Abstract
Remote ischemic preconditioning (RIPC) is an innovative treatment strategy that alleviates ischemia-reperfusion injury, whereby short episodes of regional ischemia and reperfusion delivered to remote organs including hind limb, kidney and intestine, and so on provide protection to the heart. The RIPC is known to reduce infarct size, serum levels of cardiac enzymes, and myocardial dysfunction in various animal species as well as in patients. There have been a large number of studies suggesting that the ATP-sensitive potassium channels (KATP channel) play a significant role as a mediator or end effector in RIPC. The present review discusses the role of KATP channels and possible mechanisms in RIPC-induced cardioprotection.
Introduction
Remote ischemic preconditioning (RIPC) has received enormous recognition for its ability to mitigate ischemia reperfusion-induced injury in various organs such as liver, kidney, brain, skeletal muscle, and heart. 1 –5 The RIPC is a treatment approach whereby regional ischemia and reperfusion episodes to remote tissues provide protection to the target organ against ischemia-reperfusion injury. Przyklenk et al documented that application of 4 episodes of 5 minutes ischemia-reperfusion to the circumflex artery significant reduced the infarct size in the left anterior descending coronary artery perfused region of dogs. The authors named the phenomenon as remote “intraorgan preconditioning.” 6 In later years, different scientists described that application of preconditioning stimuli to the noncardiac tissues such as kidney, liver, hind limb, and intestine also protect myocardium from the detrimental effects of ischemia-reperfusion and this approach has emerged as “interorgan protection.” 7 –12 Although, the biphasic effects of RIPC have been indicated, 13 yet these have not been rigorously investigated as in ischemic preconditioning. 14 However, studies have established that remote preconditioning confers cardioprotection in biphasic manner similar to ischemic preconditioning. It induces early phase of protection that persists up to 2 to 4 hours 13,15 and the second window of protection reoccurs at 24 hours after the remote preconditioning stimulus. 16,17
The clinical applications of remote preconditioning have been expanded due to noninvasive application of preconditioning stimulus in form of upper limb ischemia using a blood pressure cuff. Clinical studies have indicated a reduction in serum levels of cardiac enzymes (troponin I, troponin T, and creatine kinase [CK]) with RIPC in patients undergoing cardiac and major vascular surgery. 18,19 Furthermore, meta-analysis of 17 clinical trials in patients undergoing cardiovascular interventions indicated that patients receiving RIPC had lower release of markers of myocardial injury (troponin I, troponin T, and CK) and reduced incidence of perioperative myocardial infarction. 20 Meanwhile, meta-analysis of 10 studies with a total of 693 participants revealed that RIPC possibly results in cardiac protection (in terms of reduced troponin release) during cardiac surgery. However, the effects of RIPC were significantly higher in studies without full blinding in comparison to fully blinded studies. 21 On the contrary, studies have shown that although RIPC is effective in reducing biomarkers of cardiac injury, yet the long-term benefits in terms of reduction in mortality have not observed. A meta-analysis of 9 clinical studies with 482 patients indicated that RIPC significantly reduces troponin levels, but did not reduce mortality and perioperative myocardial infarction in the patients undergoing cardiovascular surgery. 22 Another meta-analysis of 46 clinical trials with 1878 patients indicated that RIPC reduces the incidence of perioperative myocardial infarction and troponin release, but failed to reduce mortality and other major adverse cardiovascular events in the patients undergoing cardiovascular interventions. 23 Accordingly, more double-blind, randomized controlled trials are required to ascertain the long-term utility of RIPC in reducing mortality and other major cardiovascular events during cardiac surgery.
