Abstract
Background:
Several trials have reported that dipeptidyl peptidase-4 (DPP-4) inhibitors are used to improve endothelial function in addition to treating type 2 diabetes (T2DM). The current study investigated the effects of vildagliptin, DPP-4 inhibitor, compared to metformin on endothelial function and blood pressure through vascular endothelial growth factor (VEGF) modulation in patients with T2DM and hypertension.
Methods:
This study was designed as a randomized controlled parallel study. A total of 120 volunteers were recruited and allocated into 4 groups: healthy volunteers, patients recently diagnosed with hypertension and diabetes, patients treated with captopril for hypertension in addition to metformin, and patients treated with captopril in addition to vildagliptin. The percentage change in body weight was calculated in addition to serum VEGF levels, blood pressure, glycated hemoglobin (HbA1c), total lipid profile, and insulin resistance.
Results:
At the end of the therapeutic period, the results showed that vildagliptin significantly decreased blood pressure and increased serum VEGF levels, while metformin was more effective at lowering body weight. In comparison with metformin, vildagliptin showed a promising action through its antihypertensive effect via elevating VEGF levels and improving physiological angiogenesis and vasculature.
What is new and conclusion:
Vildagliptin showed a promising action through its blood pressure-regulating effect via modulating VEGF levels and improving physiological angiogenesis and vasculature, in addition to improving the lipid profile of patients, while metformin was better in reducing body weight.
Introduction
Essential hypertension and diabetes mellitus (DM) are both very common conditions, and therefore, their coexistence is extremely prevalent. In addition to lifestyle modification, metformin is considered a first-line treatment modality for type 2 DM (T2DM) when used as a mono-therapy or combined with dipeptidyl peptidase 4 (DPP-4) or glucagon-like peptide 1 in many updated guidelines and the addition of insulin therapy is recommended for patients with inadequate diabetic control. 1,2
In addition to their therapeutic effects in metabolic disorders, both metformin and vildagliptin have been reported to have beneficial effects on cardiovascular (CVS) risk in both diabetic and nondiabetic rats. 3,4 Some recent studies have highlighted that both metformin and vildagliptin have a positive effect in reducing insulin resistance and attenuating the CVS complications of T2DM. 5,6
Vascular endothelial growth factor (VEGF) is an endothelium-specific peptide that stimulates angiogenesis. 7 The introduction of treatment with a VEGF signaling pathway inhibitor has highlighted the role of baseline VEGF activity in blood pressure regulation; consequently, active monitoring of blood pressure and cardiac toxicity throughout treatment is recommended in patients receiving VEGF signaling pathway inhibitors. 8
Many different treatment guidelines recommend a target glycated hemoglobin (HbA1c) of less than 7% and therapy tailored for various factors, such as disease duration, patient age, vascular complications, life expectancy, and other comorbidities. 9
Methods
This was a prospective randomized parallel study with an equal number of patients in each group and balanced characteristics. The patient populations were matched for demographics of age, weight, and height. Informed consent was obtained from all subjects after the nature, purpose, potential risks of the study, and study duration (6 months) were explained. The study was approved by the research ethics committee of Cairo University (IDE00217) following the tenets of the Declaration of Helsinki 10 and was performed from June 2016 until March 2017.
Study Design
The study was designed as a randomized controlled parallel study. A total of 120 volunteers (90 patients and 30 healthy volunteers) were recruited per the following inclusion and exclusion criteria:
Inclusion criteria: Male and female patients suffering from stage 1 hypertension, according to American JNC-8, and DM with an HbA1c ≥ 7, age range between 40 and 60 years, and treatment with diet alone or any combination of oral antidiabetic agents and/or insulin before admission.
Exclusion criteria: Patients who had clinical evidence of ischemic heart disease or chronic obstructive pulmonary disease, the presence of diabetic ketoacidosis, were admitted to the intensive care unit (ICU), expected to undergo surgery during the study period, or had clinically relevant hepatic disease which is a clinically significant liver disease including liver cirrhosis and fibrosis (Child Pough score B and C), 11 in addition to hepatitis B and C, impaired renal function (serum creatinine ≥3.0 mg/dL), systemic infections, or pregnancy. Additionally, patients on medications known to interfere with the blood glucose level (either increasing or decreasing) were excluded from the study.
Randomization and Study Intervention
Randomization was done using a computer-validated software (Random allocator). Thirty individuals with T2DM and hypertension were evaluated in both the vildagliptin and metformin groups before and after 24 weeks of treatment.
