Abstract
Background:
Cardiovascular guidelines do not give firm recommendations on statin therapy in patients with gout because evidence is lacking.
Aim:
To analyze the effectiveness of statin therapy in primary prevention of coronary heart disease (CHD), ischemic stroke (IS), and all-cause mortality in a population with gout.
Methods:
A retrospective cohort study (July 2006 to December 2017) based on Information System for the Development of Research in Primary Care (SIDIAPQ), a research-quality database of electronic medical records, included primary care patients (aged 35-85 years) without previous cardiovascular disease (CVD). Participants were categorized as nonusers or new users of statins (defined as receiving statins for the first time during the study period). Index date was first statin invoicing for new users and randomly assigned to nonusers. The groups were compared for the incidence of CHD, IS, and all-cause mortality, using Cox proportional hazards modeling adjusted for propensity score.
Results:
Between July 2006 and December 2008, 8018 individuals were included; 736 (9.1%) were new users of statins. Median follow-up was 9.8 years. Crude incidence of CHD was 8.16 (95% confidence interval [CI]: 6.25-10.65) and 6.56 (95% CI: 5.85-7.36) events per 1000 person-years in new users and nonusers, respectively. Hazard ratios were 0.84 (95% CI: 0.60-1.19) for CHD, 0.68 (0.44-1.05) for IS, and 0.87 (0.67-1.12) for all-cause mortality. Hazard for diabetes was 1.27 (0.99-1.63).
Conclusions:
Statin therapy was not associated with a clinically significant decrease in CHD. Despite higher risk of CVD in gout populations compared to general population, patients with gout from a primary prevention population with a low-to-intermediate incidence of CHD should be evaluated according to their cardiovascular risk assessment, lifestyle recommendations, and preferences, in line with recent European League Against Rheumatism recommendations.
Keywords
Introduction
Gout is the most frequent inflammatory arthritis. Both the prevalence and incidence of gout are higher in men than in women and are rising in developed countries, 1 and it is associated with increased cardiovascular mortality and morbidity 2 -4 and cardiovascular risk factors such as hypertension, 5 chronic kidney disease (CKD), 6 and metabolic syndrome. 7 The coexistence of gout with these diseases suggests commonalities in the inflammatory pathogenic mechanisms related to the atherogenic process 8,9 and in disease-related treatment mechanisms such as non-steroidal anti-inflammatory drugs (NSAIDs) or corticosteroids, 10 which have also been associated with an increase in cardiovascular risk. 11
In 2016, the European League Against Rheumatism (EULAR) task force recommended treatment of gout flares. Colchicine, NSAIDs, and corticoids are the first-line therapies of the acute episodes because of their anti-inflammatory effects, and urate-lowering drugs are the first choice at the new onset of this disease. 12 In addition to the active treatment of the disease, identification of cardiovascular risk factors and active cardiovascular risk management were particularly recommended. However, no specific recommendations on statin therapy were made. Moreover, local cardiovascular guidelines do not give firm recommendations in patients because evidence is lacking. The potential benefits of statins in relation to their pleiotropic effect has not been evaluated in individuals with gout; most studies involving statins have focused on other inflammatory arthritis, 13,14 with no studies on their effectiveness in primary prevention of cardiovascular diseases (CVDs) in these patients. Evidence from electronic medical records data can be useful in cardiovascular risk management decision-making and in evaluating treatment effectiveness in clinical settings.
The present study aimed to analyze the effectiveness of statin therapy in primary prevention of coronary heart disease (CHD), ischemic stroke (IS), and all-cause mortality in a population with gout.
Methods and Materials
Study Design and Data Source
Retrospective population-based cohort study using a research-quality set of anonymized longitudinal patient records from the Information System for the Development of Research in Primary Care (SIDIAPQ), 15 which contains anonymized longitudinal data on approximately 2 million patients, attended by 1365 General Practitioners (GPs) and has been used in numerous epidemiological studies. 16 -19 The full database contains records for ∼6 million people (80% of the Catalan population, constituting 10.2% of Spain’s population).