An ATP-sensitive K+ channel (KATP) is a member of ATP-binding cassette transporter superfamily and was first identified in the sarcolemma of cardiac myocytes by Noma et al. 24 Apart from heart, these channels are present on different organs including pancreatic β-cells, 25 skeletal muscles, 26 brain, 27 smooth muscles, 28 kidney, 29 and pituitary. 30 This channel is believed to play a role in physiological as well as pathological processes such as maintaining the vascular smooth muscle tone, 31 hormonal secretion, 32 stress adaptation, 33,34 skeletal muscle fatigue, 35 neuronal degeneration, 36 and regulation of neuronal excitability. 37
In the myocardium, 2 subtypes of KATP channels are present, one on the sarcolemma (sarcolemmal KATP) and another on the inner membrane of the mitochondria (mitochondrial KATP) 24 and are involved in the regulation of physiological and pathophysiological processes in the heart. Different scientists have employed pharmacological modulators of KATP channels to demonstrate their involvement in ischemic preconditioning against ischemia-reperfusion injury. 38,39 Furthermore, researchers have also explored the significant involvement of activation of mitochondrial KATP channels during ischemic postconditioning in providing cardioprotective effects against ischemia-reperfusion injury. 40,41 Besides, a number of studies have shown that RIPC protects the myocardium from ischemic injury via opening of KATP channels. 42 –44 The rationale behind this review is that till now, there has not been an integrative review indicating the involvement of KATP channels in RIPC-induced cardioprotection. The literature survey was done using various keywords like ischemia, RIPC, KATP channels, and heart to find out the relevant studies. The present review describes the key role of KATP channels in RIPC-induced tissue protection with possible mechanisms.
ATP-Sensitive Potassium Channels in Remote Preconditioning
There have been a number of studies documenting the key role of KATP channels in RIPC-induced protection in different tissues (Table 1). Hu et al described that RIPC improves ejection fraction, myocardial performance index, neurological deficit score, and survival duration in hemorrhagic shock model of rats. However, the cardioprotective and neuroprotective effects were abolished in the presence of glibenclamide (nonselective KATP channel inhibitor) suggesting the possible involvement of KATP channels. 45 A recent study of Noorbakhsh et al showed that blockade of KATP channels by sodium 5-hydroxydecanoate (5-HD, mito KATP channels blocker) and glibenclamide suppress the ability of remote liver preconditioning in attenuating ischemia reperfusion-induced arrhythmia in isolated rat hearts. 46 An earlier study of Pell et al reported a reduction in myocardial infarct size and preservation of blood pressure in in vivo rabbit model of acute myocardial infarction in response to remote renal preconditioning. Furthermore, they found that the anti-infarct effects of renal preconditioning were abolished by a specific mitochondrial KATP channels blocker, 5-HD, and 8-(p-sulfophenyl)-theophylline, 8-SPT (nonselective adenosine receptor blocker). It suggests the potential role of adenosine and mitochondrial KATP channels in renal remote preconditioning-induced cardioprotection. 42 Rehni et al documented that 15 minutes occlusion-reperfusion of mesenteric artery protects the brain from ischemia-reperfusion injury in terms of improvement in motor coordination, short-term memory, and reduction in infarct size. These protective effects of RIPC were blocked by caffeine (adenosine blocker) and glibenclamide indicating the neuroprotective effects of RIPC are mediated via activation of adenosine receptors and subsequently KATP channel opening. 3 Sato et al reported that in intact rabbit ventricular myocytes, mitochondrial KATP channels are activated in the presence of adenosine. 47 Based on these, it may be hypothesized that during RIPC adenosine triggers the opening of mito KATP channels which may in turn impart cardioprotection. 42
Preclinical Studies Indicating the Involvement of KATP Channels in RIPC-Induced Tissue Protection.

Abbreviations: CK, creatine kinase; 5-HD, 5-hydroxydecanoate; KATP, ATP-sensitive potassium; LDH, lactate dehydrogenase; NF-κB, nuclear factor-kappaB; NO, nitric oxide; RIPC, remote ischemic preconditioning; ROS, reactive oxygen species.