Study Outcomes
The primary outcome of this study was to modulate VEGF levels in patients suffering from both DM and hypertension using an appropriate glucose-lowering therapy helping them to regulate elevated blood pressure. The secondary study outcome was to investigate the drug effects on HbA1c levels, lipid profile, and body weight.
Patient Grouping and Schedule of Pharmacological Treatments
One hundred fifty participants were recruited in the study, 30 of them were excluded as they didn’t meet the inclusion and exclusion criteria of the study then the remaining 120 participants were rolled on. After informed consent was obtained from the patients, they were randomized into 3 groups of patients in addition to a healthy volunteer group. Each group consisted of 30 subjects who completed the study without dropping out. Group I: Thirty healthy volunteers Group II: Thirty patients recently diagnosed with hypertension and DM at the time of the study. This group of patients was clinically assessed and received the proper medication accordingly. Group III: Thirty diabetic hypertensive patients treated with metformin (1000 mg twice daily) in addition to captopril (25 mg/d). Group IV: Thirty diabetic hypertensive patients treated with vildagliptin (50 mg twice daily) in addition to captopril (25 mg/d).
Treatment was continued until the end of the study (Fig. S1).
Patients previously treated with antihyperglycemic drugs other than drugs used in their study group, needed a washout period of 15 days, to allow patients to discontinue from their previous antihyperglycemic agents and switched to insulin therapy. All patients were counseled about diet control. Patients of group II were diagnosed for the first time with their hypertension and DM at the beginning of the study, blood samples were taken from them then suitable medications, other than drugs of this study, were prescribed for them, so they took their proper treatment just after blood sampling.
Parameters Assessed
Patients were monitored weekly for any discomfort via telephone calls. Serum VEGF levels were recorded. Blood pressure was measured monthly and an average was taken. Total lipid profile, including total cholesterol level, low-density lipoprotein (LDL), high-density lipoprotein (HDL), and triglycerides, was recorded. Serum blood glucose levels and insulin levels of those in the metformin-treated and vildagliptin-treated groups were measured. Glycated hemoglobin was recorded at the beginning and end of the study period. Body weight was recorded at the beginning and end of the study period and the percent difference in body weight was calculated. All chemical parameters were determined at the beginning and end of the study period. Blood samples were collected and centrifuged at 2000×g for 15 minutes within 30 minutes after collection. Then, sera were separated and collected into 2 clean Eppendorf tubes and stored at −20°C until used for different assays.
Enzyme-Linked Immunosorbent Assay Kits
Determination of serum insulin levels was performed using an enzyme-linked immunosorbent assay (ELISA) kit for insulin (Biorbyt, United Kingdom) following the manufacturer’s protocols.
Determination of serum VEGF levels was performed using the Human VEGF Piokine ELISA Kit (Pleasanton). Determination of serum HbA1c levels was performed using a HbA1c Test Kit purchased from Green Cross Medis Corp. (Chungcheong nam-do, Korea).
Enzymatic Colorimetric Assay
Determination of serum HDL cholesterol level was performed using reagents purchased from the Egyptian Company for Biotechnology (Cairo, Egypt). Determination of serum levels of total cholesterol was performed using reagents purchased from ELITech clinical systems, Zone Industrielle (France).
Determination of LDL cholesterol level was performed using reagents purchased from Centronic GmbH (Wartenberg, Germany). The LDL cholesterol assay was intended for the in vitro quantitative determination of LDL cholesterol in human serum.
Insulin resistance
Insulin resistance was estimated using the homeostasis model assessment for insulin resistance (HOMA-IR) index using the formula
HOMA-IR index = [fasting glucose (mmol/L) × fasting insulin (μU/ml)] / 22.5).
Lifestyle Modification
All patients received instructions to modify their lifestyle, including structured advice for physical activity and dietary advice for adjustment of their intake of carbohydrates and fat.
Subject Compliance and Safety Analysis
Adherence to study medication and adverse events were assessed by weekly phone calls and monthly interviews during and at the end of the study period. Compliance was evaluated by counting the consumed pills in each patient’s strips every routine visit, as pill count is a superior method of medication adherence assessment, 12 compliance was considered adequate when the patient consumed the actual calculated number of pills. Adverse events were reversible and generally mild in nature, including some gastrointestinal discomforts and diarrhoea and that was explained as a minor side effect due to metformin or vildagliptin administration. Throughout the study period, no severe adverse events were observed. Neither dry cough nor angioedema were reported. Potassium monitoring throughout the study was checked and no significant changes were observed among all patients.
