Abstract
Ca2+/calmodulin-dependent protein kinase IIδ (CaMKIIδ) plays a vital role in cardiovascular system. However, the potential protective role of inhibitor 1 of protein phosphatase 1 (I1PP1), which can regulate CaMKII, on myocardial ischemia–reperfusion (I/R) injury remains unknown. In the present study, expression of CaMKIIδ variants was detected by quantitative real-time polymerase chain reaction. I1PP1 was overexpressed by pericardial injection of recombinant adenovirus. Two weeks later, rats were subjected to left anterior descending ligation for 30 minutes followed by reperfusion. Myocardial infarct size was assessed by Evans blue/triphenyl tetrazolium chloride staining. Serum creatine kinase (CK) and lactate dehydrogenase (LDH) activity as well as myocardial pathological structure were detected. CaMKII activity was evaluated by phosphorylation of phospholamban (PLB) and oxidation of CaMKII. Expression of dynamin-related protein 1 (DRP1) and optic atrophy 1 (OPA1) in the mitochondria was measured by Western blot. We found that CaMKIIδA and CaMKIIδB expression decreased, while the expression of CaMKIIδC increased after myocardial I/R. Moreover, after 30-minute ischemia followed by 6 hours of reperfusion, I1PP1 overexpression reduced myocardial infarct size, decreased serum CK and LDH activity, ameliorated myocardial pathological structure, inhibited PLB phosphorylation at Thr17, suppressed CaMKII oxidation, elevated CaMKIIδA and CaMKIIδB variants but reduced CaMKIIδC variants, attenuated myocardial oxidative stress, improved myocardial mitochondrial ultrastructure, increased mitochondrial number and mitochondrial DNA copy number, and decreased DRP1 but increased OPA1 protein expression from the mitochondria in rats. Thus, I1PP1 regulated CaMKII, protected mitochondrial function, reduced oxidative stress, and attenuated myocardial I/R injury.
Keywords
Introduction
During myocardial ischemia–reperfusion (I/R) injury, ischemic myocardium exhibits more severe damage or undergoes more serious dysfunction and promotes arrhythmia or infarction after blood reperfusion. 1 The underlying pathophysiological mechanisms of I/R injury have not been well elucidated. It is generally recognized that oxidative stress, intracellular calcium overload, or redistribution during reflow may be involved in myocardial I/R injury. 2 However, oxygen-derived free radicals and calcium overload are not the only culprits responsible for I/R injury. Many other factors including cardiomyocytes death, inflammation, noncoding RNA, mitochondrial dysfunction and autophagy, microvascular perfusion defect, platelet hyperactivity, and neutrophil-mediated injury may also contribute to I/R injury. 3 –10
Previous study confirmed that Ca2+/calmodulin (CaM)-dependent protein kinase (CaMK), including CaMKI, II, and IV is a multifunctional threonine/serine protein kinase to mediate intracellular calcium ion channels. 11,12 CaMKII is encoded by 4 different genes: α, β, γ, and δ. CaMKIIα and CaMKIIβ are mainly expressed in nervous tissues, while they are generally absent or less expressed in the heart. 13 CaMKIIγ and CaMKIIδ are widely expressed in the body. CaMKIIδ is the main subtype in the myocardium, while CaMKIIγ is a minor subtype. 14 CaMKIIδ has an important regulation effect on the heart. Moreover, there is an alternative splicing of CaMKIIδ messenger RNA (mRNA) precursor. In the existence of mRNA cleavage enzymes, different isoforms of CaMKIIδ variants are produced in the heart. Previous research found that CaMKII activation was closely related to its subcellular localization, subtypes, and splicing variants. 15 Moreover, after alternative splicing, CaMKIIδA, CaMKIIδB, and CaMKIIδC are produced. 16
Protein phosphatase 1 (PP1) is a multifunctional phosphatase, which decreases the phosphorylation of splicing factors to regulate alternative splicing of CaMKIIδ. 16 However, PP1 inhibits phosphorylation of most proteins to limit the clinical application value. 17 Moreover, it was found that PP1 activity significantly increased in the myocardium of patients with myocardial infarction or angina, while the expression of inhibitor 1 of protein phosphatase 1 (I1PP1, an endogenous inhibitor of PP1) significantly decreased. 18 Our previous study suggested that PP1 regulated CaMKIIδ alternative splicing to alter CaMKIIδA, CaMKIIδB, and CaMKIIδC variant production by binding with splicing factors. 16 Overexpression of PP1 enhanced CaMKII activity, increased cardiac mass index and left ventricular index, and inhibited cardiac function after abdominal aortic constriction. 19 Our primary study found that expression of CaMKIIδ variants was disordered in myocardial I/R injury of rats. However, whether the disorders of CaMKIIδ variants were the key mechanisms to mediate I/R injury remains unclear. Moreover, the potential protective role of I1PP1 on CaMKIIδ alternative splicing to protect against myocardial I/R injury remains unknown.
Based on these findings, we aimed to clarify whether I1PP1 overexpression was capable to reverse CaMKIIδ alternative splicing and CaMKII activity to alleviate I/R injury. The protective effect and mechanism on myocardial I/R injury were explored after CaMKII regulation by I1PP1 overexpression. It is beneficial to propose novel strategies to alleviate myocardial I/R in clinic.
