortality by 26% compared to placebo/control in AF.
Pharmacology
Vitamin K Antagonists
The VKAs, such as warfarin, are highly water soluble and thus absorbed from the gastrointestinal tract with high bioavailability. They are then cleared from the bloodstream through oxidative metabolism by liver enzymes of the cytochrome P450 system. The VKAs differ mainly in their half-lives, ranging from 8 to 270 hours, warfarin having an intermediate half-life (40 hours). 6 The VKA dose required to achieve therapeutic anticoagulation as well as the response to a fixed dose is varied, as this depends on multiple factors, including various food, drug, and alcohol interactions as well as age-related alterations and genetic variations (polymorphisms affecting either cytochrome p450 system, as 2CYP9 *1, *2, and *3, or VKORC, as rs2323991). In that sense, pharmacogenetic information could help to assess the initial dose, potentially being the path to a personalized medicine. 7 However, VKAs are highly susceptible to drug interactions, which can boost or decrease the anticoagulant effect by inhibiting or enhancing, respectively, its clearance and therefore, close monitoring is always needed. 8 Situations inhibiting the synthesis of vitamin K-dependent factors (such as liver impairment) entail an increase in the anticoagulant effect as well as the ones that interfere with hepatic metabolism (as drug competition or liver congestion). Furthermore, the achieved anticoagulation response depends also on the fluctuations in the dietary intake of vitamin K as well as on endogenous production of vitamin K by gastrointestinal bacteria. Its narrow therapeutic window leads to the need for regular, close monitoring and for dose adjustments to achieve the desired international normalized ratio (INR) target of 2.5 (range 2.0-3.0), whose maintenance within the therapeutic range helps to reduce the risk of adverse events, both thrombotic and hemorrhagic ones.
Indeed, the efficacy and safety of VKAs strongly depend upon the time in the therapeutic range (TTR; with maximum benefits shown when the TTR is between 70% and 80%). 9,10 Poor control of anticoagulation intensity increases the risks for both thrombotic and hemorrhagic events. 10
The inherent limitations of VKA therapy complicate its management 11 and have promoted the development of novel oral anticoagulants (NOACs), which may offer efficacious and safer alternatives (with appropriate patient selection). 12 Unlike the VKAs, NOACs are low-molecular-weight synthetic molecules with a specific target in the coagulation pathway. With a rapid action onset and offset, they directly inhibit a specific coagulation factor (such as thrombin for dabigatran and factor Xa for both rivaroxaban and apixaban), and their predictable pharmacology (as shown in Table 1) leads to a stable dose-related anticoagulant effect, allowing a fixed dose and making regular monitoring unnecessary. 17
Characteristics of Main Oral Anticoagulants.

Abbreviations: BID, twice daily; INR, international normalized ratio; OD, once daily; SC, subcutaneous; VKAs, vitamin K antagonists; VTE, venous thromboembolism; AF, atrial fibrillation.
a Approved only in Europe.
Dabigatran
Being highly polar and lipophobic, dabigatran, the oral direct thrombin inhibitor, is not absorbed well from the gut. Thus, dabigatran is administrated as an absorbable prodrug, dabigatran etexilate, containing a tartaric acid pellet that creates an acid microenvironment that enables gut absorption. 18 Once absorbed, dabigatran etexilate is converted into the active drug (dabigatran) by blood esterases, with a metabolism independent of the P450 cytochrome, and thus subject to less drug interactions. 19 The high rate of renal excretion (85%) makes dose adjustment necessary in patients with renal impairment. 20
Factor Xa Inhibitors
Rivaroxaban
With a high bioavailability (80%), rivaroxaban, is metabolized via oxidative and hydrolytic pathways involving different classes of enzymes, with a half-life of 5 to 12 hours. 21,22 Due to its mechanisms of elimination, rivaroxaban is contraindicated in patients with a creatinine clearance (CrCl) <15 mL/min and should be administrated with some caution in patients with impaired renal or liver function. 22
Apixaban
In contrast to rivaroxaban and dabigatran, apixaban (also with an oral bioavailability about 50%) is excreted mostly in the feces (only 25% is excreted by the kidneys and therefore more suitable in cases of renal impairment) after a half-life of 8 to 15 hours. 23
Effectiveness
Atrial Fibrillation
In the Randomized Evaluation of Long-term Anticoagulation Therapy (RE-LY) trial, dabigatran showed its noninferiority to warfarin for preventing stroke or systemic embolism (rate of 1.53% for dabigatran 110 mg arm and 1.11% in dabigatran 150 mg group vs 1.69% for the warfarin-treated patients; P < .001 in both the cases; Table 2). 13
Stroke Prevention in Patients With AF.a

Abbreviations: BID, twice daily; OD, once daily; AF, atrial fibrillation; TTR, time in the therapeutic range.