Kristiansen et al have documented that 4 cycles of limb preconditioning contribute to cardioprotection in the explanted heart by virtue of activation of mitochondrial KATP channels. The RIPC and diazoxide (KATP channel opener) imparted cardioprotection in similar magnitude in terms of reduction in infarct size and improvement in left ventricular performance. Administration of glibenclamide and 5-HD attenuated the cardioprotective effect of RIPC and diazoxide suggesting that mitochondrial KATP channels mediate RIPC-induced cardioprotection. 48 Furthermore, Konstantinov et al observed that RIPC stimulus (4 cycles of hind limb preconditioning) in the recipient decreases ischemia-reperfusion injury in the donor heart following orthotopic heart transplantation. The reduction in infarct size was abrogated by pretreatment with glibenclamide, which showed that the protection afforded by RIPC is dependent on opening of KATP channels. 43 Moses et al described the principal role of sarcolemmal KATP as well as mitochondrial KATP channels in early and delayed remote preconditioning both as a trigger and mediator, respectively. The anti-infarct effects of remote hind limb preconditioning or sarcolemmal KATP channel opener P-1075 in latissimus dorsi muscle flaps were abolished by glibenclamide or HMR-1098 (sarcolemmal KATP channel blocker), administered 10 minutes before remote preconditioning, suggesting that sarcolemmal KATP channels act as trigger during RIPC. It was also found that treatment with 5-HD or glibenclamide 24 hours after RIPC also inhibited the infarct limiting effect of late RIPC, which suggests that mitochondrial KATP channels may also act as mediator. Furthermore, the infarct-sparing effect afforded by a mitochondrial channels opener BMS-191095 in muscle flaps was accomplished by increased content of ATP (during sustained ischemia) and reduced neutrophil accumulation (during reperfusion) as in the remote hind limb preconditioning. It suggests that the opening of KATP channels may preserve the ATP content and decrease the inflammatory process during ischemia-reperfusion injury. 4
The findings of our laboratory showed that remote preconditioning and erythropoietin preconditioning elicit cardioprotection in a similar fashion via activation of KATP channels. Four episodes of brief renal artery occlusion (5 minutes) resulted in decrease in infarct size, improvement in cardiac function and decrease in the levels of lactate dehydrogenase (LDH) and CK in coronary effluent. The beneficial effects of renal and erythropoietin preconditioning were blocked by administration of a selective nuclear factor-kappaB (NF-κB) inhibitor, diethyldithiocarbamic acid (150 mg/kg intraperitoneal [ip]), and glibenclamide (5 mg/kg ip). Furthermore, treatment of minoxidil (1 mg/kg intravenous) in diethyldithiocarbamic acid pretreated rats ameliorated cardioprotective effects of remote preconditioning and erythropoietin preconditioning. Therefore, it was concluded that KATP channels are downstream mediator of NF-κB activation and contribute to cardioprotection during remote preconditioning and erythropoietin preconditioning. 49 The involvement of NF-κB as an upstream mediator is further substantiated by a previous study, whereby administration of NF-κB blocker Diethyldithiocarbamate (DDTC) significantly inhibited the pharmacological preconditioning effects of diazoxide in the isolated mice hearts. 50
Wang and coworkers reported that RIPC via occluding the superior mesenteric artery for 25 minutes followed by 15 minutes reperfusion led to significant reduction in the myocardial infarct. However, intestinal preconditioning-induced cardioprotective effects were significantly abolished in the presence of 5-HD indicating the involvement of mitochondrial KATP channels in mediating cardioprotection.