Statistical Analysis
Sample size was calculated using the following site https://www.anzmtg.org/stats/PowerCalculator/PowerANOVA
considering a 20% change in systolic blood pressure (SBP) and diastolic blood pressure (DBP) between treatment groups as clinically relevant. Assuming a standard deviation of 0.25, a total of 96 patients would be required to detect a 20% change in BP with a power of 80% at a significance level of .05. However, considering a potential drop-out or lost to follow-up rate, a total of 120 patients (30 for each group) were enrolled. All results were tabulated and after data were checked for normality, normally distributed data were expressed as mean (SD), comparisons between groups were performed using one-way analysis of variance (ANOVA) followed by Tukey post-hoc test, and non-normally distributed data were presented using median and interquartile range, Kruskal-Wallis test was used followed by Dunn test for multiple comparison. Differences between both groups at baseline or at the end of the study were evaluated using the unpaired t test while changes from baseline to 24 weeks of follow-up in both groups were evaluated using the paired t test for continuous variables. Data analysis was performed using the statistical package for social science, version 17 (SPSS Software, SPSS Inc., Chicago). The P ≤ .05 was considered significant.
Results
The baseline demographic and clinical characteristics of all patients and the effects of each treatment group were shown in Table 1. There were no differences between the 2 treatment groups in respect to age, duration of T2DM or hypertension, BP level, BMI, lipid profile, HbA1c level at baseline. The data yielded from the study indicated that serum VEGF levels in group II, the diabetic control, 193 (166.5, 210 interquartile range [IQR]) and vildagliptin-treated group 153 (134.7, 180.5 IQR) were significantly higher than those in the healthy controls 53.5 (47, 60 IQR) P < .05. The metformin-treated group 50.45 (30, 64.5 IQR) showed a serum VEGF level that was significantly lower than its level in the diabetic control group. In addition, the VEGF serum level of the vildagliptin-treated group was significantly higher than its level in the metformin-treated group (Figure 1). On the other hand, VEGF serum levels of metformin group at the end of the study were significantly lower than its level at baseline, while vildagliptin treated group showed a significant increase in VEGF serum levels at the end of the study when compared to baseline levels (Table 1).
