Abstract
This study evaluated the effect of telmisartan, ramipril, and amlodipine on atrial fibrillation (AF) recurrence and severity in hypertensive patients with metabolic syndrome. A total of 391 hypertensive outpatients with metabolic syndrome, in sinus rhythm but with at least 2 episodes of AF in the previous 6 months were randomized to telmisartan, ramipril, or amlodipine for 1 year. At the first AF, ventricular rate (VR) and plasma cardiac troponin I (TnI) were evaluated. P-wave dispersion (PWD) and procollagen type I carboxy-terminal peptide (PIP) were evaluated before and after 12 months of treatment. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were similarly and significantly reduced by all treatments (P < .001). In all, 49% of patients treated with amlodipine had a recurrence of AF as did 25.5% of patients with ramipril and 12.9% of patients with telmisartan (P < .01 vs amlodipine and P < .05 vs ramipril). Ventricular rate and TnI at the first AF recurrence were significantly lower with telmisartan and ramipril than with amlodipine. P-wave dispersion was reduced by ramipril (−5.1 ms, P < .05) and even more by telmisartan (−11 ms, P < .01). Telmisartan and ramipril induced a similar PIP reduction (−52.8 and −49.8 µg/L, respectively, P < .01). These findings suggested that in these patients telmisartan was more effective than ramipril in reducing AF recurrence and severity as well as in improving PWD, despite a similar BP reduction and a similar improvement in cardiac fibrosis. This could be related to a specific effect of telmisartan on atrial electric remodeling.
Introduction
Atrial fibrillation (AF), a cardiac arrhythmia which greatly increases the risk of stroke, coronary events, and total mortality,1,2 has been frequently observed in patients with metabolic syndrome.3,4 This condition is characterized by a cluster of cardiovascular risk factors, including hypertension, obesity, insulin resistance, and dyslipidemia,5,6 many of which are also risk factors for the development of AF.7,8 Inflammation and oxidative stress have also been implicated in the pathogenesis of both the metabolic syndrome and AF through their effect on atrial remodeling,9,10 which is the most significant factor underlying AF development and recurrence.11,12 Atrial remodeling is characterized by both anatomic changes in atrial structure (fibrosis, atrial dilatation) and electrical inhomogeneity (shortness of refractory period, dispersion of refractoriness, abnormal automatic, and anisotropic conduction).11–14 Activation of the renin–angiotensin system (RAS), which coexists with both hypertension and metabolic syndrome, is also thought to be involved in the pathophysiology of AF.15–17 Angiotensin II is known to modulate cardiac ionic currents and sympathetic tone and to stimulate fibroblast proliferation and cardiomyocte apoptosis.15–17 Furthermore, there is evidence for a role of angiotensin II (Ang II) in mediating inflammatory responses, which are additionally involved in AF.9,10 Genetic variation in the RAS and altered expression of Ang II receptors are also associated with AF, further indicating an important role in AF.18,19 Finally, a strong argument for a critical role of RAS in AF comes from both experimental and clinical trials, showing that RAS blockade may prevent new onset as well as recurrence of AF.20–27 Some recent meta-analysis from hypertension and heart failure clinical trials reported that both angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) were effective in the primary prevention of AF, although this benefit appeared to be limited to patients with systolic left ventricular dysfunction or left ventricular hypertrophy.21,22 Also there were some reports to suggest a role for RAS blocking in secondary prevention of AF.23–27 However, the recently published results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico-Atrial Fibrillation (GISSI-AF) trial failed to demonstrate a reduction in the incidence of recurrent AF with the ARB valsartan. 28 Besides, few and inconsistent findings exist about the comparative effects of ACEI and ARB on the incidence of AF.29,30
The aim of this study was to compare the effect of antihypertensive treatment with the ARB telmisartan, the ACEI ramipril, and the calcium channel blocker (CCB) amlodipine on the recurrence and the severity of AF in hypertensive patients with metabolic syndrome and a history of recent paroxysmal AF. The effects on P-wave dispersion (PWD) used as a marker of inhomogeneous atrial propagation of sinus impulses31,32 were also evaluated as were the effects on serum procollagen type I carboxy-terminal peptide (PIP) levels, used as a marker of extracellular collagen type I synthesis and myocardial fibrosis,33,34 and on carboxy-terminal telopeptide of collagen type I (CITP), used as a marker of extracellular collagen type I degradation.34–36
Patients and Methods
This was a prospective, randomized, double-blind, parallel-arm study. Between April 01, 2007, and November 30, 2009, the study population was selected according to the following inclusion criteria: male and female consecutive outpatients, with mild essential hypertension (systolic blood pressure [SBP] ≥140 and <160 mm Hg and/or diastolic blood pressure [DBP] ≥90 and <100 mm Hg) and metabolic syndrome (ATP III 2005 criteria),5,6 in sinus rhythm but with at least 2 electrocardiogram (ECG)-documented episodes of symptomatic AF in the previous 6 months and without any antiarrhythmic treatment. Previous AF episodes could be self-terminating or terminated after pharmacological and/or electrical cardioversion; the latter, however, had to be performed between a maximum of 6 months and a minimum of 8 weeks before enrollment. Patients with secondary hypertension, congestive heart failure, unstable angina, valvular disease, a left atrium size >46 mm, history of myocardial infarction or stroke, or cardiac surgery within 6 months prior to the study, cardioversion in the last 8 weeks, diabetes mellitus, hypercholesterolemia (total cholesterol ≥200 mg/dL), pregnancy, and any severe disease likely to interfere with the conduction of the study were excluded as were those previously treated with ARBs, ACEIs, CCBs, or antiarrhythmic agents and those with known hypersensitivity or contraindications to the study medications.