a Results from noninferiority trial and as-protocol groups. Data from ENGAGE AF-TIMI pending publication.
The Rivaroxaban Once daily oral direct factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in AF (ROCKET-AF) trial showed the noninferiority of rivaroxaban to warfarin, with rates of stroke and systemic embolism of 1.7% in rivaroxaban-treated patients (vs 2.2% in warfarin treated). 14
Apixaban was found to be superior to warfarin in Apixaban for Reduction In STroke and Other ThromboemboLic Events in atrial fibrillation (ARISTOTLE) trial, for both stroke prevention (1.6% vs 1.27%; P = .01) and lower all-cause mortality (3.52% vs 3.94% in warfarin-treated patients; P = 0.046). 15
Venous Thromboembolism
Venous thromboembolism is common (with an estimated annual incidence of approximately 1-2/1000 persons 24 ), and much attention has been directed toward understanding its risk factors and optimal treatment strategies in order to prevent recurrence, which is fatal in 3.6% to 10% 24 of patients.
Classic risk factors for VTE include an elderly age (≥70 years), active cancer, recent (≤1 month) surgery or major trauma, immobilization, pregnancy and postpartum, use of oral contraceptives or hormonal treatment, and antiphospholipid antibody syndrome, 25 but some other medical conditions related to inflammatory disorders (as acute infectious disease, rheumatologic disorders, and inflammatory bowel disease) have also shown a clear association with VTE risk. 26 Acute congestive heart failure (New York Heart Association Class III or IV) and acute respiratory disease (respiratory failure or an exacerbation of chronic obstructive pulmonary disease) are also well-recognized risk factors for VTE. 26
Given what life-threatening event might result a VTE, anticoagulant therapy pursues 2 objectives: active treatment of the acute episode (for the 3 first months) and prevention of new ones. Although low-molecular-weight heparins followed by VKAs remain the first-line treatment, 27 some NOACs can also be used for VTE primary prophylaxis (Table 3), and only rivaroxaban has Food and Drug Administration (FDA) approval (for primary prophylaxis, acute treatment, and long-term prophylaxis of deep vein thrombosis [DVT]), which additionally offers the advantage of a single-drug treatment for the whole period, including acute and extended treatment (see Table 4). 31
Clinical Trials With New Oral Anticoagulants Regarding Venous Thromboembolism—Venous Thromboembolism Prophylaxis.
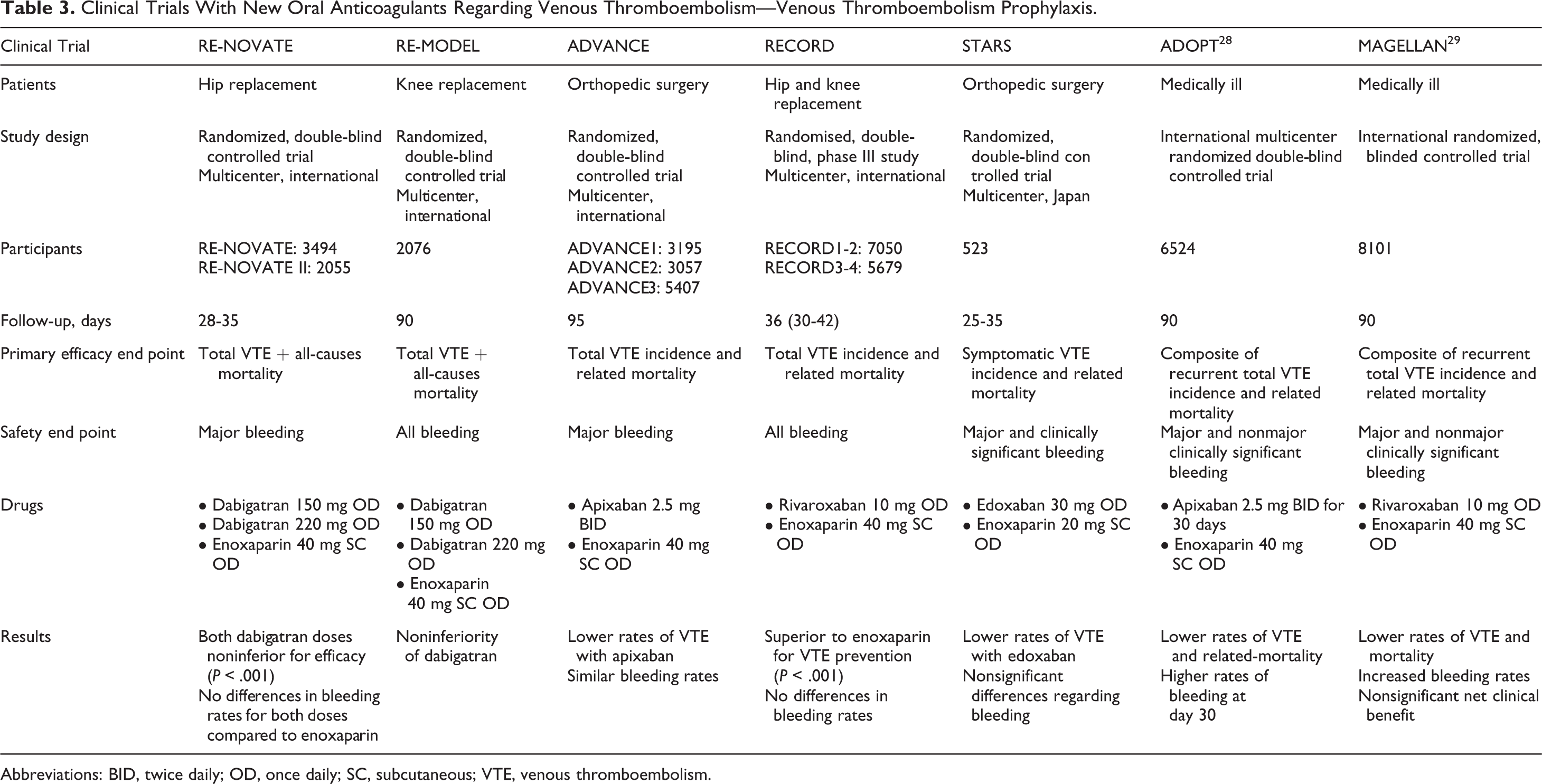
Abbreviations: BID, twice daily; OD, once daily; SC, subcutaneous; VTE, venous thromboembolism.
Clinical Trials With New Oral Anticoagulants Regarding Venous Thromboembolism—VTE Treatment.

Abbreviations: BID, twice daily; OD, once daily; SC, subcutaneous; VKA, vitamin K antagonists; VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism.
