Abstract
Cardiac steroids (CSs), such as ouabain and digoxin, increase the force of contraction of heart muscle and are used for the treatment of congestive heart failure (CHF). However, their small therapeutic window limits their use. It is well established that Na+, K+-ATPase inhibition mediates CS-induced increase in heart contractility. Recently, the involvement of intracellular signal transduction was implicated in this effect. The aim of the present study was to test the hypothesis that combined treatment with ouabain and Akt inhibitor (MK-2206) augments ouabain-induced inotropy in mammalian models. We demonstrate that the combined treatment led to an ouabain-induced increase in contractility at concentrations at which ouabain alone was ineffective. This was shown in 3 experimental systems: neonatal primary rat cardiomyocytes, a Langendorff preparation, and an in vivo myocardial infarction induced by left anterior descending coronary artery (LAD) ligation. Furthermore, cell viability experiments revealed that this treatment protected primary cardiomyocytes from MK-2206 toxicity and in vivo reduced the size of scar tissue 10 days post-LAD ligation. We propose that Akt activity imposes a constant inhibitory force on muscle contraction, which is attenuated by low concentrations of MK-2206, resulting in potentiation of the ouabain effect. This demonstration of the increase in the CS effect advocates the development of the combined treatment in CHF.
Introduction
Cardiac steroids (CSs), such as ouabain and digoxin, increase the force of contraction of heart muscle and are used in the clinic for the treatment of heart failure. Their negative chronotropic effect is further exploited for heart rate control in atrial arrhythmias. 1 The application of CS is limited owing to their extremely small therapeutic window; whereas about 1 nM digoxin is considered beneficial, significant signs of toxicity are observed already at 3 nM. 2 Ouabain, which in the past was frequently used in the clinic, 3 also has an extremely narrow therapeutic window. 4 The comprehensive DIG study showed that digoxin did not reduce overall mortality, but rather the rate of hospitalization. 5 Accordingly, it was recently reported that the use of digoxin is associated with an increased risk of death from any cause in patients with atrial fibrillation. 6 Similarly, it was found that digoxin treatment in chronic heart failure was associated with a slightly increased risk of death. 7 In contradiction to the above, recent studies have shown that digoxin use is beneficial and not associated with increased mortality. 8 Hence, although the beneficial effect of CS in a clinical setting is still debatable, the significance of increasing the therapeutic window of these steroids is evident.
The recognized mechanism of CS action involves the inhibition of the plasma membrane transporter Na+, K+-ATPase, resulting in an increase of intracellular Na+, which in turn attenuates the activity of the Na+/Ca2+ exchanger, leading to an increase in intracellular Ca2+ concentration and consequently greater contractility. 9 In the last decade, it was established that in addition to pumping ions, Na+, K+-ATPase participates in intracellular signaling processes. 10,11 The interaction of CS with Na+, K+-ATPase leads to the activation of intracellular signaling cascades that are involved in the regulation of gene expression, cell viability, differentiation, and smooth muscle and heart muscle contractility. 12 –15 One of the cascades stimulated by CS is the PI3K1A/PDK/Akt pathway, which plays a role in neonatal cellular proliferation, hypertrophy, heart remodeling, energy production, and contractility. 16 It was further demonstrated that ouabain-induced heart hypertrophy in mice depends on PI3K1A/PDK/Akt pathway activity. 10,17 We recently tested the involvement of intracellular signaling in CS-induced positive inotropy, in vivo and ex vivo, in a zebra fish model. 15 This study demonstrated that Akt inhibition by MK-2206 enhances the CS-induced increase in heart contractility, suggesting that this protein plays a negative regulatory role in CS effects.
Validation of these results in a mammalian experimental system and consequently in human would suggest that the coadministration of CS with Akt inhibitor will lead to a reduction in the effective dose of CS and hence an expansion in their therapeutic window. In the present study, we assessed the beneficiary effect of combined treatment of ouabain and Akt inhibitor MK-2206 on ouabain-induced increase in heart contractility in 3 established experimental models, primary cardiomyocytes, the Langendorff preparation, and myocardial infarction (MI) in the rat driven by the LAD ligation.
Methods
Preparation of Neonatal Rat Primary Cardiomyocytes
A Pierce Primary Cardiomyocyte Isolation Kit (Thermo Scientific, Rockford, Illinois) was used to prepare primary cardiomyocytes from 1- to 2-day-old neonatal male Wistar rats. The rats were anesthetized with isoflurane and the hearts were dissected in cold Hank balanced salt solution (HBSS) and washed twice. The harvested hearts were diced into pieces of about 1 to 3 mm3 and incubated for 30 minutes at 37°C in 0.2 mL of a mixture of isolation enzyme solution. Following 2 washes with cold HBSS, 5 × 105 cells/well were cultured in a sterile 24-well plate (Thermo Scientific) containing 0.5 mL Dulbecco modified Eagle medium supplemented with 10% heat-inactivated FBS (Gibco, Fort Worth, Texas) and 1% penicillin/streptomycin in 5% CO2 at 37°C. Following 24-hour incubation, the medium was removed and fresh medium containing 1:1000 “growth supplement solution” was added. The medium was replaced every 72 hours as previously described. 18,19 The Hebrew University Animal Care Committee approved the use of the animals and the experimental protocols used in this study (Approval #MD-13-13861-4).
Cell Viability
Cardiomyocytes were exposed to different concentrations of ouabain (1 nM-1 mM) or MK-2206 (0.1-100 nM) or combinations of the two for 3 days. Cell viability was determined with the conventional 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The MTT was dissolved in PBS at 5 mg·mL−1. A 25 µL volume of this solution was added to each well, and the plates were incubated at 37°C for 30 minutes. The assay was terminated by the addition of 100 µL/well of an aqueous solution of 20% wt/vol sodium dodecyl sulfate, 50% vol/vol N, N,-dimethylformamide, and incubation at 37°C for 1 hour. Absorbance was measured with an enzyme-linked immunosorbent assay plate reader (BioTek Instruments, Winooski, Vermont) at 570 nm to quantify the amount of formazan product, which reflects the number of viable cells in the culture. 20,21 Each experiment was repeated 4 to 7 times on different cardiomyocytes preparation from 10, 1-day-old neonatal rats.