Materials and Methods
Myocardial I/R Surgery
Male Sprague-Dawley rats (180-220 g) were supplied by the Experimental Animal Center of Nantong University (Certificate number: SYXK-SU-2017-0046). Ischemia–reperfusion models were induced by left anterior descending (LAD) ligation. After fasting without water limitation for 12 hours before surgery, the rats were anesthetized with sodium pentobarbital (60 mg/kg, intraperitoneally [IP]) and kept under anesthesia condition during the whole surgery with supplemental sodium pentobarbital (25 mg/kg, IP) administration upon evidence of the paw pinch reflex. Anesthetized rats were mechanically ventilated with room air by a positive pressure ventilator. 20 The left costal margin of rats was shaved, and an oblique incision was made (2 cm) and then the muscle between third and fourth ribs on the left was cut. The pericardium was cut open, and the vessel was ligated between the left atrial appendage and the pulmonary conus about 2 to 3 mm below the left atrial appendage with 3 to 4 mm needle span and 1.5 mm depth. After 30 minutes, the line was released and the chest was closed immediately. After reperfusion for different times, the myocardium was collected for further study (6 rats per time point). All the procedure except LAD ligation was performed as a sham control.
All experimental procedures complied with Guide for the Care and Use of Laboratory Animals from the Institute for Laboratory Animal Research, National Research Council, Washington, DC, National Academy Press, 2011, and any updates. The protocol was approved by the Instructional Animal Care and Use Committee of Nantong University (approval no. NTU-20170910).
Animals Treatment
Recombinant adenovirus solutions (Hanbio Biotechnology Co, Ltd, ShangHai, China) were directly injected into the pericardial cavity as described previously. 16 After 2 weeks, myocardial I/R was performed and 4 experimental groups (12 rats per group) were set up: (1) SHAM group, rats were subjected to sham surgery with 50 μL normal saline injection; (2) I/R group, rats were subjected to I/R surgery with 50 μL normal saline injection; (3) Ad-I1PP1+I/R group, rats were subjected to I/R surgery with 50 μL recombinant adenovirus solution carrying I1PP1 gene (1 × 1011 PFU/mL) injection; and (4) Ad-GFP+I/R group, rats were subjected to I/R surgery with 50 μL recombinant adenovirus solution carrying GFP gene (1 × 1011 PFU/mL) injection.
Immunofluorescence
Frozen sections of myocardium were incubated with anti-I1PP1 (1:100; Santa Cruz Biotechnology, Santa Cruz, California) antibodies at 4°C overnight followed by Cy3-conjugated immunoglobulin G (500; Beyotime, Shanghai, China) at 37°C for 1 hour. The nuclei were counterstained with 4′,6-diamidino-2-phenylindole for several seconds. Tissue sections were photographed with a fluorescence microscope.
Assessment of Myocardial Infarct Size
After 30 minutes of ischemia and 6 hours of reperfusion in rats, myocardial infarct size (IS) was measured with Evans blue/triphenyl tetrazolium chloride (TTC) staining. Briefly, the heart was taken out and perfused with iced 0.9% sodium chloride into coronary vasculature on a Langendorf system to wash out the blood, and 1.5% Evans blue was perfused. Finally, the heart was sliced and incubated with 1% TTC in Tris-Buffered Saline (pH 7.8) for 15 minutes at 37°C. The viable nonischemic myocardium exhibited blue with Evans blue staining, ischemic but viable myocardium exhibited red with TTC staining, and IS of myocardium exhibited white. The myocardium stained with red was regarded as the area at risk (AR). The stained slices of the myocardium photographed to analyze the AR/left ventricle (LV) and the area of IS/AR.
Detection of Serum Creatine Kinase and Lactate Dehydrogenase Activity
Activity of creatine kinase (CK) and lactate dehydrogenase (LDH) in the serum was detected using commercial assay kits (Nanjing Jiancheng Bioengineering Institute, China) according to the manufacturer’s instructions.
Hematoxylin and Eosin Staining
Left ventricles below the ligation line were fixed with 4% paraformaldehyde for 24 hours followed by hematoxylin and eosin (HE) staining. Then, the slices were photographed and examined with a light microscope.
Measurement of Oxidative Stress in the Myocardium
Superoxide production in myocardium was measured with the fluorescent probe dihydroethidium (DHE) according to a previously described method. 21 The myocardium was taken and embedded in optimal cutting temperature compound and stored at −80°C. Sections were cut about 5 μm followed by DHE (Beyotime; 2 μmol/L) incubation in Krebs’ 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer (NaCl 99 mmol/L, KCl 4.7 mmol/L, MgSO4 1.2 mmol/L, KH2PO4 1.0 mmol/L, CaCl2 1.9 mmol/L, NaHCO3 25 mmol/L, glucose 11.1 mmol/L, Na HEPES 20 mmol/L; pH 7.4) at 37°C for 30 minutes without light. Then, myocardial sections were examined with fluorescence microscope (Nikon, Tokyo, Japan) with excitation and emission wavelengths at 480 nm and 610 nm, respectively.