The local Ethics Committee approved the study protocol, and all patients gave their informed consent to participate in the study before enrollment. After a 2-week washout period during which antihypertensive drugs were discontinued and placebo was administered, patients who fulfilled the inclusion/exclusion criteria were randomized to telmisartan 80 mg once daily (od) or ramipril 5 mg od or amlodipine 5 mg od, according to a computer-generated randomization list. To maintain blindness, the study drugs were given in capsules of identical appearance (same color, size, and taste) stored in coded bottles. Patients were asked to take trial medications in the morning between 8.00 and 10.00
Patients were checked monthly for 1 year. At each visit, clinic BP values, a resting 12-lead surface ECG, and a 24-hour ECG were recorded. After a 10-minute resting period in a quiet room, sitting BP was measured on the right arm in each patient using a standard mercury sphygmomanometer (Korotkoff I and V) with a cuff of appropriate size. Measurements were always taken in the morning before daily drug intake (ie, 24 hours after dosing, at trough). An average of 3 successive BP readings taken at 1-minute interval was used for analyses. To identify asymptomatic AF episodes, 24-hour ambulatory ECG monitoring was performed every 4 weeks using a Syneflash Holter recorder (Ela Medical, Paris, France). Recordings were always started after drug intake and were performed throughout a 24-hour period, during which patients followed their normal daily routine after they left the laboratory. Patients were also required to report any episode of palpitations, to take their pulse and, in presence of arrhythmia, to reach as early as possible the nearest hospital to perform an ECG and a blood drawn for plasma cardiac troponin I (TnI) evaluation. Criteria to define AF recurrence included both nonsustained episodes of less than 3 minutes detected on the 24-hour Holter and the longer lasting sustained episodes detected on ECGs. Palpitations alone were not taken into consideration nor were patients’ subjective appraisals. At the first recurrence of AF, ventricular rate (VR) and plasma cardiac TnI were evaluated as indexes of AF severity.
At the end of the placebo period and of each treatment period, in patients who completed the 12-month study without any AF recurrence, PWD was evaluated and serum PIP and CITP levels were determined.
P-wave analyses measurements were calculated in 12-lead ECG recordings with a paper speed of 50 mm/s and a signal size of 10 mm/mV. Electrocardiogram recordings were transferred into a computer and opened with a high-performance graphic program. Manual measurements of P-wave duration were performed with digital calipers on a high-resolution computer screen by 2 cardiologists blinded to the patients’ clinical data. For each lead, 4 cycles were measured. The P-wave onset was defined as the first atrial deflection from the isoelectric line and the offset was the return of the atrial signal to baseline. Mean P duration was calculated as the mean value in each lead. The difference between maximum and minimum P duration was defined as PWD. Intra- and inter-observer variabilities were 3.3% and 3.8%, respectively, for P-wave duration and 2.9% and 3.6%, respectively, for PWD.
At the end of the placebo period and of each treatment period, blood samples for PIP and CIPT evaluation were taken in the morning, after an overnight fast, and stored at –40°C until determination. Serum PIP was determined by a rapid equilibrium radioimmunoassay, according to the method of Meikko et al 37 using commercial antisera specifically directed against the terminal carboxy-terminal peptide. The sensitivity (lower detection limit) was 1.1 µg of PIP/L. The intra- and inter-assay coefficients of variation were 4% and 7%, respectively. Serum CITP was also determined by a specific radioimmunoassay with the use of specific antisera (Orion Diagnostica, Espoo, Finland), according to the method of Risteli et al. 35 The sensitivity (lower detection limit) was 0.5 µg of CITP/L. The intra- and inter-assay variations were 6% and 8%, respectively.
Serum cardiac Tn-I was measured by a fluorimetric enzyme immunoassay using the Stratus CS system (Dade International, Derfield, IL). 38
The primary endpoint of the study was to assess the efficacy of telmisartan as compared to ramipril and amlodipine with regard to the cumulative number of patients relapsing into documented AF. Secondary endpoints were the time to a first recurrence of AF (as documented on an ECG), the severity of AF, as assessed in terms of VR and cardiac Tn-I levels at the first recurrence of AF, the changes in PWD, and the changes in PIP and CITP.