Primary Cardiomyocyte Contractility Filming
Cell culture imaging was performed with an Olympus CKX41 (Tokyo, Japan) upright microscope (×20 magnification) and integrated incandescent illumination. A FastCam IMI tech (Gyeonggi-do, Korea) high-speed digital camera with a 640 × 480 pixel gray scale image sensor was mounted on the microscope with ImCam software (IMI Technology, Co, Ltd, Gangnam-gu, Seoul, South Korea) for high-speed video recording at 80 fps during 10 seconds with a shutter speed of 0.016 seconds. Six cell clusters from each well were filmed for 10 seconds. Each experiment was repeated 4 times on different cardiomyocytes preparation from 10, 1-day-old neonatal rats.
Relative Quantification of Primary Cardiomyocyte Contractility
Image analysis application ImageJ (NIH, Bethesda, Meryland) was used, allowing delineation of the primary cell culture area at the end of contraction or at the end of relaxation periods. Sequential still frames were analyzed to capture end-contraction and end-relaxation images. The area and intensity differences between end-contraction frame and end-relaxation frame were measured. According to a nonoverlapping algorithm, the nonoverlapping area values were normalized with the overall primary cell culture area at the chosen frames. The resulted value was considered a quantitative parameter reflecting the force of primary cardiomyocytes culture contraction. In addition, we assumed that the increase in force of contraction is also reflected by changes in the intensity of light transition through the cells (integrated intensity). The intensity of the overlapping area at the end of contraction is increased due to area opacification, serving an indirect measure of the force of contraction. Six different cell clusters from each well containing a tested compound were quantified.
Langendorff Preparations
Conventional Langendorff isolated heart preparations perfused at constant flow were set up as previously described (Pierre et al, 2007, PMID: 17157283; Belliard et al, 2016, PMID: 27702882). Male Sprague Dawley rats (320-350 g, strain code 001, 5 animals per group) were anesthetized by intraperitoneal injection of sodium pentobarbital (80 mg·kg−1). Hearts were excised and rapidly placed into ice-cold Krebs-Henseleit (KH) buffer. Within 40 seconds, hearts were perfused in the Langendorff mode with oxygenated KH buffer containing (in mmol·L−1) NaCl (118.0), KCl (4.0), CaCl2 (1.8), KH2PO4 (1.3), MgSO4 (1.2), ethylene glycol bis (2-aminoethylether)-N, N, N′, N′-tetraacetic acid (0.3), NaHCO3 (25), and D-glucose. 11 Hearts were paced at 4.5 Hz throughout the experiment. The isovolumic left ventricular developed pressure (LVDP) was measured through a water-filled latex balloon inserted into the left ventricle (LV) and analyzed using Lab Chart software (ADInstruments, Oxford, UK). Following 5 to 10 minutes of stabilization, each heart was perfused for 15 minutes with the control buffer followed by 15 minutes with or without MK2206 (5 nM) before ouabain (1 µM) was added for another 15 minutes. To determine ouabain effects, the control perfusion buffer was switched to the same buffer containing the indicated ouabain concentrations. The change in LVDP was calculated relative to baseline conditions (control buffer or buffer containing MK-2206).
Left Anterior Descending Coronary Artery Ligation
Male Wistar rats (150-175 g) were used, and LAD ligation was performed in all animals as previously described. 22 The rats were anesthetized with 10% ketamine and 2% xylazine (0.1 mL·kg−1) and ventilated with a small animal respirator (Harvard Inc, Holliston, Massachusetts). The hearts were exposed via left sternotomy, and the LAD ligation was induced by placing a 6-0 silk suture around the left anterior descending coronary artery near the atrial auricle. The animals were allowed to recover and treated with subcutaneous injection (SC), immediately and 24 hours post-LAD ligation, with 5 mg·kg−1 tramadol hydrochloride. The rats were divided into 4 groups (5-8 animals/group) that were treated daily with SC injection of saline (0.5 mL·kg−1), ouabain (0.8-8 mg·kg−1), MK-2206 (12 mg·kg−1), or a combination of MK-2206 and ouabain. All injected solutions contained 9% dimethyl sulfoxide. Heart contractility in vivo was monitored with echocardiography as described below. At the end of the treatment period, the animals were killed, hearts harvested, and blood samples were drawn for analysis. Blood samples were also collected (via the orbital plexus) at baseline and 24 hours post MI. Blood samples were centrifuged and serum stored at −20°C for future measurements. Troponin-T (Trop-T) and creatine phosphokinase (CPK) were determined at the Clinical Biochemistry Department of the Hadassah Hospital, using the IMMULITE 2500 immunoassay system (Siemens Healthcare Diagnostics, Tarrytown, New York), based on validated immunoassays.
Echocardiographic Measurements
Echocardiography assessments were performed by an experienced echocardiographer, blinded to group assignment. All rats underwent baseline echocardiography assessment, 24 hours before the LAD ligation and at 1, 3, 6, and 10 days post MI. The rats were anesthetized with 2% isoflurane, and measurements were made with a commercially available ultrasound system (GE Vivid 33 equipped with a 13.3 MHz transducer; General Electric—Vingmed Technology, Horton, Norway). Images were acquired in the left parasternal short-axis view of the LV at the level of the papillary muscles in 2-dimensional mode (B-Mode) and motion mode (M-Mode). Left ventricle function was assessed in accordance with the American Society of Echocardiography definitions. 23,24 In M-Mode, 3 consecutive measurements of the internal diameter of the LV at the end of diastole and at the end of systole were obtained, averaged, and then used to calculate fractional shortening (FS), according to the equation:
where LVIDd denotes left ventricular internal diameter at diastole and LVIDs denotes left ventricular internal diameter at systole.
