Abstract
Background:
Proper anticoagulation is a crucial therapeutic regimen in atrial fibrillation (AF).
Objectives:
To evaluate the real-life anticoagulation prescriptions of AF patients upon hospital discharge.
Methods:
We studied 768 patients with comorbid AF who were discharged from the cardiology ward of a tertiary hospital. We assessed the appropriateness of oral anticoagulation (OAC) regimens at discharge based on stroke risk (CHA2DS2-Vasc score), SAMe-TT2R2 (sex, age, medical history, treatment, tobacco, race) score for vitamin K antagonists (VKA), and European labeling for nonvitamin K oral anticoagulant (NOAC) dosing. Logistic regression identified factors associated with suboptimal OAC use.
Results:
Of 734 patients at significant (moderate or high) stroke risk, 107 (14.6%) were not prescribed OAC, which was administered to 23 (67.6%) of 34 patients at low risk. Nonprescribing of OAC to high-risk patients was associated with paroxysmal AF (adjusted odds ratio [OR]: 2.42, 95% confidence interval [CI]: 1.47-3.99, P < .001), history of major bleeding (adjusted OR: 1.89, 95% CI: 1.03-3.47, P = .039), and concomitant antiplatelet use (adjusted OR: 5.78, 95% CI: 3.51-9.51, P < .001). Anticoagulation control was inadequate (SAMe-TT2R2 score > 2) in 102 (50.2%) VKA-treated patients. Off-label dosing was evident in 118 (28.9%) NOAC-treated patients and was associated with a prior stroke/transient ischemic attack (adjusted OR: 2.06, 95% CI: 1.10-3.85, P = .023). Both outcomes were independently associated with low creatinine clearance.
Conclusions:
One of 6 patients with AF newly discharged from the hospital was treated discordantly for the corresponding risk of stroke. Suboptimal OAC use was evident in half of VKA regimens, twice as common compared to NOACs, and could be predicted by several clinical parameters.
Introduction
Proper use of oral anticoagulants (OAC) is crucial to the management of patients with atrial fibrillation (AF). 1 Following the introduction of nonvitamin K oral anticoagulants (NOACs), stroke-prevention options in nonvalvular AF have expanded.
Still, more options may add greater complexity in deciding on the right anticoagulation regimen for the right patient. This increases the interest in exploring suboptimal OAC use in the real world, namely, failure to (1) apply OAC according to stroke risk, as conveyed by the CHA2DS2-Vasc (congestive heart failure, hypertension, age ≥ 75 years, diabetes mellitus, stroke/transient ischemic attack, vascular disease, age 65 to 74 years, sex category) score; (2) achieve an adequate time in therapeutic range (TTR, ie, time with INR [international normalized ratio] between 2 and 3) when using vitamin-K antagonists (VKAs); and (3) prescribe NOACs in appropriate and licensed doses. Observational studies that rely on complex populations are ideal for demonstrating these flaws in AF treatment. Real-life data may also draw clinicians’ attention toward better implementation of evidence-based strategies.
In this study, we aimed to analyze a “real-world” contemporary cohort of hospitalized patients with coexisting nonvalvular AF. Our primary aim was to assess the discharge anticoagulation regimens on the basis of stroke risk (CHA2DS2-Vasc score) and drug-specific parameters (projected TTR for VKAs, European labeling for NOAC dosing). Secondarily, we explored factors associated with suboptimal OAC use.
Methods and Materials
Study Design and Setting
This is an ancillary cross-sectional study to MISOAC-AF (motivational interviewing to support oral anticoagulation adherence in patients with nonvalvular AF, ClinicalTrials.gov identifier: NCT02941978). The latter is an ongoing, prospective, randomized controlled trial, conducted in AHEPA University Hospital. Briefly, MISOAC-AF seeks to establish a role for strengthening compliance of AF patients to OACs via tailored patient–physician interviews and improved scripted guidance.