The information recorded includes demographic and lifestyle factors relevant to primary care settings (body mass index [BMI], smoking status, alcohol use); clinical diagnoses, outcomes, and events, coded by International Classification of Diseases, Tenth Revision (ICD-10); referrals and hospital discharges (coded by ICD-9); laboratory tests; and prescribed medications dispensed by community pharmacies. Ethics approval for observational research using SIDIAPQ data was obtained from our local ethics committee.
Study Population
All patients aged 35 to 85 years with an active gout code diagnosis (ICD-10 codes M10) were eligible for study inclusion. Exclusion criteria included active diagnostic codes at baseline for cancer, dementia, paralysis, organ transplant, dialysis or institutionalized care, missing data for MEDEA deprivation index score, 20 and previous history of symptomatic peripheral arterial disease, CHD, IS or hemorrhagic stroke, revascularization, heart failure or cardiac therapy (Anatomical Therapeutic Chemical Classification code C01), or cholesterol-lowering drugs other than statins taken between July 2006 and December 2008.
Study Enrollment
Patients were enrolled from July 2006 to December 2008 and censored at the date of transfer out from SIDIAPQ or the end of follow-up, December 31, 2017.
Statin Exposure
To prevent survivor bias and covariate measurement bias among users of statins (simvastatin, pravastatin, lovastatin, fluvastatin, rosuvastatin, atorvastatin, pitavastatin), only new users were selected and their first date statins were dispensed was set as index date; those same dates were then randomly assigned to include nonusers. Individuals with fewer than 2 invoices for statins during the enrollment period were excluded.
Main analyses were performed considering statin exposure as new users versus nonusers. In descriptive analysis, we classified patients’ exposure to statins according to the drug’s cholesterol reduction capacity, as follows: low, <30%; moderate, 30%-40%; and high, >40%. 21
Outcomes
The SIDIAPQ codes for CVD, previously validated for research use, 19 were identified in both primary care (ICD-10) and hospital discharge records (ICD-9). Primary outcomes were CHD (a composite of Acute Myocardial Infarction (AMI) and angina), IS, and all-cause mortality recorded during follow-up.
Adverse Effects
Liver toxicity and myopathy occurring within 12 months of initiating statins therapy were attributed to the treatment. New-onset diabetes, cancer, and hemorrhagic stroke diagnosed at least 12 months after the first date statins were dispensed were also considered as associated with statin exposure. 22
Baseline Covariates
Baseline period was defined as 1 year before the index date. The following covariates that may have influenced prescription decisions and study outcomes were considered: age, sex, deprivation index developed for Spain by the MEDEA study, 20 systolic and diastolic blood pressure (SBP, DBP; mm Hg), and dichotomous (yes/no) variables for high-risk alcohol intake, smoking, diabetes or record of antidiabetic drug use, hypertension or record of antihypertensive drug use, dyslipidemia, and BMI > 30 kg/m2. Laboratory results were considered for fasting glucose, total cholesterol, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, and triglycerides. Comorbidities were noted (yes/no): atrial fibrillation, CKD, chronic obstructive pulmonary disease, asthma, benign neoplasms, hypo- and hyperthyroidism. Finally, the number of GP visits in the 12 months before index date and other drug uses were recorded: antiplatelets, anti-inflammatory drugs, gout treatments, psychoanaleptics, and psycholeptics. Ten-year CHD risk was calculated using the Framingham function adapted and validated in the Spanish population by the REGICOR study in the population aged 35 to 74 years. 23
Statistical Analysis
Results are expressed as percentages for categorical variables and otherwise as mean (standard deviation, SD) or median [quartiles]. Multiple imputations by chained equations 24 were used to replace missing baseline values for total cholesterol, HDL cholesterol, LDL cholesterol, triglycerides, glucose, SBP, DBP, and BMI (weight and height), as detailed in the Supplementary File.
Due to nonrandom treatment allocation, a propensity score (PS) for statin treatment was calculated, using a logistic model based on potential confounding covariates (contained in Supplementary File). Baseline characteristics before and after PS adjustment were compared using standardized differences, with values <0.10 indicating well-balanced variables. Variables not balanced between users and nonusers of statins were further included in the models. Multivariate analysis was restricted to individuals with a common PS range for nonusers and new users. Supplementary Table 1 shows the baseline characteristics of the individuals out of the common range. Ten PS and 10 hazard ratio (HR) values were calculated in each imputed data set. Pooled HR was then calculated according to Rubin rules in White et al. 24 with quadratic PS as covariate.