Similarly, using B-Mode acquisitions, 3 repeated measurements of the LV area at the papillary muscles level at the end of diastole and at the end of systole were measured, averaged, and used to calculate the fractional area change (%FAC) using the equation:
where LVAd denotes left ventricular area at diastole and LVAs denotes left ventricular area at systole. 24
Masson Trichrome Staining
Masson trichrome staining was used to evaluate the fibrosis as expressed by the presence of collagen fibers (stained blue) as a parameter of scar tissue present in LV myocardium. Heart muscles were placed in 4% paraformaldehyde for 72 hours. Paraffin-embedded sections of 5-µm thickness were made from the ligation area to the apex. Tissue sections were deparaffinized, rehydrated with graded alcohols, immersed in Bouin solution, and incubated overnight at room temperature. Staining was performed with the aid of a commercial kit (Sigma-Aldrich, HT15, St Louis, Missouri). After washing with running tap water for 5 minutes, the nuclei in the heart sections were stained (black) with Weigert hematoxylin for 5 minutes, washed in running tap water for 5 minutes, and rinsed with deionized water for 5 minutes. Heart muscle was stained (red) by incubation with Biebrich Scarlet-Acid-Fuchsin for 5 minutes following a rinse with deionized water for 5 minutes and immersion in phosphomolybdic–phosphotungstic acid for 5 minutes. Subsequently, collagen was stained (blue) by incubation in Aniline Blue for 5 minutes and rinsed in 2% acetic acid for 2 minutes. Finally, the tissues were rehydrated with ethanol and mounted with VectaMount (Vector Laboratories, Burlingame, California). The slides were viewed with a Nikon TL microscope Diaphot 200, (Agentek, Tel-Aviv, Israel), photographed, and the total section area of the myocardium in the tissue sections was measured with ImageJ software. Three selected sections were quantified for each animal. After determining the area of each heart myocardium and the fibrosis region, the relative area of the fibrotic tissues was calculated.
Western Blotting
Protein extraction and separation on SDS-PAGE electrophoresis, followed by transfer to a polyvinylidene fluoride membrane were carried out as previously described. 25 The membranes were incubated for 1 hour at room temperature with specific antibodies against Phospho-Akt (Ser473; 193H12) and rabbit mAb #4058 (Cell Signaling, Ornat Biochemicals, Rehovot, Israel), at a 1:1000 dilution in Tris-buffered saline (TBS) containing 0.1% Tween. The wash with TBS containing 0.1% Tween and exposure to horseradish peroxidase–conjugated secondary goat antirabbit IgG antibody (1:50 000) as well as membrane stripping before exposure to a different antibody were performed as previously described. 25 Detection was carried out with the aid of a Luminata Crescendo Western HRP substrate (Darmstadt, Germany) according to the manufacturer’s instructions. Preliminary experiments verified that the stripping and reblotting procedures did not affect the quantification of any of the proteins.
Statistics
All data are expressed as the mean ± SEM. Data were analyzed using SPSS 24.0 software (IBM, Inc, Armonk, New York). Statistical differences in Figures 1, 2, and 5 to 7 were performed using Student t test. P < .05 was considered statistically significant. Since some of the experiment included 4 treatment groups (control, ouabain, MK-2206, and mix), and each rat was measured on 4 indicators Fractional Area Change (FAC), Ejection Fraction (EF), Shortening Fraction (SF), and Heart Rate (HR) across 4 time points (24 hours, 3, 6, and 10 days post LAD ligation), a mixed within-(time) between-(group) doubly repeated (indicator) multivariate analysis of variance was used to test significance (in Figures 3 and 4). The analysis partitions the overall variance across all indicators in the sample to variance that is due to treatment effects (belonging to 1 of the 4 treatment groups), variance that is due to the passage of time (signifying recovery over time), and variance that is due to possible interactions between time and treatment effects (indicating differential recovery between groups). The focus of our analysis is the interaction effects. The conservative Bonferroni adjustments were used to control for potential type I error due to multiple comparisons (between treatments groups, between time points, and with their interactions) in the analysis.

Effect of ouabain, MK-2206, and combination of the two on rat cardiomyocyte contractility. Primary rat cardiomyocytes were prepared as described in Materials and Methods. Spontaneously contracting cells were exposed to ouabain, MK-2206, or a combination thereof for 1 hour at 37°C. The cells were photographed for 5 seconds and the change in cell contraction was deduced from the difference in area change and light density between relaxation and the contraction peak. Six cell clusters in 3 wells were photographed and measured for each experimental condition. Quantification of motility is displayed as the cell area change (A and C) and the integrated intensity (pixel intensity/area). B and D, Dependence of contractility on ouabain doses is depicted in A and B, and the combined effect of ouabain and MK-2206 (MIX) is depicted in C and D. Results are expressed as means ± SEM from 4 independent experiments. *Significantly higher than control, P < .05. #Significantly higher than ouabain-treated group, P < .05.