Study Population
Our study population consisted of consented patients (aged >18 years) who were discharged from the cardiology ward of our hospital having comorbid, electrocardiographically confirmed nonvalvular AF or atrial flutter (first diagnosed, paroxysmal, persistent, or permanent). Patients were included irrespective of prior anticoagulant exposure from baseline participants of MISOAC-AF. The principal trial did not affect anticoagulation choices at discharge; these were at the discretion of the attending physician. Exclusion criteria were moderate-to-severe mitral stenosis or mechanical valves 2 and no information regarding anticoagulant use at discharge. We also excluded patients with an indication for anticoagulation other than AF (eg, treatment of an acute episode or long-term prevention of deep vein thrombosis or pulmonary embolism)
Data Sources
We used data from the MISOAC-AF electronic registry, including baseline demographics, medical history, laboratory values, discharge medication, and discharge diagnoses. These data were collected into standardized templates by trained, independent medical researchers, who interviewed patients during their hospitalization and reviewed discharge notes. All data used for the current study preceded any interventions of the MISOAC-AF trial. This study was approved by the institutional review board of Aristotle University of Thessaloniki, Faculty of Medicine and conformed to the Declaration of Helsinki.
Study Variables
We assessed thrombotic risk by combining comorbidity information into the CHA2DS2-Vasc score. 3 We categorized patients as at “low,” “moderate,” and “high stroke risk” based on the presence of zero, one, or more than one nongender-related stroke risk factors. For these strata, the 2016 European Society of Cardiology (ESC) AF Guidelines disfavor, favor, and strongly recommend OAC, respectively. 2 Similarly, we calculated the SAMe-TT2R2 (sex, age, medical history, treatment, tobacco use, race) score. Serving as a TTR surrogate, the score allows a priori identification of patients deemed to fail VKA therapy. Since presented by Apostolakis et al in 2013, 4 the score was validated in multifaceted coumadin-anticoagulated populations 5 and is supported by a recent ESC consensus article. 6 We also calculated the HAS-BLED (hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile INR, elderly patients, drugs/alcohol) score to assess bleeding risk. 7 Details regarding the scores featured in this analysis can be found in the Supplemental Tables 1 to 3.
We took discharge diagnoses into account and coded additional comorbidities and points into the scores, as needed. We estimated creatinine clearance (CrCl) by inputting the serum creatinine value closest to discharge to the Cockcroft formula. 8 Hence, all calculations reflect the clinician-utilized parameters when prescribing an OAC for a patient leaving the hospital.
Study End Points
First, we assessed the percentages of OAC (VKA [acenocoumarol], NOAC [dabigatran, rivaroxaban, apixaban]) versus no OAC (antiplatelet therapy [AP; aspirin, clopidogrel, ticagrelor or combinations], low-molecular-weight heparin (LMWH), and no antithrombotic therapy) prescriptions at discharge across the stroke risk spectrum (low, moderate, and high risk).
Second, we assessed the quality of individual OAC regimens prescribed to patients at discharge (Supplemental Table 3): For VKA prescriptions, we used the SAMe-TT2R2 score to discern patients expected to achieve good anticoagulation control (score 0-2, ie, projected TTR ≥ 70%) from patients who may be unable to achieve good anticoagulation control (score > 2, ie, projected TTR < 70%).
6
For NOAC prescriptions, we referenced the approved European labels that were refined in the 2018 European Heart Rhythm Association Practical Guide on NOAC use in AF.
2,9
Accordingly, we categorized NOAC-treated patients by dose: “Dosing consistent with labeling,” including “appropriate, standard dosing” (ie, no indication for reduced dose, received standard dose) and “appropriate, reduced dosing” (indication for reduced dose and received reduced dose) categories. “Potential off-label dosing,” including “potential underdosing” (ie, indication for standard dose, received reduced dose) and “potential overdosing” (ie, indication for reduced dose and received standard dose).
Statistical Analysis
Patients were grouped according to the study’s end points. Differences between variables with normal and skewed distributions were assessed with Student t test and the Mann-Whitney U test, respectively. Categorical variables were tested by the χ2 or Fisher exact test as appropriate. A 2-sided P value of <.05 was accepted as statistically significant.
We used univariate and separate logistic regression models to investigate clinically plausible factors associated with prescribing (1) no OAC to high stroke risk patients and (2) an off-label-dosed NOAC. These factors included age, gender, AF subtypes (paroxysmal and first diagnosed), history of stroke/TIA, major bleeding and heart failure, CHA2DS2-Vasc and HAS-BLED scores, and CrCl, antiplatelet, and rhythm control medication at discharge. We forced variables of major interest into the first step of the multivariate analysis. Next, we filtered the rest into the final model using backward elimination (retention threshold P value < .10). The composite variable HAS-BLED or its collinear covariates (eg, age) were alternatively inserted between steps, resulting in a primary and secondary model for each outcome. The secondary models, where the composite variable was initially controlled for, served as sensitivity analyses. The models’ predictive ability was assessed according to pseudo-R 2 and C-statistics. Calibration was evaluated with the Hosmer and Lemeshow test.