Statistical Analysis
Given an estimated efficacy at 1 year of 80% for telmisartan, 65% for ramipril, and 50% for amlodipine, a significance level of .05 and a test power of 0.80, the calculated sample size was of 91 patients for each treatment group. A 15% risk of patient loss at follow-up was assumed. Data for continuous variables are reported as means ± SD, while categorical variables were measured as frequencies. Statistical significance for baseline characteristics were examined using a Student t test for continuous variables. The Fisher exact test was used for categorical variables. The endpoint was analyzed on an intention-to-treat basis. The time to first AF recurrence was analyzed using the Kaplan-Meier method and compared with the log-rank test. The comparison of the number of days to AF relapse (median and range) among the treatment groups was performed by the nonparametric Wilcoxon test.
Results
A total of 456 consecutive hypertensive patients, 198 untreated and 258 previously treated for hypertension, were referred to our hypertension center with a history of paroxysmal AF. Of them, 391 were finally randomized to participate in this study (Figure 1 ). A total of 47 patients were excluded from this protocol because they did not meet the inclusion/exclusion criteria. Eighteen patients refused to participate. As shown in Table 1 , the 3 treatment groups were well matched with regard to baseline demographic and clinical characteristics. In all, 30 patients in the telmisartan group, 29 patients in the ramipril group, and 25 patients in the amlodipine group underwent electrical cardioversion before entering the study, whereas pharmacological cardioversion was performed in 65 patients in the telmisartan group, 64 patients in the ramipril group, and 66 patients in the amlodipine group.
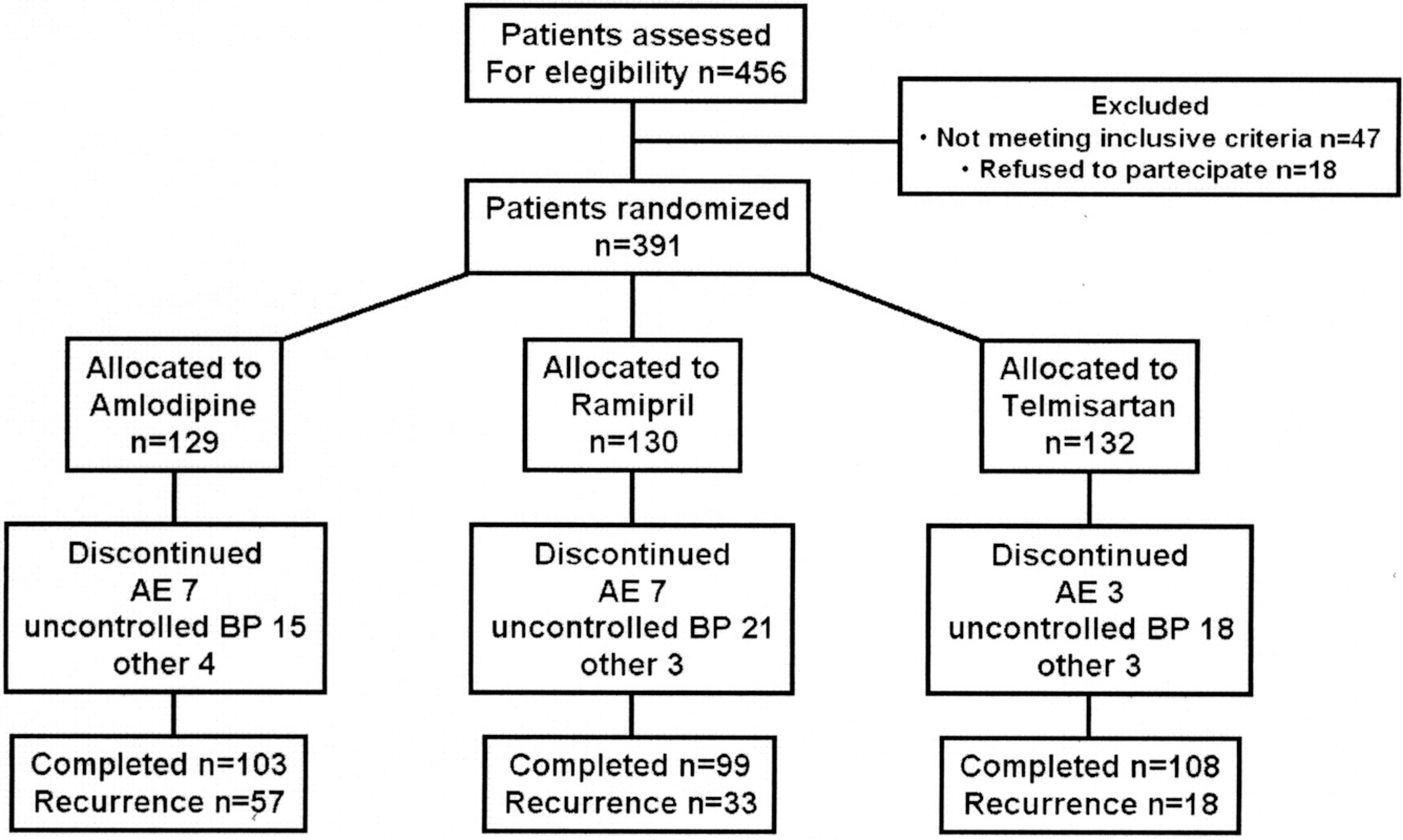
Flow diagram of the study.