Malondialdehyde (MDA) level in the myocardium was measured with Thiobarbituric Acid method and was represented as nmol/mg protein. 22 Total antioxidant capacity (T-AOC) in the myocardium was assessed with 2,2′-azino-bis(3-ethylbenzthi-azoline-6-sulfonic acid) method (Beyotime) and was represented as μmol/mg protein. 22 The detailed experimental procedure was performed according to the instructions of the kits.
Ultrastructural Examination of the Myocardium
The myocardium was cut into pieces of approximately 1 mm3, fixed with 4% glutaraldehyde, and then 1% citric acid. After fixation, the myocardium was dehydrated with acetone, embedded with Epon812, and stained with toluidine blue. Finally, the myocardium was cut into sections of approximately 70 nm and stained with uranyl acetate and lead citrate. The ultrastructure of the myocardium was examined by transmission electron microscopy (JEM-1230, JEOL Ltd., Japan). The mitochondria numbers were counted.
Real-Time Polymerase Chain Reaction
After 30 mg of LV was cut into pieces, the total RNA of myocardium was extracted by Trizol separation reagent. RNA samples were subjected to reverse transcription with the following procedure: 37°C 15 minutes, 85°C 5 seconds, and 4°C forever. The complementary DNAs were amplified with SYBR Green Fast qPCR mix (Takara, Otsu, Shiga, Japan) using ABI Stepone polymerase chain reaction (PCR) System (ABI, Carlsbad, California). The primer synthesis sequences (Sangon Biotech Co, Ltd, Shanghai, China) were listed as follows: CaMKIIδA-F 5′-CGAGAAATTTTTCAGCAGCC-3′, CaMKIIδA-R 5′-ACAGTAGTTTGGGGCTCCAG-3′; CaMKIIδB-F 5′-CGAGAAATTTTTCAGCAGCC-3′, CaMKIIδB-R 5′-GCTCTCAGTTGACTCCATCATC-3′; CaMKIIδC-F 5′-CGAGAAATTTTTCAGCAGCC-3, CaMKIIδC-R 5′-CTCAGTTGACTCCTTTACCCC-3′; 18S-F 5′-AGTCCCTGCCCTTTGTACACA-3′, 18S-R 5′-CGATCCGAGGGCCTCACTA-3′. Experimental cycle threshold values were normalized to 18 S, and relative mRNA expression was calculated versus a control sample. 23
Western Blot
About 30 mg of LV tissue was cut into pieces, and 300 μL of lysate (NP40/phenylmethanesulfonyl fluoride, = 100:1) was added. After complete grinding on the ice, the mixture was further lysed on ice for 40 minutes. Then, the lysates were centrifuged at 12 000g for 15 minutes, loading buffer was added into the supernatant, and boiled for 5 minutes. Mitochondrial protein from the myocardium were extracted with Tissue Mitochondria Isolation Kit according to the instructions of the kits (Beyotime, Shanghai, China) followed by total protein extraction. Total proteins or mitochondrial proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to a polyvinylidene fluoride (PVDF) membrane (Millipore, Billerica, Massachusetts). After blocking with 5% nonfat milk for 2 hours, the PVDF membranes were incubated overnight with anti-I1PP1(1:1000), anti-PP1(1:1000), anti-phospholamban (PLB, 1:1000), anti-phosphorylation-PLB Thr17 (1:1000), anti-voltage-dependent anion-selective channel proteins 1 (VDAC1, 1:1000; Santa Cruz Biotechnology), anti-phosphorylation-PLB Ser16 (1:1000), anti-ox-CaMKII (1:1000; Millipore, Kenilworth, New Jersey), anti-CaMKII (1:1000; Abcam, Cambridge, United Kingdom), anti-dynamin-related protein 1 (DRP1; 1:1000), anti-optic atrophy 1 (OPA1; 1:1000; Cell Signaling Technology, Danvers, Massachusetts), and anti-GAPDH (1:5000; Sigma-Aldrich, St Louis, Missouri) antibodies at 4°C followed by horseradish peroxidase-conjugated secondary antibody at room temperature for 2 hours. Enhanced chemiluminescence (Thermo Fisher Scientific Inc, Rockford, Illinois) was added to visualize the protein bands.
Mitochondrial DNA Copy Number
The mitochondrial DNA (mtDNA) copy number was determined using real-time PCR as previously described. 24 The primers were 18S (5’-AGTCCCTGCCCTTTGTACACA-3’ and 5’-CGATCCGAGGGCCTCACTA-3′) or mtDNA (5’-TGGTTCATCGTCATACGTT-3’ and 5 ‘-TGACGGCTATGTTGAGGAAG-3’). The relative mtDNA copy number was represented as mtDNA content normalized by 18S.
Statistical Analysis
All the data were expressed as mean ± standard error of mean (SEM) and analyzed using 1-way analysis of variance followed by Student-Newman-Keuls test (Stata 13.0). P < .05 was considered as statistically significant.
Results
Expression of CaMKIIδ Variant Disorders in Myocardial I/R Injury of Rats
Since there is no specific antibody for CaMKIIδ variants, the mRNA levels of CaMKIIδA, CaMKIIδB, and CaMKIIδC at different reperfusion points after myocardial ischemia for 30 minutes were detected by quantitative real-time PCR. The expression of CaMKIIδA and CaMKIIδB decreased from 6 hours after reperfusion, while the expression of CaMKIIδC increased, suggesting that there was a significant disorder on expression of CaMKIIδ variants during myocardial I/R injury (Figure 1).