While all clinical guidelines advocate the use of anticoagulant therapy as VTE prophylaxis and initial treatment, the optimal duration remains uncertain, needing to achieve a balance between the estimated risk of recurrence and the risk of bleeding complications. Venous thromboembolism caused due to a transient risk factor (as surgery, trauma, or pregnancy) have a lower risk of recurrence and therefore do not benefit from prolonged treatment. 24 In contrast, among patients with unprovoked VTE, the risk of recurrence has been shown to be highest in the first 6 to 12 months after the initial episode (reaching 10% in some cases 24 ), but some level of risk may continue for 10 years. 33
In that sense and given the results of the clinical trial on NOACs, extended treatment regimens have been recommended. These trials highlighted that the risk of VTE increases beyond the time of hospital discharge, 29 but unfortunately higher rates of clinically relevant bleeding could arguably lead to no net clinical benefit of extended therapy. 30 Therefore, the length of anticoagulant treatment after a VTE episode remains controversial, although some meta-analysis support the use of prolonged thromboprophylaxis. 34
Safety Profile
Poor control of anticoagulation intensity using VKAs increases the risks for both thrombotic and hemorrhagic events. 10 The efficacy and safety of VKAs strongly depend upon TTR, with INR between 2.0 and 3.0. Maximum benefits from VKAs are achieved when the TTR is more than 70% (and TTR <40% offsets the benefit of VKA therapy 4 ), but this is seldom accomplished in everyday clinical practice, 9,10 and efforts have been made to improve TTRs. Well-controlled INRs are associated with a significant decrease in the annual rates of stroke and mortality in patients with AF taking warfarin. 3,35
Bleeding remains the main complication of OACs. Indeed, among physicians, the fear of bleeding in patients leads to a lack of prescription of OACs and reduced uptake particularly in patients who may otherwise benefit greatly from its use. 36 –39 This underuse is particularly pronounced in elderly patients (>80 years old) and patients with renal dysfunction, owing to the concern that the bleeding risk could outweigh the benefits of VKAs in these populations. 40,41 Patients with mild–moderate chronic kidney disease might still be suitable for some NOACs, even after dose reduction (for specific doses, refer to Figure 1). However, they should be avoided in severe renal impairment (CrCl < 15 mL/min) pending more clinical trial data. 42

Dose reduction according to renal function. *only approved in the United States, not in Europe. BID indicates twice daily; INR, international normalized ratio; OD, once daily; VKAs, vitamin K antagonists.