Main Demographic, Clinical, and Echographic Characteristics of Patients in the 3 Treatment Groups

NOTE: AF = atrial fibrillation; DBP = diastolic blood pressure; EDLV = end-diastolic left ventricular; eGFR = estimated glomerular filtration rate; FPG = fasting plasma glucose; HDL-C = high density lipoprotein-cholesterol; HR = heart rate; ns = not significant; SBP = systolic blood pressure; TC = total cholesterol; Tg = triglycerides.
A total of 132 patients were randomized to treatment with telmisartan, 130 to treatment with ramipril, and 129 to treatment with amlodipine. Systolic blood pressure and DBP values were significantly reduced in the 3 treatment groups. At the end of follow-up, SBP decreased by 15.9 mm Hg (P < .001 vs baseline) in the telmisartan group, by 15.6 mm Hg in the ramipril group (P < .001 vs baseline), and by 16.8 mm Hg in the amlodipine group (P < .001 vs baseline), with no significant difference among treatments. Corresponding changes for DBP were 12.5, 12.3, and 13.1 mm Hg (P < .001 vs baseline), respectively, again without any significant difference among treatments. The heart rate (HR) did not show any significant change from baseline in any treatment group.
The AF recurrence data are shown in Table 2 . At the end of titration period, 35 patients had a recurrence of AF: by intention-to-treat analysis, the occurrence rate was significantly lower in the telmisartan group (6 patients) than in the amlodipine group (18 patients). Kaplan-Meyer analysis demonstrated a 12-week probability of 96% for maintaining sinus rhythm in patients who received telmisartan compared with 92% in patients who received ramipril and 86% in patients who received amlodipine (P = .02).
Results: Intention-to-Treat Analysis

a P < .05.
b P < .01 versus amlodipine.
c P < .05 versus ramipril.
At the end of the follow-up (median 258 days [range 29-360]), 57 (44.2%) amlodipine-treated patients had a recurrence of AF, as did 33 (25.5%) ramipril-treated patients (P < .01 vs amlodipine) and 17 (12.9%) telmisartan-treated patients (P < .01 vs amlodipine and P < .05 vs ramipril). The Holter-detected AF episodes were 3 in the amlodipine group and 1 in the ramipril group; the others were all symptomatic ECG-detected AF episodes. Of the 57 