Proportionality of hazards assumption was tested by calculating the median of the χ2 tests of the models fitted for the 10 imputed data sets. Five-year number needed to treat (NNT) for one additional patient to survive was also calculated. A sensitivity analysis compared complete case with multiple imputation results (Supplementary File). We analyzed the data using a simulated “intention-to-treat” scenario where subsequent treatment of the exposed and unexposed patients is assumed to be the same as defined in the baseline. Competing risk analysis was performed to discard survival bias (Supplementary File). Statistical analysis used R-software version 3.4.3. 25
Results
Baseline Characteristics
During the enrollment period, 8018 patients met inclusion criteria and 736 (9.1%) were new users of statins. Losses to follow-up were 106 (1.3%), all of them due to transfer out of the SIDIAPQ database. The study flowchart is detailed in Figure 1. Median follow-up was 9.8 years (9.1, first quartile; 10.5, third quartile).
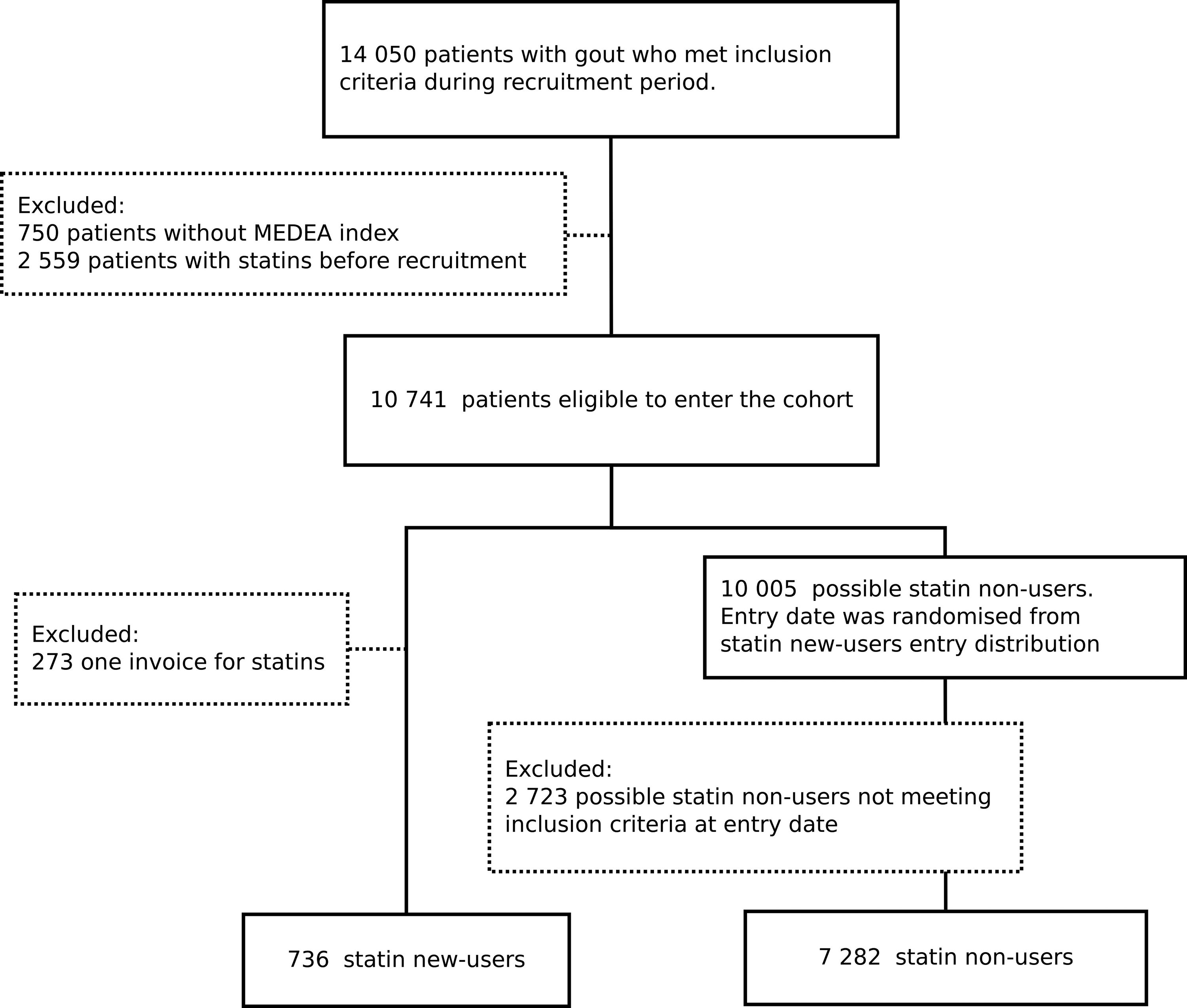
Flowchart of participant selection.