Missing data in the multivariate models were replaced by means of multiple imputation, using chained equations. Combined results from 5 imputations are reported, as odds ratios (OR) with 95% confidence intervals (CI; further elaboration in Supplemental data online, along with Supplemental Table 6). Complementary sensitivity analyses containing nonimputed data were conducted.
IBM SPSS Statistics version 24.0 (IBM Corp, Armonk, New York) was used for data coding and analysis and GraphPad Prism version 7.0 (GraphPad Software, San Diego, California) for graphing.
Results
Patient Characteristics
A total of 768 unselected patients with nonvalvular AF (mean age 73.6 [11.0] years; 53.9% male), discharged between December 2015 and November 2017, were studied (flowchart shown in Supplemental Figure 1). Postdischarge clinical characteristics of patients, divided by OAC use, are presented in Table 1. Further discrimination between different stroke risk strata is explored in Supplemental Table 4. Overall, the mean CHA2DS2-Vasc score was 4.4 (1.9), mean SAMe-TT2R2 score 2.6 (1.1), and mean HAS-BLED 1.9 (1.1). Comorbidities aggregated in patients who were prescribed OAC, paralleling an increase in stroke risk.
Clinical Characteristics of AF Patients According to OAC Use at Discharge.a
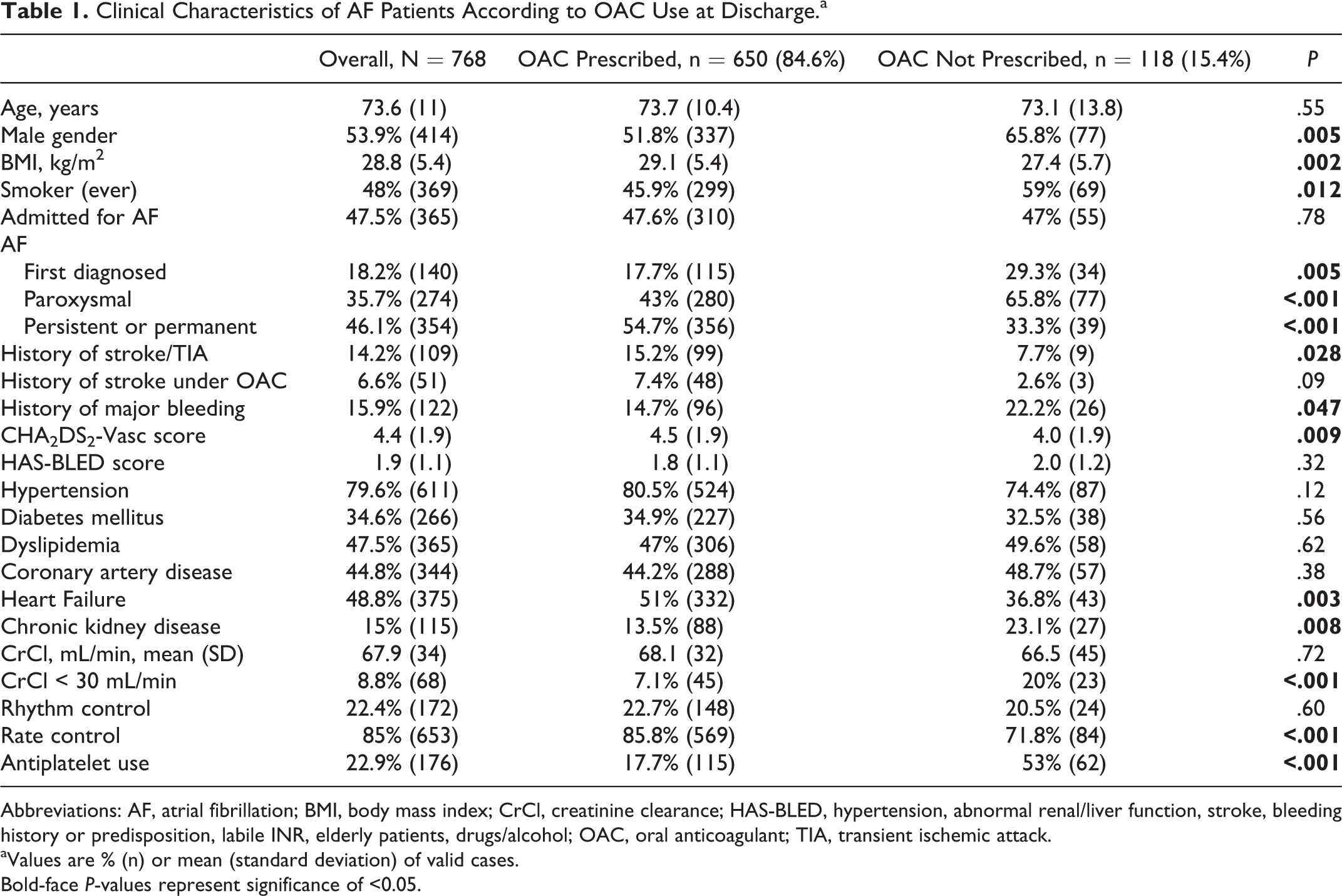
Abbreviations: AF, atrial fibrillation; BMI, body mass index; CrCl, creatinine clearance; HAS-BLED, hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile INR, elderly patients, drugs/alcohol; OAC, oral anticoagulant; TIA, transient ischemic attack.