In their large phase 3 clinical trials of stroke prevention in AF, there was a consistent significant reduction in hemorrhagic stroke and intracranial bleeding with all 3 NOACs, and this is most probably a key benefit of the NOACs compared with warfarin. 13 –15,43,44 Moreover, the choice of a lower dose in patients at high bleeding risk might be a suitable option, and recent European guidelines 45 recommend dabigatran 150-mg dose for patients at low bleeding risk (assessed by a Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile INR, Elderly, Drugs/alcohol concomitantly [HAS-BLED] score <2) and the 110-mg dose for those patients at higher bleeding risk (HAS-BLED >3), although the last one is not available in the United States, and thus this recommendation is not applicable worldwide. However, when prescribing any anticoagulant, physicians should remain aware of the inherent bleeding risk, and NOACs could perhaps transmit a false feeling of security. The European Heart Rhythm Association has recently published a practical guide for the clinical use of NOACs as well as recommendations for follow-up and the management of overdose or bleeding. 42
The benefit from NOACs could be particularly enhanced where there is poor anticoagulation control on warfarin arms. In the phase 3 trials, the mean TTR varied widely by countries and clinical center (ranging from 44% to 78%), reflecting the wide variability in clinical care compared to in the global trials (RE-LY, 13 ARISTOTLE, 15 and ROCKET 14 reported overall mean TTRs of 64%, 62%, and 55%, respectively). Ancillary analyses 46,47 have shown benefits in reduction in both stroke and bleeding events with NOACs across the range of predicted TTR, showing that they are not simply superior or noninferior to warfarin because of poor INR control.
Managing Bleeding
As NOAC prescriptions are increasing, clinicians need to be prepared to deal with bleeding complications (see Figure 2) as well as with needed surgeries. While bleeding events are a known adverse effect of all anticoagulants, some safety concerns have been raised regarding the NOACs due to the lack of any specific antidote for a rapid reversal in case of emergencies. Management of bleeding while on NOACs depends on the elimination half-life of the agent, which ranges from 8 to 17 hours (see Table 1); and therefore, stopping the medication drops rapidly the blood levels, with an expected restoration of hemostasis within 12 to 24 hours after the last dose. 42 Withholding the drug is usually enough for most cases of mild bleeding, but in patients with major, life-threatening bleeding, other measures might be necessary. Fluid replacement and blood transfusion would be part of supportive management (as most of NOACs have a renal excretion, maintaining renal perfusion should be a priority goal), and in very severe cases, the use of nonspecific reversal agents such as fresh frozen plasma (FFP), prothrombin complex concentrates (PCCs, which provide more prothrombin to increase thrombin generation), recombinant-activated factor VII (rFVIIa), activated PCCs (FEIBA, consisting mainly of activated factor X and prothrombin), and so on. 48 However, the safety and usefulness of FFP or PCC are still to be established, because the drug present in plasma may block newly administered coagulation factors as well, and the use of rFVIIa may result in a potentially high risk for arterial thrombosis. 49 Regarding dabigatran, haemodialysis can help reduce drug blood levels, 50 but multiple sessions may be required due to its large distribution volume; the risk of bleeding at venipuncture site should be considered, and thus is more suitable when overdoses rather than to stop an active bleeding. Of note, rivaroxaban and apixaban are not dialyzable due to their high plasma protein binding capacity. 42,51,52

Management of bleeding events in patients under novel oral anticoagulants (NOACs), FEIBA indicates activated prothrombin complex concentrates; PCCs, prothrombin complex concentrates; Adapted from Heidbuchel et al. 42
Specific antidotes, as PRT064445, 53 or monoclonal antibodies (like aDabi-Fab 54 ) are currently under development but still not ready for clinical use.