Missing data for incomplete variables and a comparison of the complete case and imputed data sets are shown in Supplementary Table 2. Overall, mean values for incomplete variables were lower after multiple imputations.
Women constituted 8% of the study population and the mean age was 59.0 (11.6) years. Diabetes was present in nearly 12% of participants, hypertension in 49%, smoking in 30%, and dyslipidemia in 30%; nonetheless, mean (SD) estimated 10-year CHD risk was low, at 4.1 (2.5). Median medication possession ratio (# days of statin supplied in 6 consecutive months/183 days) was 77% [first quartile, 46%; third quartile, 100%].
Baseline characteristics for new users and nonusers and standardized differences before and after adjusting for PS are presented in Table 1. More than 80% of new users were treated with a statin of moderate LDL reduction capacity.
Baseline Characteristics of the Total Population Before and After Adjusting for PS.

Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin II receptor (AT-II) blockers; CHD, coronary heart disease; COPD, chronic obstructive pulmonary disease; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PS, propensity score; SD, standard deviation; SDiff, standardized differences.
aHigh-risk alcohol intake: >28 UBE units for men and >17 UBE units for women. 1 UBE unit = 10 g alcohol.
Supplementary Table 3 shows the results in the complete data set. Overall, the population with complete data was older, with a worse cardiovascular risk profile.
Outcomes and Effectiveness of Statins
For 2006 to 2017, overall crude incidence per 1000 person-years at risk of CHD, IS, and all-cause mortality was 6.43 (95% confidence interval [CI]: 5.85-7.01), 5.51 (95% CI: 4.97-6.04), and 13.17 (95% CI: 12.35-13.98), respectively. Crude incidence, adjusted HRs, and 5-year NNTs for all primary outcomes by statin use in people within the PS common range (n = 7186) are shown in Table 2.
Adjusted Hazard Ratios (HRs) of Incident Cardiovascular Events, Mortality, Adverse Effects, and the 5-Year Number Needed to Treat (NNT) to Prevent One Event by Use of Statins.a