aValues are % (n) or mean (standard deviation) of valid cases.
Bold-face P-values represent significance of <0.05.
Oral Anticoagulant Use Related to Stroke Risk
The distribution of antithrombotics provided to the discharged AF patients, stratified by stroke risk, is illustrated in Figure 1. Clinicians chose not to prescribe OAC to 107 (14.6%) of 734 patients at significant stroke risk. In specific, 101 (14.6%) of 693 patients at high stroke risk and 6 (14.6%) of 41 patients at moderate stroke risk were given antiplatelets, LMWH, or no antithrombotic instead of OAC. Conversely, OAC prescriptions were identified in 23 (67.6%) of 34 patients at low risk. In total, 130 (16.9% of total) patients were treated discordantly to their corresponding stroke risk.

Stratification of antithrombotic discharge regimens according to stroke risk OAC-treated groups are marked with brackets. AP indicates antiplatelet; LMWH, low molecular weight heparin; NOAC, nonvitamin K antagonist oral anticoagulant; OAC, oral anticoagulant; VKA, vitamin K antagonist.
Oral anticoagulation nonprescribing in high-risk AF showed a strong unadjusted relation (P < .05) with all variables tested, except from age and stroke/TIA (Supplemental Figure 2). In the multivariate analysis, independent predictors of this outcome were presence of paroxysmal AF (OR: 2.42, 95% CI: 1.47-3.99, P < .001), a prior major bleeding episode (OR: 1.89, 95% CI: 1.03-3.47, P = .039), antiplatelet use (OR: 5.78, 95% CI: 3.51-9.51, P < .001), and a lower CrCl (OR: 1.16 per 10 mL/min decrease, 95% CI: 1.05-1.29, P = .005) when controlling for age, gender, history of stroke/TIA, and heart failure. When forcing the HAS-BLED score (but not its covariates) along with the rest of the variables of interest into the first step, the secondary model indicated the same independent predictors, apart from major bleeding (see Figure 2). The models’ performance across the original and imputed data sets was essentially equal (all C-statistic 0.79, 95% CI: 0.74-0.84, pseudo-R 2: 14% to 25%, Hosmer and Lemeshow test P = .67-.99).

Multivariate analysis of factors related to nonprescribing of OAC to high stroke-risk AF patients. Variables forced into the first step of the primary and secondary model are marked as (*) and (†) respectively. The remaining variables’ contribution was assessed with backward regression. AF indicates atrial fibrillation; CI, confidence interval; CrCl, creatinine clearance; OAC, oral anticoagulant; OR, odds ratio; TIA, transient ischemic attack.
We observed that 22% of high stroke-risk patients who did not receive OAC had severe renal impairment (ie, CrCl < 30 mL/min, see Supplemental Table 5), which could influence the choice to anticoagulate per se. Thus, we performed an additional sensitivity analysis excluding these patients, whereby CrCl was now listed as a confounder in the primary model and barely reached significance in the secondary one (Supplemental Figure 3).