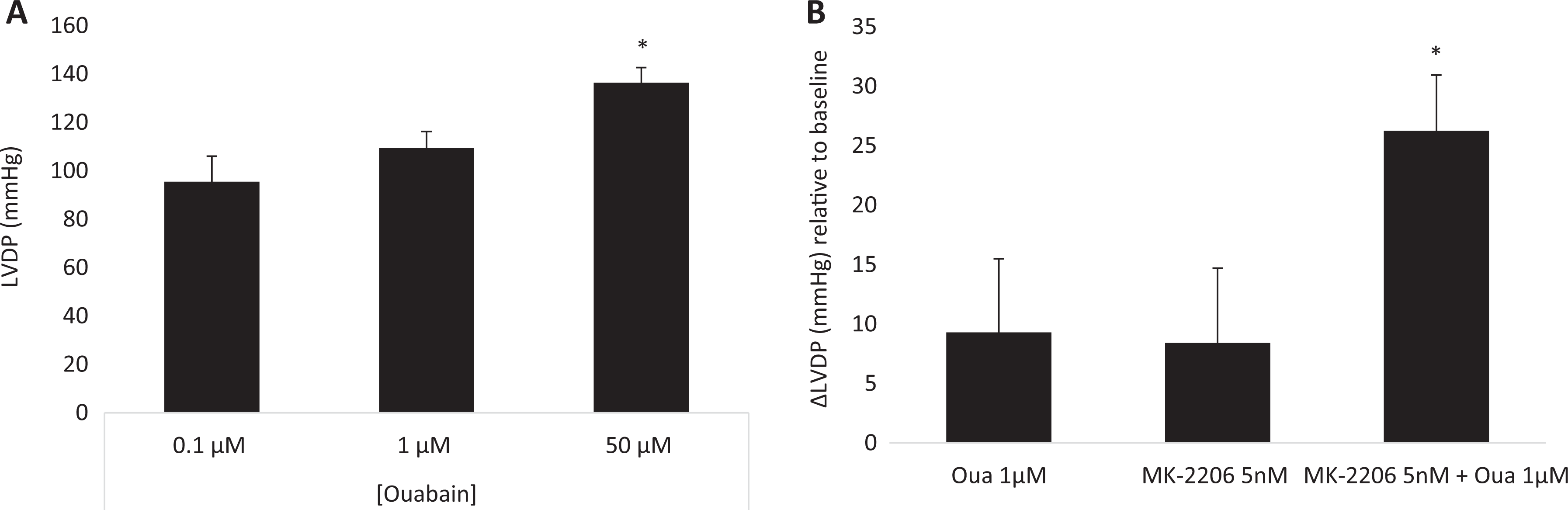
Ouabain-induced increase in left ventricular pressure in Langendorff-perfused rat heart in the presence and absence of MK-2206. A, Hearts were perfused with Krebs-Henseleit buffer for 15 minutes followed by 15 minutes of ouabain at the indicated concentration. B, The ouabain group (Oua) was perfused with Krebs-Henseleit buffer for 15 minutes followed by 15 minutes of ouabain 1 µM. The MK-2206 + Oua group was perfused with MK-2206 5 nM for 15 minutes followed by 15 minutes of perfusion with MK-2206 5 nM + ouabain 1 µM. Results are expressed as means ± SEM from 4 to 7 independent experiments for each group. *P < .05 versus ouabain alone.
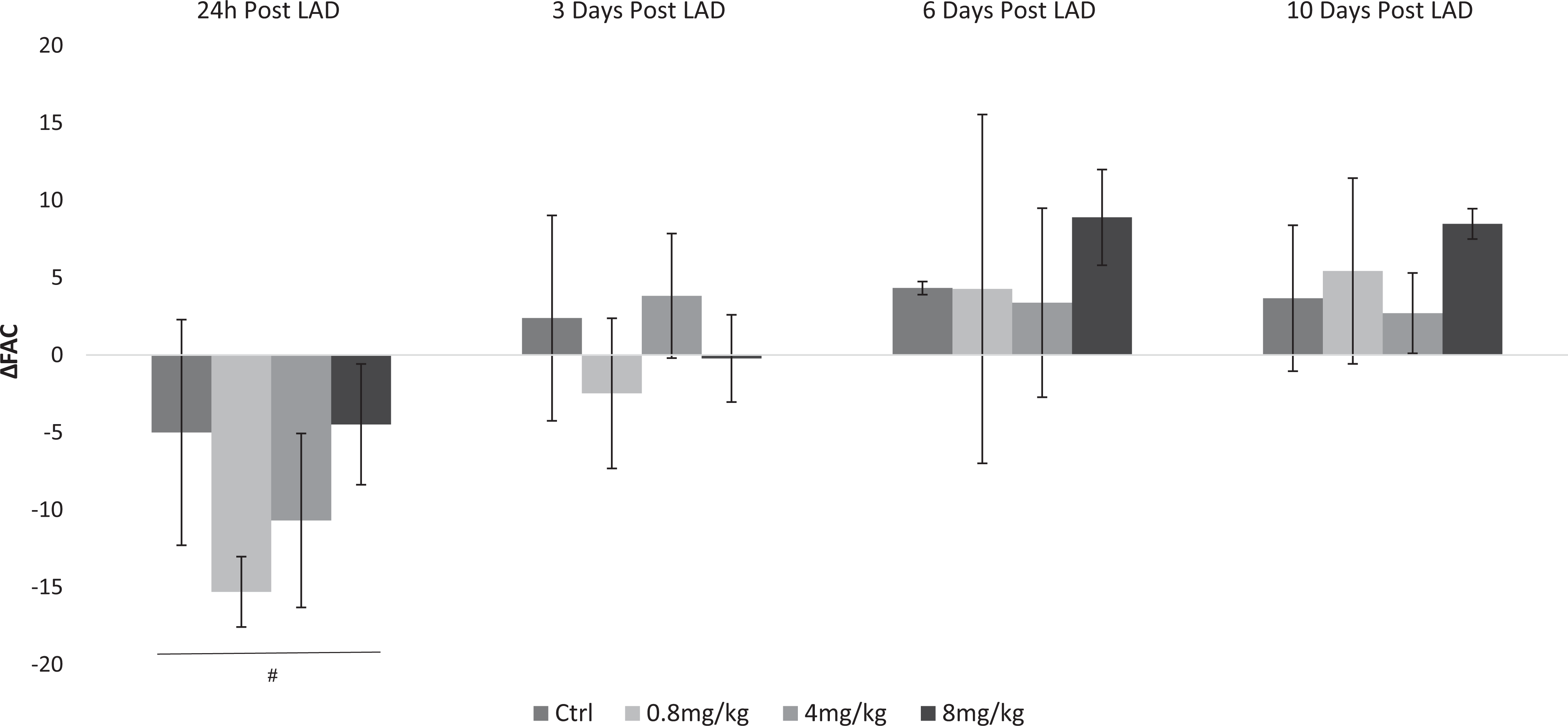
Effect of ouabain on force of contraction following myocardial infarction (MI) in rats. Left anterior descending coronary artery (LAD) ligation was performed in male Wistar rats as described in Materials and Methods. The control group (Ctrl) received an intraperitoneal (IP) injection of saline (0.5 mL·kg−1·d−1). Ouabain 0.8, 4, and 8 mg/kg/d were injected every 24 hours. Heart contractility in vivo was monitored with echocardiography at baseline, 24 hours, and 3, 6, and 10 days post MI. The difference between the fractional area change (ΔFAC) post MI during tested time period at each experimental condition is depicted. Results are expressed as means ± SEM from 6 animals for each experimental group. #Significantly lower than baseline (P < .05).