Clinical and Biochemical Parameters Before and After 24 Weeks of Treatment in the Treated Groups of the Study.a

Abbreviations: BMI, body mass index; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein, VEGF, vascular endothelial growth factor.
aDifferences between both groups at baseline or at the end of the study were evaluated using the unpaired t test while changes from baseline to 24 weeks of follow-up in both groups were evaluated using the paired t test for continuous variables.
bStatistically significant differences within each group after treatment when P ≤ .05.
cStatistically significant differences between the 2 groups when P ≤ .05.
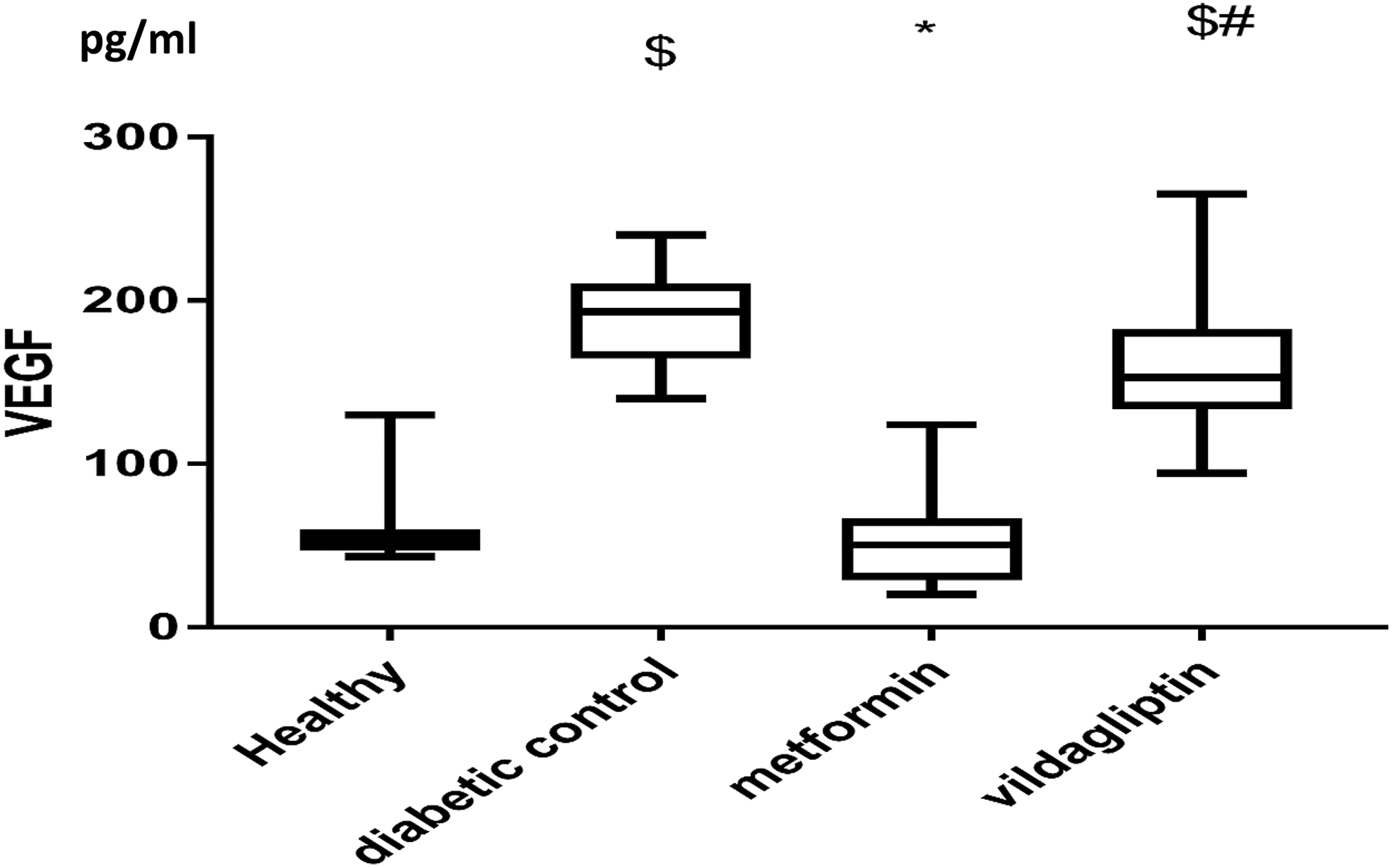
Effect of metformin or vildagliptin in addition to captopril on VEGF serum levels of diabetic hypertensive patients compared to the 2 control groups. Results are expressed as median and interquartile range and analyzed using Kruskal-Wallis followed by Dunn multiple comparisons test. $Significantly different from healthy group. *Significantly different from diabetic control group. #Significantly different from metformin treated group. VEGF indicates vascular endothelial growth factor.
The diabetic control group 168 (165, 171 IQR), 95 (95, 98 IQR) mm Hg, metformin-treated group 151.5 (149, 156 IQR), 94.5 (90, 95 IQR) mm Hg, and vildagliptin-treated group 142.5 (140, 145 IQR), 85 (85,89 IQR) mm Hg, had significantly higher systolic and diastolic blood pressures than the normal controls 120 (120, 122 IQR) mm Hg. Furthermore, the systolic and diastolic blood pressures of the vildagliptin-treated group and metformin-treated group were significantly lower than those of the diabetic controls. However, the vildagliptin-treated group recorded a significantly lower systolic and diastolic blood pressure than the metformin-treated group (Figure 2). In addition, metformin showed a significant decrease in SBP of patients when compared to baseline readings while vildagliptin showed a decreased SBP and DBP of patients at the end of the study when compared to baseline (Table 1).