Assessing Anticoagulation Effect on NOACs
In some urgent situations, as prior to the unexpected surgery, the accurate estimation of anticoagulant effect is also essential. Although NOACs prolong some laboratory coagulation tests (as the prothrombin time with rivaroxaban or the activated partial thromboplastin time or hemoclot [dilute thrombin time] with dabigatran), only qualitative measures can be made. 17,55 In fact, the activated partial thromboplastin time shows a curvilinear dose response to dabigatran and may be used to reflect that a patient is under dabigatran treatment, but it does not provide a quantitative measure. 56 The prothrombin time shows better linear dose–response to both dabigatran and rivaroxaban, although standardization among different laboratories is needed before clinical use. 49
When interpreting the results of coagulation assays, and unlike to what we are used to with warfarin, it is mandatory to know the exact time lapse between drug intake and performance of the test, as their impact on coagulation test varies roughly with the peak level. 42,56 There is also some dependence upon the specific coagulometer and reagent used in each laboratory, and the high variability in reagent’s sensitivity leads to a lack of standard measures to quantify their effect, 57 although assay-specific calibrators and calibration curves can be made at each center. 42 Thus, the anticoagulant intensity of NOACs should not be monitored with any of these laboratory tests, although a normal prothrombin time ratio usually excludes an anticoagulation effect due to rivaroxaban, and a normal activated partial thromboplastin time could exclude one due to dabigatran (Table 1).
The degree of anticoagulation reached can partly be measured by noncoagulation methods (such as Ecarin clotting time or chromogenic methods), which are only available for research purposes, 22,58 as well as the HEMOCLOT test (Hyphen BioMed®), which measures the effect of dabigatran through a modified thrombin clotting time. 51
Despite important advantages over VKAs and their impressive performance in clinical trials, the long-term efficacy and safety of NOACs still require evaluation. Concerns have been raised over a numerical increase (nonsignificant) in the rates of myocardial infarction (MI) with dabigatran 150 mg (0.74%) compared with warfarin (0.53%), initially reported in the RE-LY trial (relative risk [RR] = 1.38; 95% confidence interval [CI] 1.00-1.91; P = .048), although the difference became insignificant after the inclusion of silent MIs (RR = 1.27; 95% CI 0.94-1.71; P = .12). 13,59
The controversy remains as some meta-analyses have reported no differences between NOACs and VKAs, 60 but further studies have reported lower MI rates with warfarin versus other treatments (RR = 0.77; 95% CI 0.63-0.95; P < .01). 61 A protective effect of warfarin against MI rather than an inherent dabigatran-related risk for MI could contribute to this relative increase in MIs. 44,61 However, a recent postmarketing nationwide Danish study of nearly 6000 patients on dabigatran did not find any excess of MIs compared to propensity-matched patients on warfarin. 62
Why would VKAs have a greater protective effect against MI? This question is not easily answered. Theoretically, thrombin inhibition might delay the activation of protein C, by reducing the binding of thrombin to thrombomodulin on the endothelial cells. Activated protein C is known to act as a natural anticoagulant, and thus decreasing its activation might result in a procoagulant phenomenon. 43
Who Should Switch?