Expression of -CaMKIIδ variants disorders in myocardial I/R injury of rat. After LAD ligation for 30 minutes, the artery ligation was released and the thoracic cavity was closed. The mRNA levels of CaMKIIδA, CaMKIIδB, and CaMKIIδC of the ischemic myocardium (below the ligature point) after reperfusion were detected by quantitative real-time PCR. 18S was serviced as a housekeeping mRNA. Means ± standard error of the mean (SEM). n = 6. **P < .01 versus 0 hours after ischemia (at the beginning of the ischemia). CaMKIIδ indicates calmodulin-dependent protein kinase IIδ; I/R, ischemia–reperfusion; LAD, left anterior descending; mRNA, messenger RNA; PCR, polymerase chain reaction.
I1PP1 Is Overexpressed by Pericardial Injection of Recombinant Adenovirus Carrying the I1PP1 Genes in Rats
However, whether the disorders of CaMKIIδ variants were the key mechanisms to mediate I/R injury remains unclear. In order to verify this hypothesis, the recombinant adenovirus carrying the I1PP1 gene was further directly injected into the pericardial cavity of the rats to regulate CaMKIIδ alternative splicing. Two weeks after injection, the staining intensity of I1PP1 with immunofluorescence and the intensity of I1PP1 band with Western blot were significantly enhanced in the myocardium compared to GFP recombinant adenovirus (Figure 2A and B). Moreover, I1PP1 overexpression successfully inhibited PP1 expression in the myocardium (Figure 2C). Abovementioned data suggested that both I1PP1 expression and I1PP1 activity were elevated after cardiac I1PP1 overexpression.

I1PP1 is overexpressed by pericardial injection of recombinant adenovirus carrying the I1PP1 genes in rats. A, 50-μL recombinant adenovirus solution carrying I1PP1 gene (1 × 1011 PFU/mL) or GFP gene (1 × 1011 PFU/mL) was injected directly into the pericardial cavity. Two weeks later, I1PP1 in the myocardium was immunofluorescence stained using Cy3 (Red) conjugated IgG. The nuclei were stained using DAPI (Blue). Scale bars: 100 μm. B-C, Expression of I1PP1 and PP1 protein was quantified in the myocardium by Western blot. GAPDH was used as a loading control. Means ± SEM. n = 6. DAPI indicates 4′,6-diamidino-2-phenylindole; GAPDH, Glyceraldehyde 3-phosphate dehydrogenase; IgG, immunoglobulin G; I1PP1, inhibitor 1 of protein phosphatase 1; PP1, protein phosphatase 1; SEM, standard error of the mean.
I1PP1 Overexpression Reduces Myocardial IS After I/R in Rats
Two weeks after I1PP1 recombinant adenovirus injection, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours of reperfusion. Evans blue/TTC double staining showed that there was no statistical difference on AR/LV among all the I/R groups, indicating that the ligation sites were almost the same (Figure 3A and B). And 30 minutes ischemia and 6 hours of reperfusion promoted myocardium infarction with enlarged white area. Moreover, I1PP1 overexpression, but not GFP, significantly reduced myocardial IS after I/R (Figure 3C). It suggested that I1PP1 played a cardioprotective role during myocardial I/R in rats.

I1PP1 overexpression reduces myocardial IS after I/R in rats. A, Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours of reperfusion. Representative staining images of heart by Evans blue/TTC after 6 hours of reperfusion were shown. The viable nonischemic myocardium exhibited blue with Evans blue staining, ischemic but viable myocardium exhibited red with TTC staining, and IS of myocardium exhibited white. B, Ratio of AR/LV was calculated after 6 hours of reperfusion. C, Ratio of area of IS/AR was calculated after 6 hours reperfusion. Means ± SEM. n = 6. ## P < .01 verses I/R. AR indicates area at risk; I1PP1, inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; IS, infarct size; LAD, left anterior descending; LV, left ventricle; SEM, standard error of the mean; TTC, triphenyl tetrazolium chloride.
I1PP1 Overexpression Decreases Serum CK and LDH Activity After I/R in Rats
A large amount of CK and LDH are released into the blood during myocardial infarction. 25 Our results showed that serum CK and LDH activity significantly increased after I/R, which was attenuated by I1PP1 overexpression (Figure 4A and B). It suggested that I1PP1 overexpression alleviated myocardial I/R injury.

I1PP1 overexpression decreases serum CK and LDH activity after I/R in rats. Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours of reperfusion. A, Serum CK activity was measured. B, Serum LDH activity was measured. Means ± SEM. n = 6. **P < .01 verses SHAM, ## P < .01 verses I/R. CK indicates creatine kinase; I1PP1, inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; LAD, left anterior descending; LDH, lactate dehydrogenase; SEM, standard error of the mean.