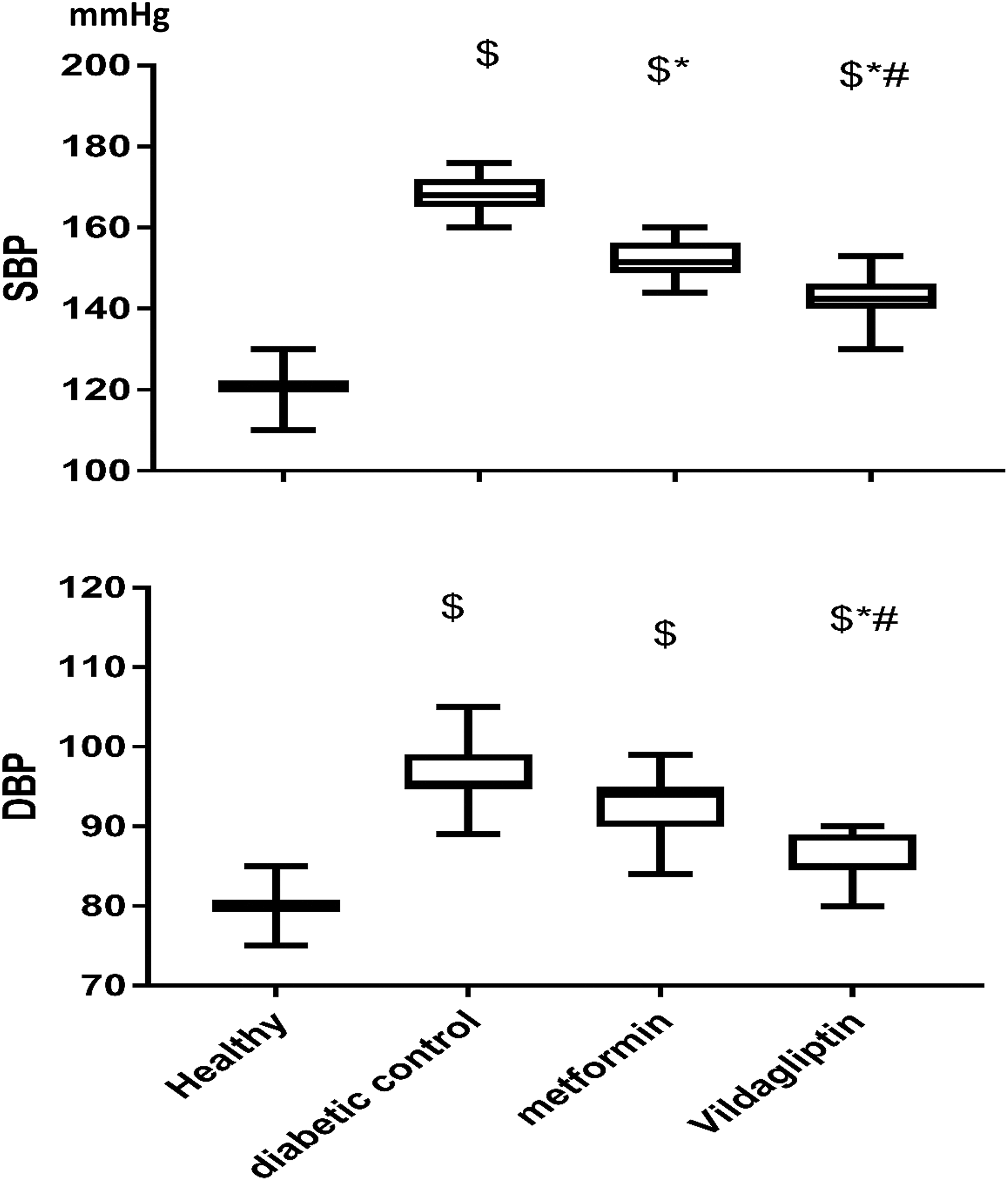
Effect of metformin (1000 mg bid) and vildagliptin (50 mg bid) in addition to captopril on serum level of systolic and diastolic blood pressure compared to the 2 control groups. Results are expressed as median and interquartile range and analyzed using Kruskal-Wallis followed by Dunn multiple comparisons test. $Significantly different from healthy group. *Significantly different from diabetic control group. #Significantly different from metformin-treated group.
The lipid profile of each group demonstrated that the total cholesterol levels of the diabetic control group 244.4 (222, 263 IQR) mg/dL, metformin-treated group 234.7 (199, 269 IQR) mg/dL, and vildagliptin-treated group 198.56 (161, 220) mg/dL, were significantly higher than their levels in the healthy controls 154.4 (127, 167 IQR) mg/dL (P < .05), as shown in Figure 3. Additionally, vildagliptin-treated group showed a significantly lower total cholesterol level than that of both diabetic control group and metformin treated group (Figure 3).

Effect of metformin (1000 mg bid) and vildagliptin (50 mg bid) in addition to captopril on serum level of lipid profile compared to the 2 control groups. Results are expressed as median and interquartile range and analyzed using Kruskal-Wallis followed by Dunn multiple comparisons test for both TGs and HDL. $Significantly different from healthy group. *Significantly different from diabetic control group. #Significantly different from metformin-treated group.
The triglyceride levels of the normal controls 155.2 (145, 165 IQR) mg/dL, were not significantly different from those of the metformin-treated group 194.99 (141, 241 IQR) mg/dL, or the vildagliptin-treated group 131.36 (130, 166 IQR) mg/dL. In contrast, the triglyceride levels of the diabetic control group 266.59 (245, 304 IQR) mg/dL were significantly higher than those of the normal control group (Figure 3). In addition, the vildagliptin-treated group showed a significant decrease in triglyceride levels when compared to the diabetic control group and the metformin-treated group (Figure 3).