51
Shahid et al explained that short episodes of occlusion-reperfusion of femoral arteries increase nitric oxide (NO) levels, open mitochondrial KATP channels, and generate reactive oxygen species (ROS). Administration of 5-HD,
Wu et al reported that 3 cycles of 5 minutes occlusion-reperfusion for 3 days exerted protective effects in the myocardium (reduced infarction and arrhythmia) that were similar to ischemic preconditioning. Remote preconditioning also reinforced the activities of manganese superoxide dismutase (Mn-SOD), superoxide dismutase (SOD), and glutathione peroxidase, diminished the xanthine oxidase activity and malondialdehyde content (MDA) in the myocardium. The protective effects were abolished by administration of 5-HD suggesting the involvement of mitochondrial KATP channels as a mediator/effector during delayed remote preconditioning. Furthermore, the improvement in the myocardial antioxidative ability has also been linked to the activation of mitochondrial KATP channels. 57 The report of this study is consistent with other studies showing the elevation in the antioxidant enzyme expression in the heart was abolished in the presence of 5-HD. 58 Interestingly, it is known that the opening of mitochondrial KATP induces ROS generation to provide cardioprotection. 52 On the other hand, the opening of mitochondrial KATP channel has also been shown to decrease the ROS generation during sustained ischemia by altering antioxidant balance. 59 It has been well documented that transient increase in free radical generation during preconditioning phase may trigger the synthesis or increase the activity of antioxidant enzymes, which subsequently decrease the deleterious effects of ROS during sustained ischemia-reperfusion injury. 60 Therefore, it can be assumed that the opening of mitochondrial KATP triggers the ROS production or arrests the ROS burst, is dependent upon the phase of preconditioning, or ischemia/reperfusion. It may be proposed that the opening of KATP channels during preconditioning phase may transiently increase ROS which in turn may increase the antioxidant enzyme activity during sustained ischemia-reperfusion. Mehrjerdi et al demonstrated that short episodes of renal ischemia followed by reperfusion causes significant neuroprotection against ischemia-reperfusion of hippocampal tissue in the brain of mice. It was found that the mechanism of endogenous protection was dependent on the opening of mitochondrial and sarcolemmal KATP channels and an increase in the antioxidative ability of hippocampus as evidenced by suppression of MDA level and improvement in the catalase activity. However, pretreatment with glibenclamide and 5-HD in the preconditioned animals reversed RIPC-induced improvement during the hippocampal injury. It further supports the above contention that KATP channel activation is linked with decrease in ROS generation and increase in antioxidant defense mechanism to provide tissue protection. 44
Taliyan et al illustrated the activation of α1 adrenergic receptor and subsequently, the opening of KATP channels accounts for cardioprotection during remote aortic preconditioning (RAPC). Four episodes of aortic preconditioning decreased the release of CK and LDH and reduced the infarct size. However, prazosin, a α1 adrenergic receptor blocker, diminished RAPC-induced cardioprotection. Furthermore, phenylephrine (20 mg/kg ip), as α1 adrenergic receptor agonist, mimicked the cardioprotective effects of RAPC. The cardioprotective effects of phenylephrine were nullified in the presence of glibenclamide or prazosin. Therefore, it may be suggested that activation of α1 adrenergic receptor induces the opening of KATP channels, which may be responsible for RAPC-induced cardioprotection. 10 This is in corroboration with another study indicating that phenylephrine administration reduces the myocardial infarct size and improves the functional recovery of heart via activation of KATP channels. 61 Moreover, Yadav et al demonstrated that pretreatment with glibenclamide and celecoxib (a selective COX-2 inhibitor) prior to RAPC mitigated the cardioprotective effects. It may be proposed that RAPC triggers the activation of COX-2, which in turn activates the mitochondrial KATP channels to induce cardioprotection. 62 The involvement of COX is further supported by a previous study indicating that pharmacological preconditioning with PGE1 or prostaglandins PGEo led to significant reduction in the infarct size and these effects were abolished in the presence of KATP channel blocker. 63 In addition to ROPC, it has been shown that activation of mitochondrial KATP channels is involved in remote preconditioning of trauma induced cardioprotection in the mice and rats. 64,65