I1PP1 Overexpression Ameliorates Myocardial Pathological Structure After I/R in Rats
Hematoxylin and eosin staining of the LV below the ligation line with a microscope revealed that the fragmented cardiomyocytes were diffusely distributed with a large number of vacuoles, distortions, and deformations in the I/R group, indicating that I/R injury impaired the pathological structure of cardiomyocytes. Compared to the I/R group, the arrangement of cardiomyocytes was relatively regular, and cell vacuolization, distortion, and deformation were improved by I1PP1 overexpression (Figure 5). It suggested that I1PP1 overexpression ameliorated myocardial pathological structure after I/R.
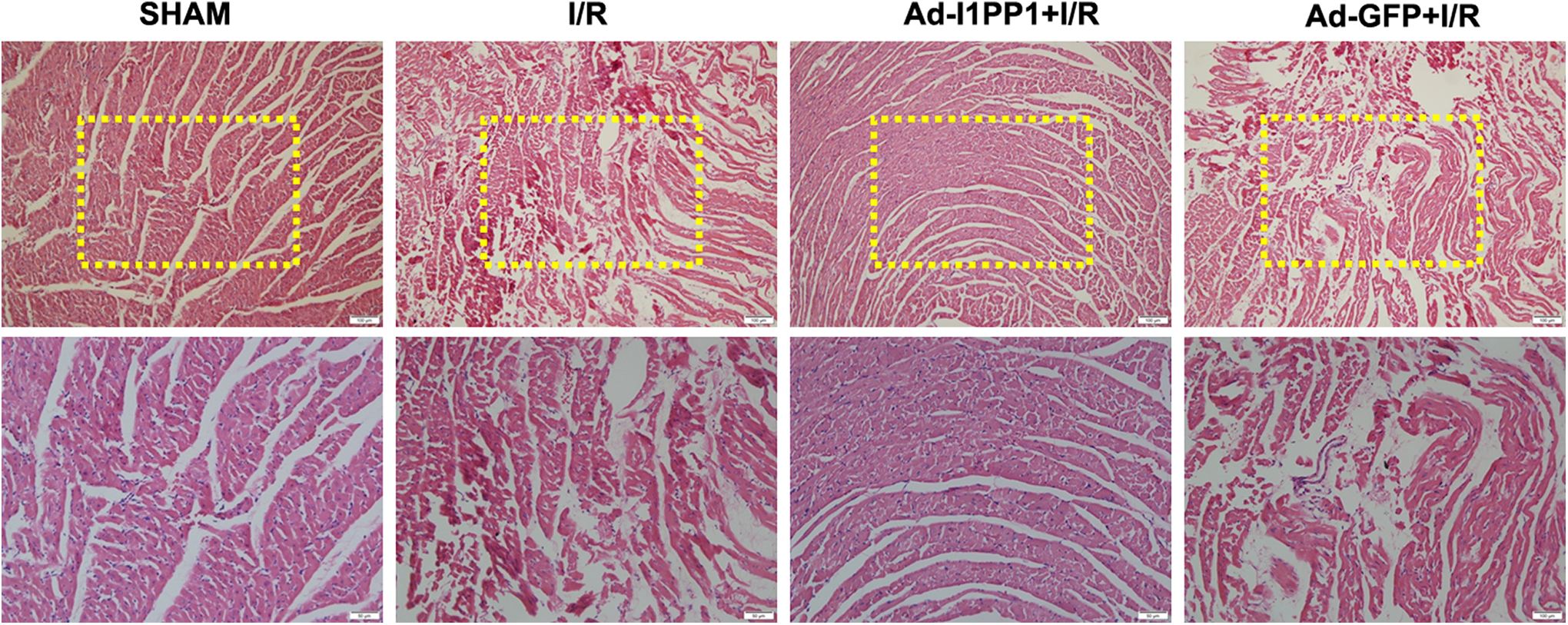
I1PP1 overexpression ameliorates myocardial pathological structure after I/R in rats. Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours reperfusion. Left ventricle below the ligation line was stained with hematoxylin and eosin and was photographed. Scale bars: 100 μm (upper) and 50 μm (lower), respectively. I1PP1 indicates inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; LAD, left anterior descending.
I1PP1 Overexpression Regulates Myocardial CaMKII Activity, CaMKII Oxidation, and CaMKIIδ Variants Expression After I/R in Rats
Previous studies confirmed that PLB phosphorylation at Thr 17 was a robust marker for CaMKII activity. 16 In our study, PLB phosphorylation at Thr17 but not Ser16 significantly increased after I/R, which was inhibited by I1PP1 overexpression. It suggested that I1PP1 suppressed CaMKII activity after I/R (Figure 6A). The present study also found that oxidation of CaMKII was enhanced after I/R injury, while I1PP1 diminished CaMKII oxidation in ischemia myocardium after I/R injury (Figure 6B). The expression of 3 CaMKIIδ variants was detected in the further experiments. It was noted that CaMKIIδA and CaMKIIδB expression significantly decreased, while CaMKIIδC expression significantly increased after I/R. Moreover, I1PP1 overexpression elevated CaMKIIδA and CaMKIIδB variants but reduced CaMKIIδC variants (Figure 6C and E). It suggested that I1PP1 overexpression corrected the enhancement of CaMKII activity and CaMKII oxidation as well as the disorder of CaMKIIδ alternative splicing after I/R.