Vitamin K Anticoagulant and NOAC Prescriptions Quality Assessment
Quality assessment of different OAC agents (VKA and NOAC) administered to discharged AF patients is illustrated in Figure 3. Acenocoumarol prescriptions in the context of a paired SAMe-TT2R2 score >2 were identified in 102 (50.2%) of 203 patients. Of 448 NOAC-treated patients, the exact dosage was available in 408 (8.9% missing). Doses consistent with European labeling were administered in 290 (71.1%) patients, whereas in 118 (28.9%) patients the dose was off-label; 97 (23.8%) were underdosed and 21 (5.1%) were overdosed. Patient-level characteristics according to each agent’s assessment are provided in Supplemental Table 5.

Assessment of VKA and NOAC discharge regimens, according to the SAMe-TT2R2 score and European NOAC-specific labeling, respectively. NOAC indicates nonvitamin K antagonist oral anticoagulant; TTR, time in therapeutic range; VKA, vitamin K antagonist.
Age, history of stroke/TIA, stroke and bleeding risk scores, and CrCl had a significant unadjusted relation to prescribing an off-label NOAC dose (Supplemental Figure 4). After multivariate adjustment, a history of stroke/TIA (OR: 2.06, 95% CI: 1.10-3.85, P = .023) and lower CrCl (OR: 1.13 per 10 mL/min decrease, 95% CI: 1.01-1.26, P = .037) were independently associated with this outcome when controlling for age and major bleeding. In the secondary model, each 1-point increase in the HAS-BLED score was independently associated with increased odds of the outcome (adjusted OR: 1.44, 95% CI: 1.14-1.80, P = .002). Inclusion of the score replaced the effects of age, stroke/TIA, major bleeding, whereas the effect of CrCl remained stable (Figure 4). Due to the high rate of missing data regarding NOAC dose, we repeated the multivariate analyses in the original data set, yielding broadly similar results to imputed data. Both models exhibited moderate to good discrimination across data sets (all C-statistic 0.66, 95% CI: 0.60-0.71) and adequate calibration (Hosmer and Lemeshow test P = .14-.68), although explaining only between 8% and 11% of the variation in the outcome.
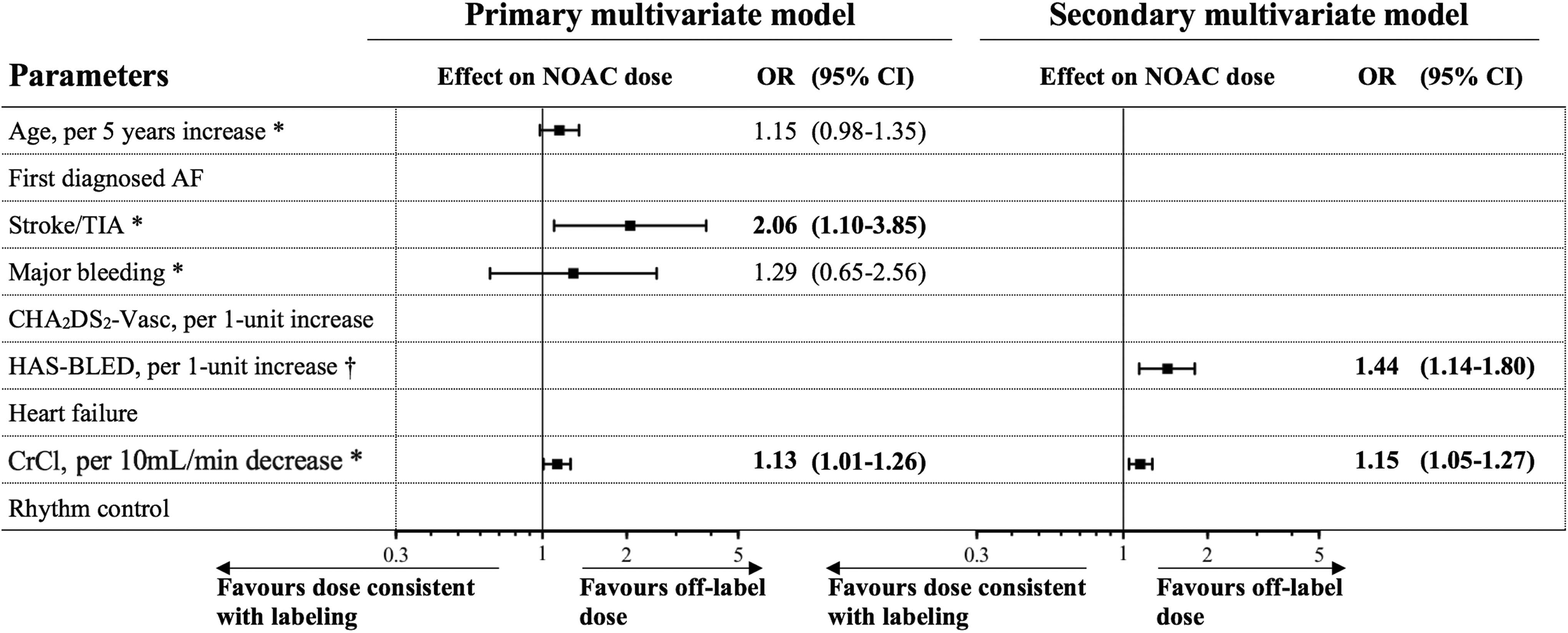
Multivariate analysis of factors related to prescribing an off-label dosed NOAC. Variables forced into the first step of the primary and secondary model are marked as (*) and (†) respectively. The remaining variables’ contribution was assessed with backward regression. AF indicates atrial fibrillation; CI, confidence interval; CrCl, creatinine clearance; NOAC, nonvitamin K antagonist oral anticoagulant; OR, odds ratio; TIA, transient ischemic attack.
Discussion
To our knowledge, this is the first comprehensive quality analysis of OAC use, accounting both for all stroke risk strata and all agents in real-world AF patients, transitioning from inpatient to outpatient care. Hereby, we report a nonnegligible proportion of patients as not receiving OAC despite significant stroke risk, receiving OAC despite insignificant stroke risk, or receiving OAC in a suboptimal fashion.