recurrent episodes of AF in the amlodipine group, 27 were paroxysmal and 29 were persistent; while in the ramipril group, 16 AF recurrences were paroxysmal and 17 persistent; and in the telmisartan group, 15 AF relapses were paroxysmal and 2 persistent. Figure 2 shows the Kaplan-Meyer AF recurrence-free survival analysis which demonstrated a significant reduction in AF recurrence in the telmisartan group (P < .001 log-rank test) as well as in the ramipril group (P < .01 log-rank test) when compared to the amlodipine group and also in the telmisartan group (P < .05 log-rank test) when compared to the ramipril group.
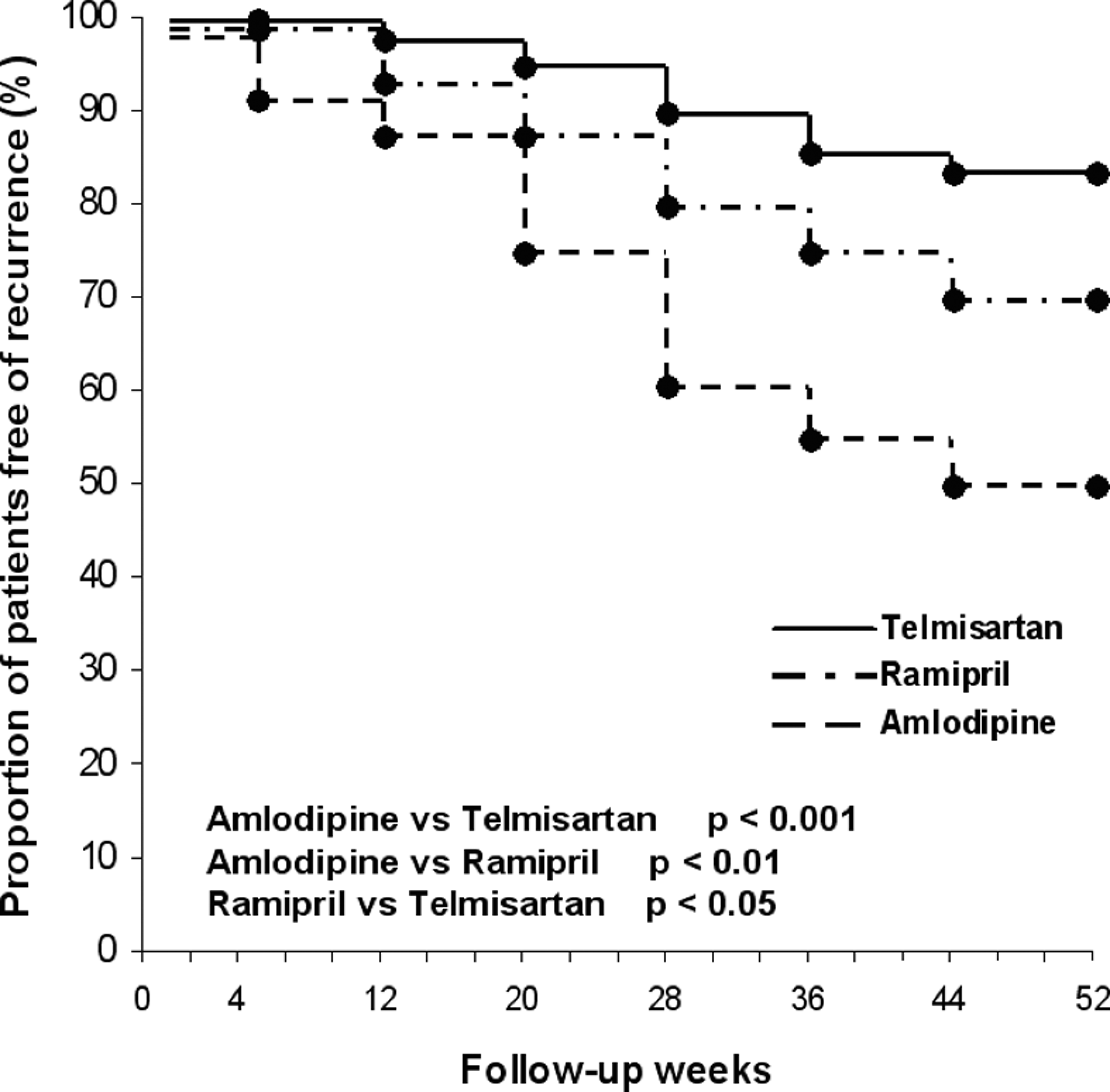
Recurrence of atrial fibrillation in the 3 study groups.
As regard the indexes of AF severity, VR at the first recurrence of AF was significantly lower in the telmisartan group (P < .05) as well as in the ramipril group (P < .05) when compared to the amlodipine group (Table 3 ). No significant difference was observed between the telmisartan and the ramipril group, with regard to this parameter. Serum cardiac TnI levels at the first recurrence of AF were significantly lower in the telmisartan group (P < .01) as well as in the ramipril group (P < .05) when compared to the amlodipine group, and also in the telmisartan group (P < .05) when compared to the ramipril group (Table 3).
Ventricular Rate and Plasma Cardiac Troponin I at the First Recurrence of Atrial Fibrillation

a P < .05.
b P < .01 versus amlodipine.
c P < 0.05 versus ramipril.
The PWD values did not show any significant change in the amlodipine group while a significant reduction was observed in the telmisartan (−11 ms, P < .05 vs placebo and P < .01 vs amlodipine) as well as in the ramipril group (−5.1 ms, P < .05 vs placebo and vs amlodipine; Table 4 ). The reduction, however, was significantly greater in the telmisartan group when compared with the ramipril group (P < .05; Figure 3 ).
Comparison of P-Wave Dispersion and Serum PIP and CITP Values Among Groups Before and After Treatment

NOTE: CITP = carboxy-terminal telopeptide of collagen type I; PIP = procollagen type I carboxy-terminal peptide; PWD = P-wave dispersion.
a P < .05.
b P < .01 versus placebo.
c P < .05
d P < .05
e P < .05 versus amlodipine.