I1PP1 overexpression regulates myocardial CaMKII activity, CaMKII oxidation, and CaMKIIδ variants expression after I/R in rats. Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours reperfusion. A, Expression of p-PLB Ser16, p-PLB Thr 17, and PLB of the ischemic myocardium were quantified by Western blot. GAPDH was used as a loading control. B, Expression of ox-CaMKII and total CaMKII in the ischemic myocardium were quantified by Western blot. GAPDH was used as a loading control. C-E, The mRNA levels of CaMKIIδA, CaMKIIδB, and CaMKIIδC of the ischemic myocardium were detected by quantitative real-time PCR. 18S was serviced as a housekeeping mRNA. Means ± SEM. n = 6. **P < .01 versus SHAM; # P < .05, ## P < .01 versus I/R. CaMKII indicates Ca2+/calmodulin-dependent protein kinase II; I1PP1, inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; LAD, left anterior descending; mRNA, messenger RNA; ox-CaMKII, CaMKII oxidation; PCR, polymerase chain reaction; PLB, phospholamban; SEM, standard error of the mean.
I1PP1 Overexpression Attenuates Myocardial Oxidative Stress After I/R in Rats
Increased oxidative stress is one of the major pathophysiological mechanisms for myocardial I/R injury. 26,27 In our study, intensity of DHE staining increased after I/R, which was weakened by I1PP1 overexpression. It indicated that I1PP1 overexpression inhibited superoxide anion production (Figure 7A). We also found that myocardial MDA levels increased, while T-AOC decreased significantly after I/R compared to SHAM group. Compared to I/R group, I1PP1 overexpression decreased MDA but increased T-AOC (Figure 7B and C). These results suggested that cardiac overexpression of I1PP1 attenuated myocardial oxidative stress after I/R in rats.

I1PP1 overexpression attenuates myocardial oxidative stress after I/R in rats. Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours of reperfusion. A, Superoxide production in the left ventricle below the ligation line was detected using a fluorescence microscope with DHE fluorescent probe. B-C, Level of MDA and T-AOC of the ischemic myocardium were measured. Means ± SEM. N = 6. **P < .01 versus SHAM; # P < .05, ## P < .01 versus I/R. DHE indicates dihydroethidium; I1PP1, inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; LAD, left anterior descending; MDA, malondialdehyde; T-AOC, total antioxidant capacity; SEM, standard error of the mean.
I1PP1 Overexpression Restores Myocardial Mitochondrial Ultrastructure and Numbers After I/R in Rats
Transmission electron microscopy revealed that myocardial mitochondria were arranged disorderly with swelling and deformation after I/R. Mitochondrial number and mitochondrial cristae decreased after 6 hours of reperfusion. All abovementioned disorders were restored by I1PP1 overexpression (Figure 8A). Mitochondrial number was evaluated with transmission electron microscopy observation and mtDNA copy number measurement. We found that I1PP1 overexpression increased mitochondrial number and mtDNA copy number in ischemic myocardium after I/R (Figure 8B and C). These data suggested that overexpression of I1PP1 improved myocardial mitochondrial ultrastructure and increased mitochondrial numbers after I/R.

I1PP1 overexpression improves myocardial mitochondrial ultrastructure after I/R in rats. Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours of reperfusion. A, Mitochondrial ultrastructure of the ischemic myocardium was examined with transmission electron microscope. Scale bars: 1 μm (upper) and 500 nm (lower), respectively. B, Mitochondrial number was evaluated with transmission electron microscopy observation. C, mtDNA copy number was measured by real-time PCR. Means ± SEM. n = 6. **P < .01 versus SHAM; ## P < .01 versus I/R. I1PP1 indicates inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; LAD, left anterior descending; mtDNA, Mitochondrial DNA; PCR, polymerase chain reaction; SEM, standard error of the mean.
I1PP1 Overexpression Regulates Myocardial Mitochondrial DRP1 and OPA1 Expression After I/R in Rats
Dynamin-related protein 1 is a mitochondrial fission-associated protein, while OPA1 is a mitochondrial fusion-associated protein. 28,29 The balance of DRP1 and OPA1 plays an important role in maintaining mitochondrial structure and function. 21,30 We found that myocardial I/R injury significantly increased DRP1 but decreased OPA1 protein expression in the mitochondria, which was restored by I1PP1 overexpression (Figure 9). It suggested that I1PP1 overexpression redressed the balance of myocardial mitochondrial DRP1 and OPA1 expression after myocardial I/R.

I1PP1 overexpression regulates myocardial mitochondrial DRP1 and OPA1 expression after I/R in rats. Two weeks after injection of I1PP1 recombinant adenovirus, the rat was subjected to LAD ligation for 30 minutes followed by 6 hours of reperfusion. Expression of DRP1 and OPA1 protein from the mitochondria of the ischemic myocardium was quantified by Western blot. VDAC1 was used as a loading control. Means ± SEM. n = 6. **P < .01 versus SHAM; # P < .05, ## P < .01 versus I/R. DRP1 indicates dynamin-related protein 1; I1PP1 inhibitor 1 of protein phosphatase 1; I/R, ischemia–reperfusion; LAD, left anterior descending; OPA1, optic atrophy 1; SEM, standard error of the mean; VDAC1, voltage-dependent anion-selective channel proteins 1.