A closer look at the data revealed that the HDL level of the control diabetic group 34.4 (32, 36 IQR) mg/dL, was significantly lower than its level in the healthy control group 53.9 (46, 55 IQR) mg/dL (P < .05; Figure 3), while there was no significant difference in HDL levels between the healthy group and the metformin-treated group 53.15 (44, 59 IQR) mg/dL (Figure 3). The current data revealed a significant increase in LDL levels in the diabetic control group 145 (130, 166 IQR) mg/dL, metformin-treated group 140 (128, 167 IQR) mg/dL, and vildagliptin-treated group 123.45 (103, 148 IQR) mg/dL compared to LDL levels in the healthy control group 100.16 (95, 111 IQR) mg/dL (P < .05). The metformin-treated group showed no significant difference in LDL levels compared to the diabetic control group, while the vildagliptin-treated group showed a significant decrease in LDL levels compared to the diabetic controls and metformin treated group (Figure 3). As reported in Table 1 metformin didn’t show any significant difference in total cholesterol or LDL serum levels at the end of the study when compared to baseline, while in the same group triglyceride and HDL levels were significantly changed at the end of the study compared to baseline. However, vildagliptin patients showed significantly different serum levels of all lipid profile components at the end of the study when compared to baseline (Table 1).
The HOMA-IR was used to estimate insulin resistance; HOMA-IR = (fasting blood glucose × fasting insulin level)/22.5. The results revealed that metformin lowered the calculated HOMA-IR after 6 months of treatment (10.82 ± 0.84 vs 2.67 ± 0.82). The vildagliptin-treated group showed a reduced HOMA-IR value after treatment as well (10.69 ± 1.16 vs 1.228 ± 0.62; Figure 4).
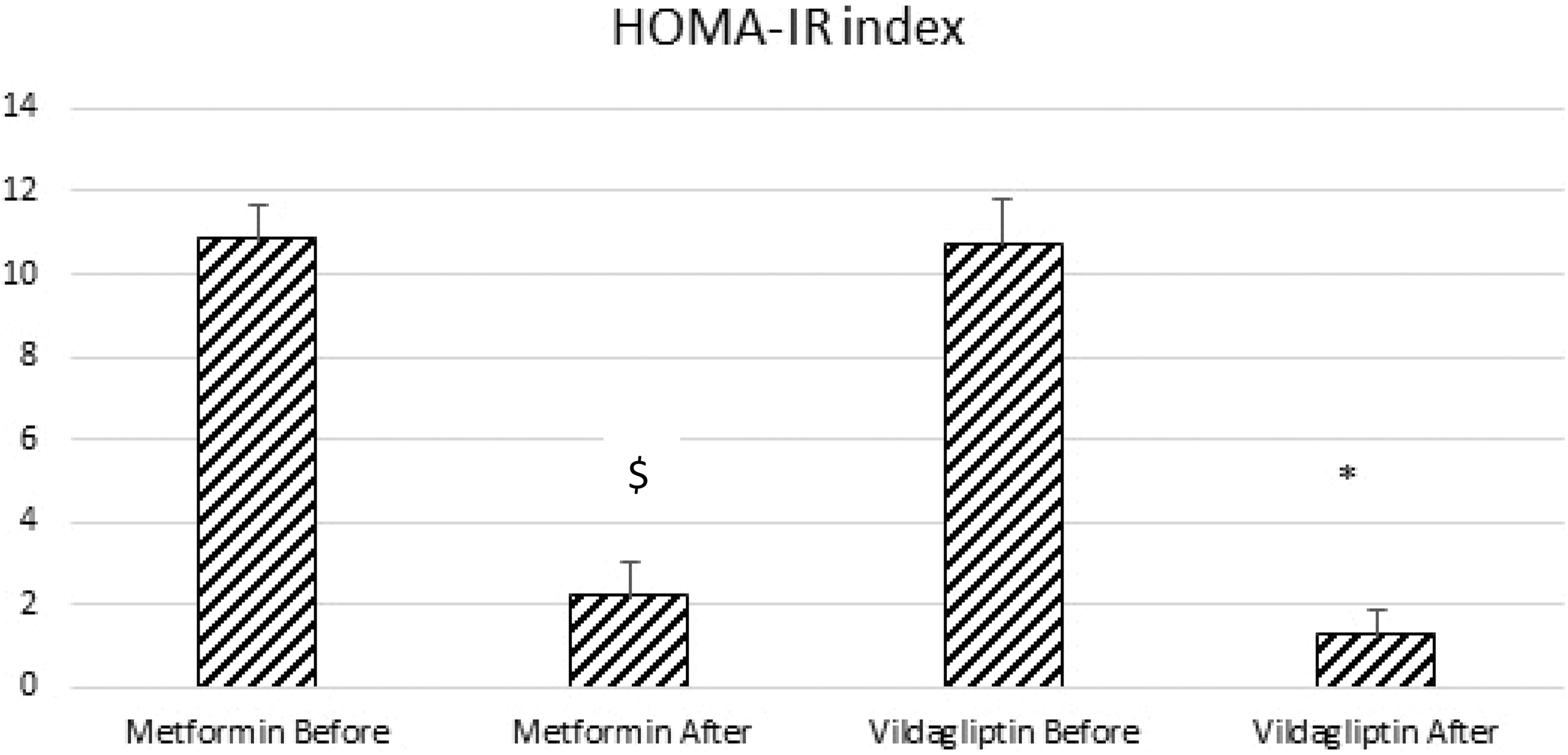
Effect of metformin (1000 mg bid) and vildagliptin (50 mg bid) in addition to captopril on HOMA-IR index of diabetic hypertensive patients compared to the 2 control groups. Results are expressed as mean ± SD and analyzed using paired t test. $ if metformin after is significantly different from metformin before group. * when vildagliptin after is significantly different than vildagliptin before group.
The HbA1c levels of the diabetic control group 10 (9.6, 11.2 IQR) %, metformin-treated group 8.9 (8.1, 10.1 IQR) %, and vildagliptin-treated group 8.2 (7.6, 9.1 IQR) % were significantly higher than the normal levels in the healthy group 4.9 (4.4, 5.3 IQR) % (P < .05). The HbA1c levels in the metformin- and vildagliptin-treated groups were significantly lower than those in the diabetic control group (Figure 5). In addition, both treatments significantly lowered HbA1c readings at the end of the study than its readings at baseline (Table 1). However, no significant difference in HbA1c levels appeared between the metformin-treated group and the vildagliptin-treated group.