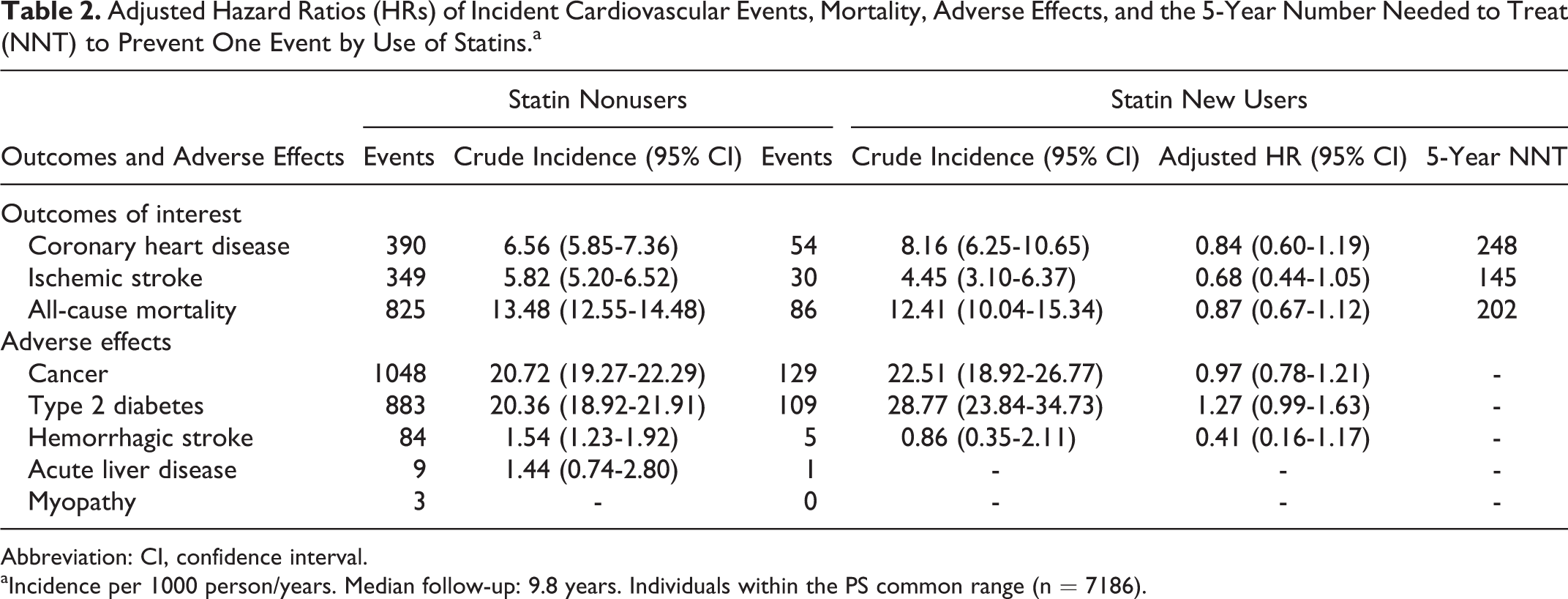
Abbreviation: CI, confidence interval.
aIncidence per 1000 person/years. Median follow-up: 9.8 years. Individuals within the PS common range (n = 7186).
Crude incidence of CHD was higher in statin users than in nonusers. Statin treatment decreased CHD risk by 16%, although the null hypothesis could not be rejected (adjusted HR: 0.84 [0.60-1.19]). The 5-year NNT for CHD was 248. There was also no significant risk reduction in IS and all-cause mortality. Further adjustment for variables not balanced after PS adjustment did not change the results.
Supplementary Table 4 shows the results in the complete data set. Coronary heart disease crude incidence was higher in statin users, whereas IS and all-cause mortality incidences were lower, compared to nonusers. Adjusted HRs were similar compared to the imputed data set.
Adverse Events
Unadjusted incidence of cancer and diabetes was higher in new users of statin than in nonusers. The increase in diabetes risk showed a weak evidence against the null hypothesis (1.27 [0.99-1.63]). There was no increase in cancer and the small sample size of the hemorrhagic stroke led to a very imprecise effect size. Supplementary Table 4 shows similar adverse event results in the complete data set.
The sensitivity analysis that considered death as competing risk showed similar results (Supplementary Table 5). Supplementary Table 6 shows the results of the proportionality of hazards assumption.
Discussion
Summary
To our knowledge, this is the first study to analyze the real-world clinical effectiveness of statins in reducing CHD, IS, and all-cause mortality among individuals diagnosed of gout and free of clinical CVD. Statin therapy decreased CHD, IS, and all-cause mortality risk by 16%, 32%, and 13%, respectively, although this effect was not significant. The 5-year NNTs ranged from 248 to 145. New users of statins had a clinically significant higher incidence of diabetes. No excess risk of cancer was recorded during follow-up.