Discussion
At present, thrombolytic therapy of acute myocardial infarction, coronary artery bypass grafting, and heart transplantation inevitably lead to myocardial I/R injury. 31 Therefore, elucidating the pathophysiological mechanism of myocardial I/R injury is beneficial to propose more effective treatment for the cardiovascular diseases. Previous study confirmed that reperfusion during the treatment of ischemic heart diseases might cause myocardial injury and induce apoptosis or necrosis and result in myocardial dysfunction. Sustained activation of CaMKII during myocardial IR causes heart failure, arrhythmia, and even sudden cardiac death. 32 –36 In addition to activation by combination with Ca2+/CaM, CaMKII activity was able to be negatively regulated by several protein phosphatases including PP1, PP2A, and PP2C. 37 CaMKII was also activated by phosphorylation at Thr287 and oxidation at Met281/282, resulting in mitochondrial permeability transition pore opening. 38 Sustained and excessive CaMKII activation usually induces heart damage. Altogether, some novel methods to modulate CaMKII activity might provide new ideas to attenuate cardiomyocytes injury. It has been found that CaMKII inhibitor KN-93 alleviated myocardial I/R injury, reduced myocardial IS, and improved cardiac function. 7 It is suggested that CaMKII pathway inhibition was beneficial to attenuate reperfusion injury of ischemic myocardium.
It is noted that after an alternative splicing of CaMKIIδ mRNA precursor, CaMKIIδA, CaMKIIδB, and CaMKIIδC are produced by alternative splicing of exons 14, 15, and 16. There are exons 15 and 16 in CaMKIIδA, which is mainly distributed in the T tube of cardiomyocytes. Overactivation of CaMKIIδA induced excessive excitation–contraction coupling and even heart failure. CaMKIIδB only encodes exon 14 containing a nuclear localization signal, which is mainly localized in the nucleus to increase antiapoptotic proteins expression and to enhance cell viability. 14 There are no exons of 14, 15, and 16 in CaMKIIδC, which is mainly expressed in the cytoplasm to regulate adaptive remodeling after myocardial infarction. Moreover, our results showed that the gene expression of CaMKIIδA and CaMKIIδB significantly decreased, while the gene expression of CaMKIIδC increased after myocardial I/R injury. CaMKIIδB and CaMKIIδC might induce pathological changes such as myocardial hypertrophy and heart failure. 19 The alternative splicing of CaMKIIδ on exon 14, 15, and 16 is mainly regulated by various splicing factors of the Ser/Arg-rich protein family. 39 Alternative splicing factors and SC35 proteins play their functions under tight regulation of phosphorylation by several kinases. 16 Our present study suggested that there was a significant disorder of CaMKIIδ variants during I/R injury. However, whether the mechanism of I/R injury was ascribed to alternative splicing of CaMKIIδ needs further study. Next, the degree of myocardial injury, as well as CaMKII activity and CaMKIIδ variants expression, was evaluated with myocardial I/R model in vivo after I1PP1 overexpression. I1PP1, an inhibitor of PP1, impairs dephosphorylation of splicing factors by PP1 and then to modulate alternative splicing of CaMKIIδ. 40 In our present study, I1PP1 was overexpressed to regulate the disorder of CaMKIIδ variants during myocardial I/R injury. I1PP1 overexpression also significantly protected mitochondrial function, reduced oxidative stress, and attenuated myocardial I/R injury.
Creatine kinase and LDH are involved in the energy metabolism. The activity of CK and release of LDH in serum is usually elevated after myocardium was damaged. 41 Our research found that CK activity and LDH release in the serum significantly increased after I/R. It is noted that abovementioned increased activity of CK and release of LDH was markedly attenuated by I1PP1 overexpression after myocardial I/R in heart, indicating that myocardial overexpression of I1PP1 alleviated myocardial I/R injury.
Phospholamban phosphorylation directly reflects the activity of CaMKII. 16 Once myocardial CaMKII activity elevated, PLB phosphorylation significantly increased, and the body was prone to manifest as calcium disorder, myocardial infarction, or even heart failure. 42,43 In addition to activation by combination with Ca2+/CaM, CaMKII was also activated by oxidation. 44 In this study, we found that myocardial overexpression of I1PP1 reduced the phosphorylation of PLB at Thr17 and suppressed oxidation of CaMKII, indicating that overexpression of I1PP1 inhibited CaMKII activity after myocardial I/R, which is beneficial to maintain calcium balance and myocardial function.
Reactive oxygen species (ROS) oxidizes and destroys intracellular lipids, proteins, and DNA to induce oxidative stress, which is closely related to atherosclerosis, tumors, diabetes, rheumatoid arthritis, liver injury, and other diseases. 45,46 Malondialdehyde is the final product of peroxidation at the presence of free radicals, which is usually measured to assess the level of lipid peroxidation. The macromolecules, small molecules, and enzymes for antioxidation constitute the T-AOC of the body. 47 Our results showed that both superoxide anion production and MDA content markedly increased after myocardial I/R, while the T-AOC significantly reduced, indicating that the mitochondrial oxidative stress was significantly aggravated. Moreover, cardiac overexpression of I1PP1 decreased the generation of superoxide anion, reduced MDA content but increased T-AOC after myocardial I/R, suggesting that modulating of CaMKIIδ alternative splicing attenuated myocardial I/R-induced oxidative stress.