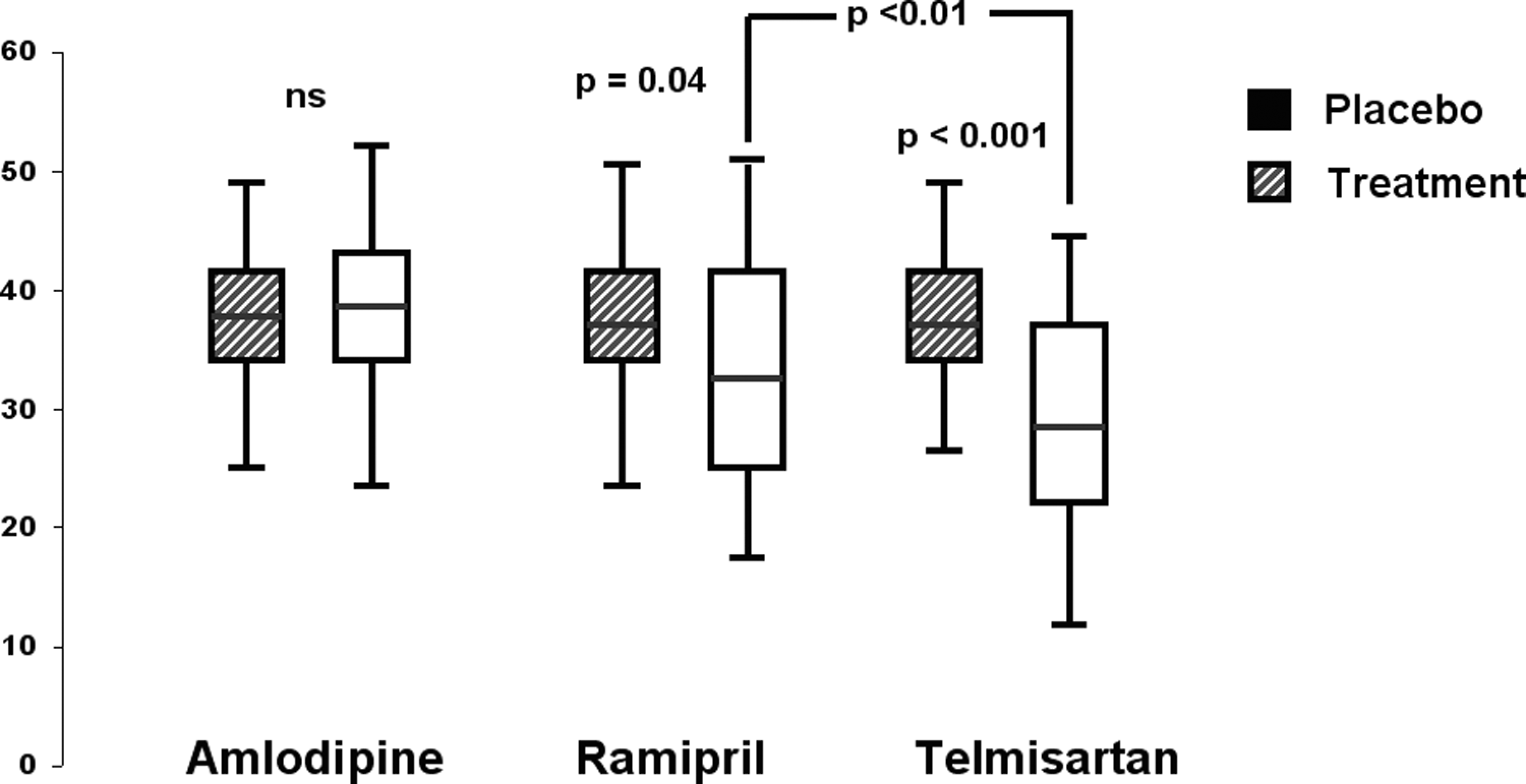
P-wave dispersion values before and after 1 year antihypertensive treatment (72 patients in the amlodipine group, 97 in the ramipril group, and 115 patients in the telmisartan group.)
As shown in Table 4, serum PIP concentration was significantly reduced (P < .001 vs placebo and P < .01 vs amlodipine), while CITP concentration was significantly increased (P < .05 vs placebo and vs amlodipine) after 12 months of treatment both in the telmisartan- and the ramipril-treated patients, with no difference between the 2 groups. No change in PIP and CITP levels has been observed in the amlodipine group.
In all, 3 patients in the telmisartan group, 7 patients in the ramipril group, and 7 patients in the amlodipine group complained of adverse events requiring treatment discontinuation. In the telmisartan group, 1 patient was removed from the trial because of hypotension, 1 of headache, and 1 of nausea. In the ramipril group, 1 patient had an atrial flutter and underwent radiofrequency ablation, 1 complained of glottis edema, and 4 discontinued because of an intolerable dry cough. In the amlodipine group, 2 patients had an atrial flutter and underwent radiofrequency ablation and 5 did not tolerate ankle edema.
Discussion
In the present study, we observed that in hypertensive patients with metabolic syndrome and a history of recurrent AF, antihypertensive therapy with both ARB telmisartan and ACEI ramipril was more effective than amlodipine therapy in reducing AF recurrence, but the preventive effect of telmisartan on AF relapse and severity was greater than that of ramipril.
At the end of the 1-year follow-up, at least 1 ECG-documented episode of AF was observed in 12.9% of patients treated with telmisartan, in 25.5% of patients treated with ramipril, and in 44.2% of patients treated with amlodipine, with a Kaplan-Meyer estimated probability for maintaining sinus rhythm of 87% in the telmisartan group vs 74% in the ramipril group vs 56% in the amlodipine group.