Effect of metformin or vildagliptin in addition to captopril on HbA1c in different groups compared to the 2 control groups. Results are expressed as median and interquartile range and analyzed using Kruskal-Wallis followed by Dunn multiple comparisons test. $Significantly different from healthy group. *significantly different from diabetic control group. #Significantly different from metformin treated group. HbA1c indicates glycated hemoglobin.
The results obtained from this study revealed that the percentage change in body weight in the metformin-treated group was significantly higher than that in the vildagliptin-treated group (−3.48 ± 0.31 vs 4.22 ± 0.34; Table 2).
Effect of Metformin (1000 mg bid) and Vildagliptin (50 mg bid) on Body Weight in Diabetic Hypertensive Patients Treated With Captopril (25 mg/day).a

aResults are expressed as mean ± SD and analyzed using ANOVA followed by Bonferroni post-hoc test. Percentage change in BWt was calculated using a formula: % change BWt = [(final BWt−baseline BWt)/baseline BWt] × 100.
bSignificantly different from metformin group at P < .05.
Discussion
This study was designed to investigate the effect of metformin and vildagliptin on regulating blood pressure in diabetic hypertensive Egyptian patients and for this reason a low dose of captopril was used (25 mg/day) to investigate the expected effect of the drugs in lowering high blood pressure. In this study vildagliptin wasn’t used as a VEGF inducer, it only prevented VEGF levels form complete normalization after treatment, as levels of VEGF in patients’ serum when treated with vildagliptin were significantly higher than its normal levels but was nonsignificant when compared to its levels in the serum of group II diabetic patients. The major findings from the study are as follows: metformin and vildagliptin attenuated insulin resistance in both groups and vildagliptin provided a superior advantage over metformin through its ability to restore blood pressure. Serum VEGF levels were significantly higher in the vildagliptin-treated group than in the metformin group. Triglycerides, total cholesterol, and serum LDL levels were more effectively decreased by vildagliptin than by metformin, so generally Vildagliptin improved lipid profile of these patients.