The mean discharge-CHA2DS2-Vasc score in our study is higher than recent similarly oriented real-world studies, which mainly have reported preadmission or outpatient values. 10 –14 This showcases that patients admitted to a cardiology ward are highly multimorbid, and the acute reason for admission may contribute to their postdischarge stroke risk. Nevertheless, we showed that these high-risk patients might sometimes be discharged without OAC therapy. This “risk-treatment paradox,” as pointed out by Cullen et al, 15 may be more prominent in acutely hospitalized patients, which forms the basis of our study population. It may be speculated that the severity of these patients’ primary illnesses draws the clinicians’ attention away from provisioning OAC treatment. Others have shown that undertreatment of high-stroke risk AF varies from 20% to more than 50% in different settings. 10 –14 In a worldwide study by Huisman et al, 16.8% of patients with a CHA2DS2-Vasc score ≥2 received no OAC, 16 which was similarly demonstrated in our study’s significant stroke risk strata. A small subset of these patients who received LMWH could have done so as a means of bridging for a scheduled intervention, although this is not encouraged in nonvalvular AF. 2,9
Patients at moderate stroke risk were largely covered with OAC, showing that cardiologists conform to the guidelines’ recommendations favoring OAC for this category. Oral anticoagulation prescribing in most of our patients with zero nongender-related stroke risk factors could have occurred for transient purposes, such as postcardioversion or post-AF ablation. Indeed, guidelines suggest a short-term anticoagulation in all patients at low stroke risk after cardioversion of AF of ≥48 hours duration. In case of AF of recent onset (<48 hours duration), it remains a clinician’s call to short-term anticoagulate a patient at low stroke risk. 9 A large multicenter registry has also reported a high rate (approximately 50%) of OAC use in low stroke risk AF, although it was focused on stable outpatients. 15
As much as half of VKA prescriptions were expected to underperform in terms of stroke prevention, which is in line with contemporary data on real-life AF patients. 17 Hospitalization is an excellent opportunity to evaluate VKA users or OAC candidates in general and opt for NOAC initiation (or switch) whether their SAMe-TT2R2 score corresponds to a TTR <70%. Likewise, a projected TTR ≥70% informs the clinician whether the previously coumadin-anticoagulated patient can stay on this regimen, without compromises in outcomes. 2,6
More than a quarter of administered NOAC regimens did not comply with the labeled recommendations regarding stroke prevention. We corroborate the findings of Steinberg et al 18 and Yao et al 19 that this is mainly an issue of underdosing. Nevertheless, these researchers report lower off-label dosing rates of 13% and 22%, respectively, albeit concerning outpatient practice. In-hospital physicians’ role is complex when opting for a dose at discharge; they must weigh evolving patient clinical profiles, such as dynamic changes in renal function, newly diagnosed comorbidities and new interacting medication as well as competing thrombotic and hemorrhagic risk. Thus, they may deliberately overdose or underdose a patient, for example, when observing a trend in renal function during hospitalization.