Mitochondria, as the main places for energy production, were vital for the energy metabolism. 48 Myocardial I/R induces apoptosis and necrosis by disrupting mitochondrial structure, promoting mitochondrial oxidative phosphorylation, and increasing oxidative stress injury. 49,50 Our study found that I1PP1 overexpression in the myocardium after I/R showed relatively regular, less swollen mitochondria with more evident cristae, suggesting that I1PP1-mediated CaMKII regulation improved the mitochondrial ultrastructure after I/R. Also, we know fission and fusion of mitochondria are critical to maintain mitochondrial function. Dynamin-related protein 1, as a core component for mitochondrial fission, promotes mitochondrial structural damage and dysfunction. 29 OPA1, as a fusion-associated protein, is beneficial to improve the efficiency of respiratory chain and maintain mitochondrial integrity and mitochondrial fusion. 28 DRP1 and OPA1, as 2 vital mitochondrial fission and fusion proteins, have been proposed as novel therapeutic targets for treating cardiovascular diseases. 21,30 Our results showed that cardiac overexpression of I1PP1 reduced DRP1 but increased OPA1 expression in myocardial mitochondria, which is beneficial to maintain the normal structure and function of mitochondria during myocardial I/R injury.
Previous study found that chronic β-adrenergic receptor stimulation persistently increased mitochondrial permeability transition pore openings through CaMKII pathway in patients with chronic heart failure. 51 Another study found that angiotensin II activated angiotensin II type I receptor (AT1R) to induce an increase in intracellular calcium then to interact with CaM. The Ca2+–CaM complex directly or indirectly activated sodium hydrogen exchanger 1 and phosphorylated CaMKII. 52 Thus, I1PP1 overexpression regulated CaMKII and attenuated myocardial I/R injury, which might be also related to β-adrenergic and angiotensin II receptor activation. These possible regulatory effect of I1PP1 overexpression on axis of β-adrenergic or angiotensin II needed to be further investigated.
One group found that CaMKIIδ mediated nuclear factor-κB activation in cardiomyocytes following I/R, which suggested that CaMKIIδ was possibly served to trigger as well as to sustain subsequent changes in inflammatory gene expression that contribute to myocardial I/R damage. 53 Furthermore, CaMKII participated in AT1R-mediated NADPH oxidase activation, leading to ROS generation, and was widely implicated in vascular inflammation and fibrosis. 54,55 CaMKII was also involved in NADPH oxidase 2-mediated angiotensin II-dependent cellular arrhythmias. 56 Associated with CaMKII regulation by I1PP1 during myocardial I/R injury, I1PP1 overexpression might also play a vital role in the abovementioned systems.
However, there are several limitations in our present study. Firstly, CaMKIIδ protein levels are high in sarcoplasmic reticulum during myocardial I/R. 57 CaMKIIδ measurement in sarcoplasmic reticulum will be greatly benefited to classify the mechanism of I1PP1 on myocardial I/R injury. Secondly, the myocardial calcium–handling profile will be beneficial to elucidate the alterations in Ca2+ homeostasis, which is critical to provide the impact of I1PP1 overexpression on myocardial H/R injury. Detection of calcium in the cardiomyocytes in vitro might be an ideal strategy in further studies. Thirdly, only about 30% cardiomyocytes were comprised in the rat heart. It is very difficult to distinguish the contribution of different heart cells for the protective effect against myocardial H/R injury.
In conclusion, our study found that CaMKIIδ alternative splicing and CaMKII activity was disordered during myocardial I/R injury. I1PP1 overexpression regulated CaMKII, protected mitochondrial function, reduced oxidative stress, and attenuated myocardial I/R injury. These results have shed new light on the molecular mechanism(s) responsible for the cardioprotective effect of CaMKII regulation in myocardial I/R injury, which is beneficial to propose novel strategies for myocardial I/R in clinic.
Footnotes
Author Contribution
Jin Yu and Yun Chen equally contributed to this work. Jin Yu, Yun Chen, Mengting Xu, Linlin Sun, Huiqin Luo, and Xiaofeng Bao contributed to acquisition, analysis, or interpretation. Guoliang Meng and Wei Zhang contributed to conception or design. Jin Yu and Yun Chen drafted the manuscript. Xiaofeng Bao, Guoliang Meng, and Wei Zhang critically revised the manuscript. Guoliang Meng and Wei Zhang gave final approval. All authors agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81670243, 81873470, 81770279), Natural Science Foundation of Jiangsu Province (BK20151276), a major project of Natural Science Research in Jiangsu Higher Education Institutions (18KJA310005), the project of “Jiangsu Six Peaks of Talent” (2018-WSN-062), a project funded by China Postdoctoral Science Foundation (2017M610342), a Jiangsu Planned Project for Postdoctoral Research Funds (1701050A), a Research and Innovation Project of Graduate Students in Jiangsu Province (KYCX18_2401), and the Nantong University Cooperative Innovation Program of Small Molecular Compound R&D (NTU2016 -1).