Discussion
There have been a number of evidences that implicate the pivotal role of KATP channels in mediating RIPC-induced tissue protection in various species. 42,43,48,57,64 The scientists have mainly employed pharmacological antagonists of KATP channels such as nonselective KATP channel blocker, glibenclamide, mitochondrial KATP channel blocker, 5-HD and sarcolemmal KATP channel blocker, and HMR-1098 to delineate the role of KATP channels in RIPC-induced tissue protective effects. 42,43,48 The myocardium possesses 2 subtypes of KATP channels: mitochondrial KATP and sarcolemmal KATP channels. 24 The involvement of mitochondrial KATP channels in mediating RIPC-induced cardioprotection has been verified in both isolated hearts 10,46,48,49,62 and in vivo animal models. 3,42,43,52,57 Furthermore, the involvement of mitochondrial KATP channels in mediating RIPC-induced neuroprotection has also been confirmed. 44,45 However, the activation of sarcolemmal KATP channels in case of the skeletal muscles has been implicated in reducing the infarct size in the latissimus dorsi muscle flaps. 4
The investigation of Konstantinov et al in the heart transplant model showing abolishment of cardioprotective effect of RIPC in the recipient animal in the presence of 5-HD or glibenclamide clearly indicates the significant role of humoral mechanism in KATP channel activation during RIPC. 43 Furthermore, KATP channels may be involved in first window of protection (early preconditioning, which lasts up to 2 hours) as a trigger and in second window of protection (delayed preconditioning, which lasts up to 24 to 72 hours) as a mediator. 3,10,49 Although, individual studies have established the linkage of KATP channels with other mediators such as adenosine, NO, norepinephrine, COX-2, NF-κB, and ROS, yet there are no studies describing the unifying mechanism of involvement of these mediators. There have been number of studies suggesting that an increase in levels of adenosine, NO, norepinephrine, COX-2, and NF-κB during the preconditioning phase may induce tissue protection by different mechanisms including decrease in inflammation and increase in antioxidant enzymes during ischemic phase (Figure 1). 66 –68

Transient ischemia during RIPC enhances adenosine discharge and activates adenosine A2 receptors, which in turn increases NO generation and subsequently opens mitochondrial KATP channels. Augmented adenosine levels may also increase nor-epinephrine release which may also activate mitochondrial KATP channels. Furthermore, increase in NO level may activate cyclooxygenase enzyme to increase the formation of prostaglandins which in turn also activates the mitochondrial KATP channels. Increased NO may also activate NF-κB during RIPC to decrease inflammation during index ischemia. Opening of mitochondrial KATP channels during RIPC increases the formation of free radicals during the preconditioning phase which in turn activates antioxidant enzymes during exposure to prolonged ischemia to decrease the inflammatory response in the heart. KATP indicates ATP-sensitive potassium; NF-κB, nuclear factor-kappaB; NO, nitric oxide; RIPC, remote ischemic preconditioning.
The report of Shahid et al describes that during RIPC, NO may trigger the opening of KATP channels, which may be followed by transient increase in ROS. 52 The redox signaling may induce tissue protection by increasing the levels of endogenous antioxidant enzymes. 60 The reports of Pell et al and Rehni et al have shown the linkage between KATP channels and adenosine and it has been tentatively proposed that adenosine may trigger the activation of KATP channels. 3,42 The literature of ischemic preconditioning suggests that adenosine discharge during ischemic preconditioning may activate adenosine A2 receptors, which in turn enhance NO generation to provide tissue protective effects. 69 Furthermore, there have been reports that pharmacological preconditioning with adenosine increases iNOS expression 70 and prevents downregulation of eNOS to provide cardioprotective effects. 71 Based on the above reports suggesting the interrelationship between adenosine, KATP channels, NO, and free radicals in RIPC, it may be stated that an increase in adenosine levels during RIPC may increase the levels of NO, which may activate KATP channels to trigger redox signaling cascade to produce tissue protection.