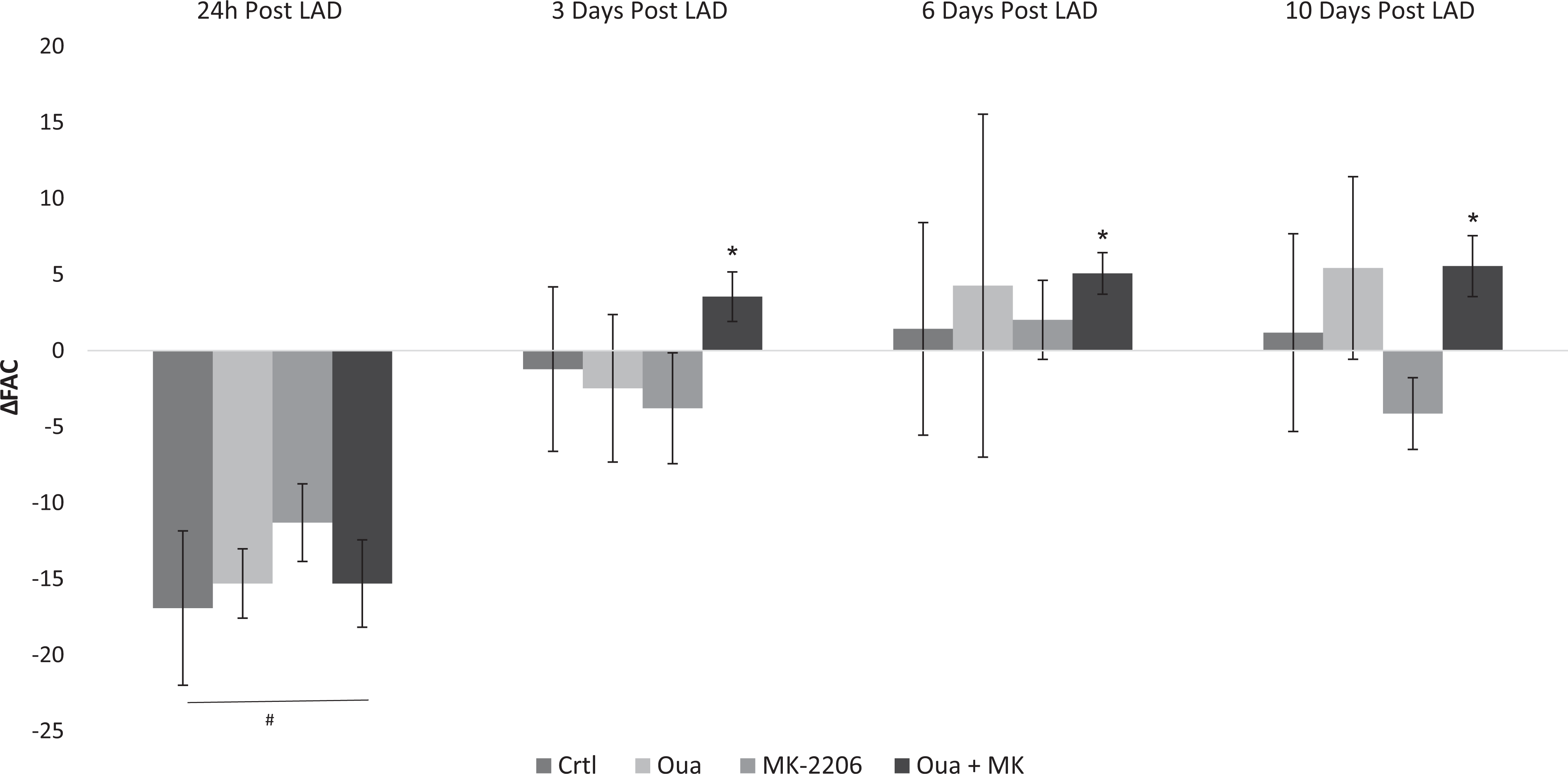
Effect of a combination of MK-2206 and ouabain on force of contraction following myocardial infarction (MI) in rats. Left anterior descending coronary artery (LAD) ligation was performed in male Wistar rats, as described in Martials and Methods. The control group (Ctrl) received an IP injection of saline (0.5 mL·kg−1·d−1), ouabain (0.8-8 mg·kg−1·d−1), MK-2206 (12 mg·kg−1·d−1, MK-2206), or a combination of MK-2206 and ouabain (Oua + MK). Echocardiography was used to monitor heart contractility in vivo. Differences between the fractional area change (ΔFAC) before LAD ligation and the experimental conditions are depicted. Results are expressed as means ± SEM from 2 independent experiments of 5 to 7 animals in each. #Significantly lower than baseline level (P < .05). *Significantly higher than 24 hours. Post LAD ligation level (P < .05).

Effect of a combination of MK-2206 and ouabain on scar tissue formation. Masson Trichrome staining was performed on rat’s heart as described in Martials and Methods. Representative Masson trichrome staining of rat cardiac tissue 10 days post myocardial infarction (MI) fibrosis and quantification of fibrosis by calculating mean percentage fibrotic area. Means ± SE were obtained from measurements in at least 7 hearts in each group. *Significantly lower than baseline (P < .05).