Comparison With Existing Literature
In our study, the higher rate of CHD incidence observed in the gout population, compared to that estimated by the risk equation, supports the consideration that these patients are at higher risk of coronary events than the general population. 2 -4 Despite this higher incidence rate, it remained close to 6.5% in statin nonusers and 8.0% in statin users at 10 years, considered an intermediate level of coronary risk. 18
Most studies on efficacy of statins in primary prevention have focused on CHD risk in the general population, reporting similar results in high and intermediate coronary risk populations, defined as a 10-year coronary risk greater than 10% and 7.5%, respectively. 26,27 Our population may be comparable to that of the studies focusing on intermediate coronary risk but has clinical characteristics that make it difficult to compare the effects of statin therapy with general population results. The effect size of our results was in accordance with the intermediate-risk studies in terms of effectiveness; however, in our gout population, statin treatment could have limited net benefit due to the large 5-year NNT: 248 to prevent 1 event. 18
The magnitude of statin effectiveness in preventing IS was lower than that of studies in general population. 18,28 The limited number of IS events observed in our sample likely influenced effect size in our results. The mechanism by which statins may be effective in reducing the incidence of CVD diseases in gout populations is not well understood. On one hand, statins and some acute gout treatments might have a joint effect. Colchicine could be associated with a reduced risk of CVD events, 29 -31 whereas NSAIDs seemed to be related to an increased risk of AMI. 11 On another hand, urate-lowering therapies, allopurinol in particular, were found to be associated with an improvement in flow-mediated dilation 32 and a reduced risk of CVD events. 33 -35 Uncertainty prevails regarding the final joint effect of anti-inflammatory gout therapies and statins on CVD incidence, but this interaction could explain the possible differences in effectiveness between the general population and our study population with gout.
In addition, further research is needed on how dyslipidemia and hyperuricemia mechanisms affect stroke risk, particularly in populations with gout. 36 No studies have evaluated the association between statin use and IS in these populations.
In our study, gout and anti-inflammatory treatments were well balanced between new users and nonusers of statins, so their potential confounding effect was minimized although we cannot rule out some residual confounding.
Studies in general populations have inspired debate about statins’ effectiveness in reducing all-cause mortality, 27,37,38 despite agreement on their moderate success (∼10%, similar to our findings) in reducing relative risk. A recent study showed a greater decrease in overall mortality in a gout population, particularly in the subgroup analyses of individuals without previous CVD. However, the healthier characteristics of the sample, compared to general population, did not preclude a bias effect. 39
We observed no increased risk of cancer or hemorrhagic stroke among new users of statins, which is consistent with the literature in general population 40,41 ; a longer follow-up might be needed to detect an association between statin use and these adverse effects. There are no studies in the population with gout.
Finally, statin treatment increased the risk of diabetes about 27%, in line with previous results. 42 This is a clinically significant result because there is also evidence that gout is associated with an increased risk of diabetes. 43,44
Strengths and Limitations
The large sample of individuals, drawn from a high-quality, internally validated database of electronic medical records that provides high external validity and clinical data from patients often excluded from trials (eg, women, older patients, individuals with diabetes), is a main strength of our study. Several general limitations are inherent to observational studies using medical records.
First, we cannot discard some risk of misclassification but the presence of cardiovascular risk factors and outcomes was previously validated in SIDIAPQ, 19 and data on statin exposures were obtained from official pharmacy invoicing records of the National Health Service. About 60% of the study population was taking gout treatment, which reinforces the accuracy of the diagnoses, although some degree of misclassification cannot be completely ruled out.
Second, residual confounding is a possibility, especially by indication. To avoid frailty bias, we excluded individuals with cancer, dementia, paralysis, organ transplant, in dialysis, or institutionalized. We used a new users design to minimize potential effect of statins on confounding factors, then adjusted for PS. In addition, nonclinical factors that may influence prescription patterns and treatment adherence were not measured. These include doctor perception of a patient’s risk profile, prescriber experiences, unreported side effects, and patient perceptions of risk and willingness to take the drug. 45
Third, missing data can influence results. To avoid selection bias, we imputed the missing values for continuous variables instead of excluding those records. The characteristics of the study population met plausibility for the missing-at-random assumption for all imputed variables except for the MEDEA deprivation index, in which its missing mechanism was completely at random; thus, exclusion of participants with missing values for this variable did not imply selection bias.
Fourth, cause of death is not available in the SIDIAPQ database, which precluded analysis of statins’ effect on CVD mortality. Fifth, acute liver diseases and myopathy could not be compared between new users and nonusers due to the low number of events, particularly in statin users. This underreporting could lead to nondifferential misclassification and reduce statistical power, biasing results toward the null hypothesis. In addition, we could not draw conclusions about the effect of statins on hemorrhagic stroke due to the width of the 95% CI.
Sixth, the present analysis did not consider variables such as degree of inflammation, uricemia levels, or elapsed time between gout diagnosis and the incidence of cardiovascular events or death. Therefore, we were not able to evaluate uricemia levels and the degree of gout activity.