As regard the severity of AF episodes, both HR and plasma levels of TnI at the first recurrence of AF were significantly lower in the telmisartan- and the ramipril-treated patients as compared to the amlodipine-treated ones. However, the telmisartan group showed significantly lower values of serum TnI as compared with the ramipril group. Although, generally speaking, elevated TnI levels are consistent with the diagnosis of acute coronary syndrome, abnormal TnI levels have been also reported after supraventricular tachycardia without evidence of coronary artery disease, pointing to minor myocardial injury.39–41 Hypothesized mechanism for such troponin elevation include the shortening of diastole with subsequent subendocardial ischemia, myocardial stretch, impairment of microvascular blood flow, and coronary vasospasm.39–41 Whatever the mechanism, the changes in myocyte membrane permeability resulting from myocardial injury could be enough for the release of cardiac TnI from the free cytosolic pool of myocytes without structural damage. The lesser increase in cardiac TnI observed in the telmisartan-treated patients during AF recurrence suggests a greater protective effect of the ARB against the arrhythmia-related myocardial injury.
While in some previous studies, treatment with ARBs or ACEIs showed an additive effect over that obtained from standard antiarrhythmic therapy in the prevention of AF relapse in hypertensive patients,23,24,27 in the present study patients on antiarrhythmic drug therapy were excluded so that the AF-preventive effect of these drugs per se was evaluated. Although a decrease in BP could play a role in the benefit observed with both inhibitors of the RAS, in the present study no significant difference in BP values was found among the 3 treatment groups. This suggests that both telmisartan and ramipril may exert an antiarrhythmic effect beyond their hypotensive action.42–44 Hypothesized mechanisms for the antiarrhythmic effects of ACEIs and ARBs in AF include (a) interference with ion-channel function, in particular K+ channel subunits and Ca2+ ion currents17,45,46; (b) modulation of refractoriness, mainly by preventing the shortening of atrial effective refractory period (AERP) and preserving AERP rate adaptation 47 ; (c) inhibition of Ang-II-mediated fibrosis48,49; (d) reduction of left atrial dilatation and overload 50 ; (e) regression of left ventricular hypertrophy 51 ; and (f) modulation of sympathetic tone.
In the present study, telmisartan and ramipril, but not amlodipine, significantly reduced PWD. This finding, which is in agreement with some previous observations by ourselves and other authors,29,52 may play an important role in the prevention of AF recurrence by these drugs. Prolongation of PWD indicates nonhomogeneous conduction in the left atrium and has been demonstrated to be an independent predictor for AF.31,32 Therefore, drugs that decrease PWD are likely to reduce AF incidence and recurrence. We found that PWD values in the telmisartan-treated patients were significantly lower than those in the ramipril-treated ones. Reasons for such a difference between the ARB and the ACEI, which resembles what observed in previous studies,29,53 are not clear. One hypothesized explanation is related to the different effect of the 2 drugs on the chimase-mediated Ang II-forming activity, which seems to be higher in the left atrium than in other cardiac chambers. Unlike ARBs, which are effective on both non-ACE and ACE-dependent Ang II pathways, ACEIs are not able to inhibit Ang II activity produced by chimase in the left atrium. Since increased chimase activity may contribute to nonhomogeneous atrial conduction, ARBs may have a greater effect on atrial electrophysiological properties than ACEIs and result in lower PWD values. 53 A peculiar effect of telmisartan on atrial electrical remodeling might also play a role. At plasma concentration levels of therapeutic doses, telmisartan has been shown to block hKv1.5 potassium channels, which are highly expressed in human atria and conduct ultrarapid delayed rectifier currents (Ikur) that contribute to action potential repolarization of human atrial myocytes. 52 Since the inhibition of Kv1.5 selectively prolong atrial but not ventricular action potential duration, Kv1.5 is considered an important molecular target for the treatment of AF.
A rise in fibrillar collagen deposition, which leads to increased interstitial and perivascular fibrosis, increases myocardial stiffness and promotes abnormalities of cardiac function, whereas its regression normalizes stiffness and function. 54 Atrial fibrosis is known to be involved in AF inducibility and maintenance, mainly through decrease in the atrial conduction velocity and heterogeneity of the conduction tissue.12,14,55 Although microscopic examination of cardiac biopsies is the most reliable method for documenting and measuring myocardial fibrosis, the use of cardiac biopsies is an invasive methodology not useful for wide-scale application. Therefore, noninvasive monitoring of myocardial fibrosis using serological markers of collagen turnover has been introduced into clinical practice. In particular, serum concentrations of PIP and CITP may be useful for assessing the synthesis and degradation, respectively, of collagen type I fibers and, although not heart-specific, they may provide indirect diagnostic information on both the extent of myocardial fibrosis and the ability of antihypertensive treatment to reduce myocardial fibrosis.33–36 In this study, both telmisartan and ramipril significantly decreased the serum concentrations of PIP and increased CITP levels, with no difference between the 2 drugs. This finding on one hand confirms previous observations showing the ability of both ACEIs and ARB to reduce the synthesis and to stimulate the degradation of collagen type I fibers,48,49 thus resulting in the reduction of myocardial fibrosis. On the other hand, it suggests that the positive effect of both telmisartan and ramipril on AF recurrence might be at least partly related to reduction in atrial fibrosis.