Both therapeutic agents in this study obviously ameliorated insulin resistance, as indicated by the decreased levels of fasting plasma insulin and glucose resulting in a reduced HOMA index. These results are strongly consistent with a previous study that similarly showed that metformin and vildagliptin decreased insulin levels and HOMA index in rats. 6
Moreover, the data of the present work highlighted the greater efficacy of vildagliptin to modulate high blood pressure in patients than that of metformin. This difference may have been due to the significantly higher VEGF levels in the serum of vildagliptin-treated patients, as vildagliptin has been reported to have a protective action on the vasculature through its ability to promote angiogenesis and endothelial cell hemostasis, as supported by William et al in their recent study. 13 On the other hand, VEGF level was most elevated in serum of the untreated group II this could be explained as VEGF was suggested to regulate the neovascularization that occur in hypoxia-induced disorders such as diabetic vascular complications. Several studies reported that in DM, VEGF expression is upregulated by hypoxia, high glucose-induced protein kinase C activation, or tumor growth factor-β mediated pathways. 14,15
An important finding in this study was the elevation in levels of serum VEGF, which is responsible for providing vascular endothelial protection and increasing cell proliferation, with vildagliptin but not metformin treatment. Sivasinprasasan and colleagues reported that metformin was harmful to endothelial membrane and nitric oxide function, resulting in increased blood pressure, 6 supporting the findings of the present study, as metformin-treated patients in this study did not exhibit a lower DBP than the diabetic controls. However, metformin mildly lowered the systolic blood pressure, which could be due to the antidiabetic effect of metformin in addition to the captopril effect. However, vildagliptin has been reported to enhance blood flow and endothelial cell function, 16 supporting the current findings and providing an explanation for the superior effect of vildagliptin in raising serum VEGF levels and modulating high blood pressure in those patients.
A closer look at the lipid profile results showed that vildagliptin more effectively decreased lipid levels than did metformin, which can generally be added to the positive outcomes of vildagliptin. These findings were supported by a recent study that indicated a beneficial effect of vildagliptin on the lipid profile of patients. 16 The effects of vildagliptin on the lipid profile could also contribute to lowering the CVS risk accompanied by both DM and hypertension.
The HbA1c results showed an improvement associated with both drugs, but the HbA1c level did not reach the target level reported in some recent guidelines, 1,2 which may by an indicator for the addition of another antidiabetic drug or a more restricted lifestyle change.
In the current study, metformin treatment was significantly better in weight reduction than vildagliptin treatment, which produced a remarkable increase in body weight. Likewise, metformin was previously reported to increase the oxidation of fatty acids and decrease weight gain. 6 Vildagliptin weight gain was also reported in a recent study 16 ; however, the weight gain reported in this study was opposed to the reports of many studies that denied an effect of vildagliptin on weight gain. 17,18 These conflicting results may be explained by variations in race among the study populations.
Together, these observations may lead to an improvement in the treatment of elevated blood pressure in patients with DM in a manner partly independent of glucose lowering.
Conclusion
It is beneficial for diabetic patients suffering from hypertension to treat their DM with a medication that can also reduce their blood pressure and CVS complications. Consequently, vildagliptin may be more beneficial in the treatment of DM in diabetic hypertensive patients, especially lean patients, as it can positively reduce blood pressure by modulating VEGF levels and improving lipid profiles in addition to its favorable effect on HbA1c and insulin resistance; however, vildagliptin can include a slight increase in weight gain. Metformin is still the first-line treatment in all newly diagnosed patients who only suffer from DM regardless of weight. The present trial has several limitations, including a small sample size, as the trial was not funded by any institution, and the relatively short treatment duration. However, the DPP-4 inhibitor vildagliptin was demonstrated to be highly efficient in achieving adequate glycemic control after short-term treatment as well as better blood pressure control when used with captopril than metformin used with captopril. Furthermore, vildagliptin was safe regarding hypoglycemic episodes. Adverse events were mild and appeared at similar rates in both study arms. Thus, vildagliptin proved to be an efficient and safe treatment option against DM comorbid with hypertension.
Supplemental Material
Supplemental Material, Fig._S1 - The Role of Vildagliptin in Treating Hypertension Through Modulating Serum VEGF in Diabetic Hypertensive Patients
Supplemental Material, Fig._S1 for The Role of Vildagliptin in Treating Hypertension Through Modulating Serum VEGF in Diabetic Hypertensive Patients by Abdel Rahman El-Naggar, Dalia Zaafar, Mohammed Elyamany, Soha Hassanin, Atef Bassyouni and Hekma Abdel-Latif in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors' Contribution
D. Zaafar and S. Hassanin contributed to conception and design, analysis, and interpretation. M. Elyamany and A. Bassyouni contributed to conception and interpretation. A. Elnaggar and H. Abdel-Latif contributed to conception and design, acquisition, analysis, and interpretation and drafted article. All authors critically revised article, gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