Effect of ouabain, MK-2206, and combination thereof on Akt phosphorylation and Na+, K+-ATPase α1 and α2 levels. Akt phosphorylation and Na+, K+-ATPase α1 and α2 isoforms levels were quantified by Western blot analysis as described in Materials and Methods. The effects of ouabain, MK-2206, and combination of the two on primary cardiomyocytes and rats heart were performed as described in the legend of Figures 1 and 4, respectively. The effects of the drugs on phospho-Akt in primary cardiomyocytes (1 hour following exposure to the drugs) are depicted in (A). Heart tissue distal to the ligated area from rats treated with saline, ouabain, MK-2206, and a combination thereof for 10 days were dissected and proteins were extracted as described in Materials and Methods. Western blot analysis was used to detect Akt phosphorylation (B) and Na+, K+-ATPase α1 and α2 isoforms (C and D, respectively). Band intensity was normalized to that of total Akt or S6 protein or α tubulin. Each bar represents the mean ± SE of 3 experiments. *Significantly higher than control, P < .05.

Effect of ouabain, MK-2206, and a combination of the two on primary rat cardiomyocyte viability. Primary cardiomyocytes were cultured as described in Materials and Methods. The cells were incubated for 3 days in medium containing 0.1 to 100 nM MK-2206, 1 nM to 1 mM ouabain, or a combination of 1 nM MK-2206 and 1 nM to 1 µM ouabain. Cell viability was assayed after 3 days according to the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) viability test. Each bar represents the mean ± SE of 3 experiments. *Significantly lower than control, P < .05.
Results
MK-2206 Potentiates Ouabain-Induced Inotropy in Rat Primary Cardiomyocytes
Our previous studies on the potentiation of CS-induced increase in heart contractility by Akt inhibition were performed on zebra fish. In the present study, we tested this phenomenon in rat heart experimental models. First, we studied primary neonatal rat cardiomyocytes.
Eight-day-old primary cardiomyocytes were exposed to Akt inhibitor, MK-2206 (0.1 nM), and ouabain (0.1-100 nM) for 1 hour. As can be seen at Figure 1, MK-2206 did not affect the force of contractions in the cardiomyocyte culture. Ouabain increased the force of contraction in the culture, in a dose-dependent fashion, with an average increase of 160%. Importantly, 0.1 nM ouabain did not affect the culture force of contraction in comparison with that of the control group. Most significantly, the addition of 0.1 nM ouabain together with 0.1 nM MK-2206 produced a 124% ± 10.26% increase in cardiomyocyte contractility. Western blot analysis (Figure 6A) showed that exposure of the cells to 0.1 nM MK-2206 alone or in the presence of 0.1 nM ouabain did not cause any change in phosphor-Akt levels. These results demonstrate that in rat cardiomyocytes, as in the zebra fish model, combined treatment of cells with Akt inhibitor, at concentrations that do not reduce total Akt phosphorylation, together with ouabain, potentiates the effect of the steroid.
MK-2206 Potentiates Ouabain-Induced Inotropy in Langendorff-Perfused Rat Heart
Ouabain was previously shown to increase the force of contraction of heart muscle in the Langendorff preparation. 26 The dose dependence of ouabain on LVDP in our preparations is shown in Figure 2A. Since 1 µM did not yield a significant effect (Figure 2), this concentration was chosen for the following experiments. The IC50 of MK-2206 to Akt1, Akt2, and Akt3 is 5, 12, and 65 nM, respectively. 27,28 Hence, 5 nM MK-2206, which has a moderate effect on total Akt activity, was used. Male Sprague Dawley rat heart preparations were exposed to 1 µM ouabain with and without preincubation with 5 nM MK-2206. As shown in Figure 2, in this set of experiments, ouabain alone did not induce a significant increase in the heart force of contraction relative to the baseline (9.29 ± 4.68 mm Hg). Furthermore, with 5 nM MK-2206, which by itself did not affect heart contractility, the steroid increased heart contractility by 26.25 ± 5.57 mm Hg, relative to baseline. These results show that in mammals, as in the zebra fish model, inhibition of Akt potentiates ouabain inotropy.
Ouabain-Induced Inotropy Following MI
Echocardiographic measurements at 24 hours post LAD ligation were performed in order to test the functional consequence of the experimental manipulation and served as a reference point to the effects of ouabain treatments. Left anterior descending coronary artery ligation caused a significant reduction in heart function manifested by a decrease in FAC (15.5% ± 3.7) and SF (14.6% ± 2.5; Figure 3 and Supplemental S1, respectively), in comparison to baseline. The damage to the heart following LAD ligation was also manifested by the appearance of measurable Trop-T and significant increase in CPK in the blood samples (Table 1).
Myocardial Infarction (MI) Biomarkers at Baseline and 24 Hours Post MI.a

Abbreviations: CPK, creatine phosphokinase; LAD, left anterior descending coronary artery; Trop-T, troponin-T.
a Blood samples were collected from rats before (baseline) and 24 hours post LAD ligation and analyzed as described in Materials and Methods.
b Significantly higher than baseline levels, P < .0001.
The effects of different doses of ouabain on heart contractility following LAD ligation were tracked for 10 days. As can be seen in Figure 3, the lower ouabain doses of 0.8 and 4 mg·kg−1·d−1 did not change FAC in comparison to that at 24 hours post LAD ligation. On the other hand, treatment with ouabain at 8 mg·kg−1·d−1 led to a significant increase in FAC at 3, 6, and 10 days post LAD ligation. Similar results were obtained in the FS measurements (Supplemental Figure S1). Interestingly, the improvement in FAC, which ranged at about 30% relative to 24 hours post-MI, was stable throughout the period tested. The control group, which received saline throughout the experiment, showed no changes in heart parameters in comparison to 24 hours post LAD ligation (Figure 3). In agreement with previous studies, 29 low dose of ouabain did not affect heart rate in comparison to 24 hours post LAD ligation (Supplemental Figure S2). The lower concentration of ouabain (0.8 mg·kg−1·d−1), which by itself did not have any beneficiary effect on heart function following MI, was selected for the subsequent experiments.