It is of interest that telmisartan has been found to stimulate peroxisome proliferator–activated receptor-γ (PPAR-γ) activity, independent of its Ang II type 1 receptor blocking activity.5,56 Proliferator-activated receptor-γ activity, beside influencing the carbohydrate and lipid metabolism, is involved in inflammatory and proliferative processes, and there is a growing body of evidence that activators of PPAR-γ improve insulin resistance and exert anti-inflammatory, antioxidative, and antiproliferative effects. 57 In particular, pioglitazone, a PPAR-γ activator, has been demonstrated to attenuate congestive heart failure–induced atrial remodeling and AF promotion in rabbits. 58 Insulin resistance with consequent hyperinsulinemia is considered the fundamental pathophysiological disturbance responsible for the metabolic syndrome and greatly contributes to the increased risk of AF development in this condition. 59 Due to its unique PPAR-γ modulating activity, telmisartan might be superior to ACEIs and other ARBs in preventing the development of AF in insulin-resistant patients, like those with the metabolic syndrome, through an insulin-sensitizing effect and the suppression of insulin-mediated atrial remodeling. 60
Results from the present study are not in agreement with those of the ONTARGET study 30 in which the incidence of AF did not significantly differ between the ramipril- (6.9%) and the telmisartan- (6.7%) treated patients. Possible reasons for such a discrepancy may be that (a) ONTARGET was a primary prevention study, whereas our trial was a secondary prevention study; (b) in the ONTARGET ramipril was always used at the dosage of 10 mg od in all patients, whereas in our study ramipril was used at dosages ranging from 5 to 10 mg od, according to the pressor response of the patients; (c) concomitant drug therapy, which was allowed in the ONTARGET but not in our study, might also have affected the results.
Concomitant drug therapy might also explain at least in part the negative results of another primary prevention study, the TRANSCEND, 61 in which telmisartan treatment did not reduce the rate of new AF (6.4%) when compared with placebo (6.3%). In the TRANSCEND study, almost 60% of the patients were receiving a β-blocker at baseline, which is likely to have attenuated additional effects of telmisartan on AF development.
Substantial differences in the characteristics of the study populations might explain the different results of our study as compared to the recently published GISSI-AF study, 28 a secondary prevention trial that failed to demonstrate the efficacy of the ARB valsartan in preventing AF recurrence. Whereas our study included only patients with hypertension and metabolic syndrome, the GISSI-AF study, besides hypertensives, included also patients with heart failure or left ventricular dysfunction, type 2 diabetes mellitus, history of stroke, and coronary artery disease or peripheral artery disease, that is, patients with more severe clinical conditions. Besides, according to the inclusion criteria, the atrial size of our patients (≤46 mm) was smaller than that of the GISSI-AF patients (>45 mm),which indicates a more preserved atrial structure in our patients. Thus, in our study a more precocious ARB administration in patients with less atrial enlargement and possibly less degree of anatomical remodeling in atrial tissues could have resulted in more effective prevention of AF recurrence. Furthermore, unlike our patients, who did not undergo cardioversion for AF within the last 8 weeks and were not receiving antiarrthythmic drugs nor RAS-blocking agents at the time of enrollment, the majority of the GISSI-AF population had undergone electrical or pharmacological cardioversion for AF within 2 weeks before randomization (thus being less electrically stable) and are receiving established therapies for the prevention of AF. In particular, 74% of the patients were receiving an antiarrhythmic drug, 58% an ACE-I, 27% a statin, and 30% a β-blocker, which are all likely to attenuate possible beneficial effect of ARB on AF. Additionally, the analysis of the baseline characteristics of the GISSI-AF patients according to study group showed that the proportion of patients with coronary artery disease was significantly higher in the valsartan group (15.4%) than in the placebo group (9.4%, P < .001) as was the proportion of patients with peripheral artery disease (5.1% vs 3.1%, P = .0047) and with pathologic Q-waves (5.7% vs 3.1%, P = .02). In other words, patients in the valsartan group were more complicated and prone to AF development than patients in the placebo group. Finally, different pharmacologic characteristic of the ARB used might have played a role in the different results obtained in our study as compared to the GISSI-AF. Telmisartan has been demonstrated to have the strongest binding affinity to Ang II type 1 receptors among various ARBs, the rank order of affinity being telmisartan > olmesartan > candesartan > valsartan > losartan. 62 Due to its higher type 1 receptor blocking ability and long plasma half-life, telmisartan might be superior to other ARBs in preventing the development of AF.
Conclusion
In hypertensive patients with metabolic syndrome and a history of paroxysmal/persistent AF telmisartan was more effective than ramipril in reducing AF recurrence and severity as well as in improving PWD despite a similar BP reduction and a similar improvement in cardiac fibrosis markers. These data suggest that telmisartans antiarrhythmia effect might be related not only to the atrial structural remodeling (similar to that induced by ramipril) but also to a specific and selective electric remodeling by improving atrial conduction disturbances.
Footnotes
This work was done at the Department of Internal Medicine and Therapeutics, University of Pavia.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