Finally, changes in the patterns of statins use, such as the increase in their prescription for primary prevention 46,47 and the use of high-potency statins, 48,49 were unlikely to have influenced the results of our study. Spanish blood lipid national and local guidelines advise no systematic prescription of statins and the overall management of the cardiovascular risk and, when recommended, low and moderate statins should be the first choice. 50,51
Conclusion
Statins were not associated with a decrease in CHD in a gout population at 10-year low-to-intermediate coronary risk. Diabetes risk was increased. No significant adverse effects were found. The high 5-year NNT to prevent 1 CHD event raises questions about recommending statin treatment in this population.
In addition, despite the higher risk of CVD in gout populations compared to general population, patients with gout from a population with a low-to-intermediate incidence of CHD should not be systematically considered at high risk and should be evaluated according to their cardiovascular risk assessment, lifestyle recommendations, and preferences, in line with the recent EULAR recommendations.
Supplemental Material
Supplemental Material, supplementary_v3_gout_(1)_revised_Elaine-updated - Effectiveness of Statins as Primary Prevention in People With Gout: A Population-Based Cohort Study
Supplemental Material, supplementary_v3_gout_(1)_revised_Elaine-updated for Effectiveness of Statins as Primary Prevention in People With Gout: A Population-Based Cohort Study by Maria Garcia-Gil, Marc Comas-Cufí, Rafel Ramos, Ruth Martí, Lia Alves-Cabratosa, Dídac Parramon, Daniel Prieto-Alhambra, Jose Miguel Baena-Díez, Betlem Salvador-González, Roberto Elosua, Irene R. Dégano, Jaume Marrugat and María Grau in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
The data sets generated and/or analyzed during the current study are not publicly available due to confidentiality policy of SIDIAPQ database but are available from the corresponding author on reasonable request. Ethics approval for observational research using SIDIAPQ data was obtained from our local ethics committee.
Acknowledgments
The authors thank the Registre del conjunt mínim de bases de dades (CMBD), Divisió de Registres de Demanda i d’Activitat, Àrea de Serveis i Qualitat, Servei Català de la Salut for Hospital Discharge data; CMBD personnel were not involved in preparing the manuscript and the paper does not necessarily reflect the agency’s opinion or point of view. The authors are solely responsible for data integrity and analysis. The authors also thank Eduardo Hermosilla for data management support. The authors also appreciate the revision of the English text by Elaine Lilly, PhD.
Author Contributions
R.R., M.G-.G., and M.G. contributed to the intellectual conception and design of the study, to the design of statistical analysis and interpretation of the results, to the manuscript writing, and to the manuscript review. M.C-.C. contributed to the design and carried out the statistical analysis. D.P-.A. contributed to the design of statistical analysis, interpretation of the results, and manuscript writing and review. R.M., L.A-.C., D.P., J.M.B-.D., B.S-.G., R.E., I.R.D., and J.M. contributed to the interpretation of the statistical analyses and to the manuscript review. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Ramos and Garcia-Gil reported collaboration in projects funded by AstraZeneca, AMGEN, and Novonordisk through IDIAP Jordi Gol. Dr Garcia-Gil has received speaker fees from Novartis. DPA’s research group has received research grants from Servier, Amgen, and UCB; speaker fees from Amgen; and consultancy fees from UCB Biopharma. These projects are unrelated to the present work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant funds were provided by Spain’s 2010 Ministry of Health call for clinical research proposals (EC10-084), 2012 Ministry of Science and Innovation call (via Carlos III Health Institute, Nets RD12/0005/0002, RD12/0042), CIBERCV (CB16/11/00229), and the 2013 Carlos III Health Institute call for Health Strategic Action Plan, 2013-2016, clinical research proposals, “Program on Research Related to Society’s Challenges” within Spain’s 2013-2016 Plan for Scientific and Technical Research and Innovation (PI13/01511, cofunded by the European Union’s Fund for Regional Development. M.G. received an FEDER contract (Carlos III Health institute: FIS CP12/03287). I.R.D. was funded by the RECERCAIXA Program (Obra Social “La Caixa”: (RE087465).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