MK-2206 Potentiates Ouabain-Induced Inotropy Following MI
To test the potential beneficiary effect of the combined treatment with ouabain and Akt inhibitor following LAD ligation, ouabain at 0.8 mg·kg−1·d−1 was administered to rats together with 10 mg·kg−1·d−1 MK-2206. Our results show that at 24 hours post LAD ligation all the animals showed reduced heart contractility, manifested by a significant decrease in FAC (20% ± 4.7%; Figure 4) and FS (11.6% ± 2.15%; Supplemental Figure S1) versus baseline. In addition, as mentioned above, the levels of CPK and Trop-T increased dramatically 24 hours post LAD ligation (Table 1), also indicating significant damage to the heart.
A possible beneficiary effect of the combined treatment of ouabain and MK-2206 on heart contractility was tested with doses of the 2 drugs which, when administered alone, have no effect on inotropy (Figure 4). The effects of the combined treatment were followed for 10 days post LAD ligation. As seen in Figure 4, the coadministration of ouabain and MK-2206 resulted in a significant increase in FAC by about 6% at all time points tested (3, 6, and 10 days post LAD ligation) versus the FAC values obtained 24 hours post MI. Similar findings were obtained upon calculating the FS from the echocardiography data (Supplemental Figure S3).
Combined Treatment of Ouabain and MK-2206 Reduces the Size of Scar Tissue Following MI
The beneficial effect of the combined treatment of ouabain and MK-2206 on heart contractility may result from a direct effect on the heart or indirectly due to neuronal or endocrine mechanisms. To address this point, the size of scar tissue in the hearts of animals following LAD ligation was determined in rats treated with ouabain or MK-2206 and a combination of the two. Hearts from the animals, 10 days post LAD ligation, were dissected, embedded in paraffin, and sectioned and stained with Masson trichrome to quantify the relative scar tissue area (Figure 5). Heart remodeling of rats treated with ouabain or MK-2206 for 10 days post LAD ligation showed no significant difference in collagen staining versus the control group. Conversely, the combined treatment of ouabain-MK-2206 led to a significant reduction of 55.98% ± 14.94% in the ratio of scar tissue to heart area in comparison with that in the control group (Figure 5).
Combined Treatment With Ouabain and MK-2206 Increases Heart Tissue α1 and α2 Isoforms of Na+, K+-ATPase Following MI
The α1 and α2 isoforms of Na+, K+-ATPase were measured in heart tissue regions distal to the ligated area, 10 days following MI. As can be seen in Figure 6A and B, whereas ouabain or MK-2206 treatment did not affect the isoform levels in the heart, the combined treatment increased α1 and α2 levels by 213% ± 21.76% and 314% ± 24.66%, respectively.
Low Doses of MK-2206 Do Not Reduce Ouabain-Induced Akt Phosphorylation Following MI
Ouabain has been shown to induce Akt phosphorylation in several experimental systems. 30,31 As can be seen in Figure 6B, treatment with 0.4 mg·kg−1·d−1 ouabain for 10 days post MI caused a 44.48% ± 11.62% increase in Akt phosphorylation in heart tissue regions distal to the ligated area. Treatment with 10 mg·kg−1·d−1 MK-2206 did not significantly alter Akt phosphorylation. Combined treatment with ouabain and MK-2206 did not reduce the ouabain-induced increase in total Akt phosphorylation. Note, the doses of MK-2206 used here were about 5% of those used for the inhibition of Akt in cancer treatment regimens. 32,33
Ouabain Attenuates MK-2206-Induced Reduction in Rat Primary Cardiomyocyte Viability
The possible additive or synergistic effect of ouabain and MK-2206 on cardiomyocyte viability was tested at different concentrations following 4 days of incubation. As can be seen in Figure 7B, high concentrations of 1 mM ouabain caused the death of the entire treated culture. Lower concentrations (up to 0.1 mM) of the steroid did not significantly affect cell viability. MK-2206 dose dependently inhibited cardiomyocyte viability (Figure 7A). Whereas 0.1 nM did not affect cell viability, a significant 31.87% ± 3.8% reduction in viability was apparent at 1 nM of the inhibitor, and higher concentrations of 10 and 100 nM caused total cardiomyocyte death. The possible cross talk between ouabain and MK-2206 was tested by exposing the cells to simultaneous additions of the 2 compounds (Figure 7C). Exposure of the cells to 1 nM MK-2206 and 1 nM or 10 nM ouabain resulted in a significant reduction of 15% ± 1.35% and 11% ± 2.70%, respectively, in cardiomyocyte viability, similar to the effects seen with MK-2206 alone. Exposure of the cardiomyocytes to 1 nM MK-2206 and 100 nM or 1 µM ouabain resulted in rescue of the cardiomyocytes from the toxic effect of MK-2206, resulting in 97% to 98% viability. These results demonstrate the protective effect of ouabain against MK-2206 toxicity and are in accord with the protective influence seen in vivo on scar tissue size following LAD ligation (Figure 5).
Discussion
Congestive heart failure (CHF) is a widespread disease that has an impact on health, worldwide. Despite advances in therapy, morbidity and mortality due to CHF remain high. 34 Advances in the understanding of the pathophysiological mechanisms of CHF and the development of new drugs for the treatment of the disease are of outmost importance. Since the canonical discovery of the beneficial effect of plants containing CS by Withering, more than 200 years ago, these steroids have been used for the treatment of CHF and atrial arrhythmia. 35 As mentioned above, the narrow therapeutic window of CS limits their clinical use. In recent years, the combined treatment of patients with CHF having CS and angiotensin converting enzyme inhibitors was found advantageous over treatment with CS alone. 36
The present study shows that combined treatment with ouabain and Akt inhibitor, MK-2206, augment the effect of the steroid. Doses of ouabain that did not affect contractility produced inotropy in the presence of MK-2206. This was demonstrated in 3 independent experimental rat models: neonatal primary cardiomyocytes (Figure 1), the Langendorff preparation (Figure 2), and rat LAD ligation model (Figure 4). Left anterior descending coronary artery ligation in rats leads to ischemia of heart muscle and is an acceptable murine model for the study of pathophysiology and pharmacological treatment of MI. 22 Although CSs are in use in the Western and Eastern medicine for centuries, there are no reports on the effect of this class of compounds in vivo, in experimental animal models of MI. This is the first demonstration of the advantage of such a treatment in a mammalian experimental model of MI.