Patients with unstable INRs (TTR < 55%) should be carefully considered for switching from VKAs to NOACs. In fact, patients with TTR < 55% or treated with interfering drugs proven to cause INR fluctuations might benefit from NOACs. 49 However, drug compliance and treatment adherence need to be assured by focusing on patient education so that they understand the factors under their control, which might affect efficacy and safety (eg, diet, alcohol, and other medications) while on VKAs. If labile INR is not caused by poor compliance, switching may be considered. Patients with poor compliance and treatment adherence may be a problem since the short half-life of NOACs will leave them unprotected at a greater degree than warfarin (in patients not monitored, the first marker of noncompliance will probably be a stroke or other thrombotic complications). 63 Once daily regimens could be related to greater adherence than twice daily regimens, which can have a major impact with NOACs. 42 Also nonadherence in patients older than 65 years, with chronic conditions such as AF, is estimated to range from 40% to 75%. 49
A new clinical score has recently being developed to assess the probability of good TTR, based upon several clinical characteristics called the SAMe-TT2R2 score, 64 which includes female Sex, Age (<60 years), Medical history (at least 2 of the following: hypertension, diabetes, coronary artery disease/MI, peripheral arterial disease, congestive heart failure, previous stroke, pulmonary disease, hepatic, or renal disease), Treatment (interacting drugs, eg, amiodarone for rhythm control; all 1 point), as well as current Tobacco use (2 points) and Race (noncaucasian; 2 points), as detailed in Table 5. This simple score (SAMe-TT2R2) can help predict poor INR control and aid decision making by identifying those patients with AF who would do well on VKA (SAMe-TT2R2 score = 0-1) or, conversely, those (ie, SAMe-TT2R2 score ≥ 2) who require additional interventions to achieve acceptable anticoagulation control. In that sense, patients with a poor predicted TTR (ie, SAMe-TT2R2 score ≥ 2) would be candidates who are more suitable for NOACs.
SAME-TT2R2 Definition.

a Defined as more than 2 of the following: hypertension, diabetes, coronary artery disease/myocardial infarction, peripheral arterial disease, congestive heart failure, previous stroke, pulmonary disease, hepatic, or renal disease.
In more severe degrees of renal dysfunction (CrCl: 15-30 mL/min), warfarin might be a preferable option, due to limited experience with NOACs in this setting (notwithstanding the FDA approval of dabigatran 75 mg twice daily and rivaroxaban 15 mg in such patients, despite the lack of trial data) and the almost complete hepatic metabolism of warfarin. This is particularly relevant for patients on hemodialysis: dabigatran would be cleared and other NOACs have not been evaluated. Therefore, these patients should remain under VKA treatment. However, renal function needs to be closely monitored (even in patients without known renal impairment), 65 especially among patients receiving dabigatran or elderly patients. 42 We should bear in mind that over time, patients get older, may undergo some renal impairment, new drugs might be added to their daily treatment, and so on, and, therefore, constant reassessment should be provided.
The NOACs have not already been evaluated in patients with heart valve prosthesis (both mechanical and bioprothesis), and 1 phase 2 trial (RE-ALIGN) with dabigatran was stopped early due to the increased risk of thromboembolism. 66 As safety and efficacy profiles in such patients cannot be determined, patients should remain on warfarin.
Conclusions
The NOACs have shown to be as effective as VKAs in preventing stroke and a trend to present fewer major bleeding events. However, bleeding still remains the main complication of chronic anticoagulation. Specific reversal agents are under development, but until then, in the case of severe hemorrhage, alternatives such as nonspecific hemostatic reversal agents must be considered.
Although monitoring is not necessary with NOACs, patients need to be followed up, and risk assessment should remain a dynamic process, with constant reassessment during the follow-up in order to assure the most accurate treatment to each patient.
While awaiting long-term safety data, the choice between all these available therapies should be based on patient preferences, compliance, and ease of administration as well as on local factors affecting cost-effectiveness. In that sense, finding an accurate way to predict patients who will remain steady on VKA and who would better fit for NOAC should be a priority, and a novel score, the SAMe-TT2R2, seems a reasonable way. But the increasing variety of therapeutic options when chronic OAC is needed can only improve the provided health care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Lip has served as a consultant for Bayer, Astellas, Merck, Sanofi, BMS/Pfizer, Daiichi-Sankyo, Biotronik, Portola, and Boehringer Ingelheim and has been on the speakers bureau for Bayer, BMS/Pfizer, Boehringer Ingelheim, and Sanofi Aventis. He serves as a DSMB committee member for the BRUISE CONTROL trial. Dr Roldán has received funding for consultancy and lecturing from Bristol-Myers-Squibb, Boehringer Ingelheim, and Bayer. Dr Gallego holds a grant from the Alfonso Martín Escudero Foundation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