Our study provides strong replication evidence of the misperception that paroxysmal AF carries a lower stroke risk burden, that a major bleeding episode should halt long-term anticoagulation efforts, and that AP is effective in stroke prevention. When dosing NOACs, physicians may overestimate the potential bleeding risk, compared to the benefits of stroke reduction, even more in the face of a previous cerebrovascular event. Also, an impaired renal function may negatively influence effective anticoagulation strategies. This may even be more pronounced below the 30 mL/min threshold, when deciding whether to apply OAC or not. Clinicians are advised to be vigilant when encountering patients with AF and these characteristics, as such patients are seemingly more prone to suboptimal anticoagulation management.
Limitations and Strengths
First, since we focused on a hospitalized cohort of patients with AF, the results may not be generalizable to the wider nonhospitalized AF population. More bias could be introduced due to the cohort being managed by university physicians with knowledge of participating in a registry forming context. However, our results reflect academic inpatient practices and, due to their magnitude, may also raise awareness for proper OAC use in the general population. Second, we could not account for all variables that might have influenced the use of OAC, which may also be reflected by the low variance explained by the regression models. Unmeasured confounders, such as clinician and patient preference, cost, or clinician-centered concerns regarding frailty, the presumed risk of falling, or anticipated nonadherence to therapy, could aid in the extraction of more robust associations had they been available. Third, our registry lacked prospective INR measurements of patients treated with VKAs. Thus, strict TTR calculation, as with the Rosendaal method, 20 was not feasible, meaning we were unable to assess the reliability of the SAMe-TT2R2 score’s projections. One should also acknowledge that this score is far less disseminated in clinical practice than CHA2DS2-Vasc and HAS-BLED. Nevertheless, it allowed us to gauge the quality of VKA regimens at the time of discharge, just as we did with NOAC regimens. Evaluation of the latter was carried out without methodology constraints, since all variables regarding categorization of dosing for AF were available. Finally, we aimed to overcome sample size restrictions and enhance extrapolation of our results. We did this by exploiting every bit of our data set with multiple imputations on missing values and by employing clinically reasonable multivariate and sensitivity analyses.
Conclusion
One of 6 AF patients newly discharged from the hospital may be treated discordantly to the corresponding risk of stroke, mainly due to undertreatment of high stroke risk AF. Half of VKA-treated patients were not expected to achieve good anticoagulation control as conveyed by the SAMe-TT2R2 score. Off-label dosing was relevant to more than a quarter of NOAC users, yet VKA regimens were used inappropriately at approximately twice the rate compared to NOAC regimens. Paroxysmal AF type, a prior stroke/TIA or major bleeding episode, antiplatelet use, and impaired renal function could be associated with suboptimal OAC use entities, such as not applying OAC to high-risk patients or NOAC underdosing.
Supplemental Material
Supplemental Material, Supplementary_File - Flaws in Anticoagulation Strategies in Patients With Atrial Fibrillation at Hospital Discharge
Supplemental Material, Supplementary_File for Flaws in Anticoagulation Strategies in Patients With Atrial Fibrillation at Hospital Discharge by Anastasios Kartas, Athanasios Samaras, Dimitra Vasdeki, George Dividis, George Fotos, Eleni Paschou, Evropi Forozidou, Paraskevi Tsoukra, Eleni Kotsi, Ioannis Goulas, George Efthimiadis, Haralambos Karvounis, Apostolos Tzikas and George Giannakoulas in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
Kartas, Tzikas, and Giannakoulas contributed to conception and design. Samaras, Vasdeki, Dividis, Fotos, Paschou, Forozidou, Tsoukra, Kotsi, Goulas, Efthimiadis, Karvounis, and Tzikas contributed to conception. Kartas and Giannakoulas contributed to acquisition, analysis, and interpretation. The authors Tsoukra, Kotsi, Goulas, Samaras, Vasdeki, Dividis, Fotos, Paschou, and Forozidou, contributed to acquisition. Efthimiadis, Karvounis, and Tzikas contributed to interpretation. All authors drafted manuscript critically, revised article and gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