The interrelationship between KATP channels with COX, NE, and NF-κB in RIPC 10,49,62 may also be ascribed secondary to adenosine and nitric oxide. Preconditioning-dependent increase in adenosine release may activate presynaptic adenosine receptors, which may subsequently increase norepinephrine discharge to provide cardioprotective effects. 72 Meanwhile, upregulation of iNOS and subsequent NO generation may also enhance COX activity during ischemic preconditioning to provide cardioprotection. 66 Moreover, an upsurge in NO level induces transcriptional factors including NF-κB to induce cardioprotection (Figure 2). 73,74 It may be proposed that during RIPC the opening of KATP channels is an important step and is interlinked with other mediators and effectors. The precise relationship of KATP channels with other mediators in RIPC needs further investigations.
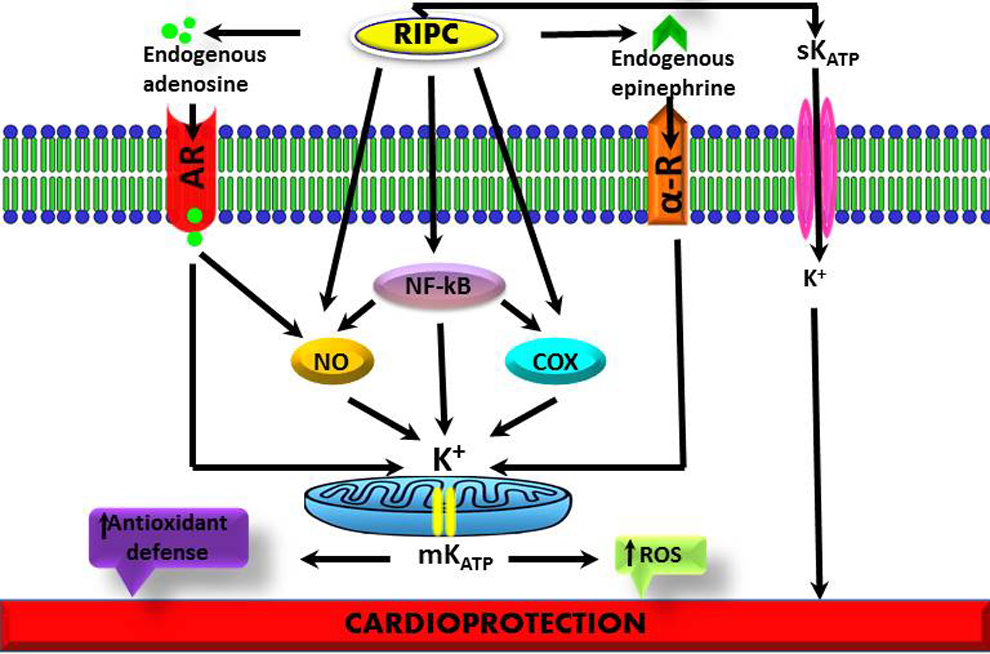
Schematic diagram showing the possible mechanism(s) involved in ATP-sensitive potassium channel-mediated RIPC. RIPC indicates remote ischemic preconditioning.
Conclusion
The preclinical studies clearly show that activation of KATP channels is involved in both early and delayed phases of RIPC. The pathways involved in producing these protective effects may include NO, adenosine, NF-κB, COX, epinephrine, and ROS.
Footnotes
Acknowledgments
The authors are thankful to Department of Science and Technology F.No. SB/SO/HS/0004/2013, New Delhi for their gratefulness for providing us financial assistance and Department of Pharmaceutical Sciences and Drug Research, Punjabi University, Patiala, India for supporting us.
Author contributions
S. Aggarwal contributed to acquisition, drafted the manuscript, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. P. Randhawa contributed to conception, design, and acquisition. N. Singh contributed to conception and critically revised the manuscript. A. Jaggi critically revised the manuscript and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Department of Science and Technology F.No. SB/SO/HS/0004/2013, New Delhi.