A large discrepancy in the sensitivity to ouabain was noted in the 3 experimental systems. The highest noneffective concentrations of ouabain on inotropy were 0.1 nM, 1 µM, and 0.8 mg·kg−1·d−1 for cardiomyocytes (Figure 1), isolated perfused heart (Figure 2), and adult rats (Figure 3), respectively. This may be due to the significant difference in the accessibility of the steroid to the cells, that is, the thicker and more complex the preparation, the higher the concentration needed to produce the effect.
Two possible mechanisms should be considered for the augmentation of CS increased contractility by the presence of MK-2206. First, the effect could be mediated by the density and activity of the Na+, K+-ATPase, and second, the effect could be mediated by changes in intracellular signaling events. 1 The density of Na+, K+-ATPase in the plasma membrane was shown to be a determinant of CS effects. For example, it was found that the ouabain effect on several tissue culture growth is a function of Na+, K+-ATPase density in the plasma membrane. 37 In addition, the basis for the adverse effects of CS observed in ischemic hearts in both clinical and experimental settings is most likely multifactorial and includes alterations of the cardiac Na+, K+-ATPase density during ischemia. 38 –40 Hence, the increased CS-induced inotropy in the presence of Akt inhibitor may result partially from an elevation in plasma membrane Na+, K+-ATPase. The present finding of an increase in heart α1 and α2 isoforms under the combined treatment (Figure 6) supports the involvement of this proposed mechanism. Furthermore, it was shown that constitutively active mutants of Akt increased Na+, K+-ATPase activity. 41 This increase in activity was abolished by Akt inhibitors. Since the combined treatment of ouabain and MK-2206 increased Akt phosphorylation (Figure 6B), the involvement of changes in Na+, K+-ATPase activity in the underling mechanisms should also be considered. 2 It is well established that CSs induce the phosphorylation of several intracellular kinase cascades that are crucial to the CS-induced biological effects. 12,42,43 For example, the ouabain-induced phosphorylation of Akt in the heart was shown to stimulate growth and hypertrophy of cardiomyocytes. 10 In addition, the stretch of striated muscle causes an increase in Akt phosphorylation, 44 implicating the involvement of this kinase in the regulation of muscle contractility. We show here that the addition of Akt inhibitor in the presence of ouabain potentiates ouabain-induced heart contractility. It is conceivable that Akt activity imposes a constant inhibitory force on muscle contraction under steady state condition. Attenuation of this force (by low concentrations of MK-2206) augment the positive inotropic effects resulting from other stimulants. MK-2206 under this condition resulted a minimal change in total Akt phosphorylation or reduced activity of specific Akt isoform, both undetectable by the Western blot analyses.
The results of the present study may have far reaching clinical implications as they suggest the possible effective use of very low doses of CS for the treatment of CHF. Our results in rats show that administration of ouabain together with MK-2206 allows the use of one-tenth of the effective ouabain dose to obtain the beneficial effect of the steroid (Figures 1,2 and 4). As ouabain and digoxin have a similar dose–response relationship in increasing heart contractility 45,46 and because humans are at least 100-fold more sensitive to CS than rodents, 47 it may be anticipated that combined treatment with MK-2206 and digoxin would allow application of 1/500 of the doses used in this study, namely, 1.6 µg·kg−1·d−1 of the steroid, to increase the force of contraction when administered together with MK-2206.
It is well established that Akt activity has a crucial role in cell survival. 48 The addition of MK-2206 to the media causes a reduction in proliferation and viability of numerous cells. 49,50 Conversely, ouabain was shown to increase the viability and growth of many cells in culture and in vivo. 20,51,52 MK-2206 caused cell death in several tissue culture and in vivo. 27,32 This established toxic effect of MK-2206 on cell survival sets a potential limitation for the clinical use of MK-2206 in CHF. In agreement with these observations, we show that the inhibitor reduced the cell survival of primary rat cardiomyocytes (Figure 7). The study of the effect of MK-2206 and ouabain on cell viability revealed that exposure of rat primary cardiomyocytes to low concentrations of the 2 compounds, surprisingly, resulted in rescue of the cells from the toxic effect of MK-2206 (Figure 7C) and reduced post-MI scar tissue size (Figure 5). These results imply that the combined treatment proposed in this study will not have a deteriorating effect on cardiac survival.
In conclusion, our novel findings on the beneficial effect of the combined treatment with ouabain and MK-2206 on heart contractility should prompt the pharmaceutical industry to study preparations based on these features.
Supplemental Material
Supplements - Augmentation of Ouabain-Induced Increase in Heart Muscle Contractility by Akt Inhibitor MK-2206
Supplements for Augmentation of Ouabain-Induced Increase in Heart Muscle Contractility by Akt Inhibitor MK-2206 by Nahum Buzaglo, Mordechai Golomb, Haim Rosen, Ronen Beeri, Hagit Cohen-Ben Ami, Fattal Langane, Sandrine Pierre, and David Lichtstein in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
David Lichtstein, Ph.D, is the Walter and Greta Chair in Heart Studies. F.L. was a participant in the Summer Research Internship for Minority Students (SRIMS) program supported by Marshall University Joan C. Edwards School of Medicine Biomedical Sciences Graduate Program.
Author Contribution
N.B. contributed to conception and design, contributed to acquisition and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; H.R. contributed to analysis, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; M.G. contributed to acquisition and drafted the manuscript; R.B. contributed to design, contributed to analysis, and drafted the manuscript; H.C.B-.A. contributed to acquisition and analysis, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; F.L. contributed to acquisition and analysis; S.P. contributed to design, contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; D.L. contributed to conception and design, contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported in part by the Sam Lazarus Fellowship, The Hebrew University, to N.B.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


